Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Eosinophils are highly specialized granulocytic effector cells that produce and store numerous biologically active mediators, including cytotoxic proteins, lipid mediators, chemotactic peptides, and cytokines ( Table 74.1 ). Under various pathologic conditions, blood eosinophils transmigrate through the endothelial layer and invade various target organs, where these cells secrete their products into the surrounding tissues, thereby triggering inflammation, toxic damage, and tissue remodeling. Since their initial characterization by Paul Ehrlich in 1879, eosinophils have been implicated in a number of systemic diseases and conditions characterized by blood and/or tissue eosinophilia ( Table 74.2 ). During the past few decades, researchers have also gained a better understanding of the unique features and functions of activated eosinophils and of the specific roles some of their granule proteins and inducible lipid mediators may play in the pathogenesis of allergic, parasitic, neoplastic, and other diseases. In addition, recognition of the eosinophil as a major pro-inflammatory and tissue-remodeling effector cell has fueled a surge of interest in this granulocyte.
| Eosinophil Product/Molecule | Relevant Function(s) |
|---|---|
| Eosinophil-Related Enzymes | |
| Nonspecific esterase | Cytotoxic effects (microbes and tissue cells) |
| Catalase | Cytotoxic effects (microbes and tissue cells) |
| Acid phosphatase | Cytotoxic effects (microbes and tissue cells) |
| Lysophospholipase | Tissue inflammation |
| Phospholipase D | Migration, adhesion, vesicle transport, secretion |
| Hexosaminidase | Tissue inflammation and tissue remodeling |
| Arylsulphatase B | Lysosomal hydrolase |
| 5-Lipoxygenase | Leukotriene production, angiogenesis |
| Leukotriene C4 synthase | Leukotriene production |
| Cyclooxygenase | Prostaglandin production |
| Histaminase | Histamine degradation |
| Eosinophil Basic Proteins | |
| Eosinophil peroxidase (EPX) | Cytotoxic effects (parasites, microbes, tissue cells) |
| Major basic protein-1 (MBP-1) | Cytotoxic effects (parasites, microbes, tissue cells) |
| Major basic protein-2 (MBP-2) | Cytotoxic effects (parasites, microbes, tissue cells) |
| Eosinophil cationic protein (ECP) | Cytotoxic effects, mucus secretion, fibrosis |
| Eosinophil-derived neurotoxin (EDN) | Cytotoxic effects (antiviral, RNase activity) |
| Chemokines | |
| Interleukin-8 (IL-8) | Leukocyte recruitment and activation |
| MIP-1-alpha (CCL3) | Leukocyte recruitment and activation |
| RANTES (CCL5) | Leukocyte recruitment and activation |
| Eotaxin (CCL11) | Further eosinophil recruitment |
| Growth Factors/Cytokines | |
| Interleukin-1-α (IL-1α) | Endothelial activation, inflammation |
| Interleukin-2 (IL-2) | Activation of T lymphocytes |
| Interleukin-3 (IL-3) | Amplification of eosinophil accumulation/activation and basophil activation |
| Interleukin-4 (IL-4) | B-cell maturation and mast cell differentiation |
| Interleukin-5 (IL-5) | Amplification of eosinophil accumulation/activation |
| Interleukin-6 (IL-6) | Lymphocyte maturation |
| Interleukin-13 (IL-13) | Bronchial hyperreactivity, mucus production, B-cell maturation |
| GM-CSF | Leukocyte/eosinophil accumulation/activation |
| TGF-α | Fibrosis, growth inhibition |
| TGF-β | Fibrosis, growth inhibition |
| TNF-α | Endothelial activation, inflammation, cachexia |
| Oncostatin-M (OSM) | Fibrosis, angiogenesis, paracrine mobilization of eosinophils via upregulated SDF-1 production in fibroblasts |
| Lipid Membrane-Derived Substances | |
| PAF | Bronchoconstriction, edema formation |
| TXB2 | Platelet aggregation |
| LTC4 | Mucus secretion |
| 15-HETE | Diverse effects on blood and tissue cells |
| PGE1 and PGE2 | Diverse effects on platelets, endothelial cells, fibroblasts and other tissue cells |
| Antifibrinolytic Mediators | |
| PAI-2 | Antifibrinolytic and prothrombotic |
| Extracellular DNA traps | Fibrin deposition, antibacterial effect |
| Reactive Conditions |
|
| Endocrinologic Conditions |
| Addison disease |
| Neoplastic Conditions Involving the Hematopoietic System |
|
| Paraneoplastic Conditions |
|
| Idiopathic Forms |
|
a Idiopathic mild eosinophilia (500 to 1500 eosinophils/μL blood = less than HE) of unknown etiology.
Studies of the biochemistry, biologic activities, and tissue localization of distinct enzymatic and non-enzymatic cationic proteins derived from eosinophils have provided convincing evidence for their role in the pathogenesis of inflammation and tissue damage in eosinophil-associated diseases. The five cationic granule proteins that may play a role in eosinophil-related pathologies include two major basic proteins (MBP-1 and MBP-2), eosinophil peroxidase (EPX), and two ribonucleases, namely eosinophil cationic protein (ECP) and eosinophil-derived neurotoxin (EDN; see Table 74.1 ). Eosinophils also have the capacity to express toxic oxidative intermediates and other mediators of inflammation, as well as molecules that may contribute to the development of thrombosis and fibrosis. In addition, eosinophils are a rich source of DNA traps that may facilitate fibrin deposition, as well as the killing of microbial invaders. A clinically important aspect is that the various mediators produced and released by (activated) eosinophils often act together to trigger thrombosis and tissue damage, especially when eosinophil expansion and activation is chronic and “treatment-resistant.”
The process of determining the cause of eosinophilia is often frustrating for both the physician and the patient, and, in many instances, the resulting diagnosis is “eosinophilia of unknown etiology.” Depending on laboratory standards and local guidelines, eosinophilia is defined as more than 0.4 to 0.5 × 10 9 /L blood, detected by microscopy.
Diurnal variations in eosinophil counts are well documented, with minimum numbers appearing early in the morning and greatest numbers appearing late at night, mirroring circadian rhythms in adrenal corticosteroids. Although these variations are usually limited to a certain extent, basal eosinophil counts should be routinely measured during the daytime. Transient mild eosinophilia is commonly seen in allergic reactions, during and shortly after a bacterial infection, and in many other reactive states. Sometimes, transient eosinophilia may be substantial or even excessive. Such transient (<4 weeks) forms of reactive eosinophilia are considered harmless in most instances but should prompt the physician to search for certain underlying diseases, such as an occult allergy or an unrecognized infection.
Eosinophil disorders and related syndromes are a heterogeneous group of diseases characterized by marked expansion and persistent accumulation of eosinophils in the peripheral blood (PB) and other organ systems. In general, eosinophil disorders can be divided into (A) neoplastic states where eosinophils are monoclonal and (B) reactive states where eosinophil expansion is considered to be “poly-clonal” and triggered by eosinotropic cytokines, such as interleukin-5 (IL-5). In both instances, hypereosinophilia (HE), defined by a persistent eosinophil count of ≥1.5 × 10 9 /L blood, is typically present. Depending on the underlying disease and other factors, the HE state may or may not be accompanied by specific (HE-mediated) organ damage, also referred to as hypereosinophilic syndrome (HES). In many instances, HES-related organ damage manifests as overt thromboembolism (thrombosis) or fibrosis. In other patients, organ damage is associated with less-specific findings, and may involve the skin, gastrointestinal (GI) tract, or the central nervous system (CNS). Based on the underlying condition, both HE and HES can be divided into familial, primary (neoplastic), and secondary (reactive) forms ( Table 74.3 ). Apart from these classical forms of HES, more specific syndromes associated with HE and HE-related organopathies have been described. Some of these syndromes are associated with a hereditary (germ-line) defect, whereas others are accompanied by distinct immunological abnormalities ( Table 74.4A ). Finally, HE may develop in the context of organ-restricted inflammatory conditions such as eosinophilic pneumonia or eosinophilic colitis ( Table 74.4B ).
| HE Variant | Abbreviation | Associated HES Variant |
|---|---|---|
| Familial/hereditary HE | HE FA | Familial HES = HES FA |
| HE of undetermined/Unknown significance | HE US a | Idiopathic HES = HES I a |
| Neoplastic/primary HE | HE N | Neoplastic HES = HES N |
| Reactive/secondary HE | HE R | Reactive HES = HES R |
| Lymphocytic variant = HES L b |
a In patients with HE US , the presence of HES is excluded by definition; as soon as HES is diagnosed in a patient with HE US , the diagnosis changes to HES I , unless an underlying etiology (condition/disease) is also found.
| Syndrome | Characteristic Features and Laboratory Abnormalities |
|---|---|
| Gleich syndrome | Cyclic recurrent angioedema, fever, weight gain, elevated polyclonal IgM, increased interleukin-5 production, evidence of clonal T cells (CD3 – T cells) a |
| Churg-Strauss syndrome (CSS) = eosinophilic granulomatosis with polyangiitis (EGPA) | Stage I: Asthma and rhinosinusitis (isolated) |
| Stage II: Eosinophilic phase (HES-like) | |
| Stage III: Vasculitic phase defined by necrotizing (small-vessel) vasculitis and HE with pulmonary infiltrates and/or neuropathy (ANCA+ and ANCA− subvariants have been described) | |
| Eosinophilia myalgia syndrome (EMS) | Severe myalgia ± fever (flu-like), edema, fatigue, weight loss, neurologic symptoms, skin abnormalities |
| Epidemic cases have been reported to result from exposure to L-tryptophan (toxic oil syndrome). | |
| Eosinophilic fasciitis (EF) = Shulman syndrome | Scleroderma-like condition with painful swelling of the skin and chronic induration (limbs and trunk) as well as laboratory signs of systemic inflammation |
| Omenn syndrome (OS) | Severe combined immunodeficiency (SCID) with well-populated lymphatic organs (OS paradox) |
| Autosomal recessive genetic (pediatric) disease (recurrent mutations in RAG1 or RAG2 ) | |
| Clinical findings: erythroderma, hepatosplenomegaly, lymphadenopathy, increased IgE, diarrhea, weight loss (GVHD-like condition with autoreactive T cells) | |
| Hyper-IgE syndrome (HIES) | Hereditary immunodeficiency syndrome with elevated IgE, recurrent severe infections, often with skin eczema and facial anomalies |
| Examples of mutations associated with HIES: | |
| Autosomal dominant variant: STAT3 mutations | |
| Autosomal recessive variant: DOCK8 mutations PGM3 mutations |
a EGPA/CSS with clonal T cells is often regarded as special variant of lymphoid variant HES (HES L ).
|
a Many other skin abnormalities can be accompanied by eosinophilia, especially when a systemic inflammatory disease process is present and triggers eosinophilia.
A thorough examination of all clinical and laboratory parameters, including radiologic and imaging studies, bone marrow (BM) investigations, karyotype, and molecular studies, are required in order to establish the correct diagnosis (underlying disease and organ damage) in patients with initially unexplained HE. This chapter provides an overview on the epidemiology, pathogenesis, course, prognosis, and clinical features of various eosinophil disorders. We also discuss developments in the field and the impact of molecular markers and targets. In addition, this chapter provides diagnostic algorithms and recommendations for the management and treatment of patients with eosinophil disorders.
Little is known about the prevalence and incidence of primary eosinophil disorders (eosinophilic neoplasms) and HE-related syndromes, including HES. Based on the available literature, most eosinophil neoplasms and all types of HES are rare, and the same holds true for other HE-related conditions, such as the Gleich syndrome or Churg-Strauss syndrome (CSS), also known as eosinophilic granulomatosis with polyangiitis (EGPA). In an attempt to estimate the incidence of HES, data collected by the Surveillance, Epidemiology and End Results (SEER) database, sponsored by the National Cancer Institute, have been reviewed. A crude incidence of 0.035 cases per 100,000 person-years was found, and the male-to-female ratio was reported to be 1.47 to 1. The average age at diagnosis was 52.5 years, and the peak incidence was recorded in individuals aged 65 to 74 years. Childhood cases of HES have been reported but are very rare. Unfortunately, all these data have serious limitations and emphasize the need for more comprehensive epidemiologic studies using currently available diagnostic criteria and robust registries. A complicating issue is that HES may coexist with an underlying primary eosinophil disorder, a lymphoma, but also an allergy or another reactive condition. In eosinophil neoplasms presenting with the FIP1L1::PDGFRA fusion gene ( F/P ), a clear male predominance is found. As a consequence, any HES registry, including patients with F/P + eosinophilic leukemia and/or other F/P + myeloproliferative neoplasms (MPNs) with eosinophilia (MPN-eo), will report an overall male predominance of HES, even if other HES types are evenly distributed between males and females. Indeed, no clear gender predominance has been reported in other groups of HE or HES. Familial forms of HE and HES have been described but are very rare. In these cases, a hyper-immunoglobulin (Ig)E syndrome or Omenn syndrome (OS) have to be excluded. No cases of familial F/P + leukemia have been reported to date.
Eosinophils contain three populations of granules, namely (1) electron-dense, round, primary progranules present mainly at the promyelocyte and myelocyte stages, (2) specific (secondary) eosinophilic granules with an electron-dense crystalloid core, specifically found in mature eosinophils, and (3) small-sized granules, which may contain catalase, acid phosphatase, and arylsulfatase. The large, specific granule is the major source of cytotoxic and pro-inflammatory cationic proteins. Eosinophils also contain lipid bodies that may serve as a source of eosinophil eicosanoide-forming enzymes, including 5-lipoxygenase, leukotriene C4 (LTC4) synthase, and cyclooxygenase (see Table 74.1 ).
The immunological phenotype of human eosinophils is well established and may assist in eosinophil detection and enumeration by flow cytometry. In common with all leukocytes, eosinophils express leukosialin (CD43), the homing cell adhesion molecule HCAM (CD44), and the pan-leukocyte tyrosine phosphatase C (CD45). In addition, eosinophils express several myeloid differentiation antigens, including LFA-1 (CD11a/CD18) and Siglec-3 (CD33). These cell-surface antigens are also expressed on other myeloid cells, including monocytes and basophils. However, a few cell-surface structures are largely restricted to eosinophils and thus serve as cell-specific markers through which eosinophils can be detected and isolated. One of these markers is Siglec-8, an inhibitory receptor that, when crosslinked, mediates eosinophil apoptosis. Apart from eosinophils, basophils also display Siglec-8 ( Fig. 74.1 ).
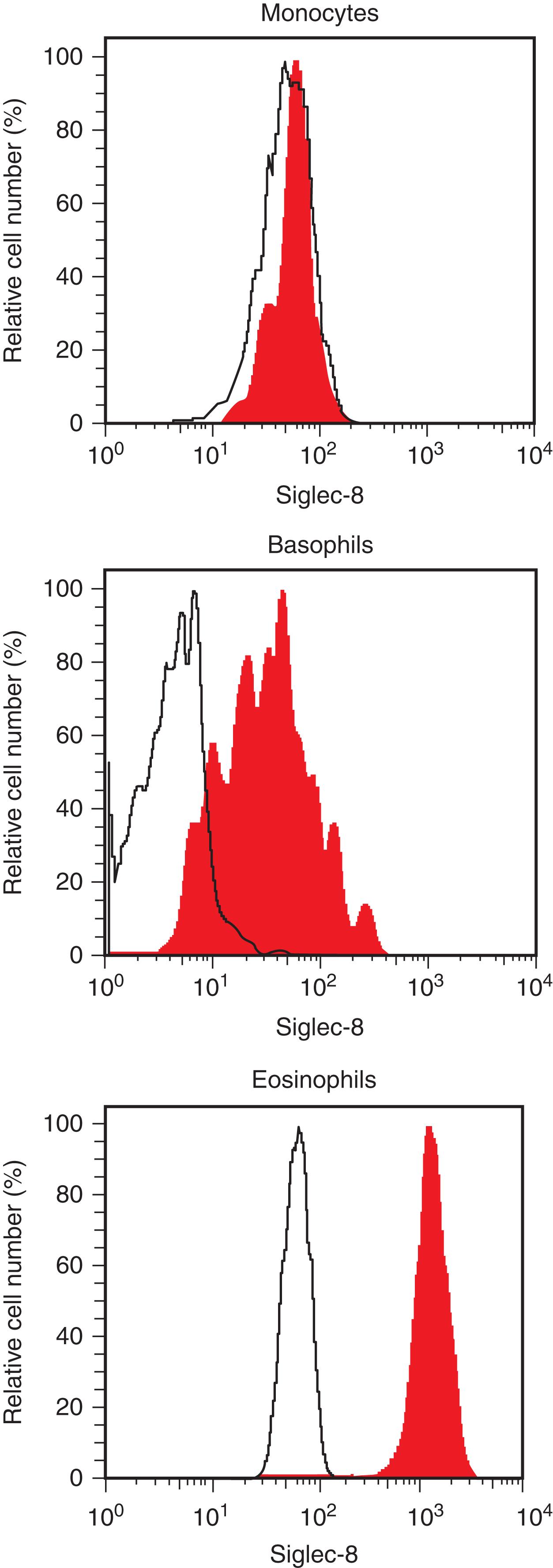
Eosinophils express several cytokine receptors, such as the IL-5 receptor, granulocyte-macrophage colony-stimulating factor (GM-CSF) receptor, and IL-3 receptor. Moreover, eosinophils exhibit various chemokine receptors, including eotaxin receptors and CXC-chemokine receptor 4 (CXCR4), a receptor for stromal cell-derived factor (SDF)-1 ( Table 74.5 ). Finally, eosinophils express diverse complement receptors (CRs), such as CR3 (CD11b/CD18) and C5aR (CD88), adhesion receptors, and Ig (Fc) receptors. Several of these cell-surface markers have been considered as potential targets of therapy. However, aberrantly expressed or disease-specific markers for neoplastic eosinophils or reactive eosinophils have not been identified yet. Several studies have shown that eosinophils in HES patients may express increased amounts of CD11b, CD16, CD25, or/and HLA-DR compared to normal blood eosinophils. However, overexpression of these antigens on eosinophils is not disease-specific.
| Receptor (R) on Eosinophils | Ligand | Effects on Eosinophils |
|---|---|---|
| IL-2RA/CD25 a | IL-2 | Activation? a Migration? a |
| IL-3R/CD123 + CD131 | IL-3 | Differentiation, survival, adhesion, migration, activation, priming |
| IL-4R/CD124 | IL-4 | Priming for effects of chemotaxins |
| IL-5R/CD125 + CD131 | IL-5 | Differentiation, survival, adhesion, migration, activation, priming |
| GM-CSFR/CD116 + CD131 | GM-CSF | Differentiation, survival, adhesion, migration, activation, priming |
| IL-10R | IL-10 | Inhibitory (activation, survival) |
| IL-12R | IL-12 | Inhibitory (activation) |
| IL-13R | IL-13 | Unknown |
| CD4 | IL-16 (LCF) | Activation, priming |
| IL-25R | IL-25 | Survival, activation |
| IL-27R | IL-27 | Survival, activation |
| IL-33R/ST2 | IL-33 | Activation, survival |
| VEGFR-1/FLT-1 | VEGF | Chemotaxis, activation |
| Tie-2/TEK | Angiopoietin-1 | Chemotaxis, activation? |
| PDGFRA/B | PDGF | Activation and growth? |
| FGFR | FGF | Activation? |
| TGFβ1R | TGFβ1 | Inhibitory (differentiation) |
| TGFβ2R | TGFβ2 | Inhibitory (differentiation) |
| IFN-α-R | IFN-α | Inhibitory (growth) |
| IFN-γ-R | IFN-γ | Inhibitory (growth, migration) |
| CCR3 (CD193) | RANTES (CCL5) | Chemotaxis, activation |
| MCP-3 (CCL7) | Chemotaxis, activation | |
| MCP-4 (CCL13) | Chemotaxis, activation | |
| Eotaxin-1 (CCL11) | Chemotaxis, activation | |
| Eotaxin-2 (CCL24) | Chemotaxis, activation | |
| Eotaxin-3 (CCL26) | Chemotaxis, activation | |
| CXCR4 (CD184) | SDF-1 (CXCL12) | Chemotaxis |
| PAF-R | PAF | Chemotaxis, activation |
| C5aR (CD88) | C5a | Chemotaxis, activation |
| TLR1, 4, 7, 9, 10 | Toll-like R-ligands | Survival, activation |
| Corticosteroid R | Corticosteroids | Inhibitory (activation) |
a Eosinophils derived from patients with hypereosinophilic syndromes (activated eosinophils) may express CD25. However, the role of CD25 in eosinophil function remains unknown.
Apart from cell surface antigens, eosinophils also express more or less specific marker antigens in their cytoplasm by which these cells can be detected in various tissues by immunohistochemistry (IHC). Useful eosinophil IHC stains include ECP, EPX, and MBP-1 ( Fig. 74.2A ). Depending on the condition and underlying neoplasm, these antibodies may also react with (immature) basophils. Otherwise, however, the staining reaction appears to be largely specific for eosinophil granulocytes. Another important aspect is that some of the eosinophil proteins, like MBP, can be detected in tissue sections as deposit material after complete degranulation of eosinophils and even after their subsequent destruction (disappearance), which is often seen in the context of chronic HE-related tissue damage (HES). Therefore, the definition of tissue HE includes not only the detection of a local eosinophil (cellular) infiltrate but also the presence of eosinophil protein deposits, even in the absence of infiltrating eosinophils.
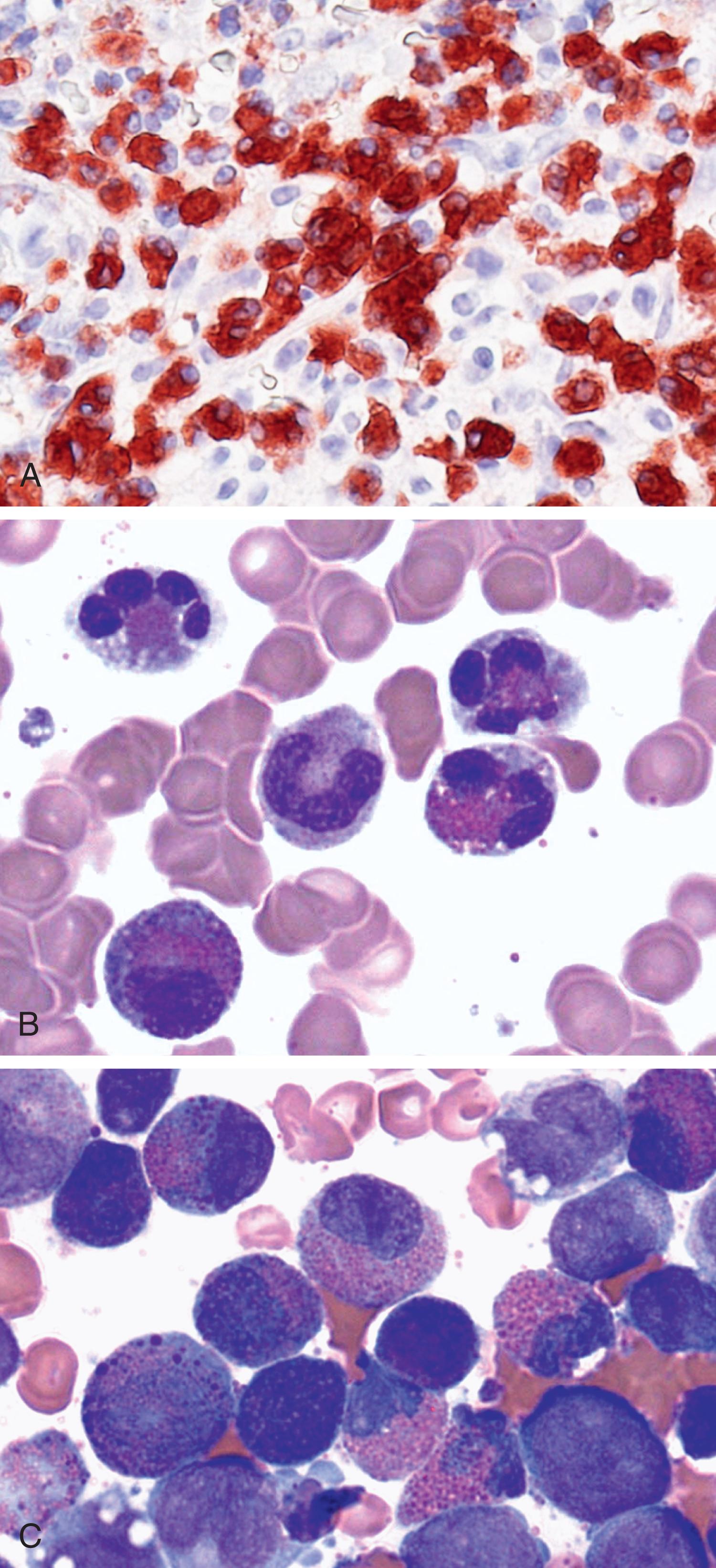
Eosinophils from the PB and tissues of patients with HE may differ from their normal counterparts by morphologic, biochemical, and functional characteristics. The most important morphologic distinction is the one between immature and mature eosinophils. In most instances, this distinction is a straightforward approach, as immature forms exhibit distinct nuclear and cytoplasmic morphologic features ( Fig. 74.2B and C ). However, sometimes the distinction may be challenging, especially in reactive states with massive inflammation and in certain myeloid neoplasms. In immature myeloid neoplasms, eosinophil precursor cells may exhibit dark basophilic granules that must not be confused with the specific granules of blood basophils. These immature eosinophils may also be misinterpreted as mixed-lineage (eosinophil/basophil) cells.
Immature eosinophils also differ from mature eosinophils in several other aspects, including density, nuclear shape, protein composition, and cell surface antigens. A difficult task is to differentiate normal eosinophils from activated (sometimes immature) eosinophils in reactive states. In previous years, researchers focused on cell density. In particular, a distinction was made between the “normodense” (normal-density) and the “hypodense” (light-density) eosinophil. The blood of normal individuals contains less than 10% eosinophils with densities less than 1.082 g/mL, whereas patients with eosinophilia can have markedly increased numbers of hypodense cells. Hypodense eosinophils often appear to be cytokine-activated cells that are characterized by increased vacuolization, decreased granule size, decreased content of granule cationic proteins, and increased numbers of cytoplasmic lipid bodies. The numbers of surface receptors for a variety of eosinophil agonists, including complement components, Igs, and platelet-activating factor (PAF), are expressed at higher levels by hypodense eosinophils. Functionally, hypodense eosinophils show increased metabolic activity and oxygen consumption, an increased capacity for the synthesis and secretion of LTC4 and certain cytokines, enhanced chemotaxis, and augmented cytotoxicity. So far, no clear relationship between the percentage of hypodense eosinophils and the etiology of eosinophilic disorders has been established. The percentage of these cells increases in most reactive forms of HE and HES, but also in patients with primary HES. Ultrastructurally, HES eosinophils may show a selective loss of secondary granule components (crystalloid MBP-containing core or granular matrix, or both), a decreased number and size of granules, and increased numbers of cytoplasmic lipid bodies and tubulovesicular structures that may be involved in eosinophil secretion during the process of piecemeal degranulation.
Eosinophils originate from pluripotent and granulocyte-committed, CD34 + , hematopoietic progenitor cells that are detectable in the BM and in the PB, and under certain conditions (local inflammation) also in various extramedullary organ sites. Eosinophil development from their multipotent and lineage-restricted progenitors is controlled by a network of transcription factors, including GATA-1, GATA-2, C/EBP-A, and C/EBP-E (see Chapter 9 ). Lineage-specific signaling as well as transcription factor expression is controlled by a network of cytokines and cytokine receptors. In fact, eosinophils develop from their progenitors in response to T-cell–derived growth factors, including IL-3, GM-CSF, and IL-5 (see Chapter 11 ). The most potent and specific eosinophilopoietic growth factor is IL-5. Eosinophils are derived from multipotent or bipotent colony-forming progenitor cell units (CFUs). Bipotent CFUs giving rise to eosinophils and basophils (CFU-eo/ba) are frequently detected in the BM and PB in healthy individuals, as well as in various disease states. By contrast, progenitor cells selectively giving rise to eosinophils and mast cells (CFU-eo/mast) are very rare. At an early stage of progenitor cell development, eosinophil-committed progenitors exhibit CD34 and CD38, and coexpress receptors for IL-3, GM-CSF, and IL-5. Later, CD34 expression is diminished, whereas the interleukin receptors continue to be expressed, and several additional, functionally important, cell-surface antigens are acquired by maturing eosinophils.
IL-3, GM-CSF, and IL-5 promote eosinophil differentiation of hematopoietic progenitor cells and eosinophil maturation of lineage-restricted precursors, as well as the recruitment, priming, activation, and survival of mature eosinophils. Whereas IL-3 and GM-CSF also affect other hematopoietic lineages, including basophils, IL-5 acts as a more lineage-restricted (eosinophil-targeting) cytokine. Therefore, IL-5 and IL-5 receptors have been recognized as major targets of therapy in diseases associated with (reactive) HE. The high-affinity receptor for IL-5 is composed of an IL-5–specific alpha chain (CD125) and a beta chain (CD131) that mediates intracellular signaling and is shared also by the high-affinity receptors for GM-CSF (CD116/CD131) and IL-3 (CD123/CD131; see Table 74.5 ) (see Chapter 11 ). Expression of high-affinity IL-5 receptors is an important prerequisite for specific cellular responses to “physiologic doses” of IL-5. Overexpression of IL-5 is observed in many reactive conditions and disease states associated with (reactive) HE. Correspondingly, IL-5 transgenic mice develop profound eosinophilia, and IL-5 knockout mice do not develop HE in response to allergic stimuli or a helminth infection. Under various reactive conditions accompanied by HE, IL-5 is produced primarily by activated Th2-type helper cells. However, although activated T-cells are considered a primary and most important source of eosinophil-triggering cytokines, like IL-5, other cell types, such as mast cells, macrophages, natural killer (NK) cells, and stromal cells, also produce these cytokines. In myeloid neoplasms with HE, clonal expansion of eosinophils usually is a growth factor-independent process. Moreover, depending on the underlying disease, neoplastic eosinophils may produce eosinophilopoietic cytokines in an autocrine manner, thereby augmenting tissue and blood HE.
In healthy individuals as well as in reactive states, eosinophils circulate only briefly in the PB, and then transit to extravascular sites. Depending on environmental factors and demand, eosinophils localize preferentially in tissues and organs exposed to external environments, principally the submucosal membranes and loose connective tissues of the skin, GI tract, genital tract, and lungs. Depending on the underlying pathology, eosinophils can migrate into any vascularized organ. During an acute or chronic inflammatory process, larger numbers of eosinophils may be recruited actively into local tissue sites. The mobilization of eosinophils from the vasculature into (vascularized) tissues is a multistep process that involves their rolling on vascular endothelial cells and their subsequent adhesion to endothelium via L-selectin, followed by binding to intercellular adhesion molecule-1 (ICAM-1 = CD54) through a CD18/CD11a/b-dependent mechanism ( Chapter 14, Chapter 16 ). Expression of selectins on endothelial cells is triggered by histamine and other mediators, whereas expression of ICAM-1 on endothelial cells is induced by various pro-inflammatory cytokines, such as tumor necrosis factor-α (TNF-α). As both histamine and TNF-α are produced and provided by (activated) mast cells, these cells are thought to play a crucial role in eosinophil recruitment in various allergic and other inflammatory reactions. However, the recruitment of eosinophils is a complex process involving many different mechanisms and molecules, such as E-selectin, endothelial-leukocyte adhesion molecule 1 (ELAM-1 = CD62E) or vascular cell-adhesion molecule-1 (VCAM-1 = CD106). Recruitment of eosinophils is also mediated by adhesion to VCAM-1 via the β1 integrin very late activation antigen-4 (VLA-4 = CD49d/CD29), which is expressed by eosinophils but not by blood neutrophils.
Eosinophil migration is regulated by several different chemotactic factors, including various chemokines (SDF-1, eotaxins, IL-8, and others), the complement fragment C5a, PAF, and several eosinotropic cytokines, such as IL-5 or GM-CSF, which also prime eosinophils for enhanced migratory responses to PAF and IL-8. In addition, IL-2, leukotriene B 4 , and the lymphocyte chemoattractant factor (LCF = IL-16) can induce eosinophil migration. At least for neoplastic eosinophils, a most potent chemoattractant appears to be SDF-1, a stromal cell-derived chemokine that binds to CXCR4 (CD184) on normal and neoplastic eosinophils. Other potent chemoattractants for (neoplastic) eosinophils are the eotaxins (eotaxin-1, -2, -3) that bind to and signal through the chemokine receptor CCR3 (CD193; see Table 74.5 ). Depending on the underlying disease, the cell type(s) involved, and the presence of comorbidities (triggering, e.g., local or systemic inflammation) all these chemotactic factors may act together to trigger eosinophil recruitment and accumulation in the affected (often inflamed) tissue sites. Likewise, in several reactive conditions, IL-5 and eotaxin may cooperate locally to promote eosinophil migration and tissue HE.
The primary source of eosinophil chemotactic factors often remains unknown. In many disease states accompanied by HE, eosinophil chemotactic factors may be produced by microenvironmental cells, including fibroblasts, endothelial cells, T-cells, macrophages, and mast cells. Several of these chemokines, like SDF-1, are produced by stromal cells in a paracrine fashion (see Chapter 12 ). For example, in neoplastic eosinophils, the F/P mutant triggers expression and release of oncostatin M (OSM), which in turn induces fibroblast and endothelial cell proliferation but also the expression and release of SDF-1 from these cells, thereby augmenting the accumulation of additional eosinophils. In this regard, it is noteworthy that the SDF-1 receptor CXCR4 is not only expressed on mature eosinophils but also on hematopoietic stem cells and eosinophil-committed precursor cells. Depending on the type of underlying neoplasm, eosinophil chemotactic cytokines may also be produced in an autocrine manner.
In various inflammatory and allergic reactions, several different interactions between eosinophils and mast cells have been postulated (see Chapter 75 ). For example, mast cells provide the immediate stimuli upon activation to initiate allergic inflammation, resulting in eosinophil recruitment during the late phase following an allergen encounter. In addition, mast cells and eosinophils are often increased in numbers in allergic reactions, but also in non-allergic inflammatory conditions and even in neoplastic states. Both cell types express a similar profile of chemotactic receptors, such as CCR3, and both respond to eotaxins and RANTES (CCL5), leading to their recruitment into local tissue sites (see Chapter 12 ). Mast cells and eosinophils also interact with each other via cell–cell contact and through several different soluble mediators. Likewise, mast cell-derived heparin binds and stabilizes various chemokines, including the eotaxins. In addition, mast cells are a source of various cytokines involved in the regulation of eosinophil adhesion, migration, and function. As previously mentioned, mast cells express and release histamine and TNF-α, and thereby facilitate eosinophil recruitment. Mast cell proteases appear to have a dual action on eosinophil functions. Mast cell-derived chymase suppresses eosinophil apoptosis and induces the release of IL-6 and various chemokines (CXCL1, CXCL8, CCL2) from eosinophils. Beta-tryptase, on the other hand, can cleave eotaxin and RANTES, and thereby may limit eosinophil chemotaxis. Apart from these interactions, eosinophils and mast cells may sometimes also be derived from the same stem or progenitor cell compartment, and therefore may express the same driver mutant, which has been documented for cases of F/P + chronic eosinophilic leukemia (CEL) with accompanying mast cell expansion, as well as for patients with KIT D816V+ systemic mastocytosis (SM) with concomitant HE (see Chapter 75 ). Therefore, mast cells and eosinophils may often appear as a dual-target cell population, both in reactive and neoplastic states, which is a critical point when considering the development of specific therapies. For example, simultaneous downregulation of eosinophil and mast cell activation might be achieved by certain inhibitory surface molecules, such as CD300a or Siglec-8. Several other target receptors, such as Siglec-3 (CD33), are expressed on eosinophils, mast cells, and basophils. In patients with F/P + neoplasms, imatinib can induce apoptosis in both eosinophils and mast cells, as both cell types express the mutant platelet-derived growth factor receptor (PDGFR) target in this neoplasm.
In the PB, eosinophils can easily be measured by microscopy or flow cytometry based on their characteristic side-scatter properties and their autofluorescence. In case of atypical (immature) cells or questionable results, immunophenotyping can be performed to confirm the presence of eosinophils. In tissue sections, the quantification of eosinophils is a more difficult task that makes routine clinical evaluations somewhat impractical. Alternative approaches such as analysis of tissue secretions from affected organs (e.g., bronchoalveolar lavage [BAL]) have been used with success in evaluating local eosinophil numbers and function in asthma. In addition to routine histochemical identification and enumeration of eosinophils and the immunochemical localization of secreted eosinophil granule cationic proteins in tissue biopsies, two other methods have been employed to demonstrate and monitor eosinophil activation and involvement: (1) the identification of activated eosinophils by staining with anti-ECP antibody EG2, recognizing a secreted (deglycosylated) form of the protein, and (2) measurement of eosinophil granule proteins, such as MBP or ECP by radioimmunoassay (RIA) or enzyme-linked immunosorbent assay in various body fluids, including serum, plasma, urine, sputum, nasal lavage, and BAL. Indeed, these antigens may serve as reliable biomarkers of eosinophil involvement and activation in allergic, parasitic, and inflammatory diseases. In addition, treatment responses can be demonstrated and measured with these assays.
In patients with documented HE, four etiologies have to be considered: (1) myeloid (and rarely myeloid/lymphoid) neoplasms where eosinophils are usually derived from neoplastic stem and progenitor cells, (2) lymphoid neoplasms or nonhematopoietic (solid) tumors where HE is a paraneoplastic phenomenon, (3) reactive conditions, such as an allergy, infection or an autoimmune disease where HE is a reactive process, and (4) rare syndromes accompanied by HE, including rare and inherited disorders (see Table 74.4A ). In patients with clonal (primary) HE, eosinophils and their progenitors often display rearrangements in PDGFRA , PDGFRB , or FGFR1 genes. Therefore, the World Health Organization (WHO) has employed these mutations as primary criteria to describe (classify) hematopoietic neoplasms accompanied by HE ( Table 74.6 ). The resulting “fusion proteins” act as prooncogenic drivers and are considered to contribute to cytokine-independent differentiation of eosinophil progenitor cells. The most frequently detected molecular abnormality is the F/P fusion gene, a mutant gene created by an 800-kb interstitial deletion on chromosome 4q12. The fusion gene is detectable by polymerase chain reaction (PCR) as well as by fluorescence in situ hybridization (FISH; deleted CHIC2 gene) but cannot be detected by conventional karyotyping. However, conventional cytogenetics may reveal translocations, deletions, or inversions on chromosome 5q31-33, indicating rearrangements of PDGFRB , or abnormalities on chromosome band 8p11-12, indicating a rearrangement of the FGFR1 gene. From a clinical point of view, it is of great importance to screen for these rearrangements in all patients using the correct techniques, as many of them, including F/P , are responsive to imatinib or other tyrosine kinase inhibitors (TKIs). However, clonal HE may also be caused by other mutations, including BCR::ABL1 , rearranged JAK2 (e.g., TK fusion partners PCM1 , BCR , and ETV6 ) or FLT3 (e.g., most common partner ETV6 ), or KIT D816V. In a substantial subset of patients with MPN-eo, no mutation is found.
|
|
|
a The table refers to the WHO classification of eosinophil neoplasm.
In paraneoplastic and reactive states, HE is usually triggered by eosinophilopoietic cytokines, including IL-5, IL-3, or GM-CSF. These cytokines, especially IL-5, may be detected in the serum of these patients and are considered to be produced by activated T cells or other activated immune cells. In a few patients with HE and HES, activated (clonal) T cells are detectable by phenotyping and PCR, but no underlying lymphoma or other underlying condition is found. In these cases, the lymphoid variant of HES (HES L ) is diagnosed. In some of these patients, an overt T-cell lymphoma or Sézary syndrome may develop in the follow-up. A number of different reactive conditions and disorders, including helminth infections, fungal infections, autoimmune disorders, allergies, or drug reactions, can lead to reactive HE and reactive HES (HES R ). These conditions are listed in Table 74.2 .
Regardless of the underlying etiology (neoplastic or reactive), sustained HE may lead to typical end-organ damage defined as HES. Even in patients with idiopathic HE, such organ damage may occur and is then called idiopathic HES (HES I ). However, not all patients presenting with HE develop organ damage even if followed over a longer time period. For example, patients with an underlying allergic disease usually fail to develop the organ damage seen in patients with typical HES. When neither an underlying disease nor HE-related organ damage is found, the diagnosis HE of undetermined significance (HE US ) is established. In general, the risk of HES is lower in reactive forms of HE, especially when the underlying disease can be treated successfully. Correspondingly, IL-5 transgenic mice with massive HE do not develop significant end-organ damage, suggesting that other factors (in addition to IL-5) are likely necessary for the occurrence of an HE-related organ damage. In patients with an underlying MPN or other underlying myeloid neoplasm, the risk of HE-related organ damage (HES) is relatively high. A particularly high risk is found in patients with mutations in PDGFR genes. Likewise, most untreated patients with F/P + MPN-eo or CEL may develop HES with cardiac involvement over time. Additional risk factors may also contribute to the development of HES in these patients, including, among others, an additional prothrombophilic state, late detection of the disease, and late initiation of specific (anti-CEL) therapy. There may also be a certain genetic predisposition for early occurrence of tissue HE in patients with F/P + CEL ( F/P + MPN-eo). In particular, the severity (extent) of tissue HE seems to correlate with a polymorphic variant (SNP in the 5′-UTR) in the IL-5RA gene. However, it remains unclear whether this SNP predisposes to HES development or progression, and thus is relevant clinically. Today, the general recommendation is to initiate therapy with imatinib early in all F/P + patients, regardless of genetic, molecular or other factors, in order to prevent any HES occurrence and to avoid disease progression (to acute leukemia).
A number of different organ systems are affected in patients with HES. The most common manifestations are summarized in Table 74.7 . Although multiple organ systems are involved and the manifestation patterns are quite complex with varying courses and outcomes, some common pathogenetic factors have been described. One common feature is the mobilization of the tissue microenvironment by eosinophil-derived mediators and cytokines, which leads to tissue remodeling, fibrosis, and increased angiogenesis. Another common factor is tissue inflammation, which is typically triggered by eosinophil-derived cytokines and chemokines. Finally, HE-related organopathies may often result from direct cytotoxic damage that is induced by the various toxic mediators produced and released by (reactive and neoplastic) eosinophils. With regard to thromboembolic events, several different eosinophil-derived mediators have been implicated, including TNF-α, plasminogen activator inhibitors (PAIs), and eosinophil-derived DNA traps (see Table 74.1 ). In addition, ECPs have the capacity to alter thrombomodulin activity. A number of different compounds produced by eosinophils, including cationic proteins and various cytokines, may induce fibrosis. Among these cytokines, OSM may play a particular role as a HES-related profibrogenic cytokine. HE-related neoangiogenesis may be triggered by IL-8, OSM, and vascular endothelial growth factor (VEGF). All these cytokines are produced and secreted by normal/reactive and also by neoplastic eosinophils. However, neoplastic eosinophils produce some of these cytokines in excess over their normal counterparts. For example, F/P + eosinophils express and release huge amounts of OSM, IL-8, and VEGF. The F/P mutant induces the production and secretion of these mediators in neoplastic cells in a STAT5-dependent manner. Concerning the mobilization of the microenvironment in various organs in patients with HES, a most critical F/P target gene may be OSM . Notably, OSM initiates not only fibrosis and angiogenesis by triggering fibroblast proliferation and tube formation in endothelial cells, but also by inducing the production of multiple cytokines and chemokines in stromal cells. Among these mediators are IL-8, SDF-1, and other chemotactic factors that can recruit additional CXCR4+ precursor cells and eosinophils to local tissue sites.
| Cardiac |
|
|
|
|
|
|
| Neurologic |
|
|
|
|
|
|
| Dermatologic |
|
|
|
|
|
|
| Pulmonary |
|
|
|
|
|
| Ocular |
|
|
|
| Connective Tissue |
|
|
|
|
|
| Gastrointestinal |
|
|
|
|
|
An unresolved question is why HE-related fibrosis and thrombosis affect certain tissues and organs, including the heart and lungs. One explanation may be that eosinophils invade heart and lung tissues via “physiologic” routes, which may not be the case in other MPNs where HE usually does not occur. An additional explanation may be that eosinophil-derived mediators are toxic to certain (tissue-specific) cell types such as cardiomyocytes. Indeed, as noted earlier, eosinophils express a number of different cationic proteins capable of inducing endothelial and endocardial damage and neurotoxicity. In addition, the eosinophil has the capacity to generate reactive oxidative species that can augment tissue damage.
The term HES was initially proposed for a syndrome characterized by idiopathic HE and HE-related organ damage, as well as by exclusion of various conditions and diseases that are typically accompanied by HE. More recently, the definition of HES has been refined as any type of HE (not just idiopathic) associated with typical HE-related end-organ damage. According to the proposal of an international working group on eosinophil disorders (ICOG-EO), the term HES should be reserved for clinical syndromes fulfilling HES criteria, but not for underlying (hematologic) malignancies or immunological disorders presenting with HE. In other words, the diagnosis HES must prompt the physician to search for (detect) an underlying (HES-triggering) disease. Accordingly, the final diagnosis is either idiopathic HES or HES based on a recognized underlying disorder, and both need to be documented in the final diagnosis. In previous WHO classifications, the term HES was sometimes used as a synonym of CEL, and sometimes to discriminate CEL from other less well-defined myeloid neoplasms with HE. However, in the 2008 edition of the WHO monograph, the term HES was no longer recommended as a synonym of WHO-defined neoplasms, a distinction that is in agreement with the proposal of the ICON group and the proposal of the ICOG-EO group.
HE is currently defined as a persistent, microscopically confirmed increase in PB eosinophils to ≥ 1.5 × 10 9 /L. In previous definitions, persistent eosinophilia over 6 months was required to establish the diagnosis HE. However, based on improved diagnostics and treatment, and the necessity to introduce such treatment rapidly to avoid organ damage, the proposed definition of HE has been revised to a 4-week observation interval by the ICOG-EO group. The term “tissue HE” has also been proposed by several experts, and it may be useful to apply it in certain conditions, especially in the context of (to demonstrate) HE-related organ damage (HES). However, isolated tissue HE (without blood HE) is rare, and the documentation of tissue HE often requires special immunostains directed against eosinophil granule proteins. Therefore, the presence of tissue HE is often overlooked or is not documented, and PB HE clearly remains the key diagnostic marker in daily practice. In some of these patients, criteria for blood HE are not met, but molecular and clinical signs are strongly indicative of a particular eosinophil disorder with or without accompanying HES. These patients should be followed closely, as they may progress to an overt eosinophil disease (neoplasm) over time. The same holds true for WHO-based neoplasms presenting with PDGFR or FGFR1 fusion genes without overt HE. In fact, many patients with FGFR1 -rearranged neoplasms do not have eosinophilia at presentation.
When arriving at the provisional pre-diagnostic “checkpoint” HE, two critical questions have to be addressed in order to approach to a final diagnosis: (1) is there any underlying disease or condition triggering HE? and (2) are there clinical signs and symptoms or laboratory abnormalities that point to the presence of HE-induced organ damage, so that the diagnosis of HES can be established? For example, the hematologic work-up in a patient with HE revealed CEL, and staging investigations showed endocardial thrombus formation: the final diagnosis in this patient is CEL with concomitant primary HES (HES N ). A diagnostic algorithm is shown in Fig. 74.3 . In patients with typical HES and typical clinical manifestation, histopathologic evaluation is generally not warranted to confirm the presence of tissue HE. However, in patients with rare or atypical manifestations, such as renal failure, isolated myocarditis or bloody diarrhea, a tissue biopsy is required to document the presence of tissue HE and to confirm HE-induced organ damage, so that the diagnosis HES can be established. The demonstration of extensive extracellular deposition of eosinophil-derived proteins supports the conclusion the organ damage is “HE-related.”
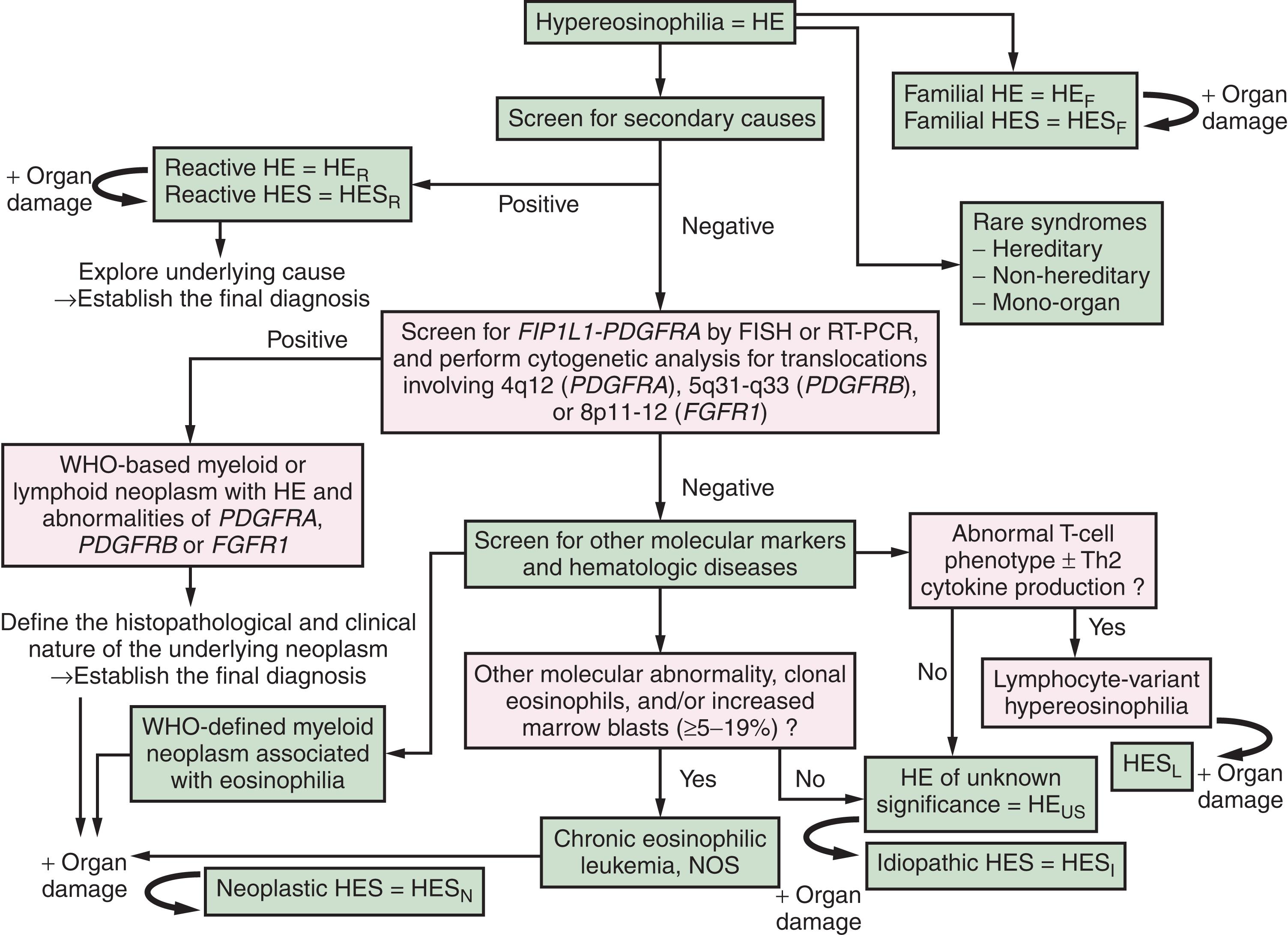
The classification of HE and HES is essentially based on the related disease and underlying etiology. Accordingly, HE and HES are both divided into a familial variant (HE F , HES F ), a primary/neoplastic form (HE N , HES N ), and reactive entities (HE R , HES R ; see Table 74.3 ). In a smaller fraction of patients, the etiology of HE and HES remains uncertain. In other rare cases, two different causative factors are detected. When the etiology of HE remains uncertain after a thorough investigation and appropriate testing and no organ damage is detected, the final diagnosis is HE of uncertain (undetermined) significance (HE US ); and if HES is diagnosed, but no underlying disease is detected, the final diagnosis is idiopathic HES (HES I ).
In general, four major groups of underlying disorders (conditions) have to be considered in HE (HES) patients: (1) myeloid (and rarely myeloid/lymphoid) neoplasms including eosinophilic leukemia (HE N ; HES N ), (2) lymphoid or nonhematopoietic neoplasms (paraneoplastic HE, a special variant of HE R ), (3) common allergic, reactive, or immunologic conditions (HE R and HES R ), and (4) clinically defined, rare syndromes and conditions accompanied by HE, including rare inherited disorders and organ-restricted inflammatory diseases with tissue HE. Overall, the classification of HE/HES remains an important step in the diagnostic algorithm (see Fig. 74.3 ). Using generally accepted criteria and parameters, this approach should lead to a final diagnosis concerning the presence or absence of HES and the presence or absence of an underlying disease.
Depending on the type of disease and other factors, a number of different organ systems may be involved in patients with HES. The most commonly involved organs are the skin, lungs, GI tract, heart, and the CNS (see Table 74.7 ). In some patients, the relationship between HE and the typical clinical signs and symptoms are pathognomonic, whereas in other cases a tissue biopsy may be required to confirm HE-related organopathy.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here