Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The human enteroviruses and parechoviruses are members of the Picornaviridae family that are commonly transmitted by enteric and respiratory routes. They cause a wide spectrum of illnesses in people of all ages.
The enteroviruses are divided among 4 species within the genus Enterovirus (i.e., human enterovirus A , B , C , and D ) based on RNA sequence relatedness. Enterovirus species C polioviruses and 3 additional genus Enterovirus species (i.e., human rhinovirus A , B , and C ) are discussed in separate chapters. Before the availability of viral genome sequencing, the human enteroviruses were distinguished from the rhinoviruses by their relative acid stability and were subdivided into several subgroups based on differences in host range and into individually numbered serotypes within each subgroup on the basis of differential neutralization by immune sera ( Table 236.1 ). The 3 poliovirus serotypes are the prototypic enteroviruses that can be transmitted to nonhuman primates and replicate in primate cell culture. The coxsackieviruses originally were distinguished from polioviruses by their ability to cause paralysis and death in suckling mice. They are divided into groups A and B according to the type of disease produced in these animals ( Table 236.1 ). Group B coxsackieviruses are isolated readily in primate cell culture, whereas most group A coxsackieviruses grow poorly in cell culture. Echoviruses (enteric cytopathic human orphan viruses) are not pathogenic for mice and monkeys but are recovered readily in primate cell culture. All enterovirus serotypes identified since 1970 are designated simply as enterovirus and are numbered sequentially. More than 100 enterovirus serotypes (including polioviruses, group A coxsackieviruses, group B coxsackieviruses, echoviruses, and numbered enteroviruses) are now distributed among the 4 human enterovirus species A through D ( Table 236.2 ).
a Rare strains (e.g., Lansing strain of poliovirus 2) have been adapted to mice.
b Coxsackievirus A23 has been reclassified as echovirus 9, which leaves 23 coxsackieviruses in group A. Antigenic interrelationships exist between coxsackievirus types A3 and A8, A11 and A15, and A13 and A18.
c Coxsackievirus A7 is pathogenic for the central nervous system of primates.
d Most coxsackievirus serotypes of group A are not readily isolated in cell cultures, but exceptions exist (e.g., types A9 and A16), and additional serotypes have been adapted to cell cultures.
e Echovirus 10 has been reclassified as reovirus 1 and echovirus 28 as rhinovirus 1A. Echovirus 34 is a variant of coxsackievirus A24; a total of 31 serotypes of echovirus therefore remain from the original 34. Antigenic interrelationships exist between echovirus types 1 and 8, 12 and 29, and 6 and 30. Echoviruses 22 and 23 are now reclassified in the genus Parechovirus .
g Hepatitis A virus was designated as enterovirus 72 but now is classified as a separate Picornavirus genus.
| Enterovirus Species | Enterovirus Serotypes |
|---|---|
| A | Coxsackievirus A serotypes 2–8, 10, 12, 14, 16 |
| Enterovirus serotypes 71, 76, 89–91, 114, 119, 120, 121 | |
| B | Coxsackievirus A serotype 9 |
| Coxsackievirus B serotypes 1–6 | |
| Echovirus serotypes 1–7, 9, 11–21, 24–27, 29–33 | |
| Enterovirus serotypes 69, 73–75, 77–88, 93, 97, 98, 100, 101, 106, 107, 111 | |
| C | Coxsackievirus A serotypes 1, 11, 13, 17, 19–22, 24 |
| Poliovirus serotypes 1–3 | |
| Enterovirus serotypes 95, 96, 99, 102, 104, 105, 109, 113, 116–118 | |
| D | Enterovirus serotypes 68, 70, 94, 111 |
Parechoviruses share many of the same biologic, clinical, and epidemiologic characteristics as enteroviruses, but they differ sufficiently in genomic sequence to be classified in a separate genus. The prototypic parechovirus serotypes 1 and 2 originally were designated echovirus 22 and 23, respectively; 18 human parechovirus serotypes are recognized currently.
The pathogenesis of enterovirus infections has been investigated most extensively with poliovirus infection in monkeys and humans. Although comparable data are not available for the nonpolio enteroviruses, it is assumed that the pathogenic events are similar, although tissue and organ tropism are highly variable. Enterovirus D68 differs from other human enteroviruses in that it is primarily an agent of respiratory disease, sharing biologic and molecular properties with human rhinoviruses, and therefore is discussed separately at the end of this section.
Human enteroviruses are acquired directly or indirectly by the ingestion of virus that is shed in the feces or via acquisition from the upper respiratory tract of infected contacts. Initial viral replication occurs in the upper respiratory tract and distal small bowel. Infectious virus is detected in ileal lymphoid tissue 1–3 days after the ingestion of virus, and fecal shedding can be detected for ≥6 weeks. Generally, enteroviruses are recovered from the gastrointestinal tract in greater density and for longer periods than from the upper respiratory tract, with the exceptions of enterovirus D68 and enterovirus D70.
Viral replication in submucosal lymphoid tissue gives rise to a transient “minor” viremia that distributes virus to reticuloendothelial tissue in distant lymph nodes, liver, spleen, and bone marrow. Early viral replication does not cause histopathologic changes in the gastrointestinal tract or lymphoreticular tissue. Further replication in these organs leads to a sustained “major” viremia and dissemination of virus to target organs such as the central nervous system (CNS), heart, and skin.
Many infected people control infection before major viremia and experience only transient symptoms or have asymptomatic infection. The onset of symptomatic disease, including fever and other systemic manifestations, coincides with the major viremia and organ-specific disease (i.e., poliomyelitis, myocarditis) is determined by tropism of the infecting serotype. Disease results from virus-induced cell necrosis and the accompanying inflammatory response. For example, animal models of myocarditis demonstrate initial viral replication in myocytes that results in scattered necrosis and focal infiltration with inflammatory cells that persists for weeks to months after disappearance of virus. The late phase of the inflammatory response is postulated to be caused by virus-induced, cytotoxic T-lymphocyte destruction of myocytes, development of a myocardial neoantigen, or cross-reactivity between viral and myocardial cell antigens. , Healing is accompanied by interstitial fibrosis and loss of myocytes. Both experimental and clinical data suggest that certain host factors enhance the severity of enterovirus infection, including exercise during the incubation period, cold stress, malnutrition, and humoral immunodeficiency.
Enterovirus D68 is isolated most frequently from respiratory specimens rather than fecal samples. Enterovirus D68 grows most efficiently at 33°C (rather than at 37°C, as do other enteroviruses) and can be acid sensitive, features that can limit survival in the gastrointestinal tract. Primarily spread through respiratory transmission, enterovirus D68 attaches to sialic acid receptors on epithelial cells of respiratory mucosal surfaces. Enterovirus D68 has been detected in serum from children with pneumonia early in the course of illness, as well as in the CNS in case reports.
Protective immunity to enterovirus infection is serotype specific. Infection leads to production of neutralizing humoral antibodies that protect against recurrent disease but not against asymptomatic reinfection. Passive antibody provided by immune globulin or by transplacental maternal antibody effectively prevents enterovirus disease. , Secretory immunoglobulin A (IgA) antibodies appear in nasal and duodenal secretions 2–4 weeks after primary infection and persist for at least 15 years. Secretion of IgA antibodies at each mucosal site depends on local virus replication. On enterovirus re-exposure, secretory IgA antibodies prevent or substantially reduce subsequent replication. Secretory IgA antibodies also are found in colostrum and milk of immune women. Antibodies in human milk can interfere with the replication of attenuated polioviruses in breastfed infants vaccinated during the first month of life.
Patients with isolated B-lymphocyte immunodeficiency syndromes develop persistent enterovirus infections, a finding suggesting that humoral antibodies are necessary for viral clearance. Administration of intramuscular immune serum globulin does not affect the course of established acute poliovirus poliomyelitis, but few controlled trials of immune globulin intravenous (IGIV) in other CNS and non-CNS enterovirus infections have been performed. Studies in a well-characterized murine model showed that inhibition of T-lymphocyte function has little effect on the course of enterovirus infection, although T lymphocytes participate in immunopathologic events contributing to the cytotoxicity that accompanies infection. These data may explain the observation that patients with T-lymphocyte immunodeficiency or immunosuppression appear to be at lesser risk for severe or prolonged enterovirus infection. In contrast, experimental studies suggested that macrophage function is critical to clearance of virus and recovery from infection. Other host factors, including human leukocyte antigen (HLA) genes, immune response gene polymorphisms, and vitamin A deficiency are linked to susceptibility and severity of enterovirus A71 disease.
Infections occur in all human populations, although rates of infection vary markedly by geography, season, age, and other host factors. Transmission occurs throughout the year in most locations but at much higher rates in temperate climates in summer and fall. Seasonal periodicity is more pronounced (i.e., higher density of infection during short seasons) with increasing latitudes in both hemispheres compared with tropical latitudes. Conversely, seasonal fluctuations diminish in tropical latitudes.
Although enterovirus infections and disease occur in all age groups, infants and young children have the highest rates. Infants aged <1 year are infected at severalfold higher rates than children and adults; and, within the first year of life, enterovirus infections are recognized most often clinically in infants aged <3 months. The risk of disease in boys exceeds that in girls by as much as 50%. Infants of low socioeconomic status have a higher risk of infection that is attributable to crowding and poor hygiene.
The contribution of different enterovirus serotypes to human infection and disease varies markedly. Thirty percent to 80% of adults have antibody to the more common enterovirus serotypes (e.g., group B coxsackieviruses 1–5 and some echoviruses). A few serotypes cause endemic disease with annual regional variation in the US. Other serotypes (e.g., echoviruses 9, 11, and 30) are responsible for widespread outbreaks in which the epidemic strain accounts for >30% of all isolated enteroviruses, followed by several years of relative quiescence. The reasons for the appearance and disappearance of particular serotypes are unknown, although accumulation of a critical mass of susceptible young children may be necessary to sustain epidemic transmission. Periodic reappearance of the same serotype is marked by strain variation as measured by oligonucleotide fingerprinting or genomic sequencing. , Between 1970 and 2008, species B enteroviruses predominated among enterovirus isolates submitted from state and local public health laboratories to the National Enterovirus Surveillance System of the Centers for Disease Control and Prevention (CDC). , From 2014 to 2016, 5 serotypes represented 75% of all enteroviruses submitted to CDC, with enterovirus D68 accounting for 55.9% of all reports ( Table 236.3 ). By contrast, some enterovirus serotypes (e.g., echovirus 1, coxsackievirus B6, and enterovirus B69) rarely are recognized clinically and appear to have little epidemic potential.
| Enterovirus Serotype | Percentage |
|---|---|
| Enterovirus D68 | 55.9 |
| Echovirus 30 | 5.7 |
| Coxsackievirus A6 | 5.5 |
| Echovirus 18 | 4.2 |
| Coxsackievirus B3 | 3.9 |
Enterovirus A71 has caused large outbreaks of hand-foot-and-mouth (HFM) disease, aseptic meningitis, and serious CNS disease in young children. Although sporadic cases and small outbreaks of enterovirus A71 infections occur globally, since the mid-1990s several genetic lineages, most recently genotype C4, have caused widespread disease in infants and young children throughout Asia.
Enterovirus D68 was uncommonly reported to CDC from 1970 to 2008. From 2008 to 2010, small regional outbreaks of enterovirus D68 were reported in Asia, Europe, and the US. In 2014, the largest and most widespread outbreak of enterovirus D68 respiratory disease occurred in the US, with more than 1100 confirmed infections. Since that time, enterovirus D68 has circulated in a biennial pattern in the US with smaller summer-fall outbreaks in 2016 and 2018 and has caused outbreaks in many locales globally. ,
Most enteroviruses are shed in the upper respiratory tract for 1–3 weeks and in the feces for 2–8 weeks after primary infection; these findings suggest both respiratory and fecal-oral routes of transmission. As noted above, enterovirus D68 is primarily spread via respiratory transmission. Studies of naturally occurring and attenuated polioviruses demonstrate that fecal (but not oropharyngeal) shedding also occurs during reinfection, albeit at lower titer and for a shorter duration. Both direct fecal-oral and indirect mechanisms of person-to-person transmission are postulated. Secondary infection rates exceed 50% in household contacts. Infants, particularly those in diapers, appear to be the most efficient transmitters of infection. Indirect transmission is abetted by poor sanitary conditions and can occur by numerous routes, including contaminated water, food, and fomites. The relative roles of virus shedding by symptomatic and asymptomatic people are not known, but both likely are important. Notable exceptions to the importance of respiratory and fecal-oral transmission are enterovirus D70 and coxsackievirus A24, the agents of acute hemorrhagic conjunctivitis, which are shed in tears and spread by fingers and fomites.
The incubation period for enteroviral infection is difficult to determine precisely and may vary according to the clinical syndrome. Brief febrile illnesses occur after an average incubation period of 3–5 days. Detailed studies of poliovirus infection have indicated that CNS manifestations (i.e., aseptic meningitis, poliomyelitis) usually are not observed until 9–12 days after exposure. The incubation period of acute hemorrhagic conjunctivitis is 1–3 days.
Most enterovirus infections in children are asymptomatic. The proportion of infections that result in illness varies widely and is affected by numerous host factors. Table 236.4 shows clinical syndromes commonly associated with nonpolio enterovirus infections in children. Some enteroviral diseases (e.g., nonspecific febrile illness, pharyngitis, and aseptic meningitis) are associated with multiple serotypes of group B coxsackievirus and echovirus, whereas others are typically associated with specific subgroups (e.g., myopericarditis with group B coxsackieviruses and herpangina with group A coxsackieviruses) or a limited number of serotypes (e.g., acute hemorrhagic conjunctivitis with coxsackievirus A24 or enterovirus D70). ,
| Group A Coxsackieviruses | Group B Coxsackieviruses | Echoviruses | Newer Enteroviruses | |
|---|---|---|---|---|
| Exanthems and Enanthems | ||||
| Herpangina | +++ | + | + | − |
| Hand-foot-and-mouth disease | +++ | + | − | +++ |
| Nonspecific rash | ++ | + | +++ | ++ |
| Central Nervous System Infections | ||||
| Aseptic meningitis | + | +++ | +++ | ++ |
| Encephalitis | +++ | ++ | ++ | ++ |
| Acute flaccid myelitis | ++ | + | + | +++ |
| Respiratory Infections | ||||
| Pneumonia/Bronchospasm | − | − | − | +++ |
| Diseases of Muscle | ||||
| Pleurodynia | + | +++ | + | − |
| Myositis | ++ | ++ | ++ | − |
| Myopericarditis | + | +++ | + | − |
| Ophthalmic Infections | ||||
| Acute hemorrhagic conjunctivitis | +++ | − | − | +++ |
| Systemic Infections | ||||
| Neonates | + | +++ | +++ | − |
| B-lymphocyte immunodeficiency | + | + | +++ | ++ |
Nonpolio enteroviruses are the most commonly identified causes of fever without an apparent focus in infants aged <3 months. , During the summer and fall, enteroviruses account for at least one-half of cases. , , Fever often occurs alone or in association with irritability, lethargy, poor feeding, vomiting, diarrhea, exanthems, or signs of upper respiratory tract infection. Approximately one-half of infants infected with an enterovirus have aseptic meningitis, often without meningeal signs. , Most infants with fever or aseptic meningitis recover within 2–10 days without complications.
Enteroviruses are common causes of a broad array of acute cutaneous eruptions and mucous membrane lesions ( Fig. 236.1 ). Exanthems can be maculopapular and resemble those of measles, rubella, or roseola, or they can be vesicular, petechial, purpuric, or urticarial. Exanthems are reported more commonly with echovirus infections than with infections with other enteroviruses.
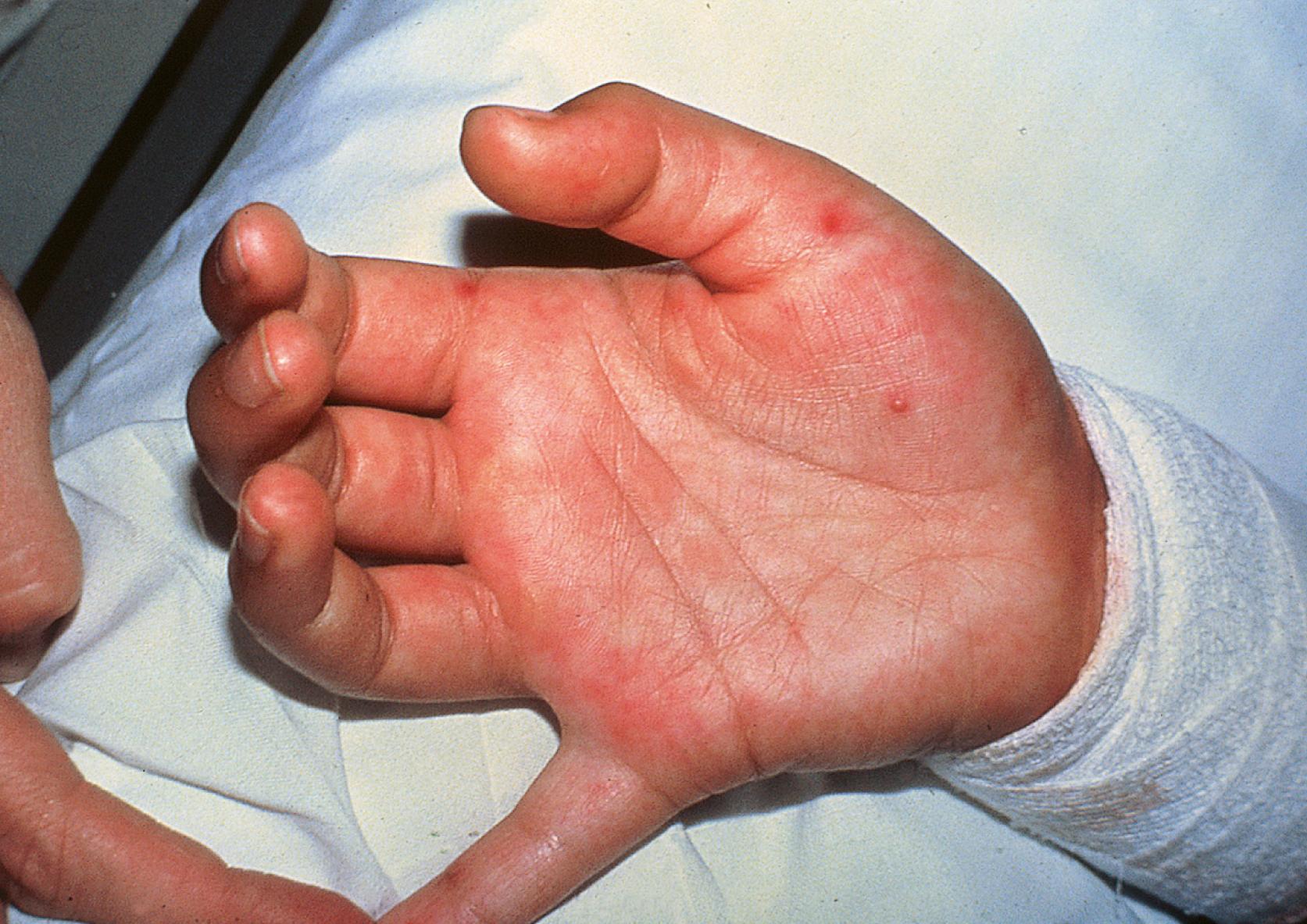
Herpangina is a distinctive enanthem characterized by painful vesicular lesions on the soft palate, uvula, tonsils, and posterior oropharynx, often accompanied by fever, headache, and sore throat. Mucosal lesions begin as punctate macules and evolve over a 24-hour period to 2–4 mm vesicular lesions that ultimately ulcerate centrally. Fever subsides in 2–4 days, but lesions can persist for a week. Coxsackie A viruses are the most common etiologic agents.
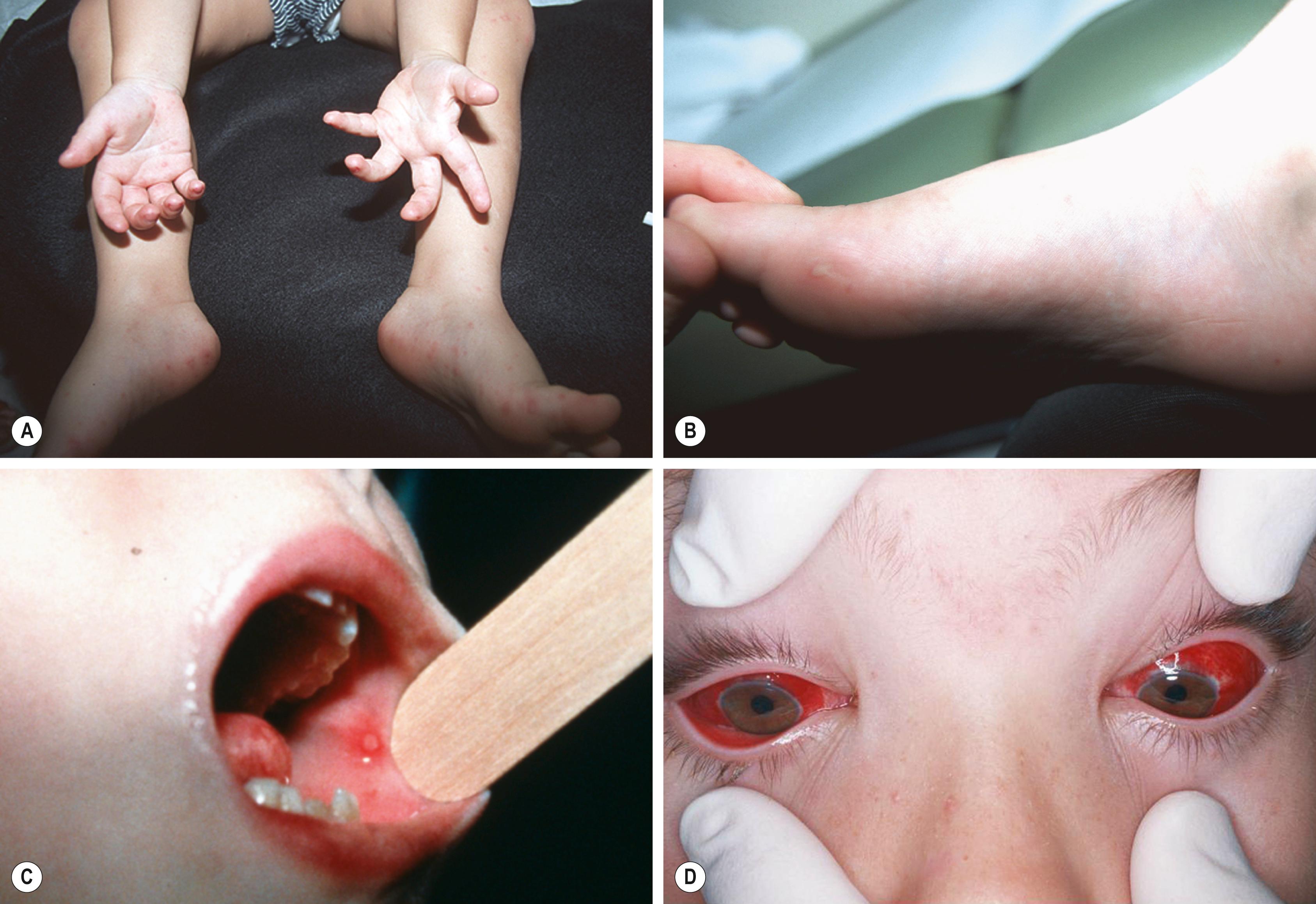
Coxsackievirus A6 and coxsackievirus A16 (and less commonly other group A and group B coxsackieviruses) and enterovirus A71 cause HFM disease, a syndrome characterized by vesicular stomatitis and cutaneous lesions of the distal extremities ( Fig. 236.2 ). Children aged <10 years are most affected. Fever, sore throat, and refusal to eat are common complaints. Oral lesions occur chiefly on the buccal mucosa and tongue and can coalesce and ulcerate by the time of the initial examination. Cutaneous lesions occur in approximately 75% of cases and are found mainly on the hands, feet, wrists, ankles, and buttocks or genitalia. HFM disease associated with coxsackievirus A6 can be extensive and additionally can involve perioral, perianal, and dorsal hand and feet lesions. The lesions of HFM disease typically are tender papules or vesicles with a surrounding zone of erythema; vesiculobullous, purpuric, and Gianotti-Crosti–like lesions and lesions accentuated in areas of eczematous dermatitis have been associated with coxsackievirus A6. HFM disease generally is self-limited, with an average 7-day duration. When caused by enterovirus A71, however, HFM disease can herald the onset of more serious CNS disease, including acute flaccid myelitis and brainstem encephalitis. , ,
Sore throat and coryza are frequent upper respiratory tract symptoms associated with the early phase of many enterovirus infections. Acute parotitis occasionally is reported in association with herpangina caused by group A coxsackieviruses and also with group B coxsackieviruses and enterovirus D70 infections. Exudative pharyngitis, otitis media, croup, bronchiolitis, and pneumonia are other less common respiratory findings associated with some enteroviruses.
Enterovirus D68 is associated primarily with respiratory disease. Although most enterovirus D68 infections are associated with mild respiratory symptoms, severe lower respiratory disease leading to hospitalization, intubation, and death is reported. Among children hospitalized with enterovirus D68 respiratory infection, one-half had a history of asthma. Most of these children presented with dyspnea, cough, wheezing, tachypnea, and hypoxia, and approximately 50% were febrile.
Most cases of community-associated aseptic meningitis are caused by viruses, 90% of which are group B coxsackieviruses and echoviruses. Although many serotypes cause meningitis, coxsackievirus serotypes B2 and B5 and echovirus serotypes 4, 6, 9, 11, 16, and 30 are most reported. Not all enteroviruses that infect young infants are equally likely to cause meningitis. In 1 study, febrile infants infected with certain serotypes (e.g., the group B coxsackieviruses and echovirus serotypes 11 and 30) were more likely to have meningitis; group A coxsackieviruses caused <5% of cases. Enterovirus D68 has been detected in cerebrospinal fluid (CSF) in case reports of aseptic meningitis.
Infants aged <3 months appear to have the highest rate of aseptic meningitis, perhaps in part because lumbar puncture commonly is performed for evaluation of fever in this age group. A small proportion of infants have clinical manifestations suggestive of neurologic disease. , , The severity of meningeal symptoms and other signs of neurologic involvement in older children and adults with aseptic meningitis varies widely. Onset can be gradual or abrupt. Typically, the patient has a brief prodrome of fever, chills, and headache. Meningismus, when present, varies from mild to severe. Kernig and Brudzinski signs are present in only one-third of older children and adults.
The CSF white blood cell (WBC) count can be normal in infants with an enterovirus detected in CSF: most commonly, CSF in enterovirus meningitis demonstrates mild pleocytosis (typically <500 WBCs/μL but occasionally >1000 WBCs/μL). Pleocytosis frequently is predominantly polymorphonuclear in the first 48 hours, then transitioning to mononuclear prominence. CSF glucose typically is normal (<40 mg/dL), and CSF protein is normal to mildly elevated (<100 mg/dL).
Most cases of enteroviral meningitis are uncomplicated. Fever and signs of meningeal irritation resolve in 2–7 days, but CSF pleocytosis can persist. A biphasic pattern of illness can occur, with fever in the initial phase followed by return of fever and meningeal signs after several days of an afebrile interlude. Adults, compared with children, appear to have more severe and prolonged symptoms. Approximately 10% of infants hospitalized with aseptic meningitis experience acute CNS complications such as seizures, obtundation, or increased intracranial pressure. , Although some early reports suggested that infants with viral meningitis at an early age had a moderate risk of long-term neurologic and cognitive sequelae, prospective studies have indicated no measurable effect, , including in infants with neurologic findings during their acute illness.
Enteroviruses are among of the most commonly identified causes of infectious encephalitis; these viruses account for 10%–20% of cases of known origin. Many nonpolio enterovirus serotypes have been identified in patients with encephalitis, including group A coxsackieviruses in infants and children with focal enteroviral encephalitis.
Clinical manifestations of enterovirus encephalitis range from mild to coma, decerebration, and death. Patients with focal encephalitis can have partial motor seizures, hemichorea, or acute cerebellar ataxia. Initial findings in some cases suggest a diagnosis of herpes simplex virus encephalitis. , Because CSF findings in enteroviral encephalitis are similar to those of aseptic meningitis, the term meningoencephalitis commonly is used interchangeably with encephalitis. Magnetic resonance brain imaging and electroencephalography usually reflect the extent and severity of disease. Encephalitis occurring as a manifestation of generalized neonatal infection often is fatal or results in permanent neurologic sequelae (see section Infection in Special Hosts). The prognosis for older infants and children is better; although static neurologic sequelae and rare deaths occur, most patients recover fully.
Nonpolio enteroviruses uncommonly cause a syndrome of acute flaccid motor weakness associated with spinal cord gray matter lesions; this syndrome, recently termed acute flaccid myelitis (AFM), is clinically indistinguishable from poliomyelitis. AFM can be caused by numerous enteroviruses, including, most commonly, enterovirus A71; enterovirus D68; enterovirus D70; group A coxsackievirus types 7, 9, and 24; group B coxsackievirus types 1 to 5; and echovirus types 6 and 9. Outbreaks of acute paralysis caused by enterovirus A71 have involved thousands of people, mostly children, in Eastern Europe and Asia, as well as smaller numbers of patients in Australia and parts of the US. , Coxsackievirus A7 has caused small outbreaks of AFM in Scotland and Russia, and AFM has been described in enterovirus D70 epidemics.
Cases of AFM and cranial nerve dysfunction in children temporally associated with an outbreak of enterovirus D68 respiratory disease were noted in the US and, to a lesser extent, in Europe and Canada in 2014. Subsequent seasonal, biennial outbreaks in the summer-fall of 2016 and 2018 coincided with periods of enterovirus D68 circulation in the US, and AFM outbreaks associated with enterovirus D68 were noted in Japan in 2015, Europe in 2016, and the UK in 2018. The term AFM was used to describe the acute onset of flaccid limb weakness associated with neuroimaging lesions in the grey matter of the spinal cord. Most children had abrupt onset of neurologic deficits approximately 1 week following an acute febrile respiratory illness. Preceding or concurrent headache, neck stiffness, and pain in the affected limb, neck, or back were common around the time of neurologic onset. Cranial nerve dysfunction was seen in a third of cases and was associated with focal lesions in cranial nerve motor nuclei in the brainstem. Limb weakness commonly was asymmetric and associated with hyporeflexia and intact sensation of the affected limb, similar to findings in poliomyelitis. Mild improvements in functional deficits were reported, but residual weakness persisted in most cases. Enterovirus D68 was identified in around half of children with AFM who had respiratory specimens collected within 1 week, but virus was uncommonly identified in CSF. Identification of high levels of enterovirus antibodies in CSF by panviral serologic assays provided further evidence of enterovirus etiology in AFM cases in 2018. ,
A distinctive, life-threatening form of brainstem encephalitis has been observed during epidemics of enterovirus A71 infection in Malaysia, Australia, Taiwan, China, and other locations. , , , Most cases occur in children aged <5 years and are associated with HFM disease or herpangina in around half of cases. Affected children have myoclonus, vomiting, ataxia, nystagmus, oculomotor palsies, and autonomic instability. The most severely affected patients die rapidly of noncardiogenic pulmonary edema, pulmonary hemorrhage, interstitial pneumonitis, or cardiac dysfunction, postulated to be caused by dysregulation of sympathetic outflow and overload of the pulmonary vascular bed. , High rates of neurologic sequelae occur among survivors of severe disease. Neuronal degeneration and mononuclear cell infiltration in the grey matter of the pons and medulla are found at postmortem examination; enterovirus A71 has been isolated from brainstem tissue, but uncommonly from CSF, and is most readily detected from stool samples. , A small number of similar cases have occurred sporadically in the US. , ,
Guillain-Barré syndrome is reported in a limited number of patients infected with group A coxsackievirus types 2, 5, and 9; echovirus types 6 and 22; and enterovirus D68. Acute transverse myelitis has been attributed to coxsackievirus A9, coxsackievirus B4, and echovirus 5.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here