Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Transport of water and salt across the gastrointestinal (GI) mucosa is essential for the survival of all mammals. In the proximal GI tract, secretion of water and electrolytes into the lumen provides an appropriate environment for the actions of digestive enzymes, solvent for water soluble nutrients allowing them to be absorbed, counter ions for nutrient transport and assists in neutralizing gastric acid. In the distal small intestine and colon, local secretion can flush toxins and bacteria from vulnerable mucosal epithelium thereby forming an active part of the mucosal barrier to external pathogens, while absorption from the lumen recovers water to maintain whole body water and electrolyte balance. The importance of the latter is clearly illustrated by the high mortality due to dehydration that occurs with diarrheal diseases such as cholera and Clostridium difficile colitis, which upset the balance of secretion and absorption.
The control of water and electrolyte transport is highly complex ( Fig. 19.1 ). It involves the nervous system; endocrine, paracrine, and autocrine systems within the mucosa; and the luminal environment including nutrients and the intestinal microbiota and their metabolic products. Each system interacts with the others so there are multiple modes of response to any given challenge with the potential for substantial feedback. In this chapter, we focus on control systems within the enteric nervous system, but these cannot be fully understood without keeping how they interact with other control systems firmly in mind.
Mucosal secretion and absorption are controlled by several interacting neural circuits. There are secretomotor neurons in the submucosal plexus of the enteric nervous system that are directly controlled by other submucosal neurons and circuitry within the myenteric plexus. There are long neural reflex pathways that pass through the prevertebral sympathetic ganglia to innervate and modulate the enteric neural circuits, while control from the central nervous system comes via parasympathetic and sympathetic pathways. Further, there is clear evidence that the enteric circuits can be directly modulated by GI primary afferents. A key point is that intestinal motility and secretion operate in concert within any region.
This chapter focuses on the small and large intestines. Our aim is to produce a general picture, but the most complete information about secretomotor pathways comes from guinea-pig ileum and colon. Accordingly, we highlight specific concepts arising from those studies and contrast these with key data from other species such as mouse, rat, and humans to identify what can be generalized across all mammals.
The primary function of the intestine is to absorb nutrients and water. The human intestine has a massive absorptive capacity, as under physiological conditions it handles about 5–10 L/day of ingested fluid, gastric juice, bile, pancreatic juice, and intestinal secretions. Most of this fluid is actually absorbed in the small intestine, but about 2 L flow through the ileo-cecal valve each day. Under optimal conditions, the colon’s absorptive capacity is more than adequate to reabsorb this residual fluid as it amounts to about 200 mL/h, about 5 L/day. Indeed, infusion into the cecum of an extra 4 l/24 h is required to generate diarrhea in healthy subjects. However, because absorption is largely a constitutive process, this only applies if there is sufficient contact time, which determines the overall balance of secretion and absorption.
It is well established that intestinal motility is important for determining the net movement of fluid and electrolytes in the colon and small intestine. The time available for absorption depends on how fast the content moves: rapid movement reduces absorption, slower movement increases water and nutrient absorption. There are two main motility programs in the small intestine: the migrating motor complex (MMC) or interdigestive motor pattern, consisting of slowly migrating segments of maximal motor activity (phase III) alternating with a substantial quiescent period; and the fed pattern, which primarily consists of mixing contractions interrupted by some propulsive patterns (see Chapter 25 for details). In the large intestine, high-resolution manometry has identified a variety of motor patterns in the fasted state most of which propagate in a retrograde direction and would retard flow in the lumen increasing time for fluid absorption. After a meal, high-amplitude propagating sequences (the equivalent of “mass movements”) appear along with an increase in retrograde contractions. The balance of these two contractile patterns will determine the duration of contact with the absorptive epithelium.
As discussed below, the enteric nervous system also directly couples motor activity and fluid secretion. Thus, the interplay between secretomotor neurons, epithelial function, and motor activity determines the net output of the system.
Both the small intestine and colon have major physiological functions other than handling nutrient absorption and transmucosal movement of fluid. For example, although the microbiotas of the GI tract have major roles in regulating host metabolism and behavior (for recent reviews see Refs. ), they are a major environmental threat to the host and must not reach the blood stream. The mucosal barrier, including mucus secreted from goblet cells in the mucosal epithelium and the tight junctions connecting adjacent columnar epithelial cells, is an important part of the host defence mechanisms. Contact between the microbiota and the immune system in the colon, where most of the microbiome resides, is reduced by a layer of firmly attached mucus adhering to the mucosal epithelium, which is sterile, and an outer, loose, layer of mucus where much of the microbiota reside. In contrast, in most of the small intestine, adherent mucus is thin or nonexistent. Both mucus secretion (for a review see Ref. ) and the properties of epithelial tight junction (for a review see Ref. ) are likely to be under neural control, potentially by the secretomotor neurons that regulate electrogenic secretion.
The transport systems of the small intestine and colon differ in some details reflecting their differing roles. There are also significant regional differences in enteric neural control of these transport systems, even between jejunum and ileum or proximal and distal colon. There are very substantial species differences in the pattern of transporter expression, so care must be taken when findings are extrapolated between segments and across species.
Net movement of water and salt depends on the balance of secretion and absorption with water movement following active ion transport, both transcellularly and via the gaps between adjacent epithelial cells, controlled by tight junctions, desmosomes, and other cell-to-cell interactions. The underlying processes are described in detail in Chapter 58, Chapter 59, Chapter 60 . Sodium and chloride are the major ions transported, but bicarbonate has a significant role in the duodenum and colon.
Available methods for measuring water transport in vivo lack spatial and temporal resolution making relationships between the enteric neural circuits that supply the mucosa and this key process difficult to identify. Standard in vitro methods, like Ussing chamber analysis of electrogenic ion transport, give greater temporal resolution at the cost of neural circuit integrity and do not detect electroneutral transport processes like sodium ion absorption. With these methods, water movement must be inferred from the electrogenic ion transport, mucosal integrity is often compromised during the course of recording, and the role of mucosal blood flow cannot be analyzed. Thus, integration of in vivo studies, which are subject to the huge number of uncontrollable variables inherent in such studies, with the higher resolution in vitro preparations that lack key physiological elements, remains a major challenge for this field.
One method that has helped bridge this gap has been to study electrogenic transport in vivo by recording the transmural potential difference (PD) generated by such transport in an intestinal segment via a luminal electrode and a reference electrode elsewhere in the body. Transmural PD provides a surrogate measure of water secretion with a high time resolution. This keeps the system relatively intact, but does not allow analysis of underlying cellular mechanisms and lacks the environmental control possible in in vitro studies. It also provides no information about electrically silent transport, for example, Na + /H + exchange, which until very recently had to be based on much lower time resolution ion flux studies. Recently developed methods for measuring bicarbonate secretion in the duodenum in vivo (for a review see Ref. ) should be adaptable to address at least some of these issues in the future.
Much is known, however, about the mechanisms of electrogenic secretion of chloride ions across enterocytes in sheets of epithelium and the counterbalancing electroneutral absorption of sodium ions across the same cells. In this section, we will identify those mechanisms relevant to neural control systems.
In considering neural control of epithelial cells, it is important to be mindful of the polarity of these cells. Neurons release their transmitters onto the basolateral membrane of the epithelial cells, so it is receptors and messenger systems in this region that are likely to be the primary sites of action. Similarly, stimuli acting on the apical (luminal) surface of the epithelium need to pass the mucosal barrier to reach the neurons under physiological conditions and hence effects on neural activity are likely to be indirect, via release of a mediator from the epithelium.
The electrochemical gradient that drives transepithelial movements of ions comes from an antiporter, an energy-dependent Na + /K + ATPase expressed in enterocyte basolateral membranes. In all species, sodium absorption occurs by electrically silent sodium/proton exchange in both small and large intestines, with NHE3 in the apical membrane being the major player (for a review see Ref. ). The apical membranes of small intestinal enterocytes also contain electrogenic Na + /solute symporters for glucose and amino acids. In the distal ileum, a Cl − /OH − exchanger is expressed along with NHE3. NHE3 is the main transporter in the proximal colon, but ENaC, an electrogenic channel, is also expressed in the distal colon.
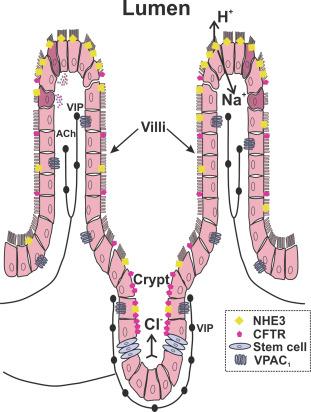
Active chloride transport via the cystic fibrosis transmembrane conductance regulator, CFTR, is the main secretory system. CFTR are located in the apical membranes of enterocytes throughout the small and large intestine. Newly differentiated enterocytes preferentially express CFTR rather than NHE3 with the balance reversing as these cells move out of the crypts, so CFTR expression is more prominent in crypt than in villus epithelium ( Fig. 19.2 ). CFTR activation inhibits ENaC and vice versa. Opening CFTR channels, usually as a result of increased intracellular cyclic AMP (cAMP), results in chloride secretion. Many mechanisms can activate adenylyl cyclase including vasoactive intestinal peptide (VIP, a major secretomotor transmitter), extracellular PGE2, or other mediators. Increased cAMP also opens basolateral K + channels including KCNQ1 (Kv7.1) with an associated regulator channel, KCNE3 and, less prominently, KNE3 and KCNN4. Opening these K + channels hyperpolarizes epithelial cells increasing the electrochemical gradient for Cl − passage through the CFTR. Raised cAMP also amplifies the cell’s maximal secretory capacity by mobilizing “resting” CFTR channels from the intracellular compartment. CFTR allow passage of both Cl − and
ions, but HCO − conductance is about 10 times lower, so Cl − secretion occurs preferentially when both are available. To overcome this, cells in the duodenum and distal colon that secrete
via CFTR express a basolateral NaHCO 3 symporter that supplies
for passage through CFTR in apical membranes. In contrast, chloride-secreting epithelia express a basolateral NKCC, sodium/potassium/chloride cotransporter.
There is another secretory system activated by increases in intracellular calcium. Several mediators produce such increases including acetylcholine (ACh, the other major secretomotor transmitter), which releases Ca 2 + from intracellular stores via activation of M3 muscarinic receptors. This opens basolateral Ca 2 + -gated potassium channels increasing the electrochemical gradient for Cl − exit through open CFTR channels so there is a transient increase in chloride secretion. Kv7.1/KCNE3 channels are also coupled to muscarinic receptor activation further enhancing the hyperpolarization. These actions at the basolateral membrane probably act together with a calcium-gated chloride channel, the CaCC, in the apical membrane that is important for rotavirus-induced hypersecretion.
Apical NHE3 activity is depressed by the intracellular mechanisms elevated cAMP, Ca 2 + , and cGMP that enhance secretion via CFTR, increased basolateral K + conductance or CaCC. There are subepithelial axon terminals from cholinergic and VIP-containing neurons in the tips of the mucosal villi in the small intestine and the surface of the colonic mucosa ( Fig. 19.2 ). Thus, absorption, like secretion, is under direct neural control.
In general, neural activity regulates epithelial transport in several ways: opening or closing ion channels in the membrane altering trafficking of transporters from the cytoplasm to the membrane and changing expression of specific transporters. Nerves also influence net secretion by altering transit time due to altered motor activity. Further, enteric neurons also influence the mucosal barrier, thereby indirectly altering mucosal permeability for water. All these aspects have to be considered when trying to understand the pathophysiology of diarrheal disease.
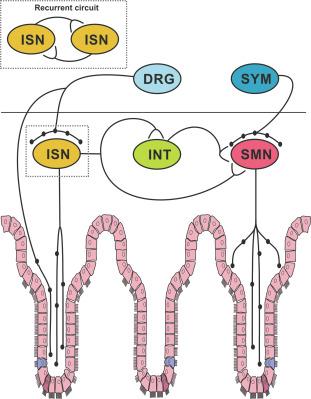
That enteric neural activity influences electrogenic chloride secretion and hence water transport across the mucosal epithelium is evidence from the many studies showing both in vivo and in vitro that blocking neural activity produces major changes in secretion. The enteric neural circuits responsible must include secretomotor neurons and sensory neurons intrinsic to the gut wall and almost certainly also involve interneurons ( Fig. 19.3 ).
There is also evidence for a role for enteric glia. Indeed, a recent study has shown that activation of enteric glia, defined as initiating calcium transients, without prior neural activity can initiate electrogenic secretion, thus implicating glia in the secretomotor control circuitry. Submucosal glia can be activated by cholinergic secretomotor neurons, implying that there are both direct and indirect mechanism operating in these pathways. However, there are very little data showing how glia couple to the neural circuitry, so we will concentrate on the neurons in the circuits in this chapter.
The identities of the different neural classes have been best characterized for guinea-pig ileum, but recent studies in other species and regions suggest that extrapolation from the circuits deduced from this tissue needs to be undertaken with care ( Table 19.1 ). Here we focus on data that are common across species with an emphasis on guinea-pig, mouse, and human.
| Gut Region | Neurochemical Coding | Function | References | ||
|---|---|---|---|---|---|
| Guinea-pig | Ileum | Cholinergic | ChAT/SP (12%) | ISN | 1, 2 |
| ChAT/calretinin (12%) | Interneuron?/vasodilator neuron?/secretomotor neuron? | ||||
| ChAT/NPY/CCK/SOM/CGRP/GAL (33%) | Secretomotor neuron/interneuron?/vasodilator neuron? | ||||
| Noncholinergic | VIP/DYN/GAL (43%) | Secretomotor neuron | |||
| Guinea-pig | Distal colon | Cholinergic | ChAT/TK/calbindin (22%) | ISN | 3 |
| ChAT/calretinin (11%) | Secretomotor neuron?/vasodilator neuron? | ||||
| ChAT/NPY/SOM/CGRP (26%) | Secretomotor neuron | ||||
| Noncholinergic | VIP/NOS (41%) | Secretomotor neuron | |||
| Mouse | Ileum | Cholinergic (41%) | ChAT/- | Unknown | 4 |
| ChAT/CGRP/SOM/calretinin | Secretomotor neuron | ||||
| Noncholinergic (59%) | VIP/NPY/calretinin | Vasodilator | |||
| VIP/NPY/calretinin/TH | Secretomotor neuron | ||||
| Neither VIP or ChAT (8%) | Unknown | ||||
| Rat | Ileum | Cholinergic (100%) | ChAT/NPY/VIP/calbindin/NK 3 (45%) | Secretomotor neuron | 5 |
| ChAT/- (NK 3 /calbindin) (55%) | Unknown | ||||
Bornstein JC, Furness JB. Correlated electrophysiological and histochemical studies of submucous neurons and their contribution to understanding enteric neural circuits. J Auton Nerv Syst 1988; 25 :1–13.
Brookes SJH. Classes of enteric nerve cells in the guinea-pig small intestine. Anat Rec 2001; 262 :58–70.
Lomax AEG, Furness JB. Neurochemical classification of enteric neurons in the guinea-pig distal colon. Cell Tissue Res 2000; 302 ,59–72.
Mongardi Fantaguzzi C, Thacker M, Chiocchetti R, Furness JB. Identification of neuron types in the submucosal ganglia of the mouse ileum. Cell Tissue Res 2009; 336 :179–98.
Mann PT, Furness JB, Southwell BR. Choline acetyltransferase immunoreactivity of putative intrinsic primary afferent neurons in the rat ileum. Cell Tissue Res 1999; 297 ,241–8.
Electrogenic mucosal secretion is directly controlled by enteric secretomotor neurons. Most secretomotor neurons have cell bodies in the submucosal plexus, at least distal to the pylorus (for reviews see Refs. ). Notable evidence for this comes from Ussing chamber studies that show marked increases in electrogenic mucosal Cl − ion secretion resulting from electrical or chemical stimulation, which depends on the presence of the submucosal plexus. Submucosal neurons innervate the intestinal mucosa in all mammals ( Fig. 19.2 ), and blocking enteric neural activity inhibits net fluid secretion in vivo .
The subepithelial layer of the small intestinal mucosa has varicose axons distributed around the crypts and running to the tips of the villi ( Fig. 19.2 ). Similarly, varicose axons innervate both crypts and surface epithelium of the large intestine. These persist even after lesions leading to degeneration of extrinsic nerves and axons from the myenteric plexus. Further, retrograde tracing of markers applied to the mucosa labels many submucosal neurons close to the injection site in guinea-pigs, pigs, and humans. However, the submucosal plexus forms a single layer in small animals, such as mice, rats, and guinea-pig, but two and three anatomically distinct layers of submucosal plexus are seen in larger animals such as pigs and humans. In humans, neurons in all three layers of SMP project to the mucosa, but the proportions and neurochemistry differ between layers, indicating some functional specificity.
There may also be myenteric secretomotor neurons. This is indicated by both retrograde tracing from the mucosa and direct tracing of the axons of myenteric neurons through full thickness preparations to the mucosa of guinea-pig ileum. Retrograde tracing in human tissues has identified VIP-immunoreactive myenteric neurons that project to the mucosa. There are also neurons in the mucosal plexus of larger mammals, notably humans, and their roles in control of secretion remain to be determined.
Two pharmacologically distinct components of neurally evoked mucosal secretion have been widely identified. One is mediated by ACh acting on M 3 muscarinic receptors on mucosal enterocytes. The other is almost certainly mediated by VIP as many submucosal neurons and mucosal nerve terminals contain VIP ( Fig. 19.2 ), which is released by stimuli that evoke neurogenic secretion and is a potent secretagogue. Use of a specific antagonist for the VPAC 1 class of VIP receptor, which is present in the basolateral membrane of mucosal enterocytes, has confirmed VIP as a secretomotor transmitter. Indeed, VIP is the predominant secretomotor transmitter acting via CFTR, with ACh playing a lesser, transient, role probably via basolateral potassium channels and apical CaCC.
In guinea-pigs and mice, the enzyme that synthesizes ACh, choline acetyltransferase (ChAT) is usually not expressed in neurons that contain VIP, although the relative proportions of these neurons differ between regions and species ( Table 19.1 ). Most submucosal ChAT neurons also project to the mucosa. Together with the pharmacological data, this led to the conclusion that there are separate populations of secretomotor neurons: those that contain and release VIP and one or more populations of cholinergic secretomotor neurons. In humans and rats ( Table 19.1 ), however, many VIP secretomotor neurons express ChAT and other markers of cholinergic neurons, implying that they can release both VIP and ACh.
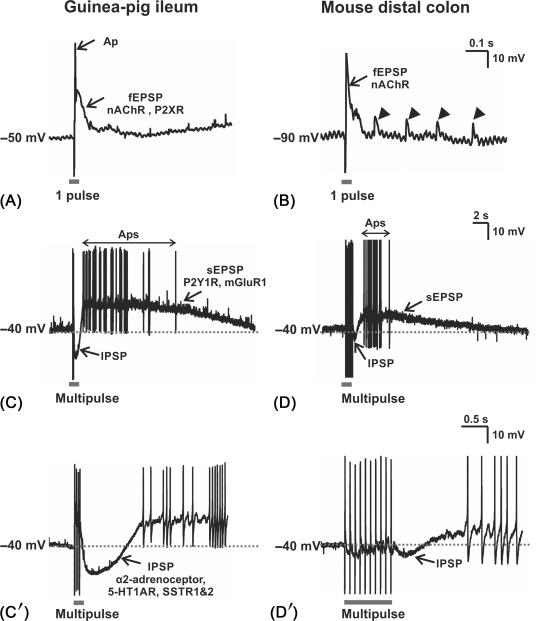
In guinea-pig ileum, distinct neurochemically identified subtypes of secretomotor neurons exhibit markedly different patterns of synaptic inputs with those to VIP neurons being substantially more complex. However, there have been no equivalent studies in other species or regions, so it is impossible to know whether these observations can be generalized. What is clear is that virtually all submucosal neurons exhibit fast excitatory synaptic potentials (EPSPs) mediated by ACh acting on nicotinic receptors, a result seen in guinea-pig small and large intestine and mouse colon ( Fig. 19.4 ) and very recently in biopsy samples of human submucosa. Many submucosal neurons also exhibit either inhibitory synaptic potentials (IPSPs) or slow EPSPs mediated by various metabotropic receptors (guinea-pig ileum and colon, and mouse colon ).
An interesting feature of the secretomotor neurons is that the distribution of their terminals is such that they might be expected to have significant roles in other aspects of mucosal function or in longer term modulation of secretion and absorption. For example, both VIP and ACh modulate the activity of the stem cells that produce enterocytes and other mucosal epithelial cells. Similarly, VIP modulates expression of key proteins within the tight junction complexes and other structures connecting enterocytes that determine transepithelial permeability and hence the flow of water across the mucosa. Furthermore, both VIP and ACh can activate release of mucin from goblet cells. Thus, it is highly likely that, in addition to their immediate roles in regulating secretion and absorption, submucosal secretomotor neurons play key roles in local regulation of the mucosal barrier function. This is consistent with the idea that one function of the secretomotor circuitry is to flush away local irritants and toxins that would otherwise damage the mucosa.
In summary : submucosal neurons releasing VIP are a final common pathway for secretomotor control. Another control system employing ACh as the secretomotor transmitter contributes to short-term changes in secretion. In some species (guinea-pigs, mice), cholinergic secretomotor neurons are distinct from the VIP secretomotor neurons. In humans, there is a clear overlap and ACh and VIP presumably act as cotransmitters, perhaps preferentially released by differing patterns of activity elsewhere in the circuits.
The neurons that regulate the activity of secretomotor neurons are not as well defined. There have been very few studies of the enteric secretomotor circuitry with enough detail to identify relevant neurons, even the picture of the guinea-pig ileum is incomplete. Some elements must be present in all species: neurons that detect the state of the intestine (sensory neurons), interneurons and secretomotor neurons. There must also be an extensive extrinsic system to integrate secretomotor activity with other parts of the digestive system and the broad needs of the organism, for example, whole body water and electrolyte balance.
There are myenteric neurons and submucosal neurons in most species that respond directly to physiologically meaningful stimuli such as amino acids, low pH or short chain fatty acids applied to the mucosa, mucosal distortion, stretch of the intestinal wall, or mechanical deformation of the myenteric plexus. The shapes of these neurons are all similar: large smooth cell bodies and two or more axons. They have common electrophysiological properties: their action potentials trigger prominent and prolonged after-hyperpolarizing potentials (AHPs) (for reviews see Refs. ). They are variously designated as AH/Dogiel type II neurons, intrinsic primary afferent neurons (IPANs), or intrinsic sensory neurons (ISNs). The term IPAN arose because these more colloquially described sensory neurons do not produce conscious sensation and are not always directly sensitive to the specific stimuli that excite them. However, they receive excitatory synaptic input, and computational modeling indicates that they can act as interneurons under some physiological circumstances. Thus, they are not always “primary” afferents. Further, other enteric neurons with the anatomy and physiological characteristics of interneurons respond transiently to mechanical stimuli and may have a “sensory” function. Here we refer to them as ISNs.
In guinea-pig small intestine, the axons of myenteric ISNs largely run circumferentially and ramify in neighboring myenteric ganglia as do those in mouse duodenum and colon. They form synapses with virtually all other classes of myenteric neurons and also send at least one axon to the mucosa. Importantly, they have synaptic connections with other ISNs, transmitting via slow excitatory synaptic potentials. ISNs are cholinergic, but often also contain tachykinins. Each ISN supplies between 10 and 50 villi and each villus receives axons from about 65 different myenteric ISNs in guinea-pig ileum. These mucosal axons are excited by mucosal application of either serotonin (5-HT) or ATP acting via 5-HT 3 and P2X receptors, respectively.
Studies of submucosal ISNs are rare even in guinea-pig. What is known is that they supply terminals to other submucosal ganglia and the myenteric plexus. Further, their activation produces fast and slow EPSPs in other submucosal neurons. In contrast, studies in mice have not revealed neurons with the morphology or electrophysiological properties of ISNs, indicating that ISNs are absent from this plexus in this species. Neurons with similar morphology have been identified in the different layers of the human SMP , but whether they have a sensory function is untested.
Interneurons are integral parts of enteric secretomotor pathways. Several types of myenteric neurons send axons to the submucosal plexus, including ISNs. In guinea-pig ileum, three readily defined classes of myenteric interneurons supply the submucosa, two contain ChAT, while the other contains VIP and neuronal nitric oxide synthase (nNOS). One class of ChAT interneurons also contains somatostatin (SOM) and may have dual functions, as SOM mediates IPSPs in VIP secretomotor neurons. Other ChAT interneurons are immunoreactive for 5-HT and provide input to both VIP and cholinergic secretomotor neurons.
There is less convincing evidence for submucosal interneurons. Submucosal neurons receive input from other submucosal neurons in guinea-pig ileum, but only ISNs have obvious synaptic connections within the plexus. VIP neurons may have sparse connections with other VIP neurons. Data from other species are lacking.
The picture is even more cloudy in the colon, which contains functionally complete circuits, but whether these involve interneurons has not been determined. Similarly, whether there are interneurons connecting the layers of the human submucous plexus is unknown.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here