Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The use of the endonasal pathway for skull base surgery was initially reported in 1909 by Hirsch, who performed his first pituitary surgery in Vienna by approaching the sella through an endonasal route using multiple-staged operations with unenhanced visualization. Despite his first endonasal transsphenoidal surgery having reported success, Hirsch subsequently converted to a transseptal submucosal approach. The first reported use of the endoscope for transsphenoidal surgery was reported by Guiot in 1963 via a sublabial approach. The sublabial and transfixional transseptal approaches became initially favored by neurosurgeons during the expansion of transsphenoidal surgery in the 1970s and 1980s, in conjunction with the operating microscope. Griffith and Veerapen revisited the endonasal pathway with the operating microscope in 1987, using a transsphenoidal retractor. Cooke and Jones reported that use of the endonasal route for microscopic pituitary surgery did not result in any increased sinonasal or dental complications. However, favoring of the direct endonasal route for pituitary surgery was facilitated by the neuroendoscope, with the initial use of sinonasal endoscopy in Europe during the 1970s. In the field of otolaryngology, the introduction of endoscopic sinus surgery kindled an evolution in surgical techniques with advancements in understanding of sinonasal pathophysiology. Successful advances in sinonasal endoscopy then enhanced interest in the use of endoscopy for transsphenoidal surgery. Endoscopic endonasal transsphenoidal surgery (EE-TS) started with endoscope use during biopsy of a sellar lesion, followed by endoscope use for visualization during insertion of transsphenoidal retractors and as supplement to microscopic removal of pituitary adenomas, and eventually the development of the pure form of EE-TS.
This chapter describes EE-TS along with related endoscopic endonasal approaches to the midline skull base such as the anterior cranial fossa (EE-ACF), optic nerve and/or cavernous sinus (EE-CS), pterygoid fossa or petrous apex (EE-Pterygoid or EE-Petrous), clivus and posterior fossa (EE-PFossa), and craniocervical junction (EE-CC junction) ( Table 15.1 ). EE-TS is specifically an operation done completely with endoscopic visualization without any transsphenoidal retractor or nasal speculum. The physical nature of an endoscope with its optics at the tip and slender shaft allows simple access to the sella through the natural nasal air pathway via a nostril. The wide-angled panoramic view, angled-lens views, and a close-up zoom view provide enhanced visualization at the surgical target site. Applying extension of principles from EE-TS also allowed the surgical treatment of midline skull base pathologies (from the EE-ACF to the clivus and EE-PFossa along with the EE-CC) and paramedian skull base pathologies (from the optic nerve and cavernous sinus regions to the pterygoid fossa and petrous tip). Endoscopic endonasal techniques can potentially be applied to nearly any lesion within approximately 2-cm width at the midline skull base from the crista galli at the anterior-superior skull base to the foramen magnum and atlantoaxial region at the posterior-inferior skull base.
| Approach | Pros | Cons |
|---|---|---|
| EE-TS (Transsphenoidal) | Most common and versatile | Physical presence of an endoscope at the surgical corridor |
| EE-ACF (Anterior Cranial Fossa) | Direct anterior access to pathology without brain retraction | Challenges in skull base repair with potential CSF leak |
| EE-CS (Cavernous Sinus) | Direct access to pathology medial to cranial nerves | Limited to easily removable tumors |
| EE-Petrous (Apex/Pterygoid Fossa) | Direct route posterior-lateral access | Access is medial petrous region and bony thickness can be limited |
| EE-PFossa (Posterior Fossa/Clivus) | Direct route P-fossa ventral access | Clival repair may have complexities |
| EE-CC (Craniocervical Junction) | Alternative to transoral | Instrument maneuverability can be limited |
All patients with pituitary adenomas undergo formal endocrine evaluations preoperatively and postoperatively. Hypopituitarism is among the important endocrine conditions requiring treatment starting preoperatively, particularly for hypocortisolism and hypothyroidism. In addition, it is important to distinguish prolactinomas, which might potentially be medically treated. For patients with preoperative visual symptoms or tumors impinging on the optic apparatus on magnetic resonance imaging (MRI), formal neuro-ophthalmologic evaluation is obtained preoperatively with follow-up visual examinations postoperatively. MRI of the brain with and without contrast enhancement (ideally with a pituitary-focused protocol and optionally with dynamic contrast imaging) is the diagnostic imaging modality of choice with best resolution for most pituitary adenomas. Patients who are unable to undergo MRI for various reasons can undergo the lesser alternative of computed tomography (CT) of the brain with pituitary focus and dynamic contrast protocol. Usually, the basic anatomy of the paranasal sinuses and any variations of an individual can be appreciated sufficiently on MRI for the purposes of transsphenoidal surgery. However, bone-window CT scans with fine-cut axial and coronal views can disclose the bony anatomy of the paranasal sinuses in detail and may be obtained as supplemental imaging depending on surgeon’s preference, or for patients who have had previous paranasal sinus or complex bony anatomy for transsphenoidal surgery. If intraoperative image-guided system imaging is desired by the surgeon for EE-TS, imaging protocol compatible with frameless stereotaxy may also be used.
Surgical indications can include goals of reducing anatomic mass effect and/or endocrine hormone abnormalities. Patients with hormonally active or functional pituitary adenomas causing acromegaly, Cushing disease, or secondary hyperthyroidism undergo transsphenoidal surgery as the primary mode of treatment. Patients with prolactinomas are operated upon only when they fail to respond appropriately to dopaminergic medications, develop intolerable side effects to the medications, or choose against the use of dopaminergic medications. Patients with hormonally inactive or nonfunctional pituitary adenomas may be operated upon when the tumors cause symptomatic compression of the optic apparatus, pituitary apoplexy, progressive hypopituitarism, or severe intractable frontotemporal headaches. Other symptomatic mass lesions or tumors at the pituitary fossa generally undergo surgery for anatomic mass effect or for histopathologic diagnosis. In contrast to conventional microscopic surgery, a large pituitary tumor with suprasellar extension can be directly visualized in EE-TS, and bony exposure can be extended rostrally if further exploration at the planum sphenoidale is required. EE-TS can thus enhance the chance for total pituitary tumor resection and potentially reduce the need for a supplemental transcranial approach.
Following hundreds of EE-TS cases, with the youngest patient being a teenager, we have yet to encounter a patient whose nasal passage was too small to undergo standard EE-TS. Notably, patients with acromegaly usually have hypertrophic turbinates and mucosal soft tissues such that the endonasal space can be disproportionately small. In addition, patients with Cushing disease often have narrow nasal airways due to swollen hypertrophic mucosa, which also tends to bleed easily. Among our patients who have undergone EE-TS, two patients with Cushing disease required a two-nostril technique with an endoscope inserted through one nostril and the surgical instruments inserted through the other. Reoperation by EE-TS for patients who have undergone previous conventional transsphenoidal surgery is not difficult if an appropriate anterior sphenoidotomy was previously performed, although reoperation can be challenging if bony sellar structures were excessively eliminated, complications were encountered during previous surgery, or distorting extensive reconstruction was performed.
To perform endoscopic pituitary surgery, the functional physiology and anatomy of the sinonasal cavity should be understood. In the paranasal sinuses (that include the sphenoid, ethmoid, maxillary, and frontal sinuses), mucociliary movement is orchestrated by the delivery of mucus flow to the sinus ostia then toward the nasopharynx. When the path of physiologic mucus flow is interrupted mechanically or functionally, the paranasal sinuses can retain stagnant mucus, which can subsequently become infected and result in sinusitis. The confluence of the draining mucus from the frontal sinus, anterior ethmoidal sinus, and maxillary sinus is located at the middle meatus. Mucosal drainage of the posterior ethmoidal sinus occurs at the superior meatus, and drainage of the sphenoid sinus is at the sphenoethmoidal recess, which is located between the posterolateral aspect of the middle turbinate, superior turbinate, and the rostrum of the sphenoid sinus.
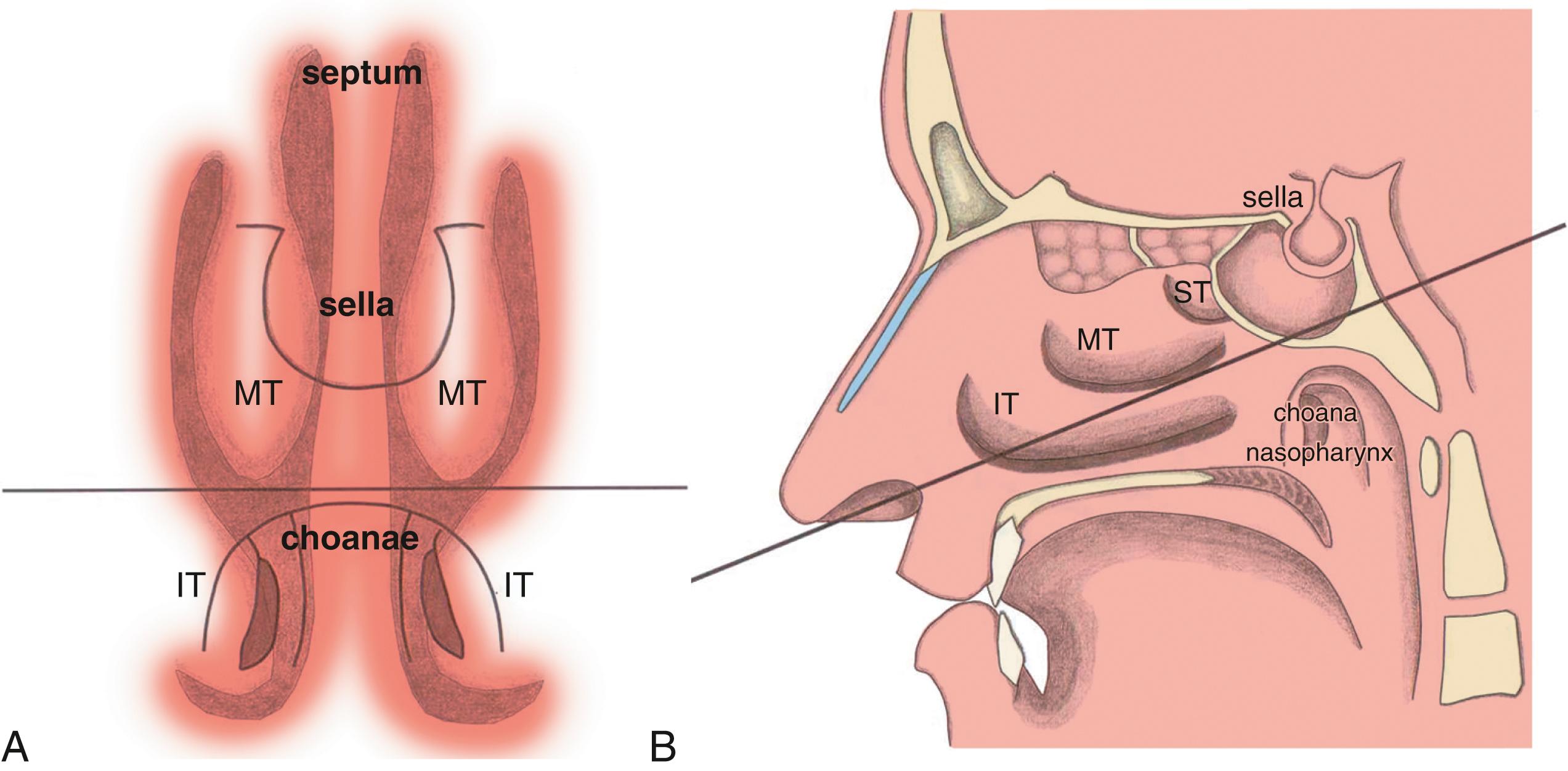
The nasal cavity itself is bordered medially by the nasal septum (composed of the septal cartilage, the perpendicular plate of the ethmoid bone, and the vomer); superiorly by the cribriform plate of the ethmoid bone and bridge of the nose (consisting of the nasal portion of the frontal bone, nasal bone, and frontal process of the maxilla); inferiorly by the floor of the nasal cavity (involving the palatine process of the maxilla and the horizontal plate of the palatine bone); and conchae or turbinates laterally (inferior, middle, superior, and sometimes supreme turbinates). The superior and middle conchae (along with the occasional supreme concha) are components of the ethmoid bone, whereas the inferior concha is a separate bone. The EE-TS procedure traverses the region medial to the middle turbinate, between the middle turbinate and the nasal septum, on the way to the sphenoid sinus then the pituitary fossa at the sella turcica. Nasal septal deviation is not rare, such that sometimes the larger nasal cavity is selected as the route for EE-TS based on preoperative imaging and intraoperative visualization.
The lateral wall of the endonasal route (with numerous projections of the ethmoid bone and individual anatomic variability) has more complex anatomy than the medial wall, which consists of the nasal septum. From a surgical and anatomic embryology standpoint, the ethmoid at the lateral wall of the nasal cavity has been divided into five sequential lamella from anterior to posterior consisting of the uncinate process, ethmoid bulla, basal lamella of the middle turbinate, basal lamella of the superior turbinate, and occasionally the basal lamella of the supreme turbinate (if present). The basal lamella of the middle turbinate divides the ethmoid sinuses into anterior and posterior air cells, and the middle turbinate attachments lie in three different planes with the anterior segment oriented along a sagittal plane (attached to the lateral portion of the cribriform plate superiorly), middle segment oriented along a coronal plane (attached to the lamina papyracea), and the posterior segment oriented nearly along an axial plane (attached to the medial wall of the maxillary sinus and perpendicular plate of the palatine bone). The normal sinonasal anatomy located laterally to the middle turbinate is referred to as the osteomeatal complex (OMC) and comprises a key set of structures for sinonasal function.
The OMC consists of the middle turbinate, uncinate process, hiatus semilunaris, ethmoid infundibulum, and ethmoid bulla. Anterolateral to the middle turbinate is the uncinate process of the ethmoid bone, behind which lies a cleft between the uncinate and ethmoid bulla called the hiatus semilunaris, which is contiguous with the ethmoid infundibulum. The hiatus semilunaris is essentially a two-dimensional (2D) crescent-shaped opening leading from the middle meatus into the three-dimensional (3D) funnel-shaped ethmoid infundibulum to which the frontal sinus, anterior ethmoid sinus, and maxillary sinus usually drain. The frontal sinus drains anteriorly at the ethmoid infundibulum (via the hourglass-shaped frontonasal recess), anterior ethmoid sinus drains into the midportion, and the maxillary sinus drains through its ostium posteriorly at the maxillary infundibulum. Along the posterior-superior bank of the hiatus semilunaris is the ethmoid bulla, which is considered one of the most constant and largest of the anterior ethmoid air cells. The ethmoid bulla is bordered medially by the lamina papyracea of the medial orbit and projects medially in the middle meatus, and the posterior-superior portion of the hiatus semilunaris connects the middle meatus with the suprabullar and retrobullar recesses (collectively called the sinus lateralis), which are defined in relation to the ethmoid bulla. There are variations and debated nuances in the anatomic terminology of paranasal sinus anatomy, including debating of the precise borders of the ethmoid infundibulum in relation to the hiatus semilunaris. Regardless, the more important point is that there are individual structural variations, which can affect paranasal sinus physiology and surgical anatomy. For instance, there are three major variations to the superior attachment of the crescent-shaped uncinate process, with the most common type attaching laterally to the lamina papyracea (∼70% in cadaveric studies) resulting in frontal sinus outflow medial to the uncinate process at the middle meatus. The two other major patterns of uncinate superior insertions include medial attachment to the base of the middle turbinate at the cribriform plate (<20%) and superior attachment to the roof of the ethmoid at the skull base (<10%) that both result in natural frontal sinus outflow lateral to the uncinate at the ethmoid infundibulum. There are other unmentioned variations of uncinate insertions, and these variations may be considered a continuous spectrum of lateral-to-medial insertions rather than merely categorical entities. Anatomic variations may also include a pneumatized or aerated turbinate, most frequently the middle turbinate, which is referred to as concha bullosa and can be enlarged. There can also be variations of a paradoxical middle turbinate with the convexity of the turbinate curve oriented laterally instead of medially, which can alter the expected configuration of the OMC. Despite these variations or the presence of any nasal polyps, the main point is that structures of the OMC should not be significantly disturbed en route to the sphenoid sinus during EE-TS. The sphenoid sinus mucosa should also be minimally disrupted by limiting the mucosal removal only to the region required at the anterior sphenoidotomy site and the anterior wall of the sella.
There can be significant variability in the structure of the ethmoid sinuses, with the ethmoid air cells having some surgically pertinent variations for EE-TS. The agger nasi is a mound or prominence on the anterior-lateral aspect of the nasal cavity formed by a mucous membrane covering the ethmoidal crest of the maxilla near the anterior aspect of the middle turbinate. The agger nasi cells are the most anterior ethmoidal cells located just anterior and lateral to the nasofrontal recess and can sometimes be involved in frontal sinus outflow obstruction. For EE-TS, the surgeon should be aware that a hyperpneumatized agger nasi cell can occasionally present as a bulge that mimics the anterior view of a turbinate. Haller cells (infraorbital ethmoid air cells or maxilloethmoidal cells) are closely related to the ethmoid infundibulum along the medial roof of the maxillary sinus, inferior-lateral to the ethmoid bulla, and extend into the inferomedial orbital floor at the inferior margin of the lamina papyracea. Because Haller cells are quite lateral, these usually do not present a problem during EE-TS, although their proximity to the ethmoid infundibulum can result in inadvertent orbital entry if not recognized. Onodi cells (sphenoethmoidal cells) are posterior ethmoidal air cells that can project superiorly into the sphenoid sinus toward the lateral side and can potentially be confused with a septated region of the sphenoid sinus. The optic nerve and/or internal carotid artery (ICA) can bulge into Onodi cells instead of the sphenoid sinus proper or occasionally may have either partial or complete bony dehiscence at the sphenoid sinus, presenting risk for injury during surgery. It is also possible to have extensive pneumatization of the anterior clinoid process at the opticocarotid recess (OCR), which can also expose the optic nerve and ICA to additional risk. It is also possible for patients who have undergone previous transsphenoidal surgery to have a mucocele at the region of the sphenoid sinus, although this is usually recognizable on the preoperative MRI with the mucocele usually having a spherical type of shape and bright T2 signal such that it can be anticipated preoperatively.
The rostrum of the sphenoid sinus is usually a relatively constant midline landmark regardless of any bowing or lateral deviation of the nasal septum and can be used as a reference for drilling. However, the sphenoid sinuses internally are divided by a highly variable intersinus septum (or configuration of septae) that can be oblique, multiple, or incomplete, and usually does not strictly respect the sagittal midline. Sphenoid sinus septations are generally oriented in a vertical direction and may attach to the regions of the optic canal or carotid artery. Horizontally oriented septations are usually boundaries between the posterior ethmoid sinus and the sphenoid sinus, which can include Onodi cells (sphenoethmoid air cells). The thickness and position of the sella turcica, plus anatomy of the presellar and retrosellar recesses, and tumor extension in the suprasellar region and laterally to the cavernous sinuses should be noted preoperatively to allow anticipatory maneuvers intraoperatively.
When the sphenoid sinus is entered with an endoscope, the complex anatomy is visualized in a panoramic fashion. The clival indentation is seen at the bottom midline, the bony protuberances covering the ICAs are located laterally from the clival indentation, the sella is at the center, the cavernous sinuses are seen lateral to the sella, the tuberculum sella is at the top, and the bony protuberances of the optic nerves are seen laterally. Surgical landmarks for endoscopic endonasal pituitary surgery consist of the choana and nasopharynx along with the inferior margin of the middle turbinate. The choana at the anterior-superior entry of the nasopharynx is always a useful landmark to confirm the middle turbinate. The extended line along the lower margin of the middle turbinate leads to the region approximately 1 cm inferior to the sellar floor. Although the sphenoid sinus ostium at the sphenoethmoidal recess may also be visualized, it is an anatomically variable structure that may not always be easily identifiable or precisely consistent in its relationship to the sella. The sphenoid sinus ostium is an inconsistent surgical landmark, whereas the choana and inferior margin of the middle turbinate are more reliable in consistency. Anterior sphenoidotomy measuring approximately 1.5 cm in diameter is performed at the rostrum of the sphenoid sinus at a location rostral to an extended line from the lower margin of the middle turbinate ( Fig. 15.1A and B ).
Following pituitary tumor removal, the thin flaps of sellar bone previously hinged open may be placed back into position to provide reconstruction at the anterior wall of the sella turcica. Alternatively, reconstruction of the sellar floor may be done using various materials or techniques, and optionally fat grafts or mucosal flaps can be used at the sella for cerebrospinal fluid (CSF) leaks or complex lesions involving significant bony defects. The sphenoid sinus is typically preserved as a natural air-filled cavity, and we do not routinely use any foreign surgical material within the sphenoid sinus itself. When an anterior sphenoidotomy is made, attention must be paid not to significantly disrupt the normal posterior mucus drainage channel at the sphenoid ostium of the sphenoethmoidal recess. Toward the conclusion of EE-TS, the middle turbinate is gently medialized back to its original position in order to avoid blockage of mucus drainage at the OMC.
The nasal mucosa is predominantly supplied by the sphenopalatine artery, with contributions from the greater palatine artery, branches of the facial artery, and the anterior and posterior ethmoidal arteries. The posterior septal artery arises from the sphenopalatine branch of the internal maxillary artery and passes to the posterior nasal septum at the inferior-medial aspect of the middle turbinate posterior margin. When surgical access is obtained between the middle turbinate and nasal septum for an anterior sphenoidotomy, coagulation of a sufficient segment of the posterior septal artery is often required. Delayed copious nasal bleeding after transsphenoidal surgery usually arises from rebleeding of the posterior septal artery branch of the sphenopalatine artery.
For endoscopic endonasal approach to the EE-ACF, the region of the ethmoid roof with the cribriform plate and the fovea ethmoidalis should be studied on preoperatively imaging. Keros categorized three types of skull base conformations along a spectrum depending on the depth of the olfactory sulcus and corresponding height of the lateral lamella, with type 1 having 1- to 3-mm depth, type 2 having 3- to 7-mm depth, and type 3 having 7- to 16-mm depth such that the ethmoid roof is significantly higher than the cribriform plate. The Keros classification is more pertinent for ethmoid sinus surgery to avoid inadvertent entry through the thin lateral lamella of the cribriform plate, especially for a deep olfactory sulcus. However, for EE-ACF or EE-CS (via the middle meatal approach with posterior ethmoidectomy), one should be aware of the potential individual variations in depth of the olfactory sulcus and the specific anatomic configuration should be noted preoperatively to assist with optimal intraoperative maneuvering. In the same region, the ethmoidal sulcus is the groove in the lateral lamella for the anterior ethmoidal artery, which branches from the ophthalmic artery and enters from the orbit in a bony canal in the ethmoid roof (anterior ethmoidal canal) or just below the level of the ethmoid roof to course anteromedially with the anterior ethmoidal nerve and to penetrate the lateral lamellae to supply the dura in the region of the olfactory sulcus. The posterior ethmoidal artery traverses the posterior ethmoidal canal within a 3-mm planar region above the cribriform plate. The anterior and posterior ethmoidal arteries usually provide the major blood supply for olfactory groove or planum sphenoidale meningiomas at the skull base.
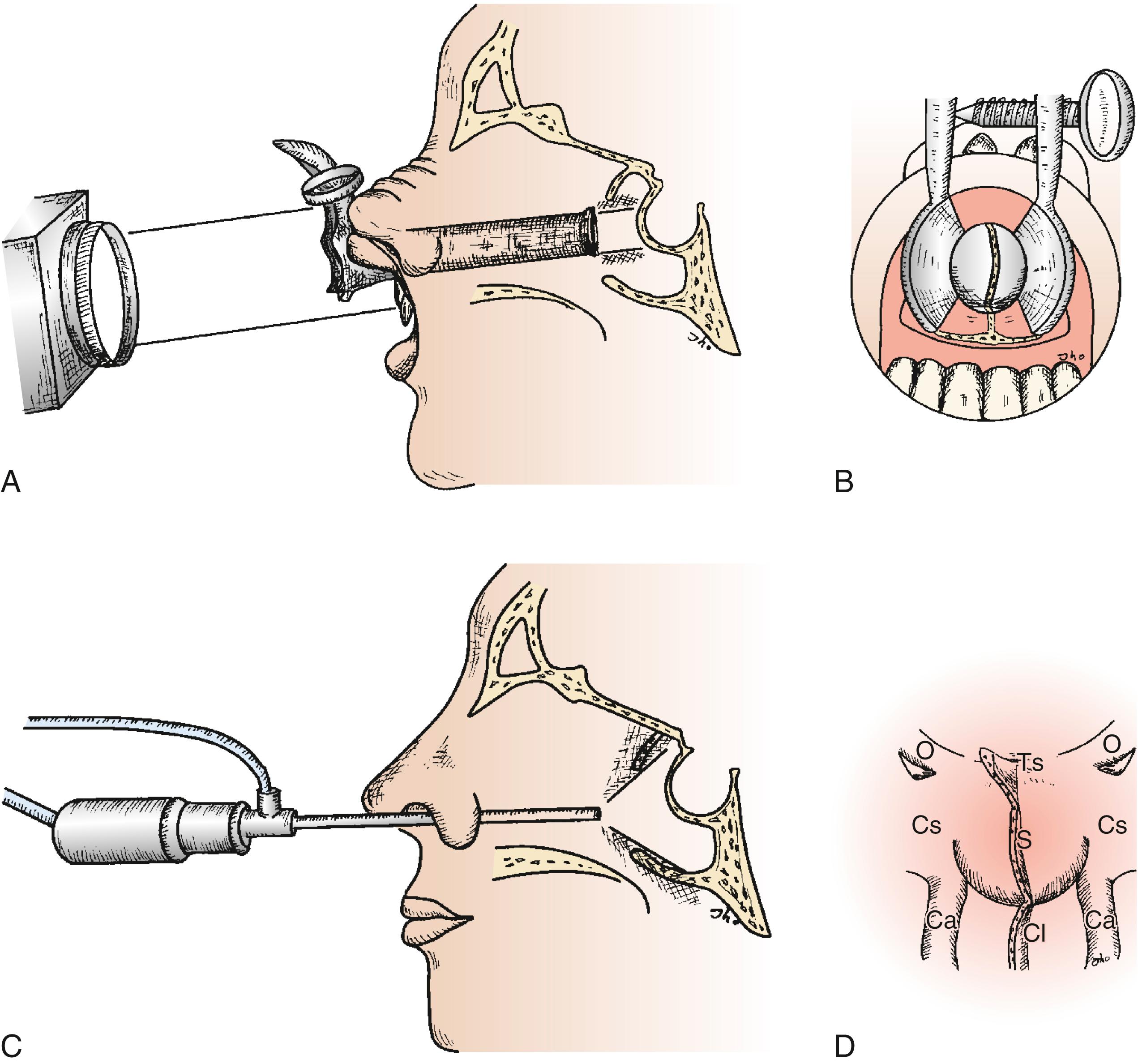
As transsphenoidal pituitary surgery evolved in the 20th century, advances in optical visualization included the adoption of the operating microscope in the 1960s and use of the endoscope in the 1990s. The operating microscope provides a magnified view with focused lighting at a central portion of the sella through a corridor exposed by the transsphenoidal retractor. The endoscope can physically enter into the sphenoid sinus and provide a wide-angled panoramic view with zooming capability. An operating microscope renders a tubular parallel beam view, but an endoscope shows a diverging flask-shaped wide-angled view ( Fig. 15.2A–D ). This wide-angled panoramic view is particularly useful for pituitary tumor surgery because it allows excellent anatomic visualization at the posterior wall of the sphenoid sinus. However, it must be recognized that the endoscopic view renders a fish eye effect with maximum magnification at the center and relative contraction at the periphery with visualization of a wide anatomic area.
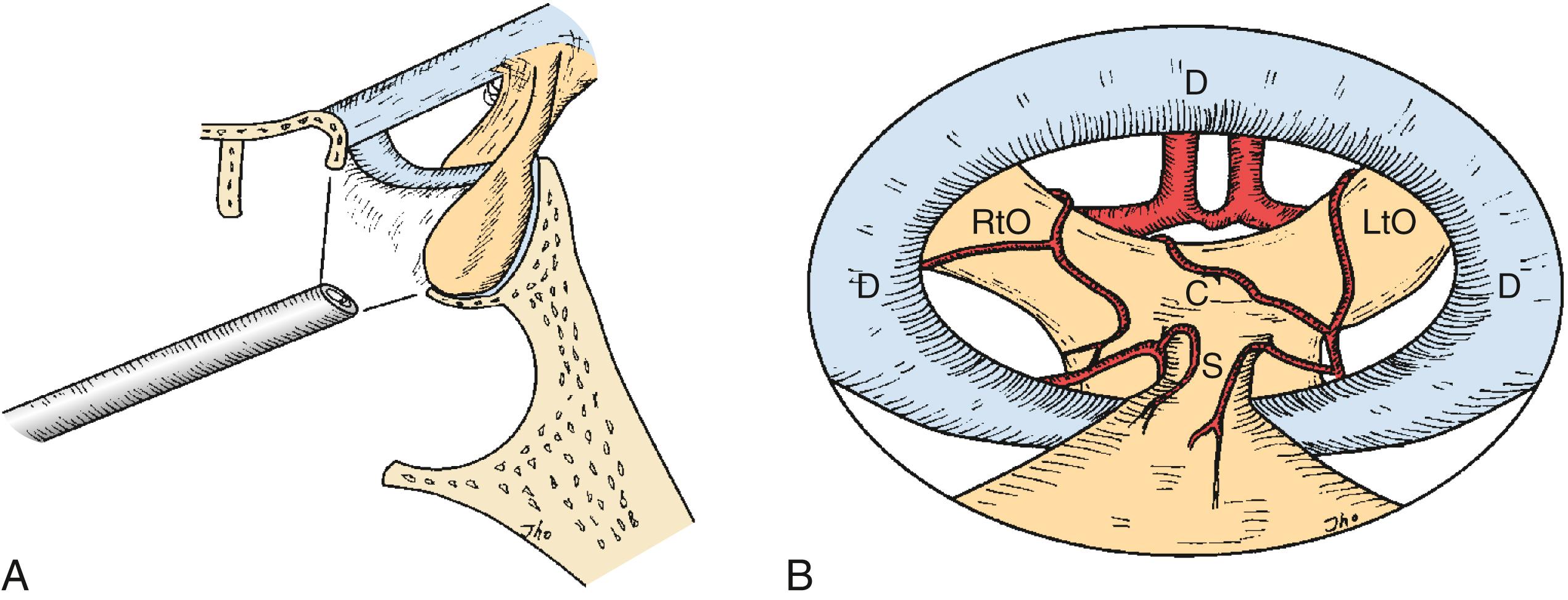
Endoscopes can be subdivided into two categories: rod-lens rigid endoscopes and fiberoptic flexible endoscopes. The image quality of the rod-lens endoscope is generally superior to the fiberoptic flexible endoscope, due to having greater numbers of fiberoptic fibers. Rod-lens endoscopes of 4-mm diameter provide full-screen images of high quality, in contrast to smaller images of lower quality for 3-mm rod-lens endoscopes. The basic endoscopes used by the authors are 4-mm rod-lens endoscopes with 0-, 30-, or 70-degree lenses. The endoscope can be advanced into the sella or suprasellar area for a close-up internal view. The close-up view can help to confirm margins or to visualize the internal anatomy at the resection site. Angled-lens endoscopes provide direct visualization of anatomic corners such as the suprasellar region or toward the cavernous sinuses. These views can be of assistance even if the endoscope is used as an adjunctive tool during microscopic surgery. When a 30-degree angled endoscope is inserted into the cavity and rotated through a full revolution, the entire circumference of the cavity can be visualized. Angled views may be advantageous when large suprasellar macroadenomas are to be removed or direct visualization of the medial wall of a cavernous sinus is required. Angled views can also assist direct surgical access to the pterygoid fossa, EE-ACF, clivus, and posterior cranial fossa in addition to the cavernous sinus ( Fig. 15.3A and B ).
Surgical equipment includes endoscopes with 0-, 30-, and 70-degree lenses ( Fig. 15.4A ) plus associated video-imaging system and light source connections, an endoscope lens-cleansing device, a rigid endoscope holder, and surgical instruments specifically customized or designed for endoscopic pituitary surgery. The length of the endoscope should be 18 cm, with shorter lengths having inadequate reach and much longer lengths being potentially awkward to maneuver. An endoscopic lens-cleansing device is recommended so that the surgeon can achieve better operating continuity ( Fig. 15.4B ). This device consists of a disposable irrigation tube, which has a loop of tubing passed through a battery-powered rotary device. The irrigation tube is connected into a warmed saline bag (the temperature prevents lens fogging), which is hung on a pole, and the motor-powered irrigation device is controlled by a foot pedal to flush saline forward. When the foot pedal is released, the motor reverses its rotary direction and draws the saline back for 1 to 2 seconds.

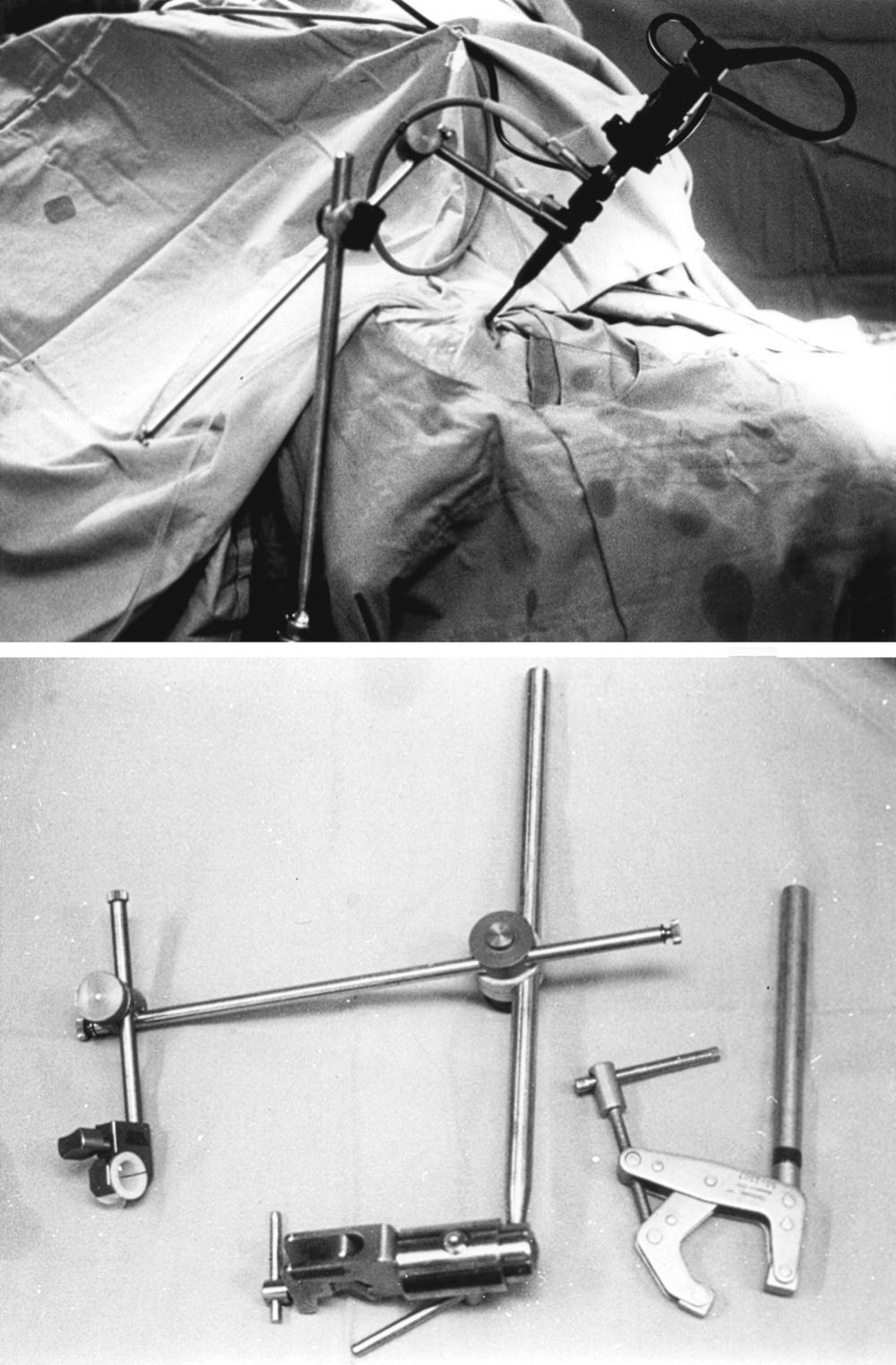
Endoscope holders provide stability of the visual image and bimanual instrument use without requiring a surgical assistant, during portions of the case such as drilling and tumor removal ( Fig. 15.5 ). There are portions of surgery during which dynamic visualization is important, such as the initial approach to the sphenoid sinus rostrum and final visualization of any tumor remnants at anatomic corners of the sella, but endoscope holders provide camera stability akin to a video camera tripod during appropriate segments of surgery. Having a surgical trainee hold and drive the endoscope can help for the trainee’s education, but the stability and localization of views during surgery may be variable depending on the level of the trainee or assistant. The endoscope holder should provide rigid fixation of the endoscope while allowing simple transition between stable fixation and manual dynamic steering. The holding terminal should be compact and slender so as to render adequate operating space around the endoscope shaft for the surgeon to maneuver surgical instruments. We routinely use a customized manual endoscope holder specifically designed for EE-TS, in contrast to a nitrogen-powered holder called the Mitaka Point Setter (Mitaka USA; Park City, Utah) for our endoscopic transcranial approaches and a nitrogen-powered holder called Unitrac (Aesculap; Tuttlingen, Germany) for our endoscopic spine surgery. The EE-TS manual holder has multiple hand-tightened joints, but only a single joint has to be loosened during a case to transition between endoscope fixation and manual driving.
Among various surgical instruments, a monopolar suction coagulator (No. 8- or 9-French cannula), a bipolar suction coagulator (No. 8- or 9-French cannula), and a single-bladed bipolar coagulator are used for hemostasis. These are all disposable, and the monopolar suction coagulator is malleable and insulated. The malleable monopolar is useful during the endonasal stage when preparing the nasal cavity for anterior sphenoidotomy. The single-bladed bipolar has one electrode at the core and the other at the shell. Suction cannulas with curved or malleable tips (No. 5- and 7-French cannulas) are typically used. Dural suturing at the sella is typically not needed, but titanium microclips and nitinol U-clips are commercially available. In addition, standard surgical sutures may be used in conjunction with specialized tying techniques if needed. Other instruments used include a micropituitary rongeur, a pituitary rongeur, ethmoid rongeurs, a high-speed drill, micro-Kerrison rongeurs, pituitary ring curettes, Jannetta 45-degree microdissector, single-bladed Kurze scissors, and a specially-designed septal opener. A slender high-speed drill is useful when the sphenoid sinus is small or has limited pneumatization.
The patient is positioned supine in lawn chair position with the chest elevated approximately 15 to 20 degrees, and the neck is mildly extended so that the head is positioned with the forehead and chin leveled horizontally. The head is typically rotated toward the surgeon by 10 to 15 degrees. The head is placed slightly higher than the heart, with the intent that the cavernous sinus venous pressure remains low to minimize venous bleeding. The leveled head positioning allows the surgeon to access the region of the middle turbinate ergonomically for EE-TS with the endoscope inserted at approximately 25 degrees cephalad. The head is alternatively further extended 15 degrees with the endoscope inserted ergonomically at a similar angle if the EE-ACF will be explored as in EE-ACF. Conversely, the head is flexed 15 degrees when the clival or EE-PFossa region is to be explored in EE-PFossa. A head pin fixation device can be used as an option but is not necessary.
The ipsilateral arm on the side of the operating surgeon is tucked. Double-layer eye protection is used to shield from prep solution, with the nose left exposed in a triangular fashion. Multiple long-handled disposable cotton swabs (routinely a total of 10 with 5 per side) are used to prepare the nasal passages. The nasal cavities and face, along with a small region on the abdomen for potential fat graft (usually the infraumbilical area is used for cosmesis), are prepped and draped in an aseptic manner. The base of the endoscope holder is attached to the operating table. The size of the nasal airways and desired lateral angle of trajectory to the target can be assessed to determine which nostril is to be used on preoperative imaging and direct views. The nasal septum is sometimes deviated to one side, or it may be easier to visualize the contralateral side of the sella and cavernous sinus from the selected nostril. In our early cases, topical application of a vasoconstrictor and local infiltration of lidocaine with epinephrine were used routinely, but this was stopped as experience accumulated. The use of vasoconstrictors may lessen initial bleeding transiently, but this may result in suboptimal intraoperative hemostasis such that bleeding may occur in a delayed fashion postoperatively.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here