Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The importance of phosphate regulation in the human body has been highlighted by research showing that phosphate dysregulation is associated with many disease conditions, including chronic kidney disease (CKD), cardiovascular disease, tumorigenesis, premature aging, and skeletal disorders. The benefit of dietary phosphate restriction in the management of CKD is well established. However, until recently, little attention has focused on the health and disease consequences of an oversupply of phosphorus in the diet of healthy adults. Recent epidemiological research has shown that mortality in healthy adults increases as dietary phosphate intake levels rise above 1400 mg per day. Paradoxically, the United States Department of Agriculture continues to recommend dietary phosphate levels exceeding 1700–1800 mg per day for a 2000-calorie diet. Thus, issues of phosphate homeostasis apply to the general population, not just to patients with CKD.
In this book chapter, we outline the physiological basics of phosphate homeostasis, focusing particularly on endocrine regulation of this essential micronutrient—the second most abundant mineral in the body, next to calcium. The first part discussed at the intestinal absorption and regulation of phosphate homeostasis through the bone–kidney axis, the next part explained in more detail at the interaction of specific endocrine regulators of phosphate homeostasis, and the last part elaborated some of the pathophysiological conditions associated with dysregulated phosphate homeostasis.
The regulation of phosphate homeostasis is biologically important because inorganic phosphorus performs many functions within the body. Phosphate is a component of nucleic acids, DNA and RNA, and it is incorporated in the structure of phospholipids in cell membranes. As an intracellular anion, phosphate is involved in the activation and inactivation of enzymes and coenzymes. Phosphate also plays roles in cell signaling through phosphorylation, in energy metabolism as ATP, and in bone mineralization as a principal element within hydroxyapatite. Endocrine regulation of phosphate depends on a delicate balance among circulating factors like 1,25(OH) 2 D 3 (calcitriol, the active form of vitamin D), parathyroid hormone (PTH), and fibroblast growth factor 23 (FGF23). Dysregulation of these factors can induce phosphorus imbalances which can affect the functionality of almost every human system, including musculoskeletal and cardiovascular systems, ultimately leading to an increase in morbidity and mortality. Through the action of PTH, vitamin D, and FGF23, phosphate homeostasis is maintained by regulating the amount of phosphate absorbed in the intestines, reabsorbed in the kidney, and resorbed from bone ( Fig. 31.1 ).
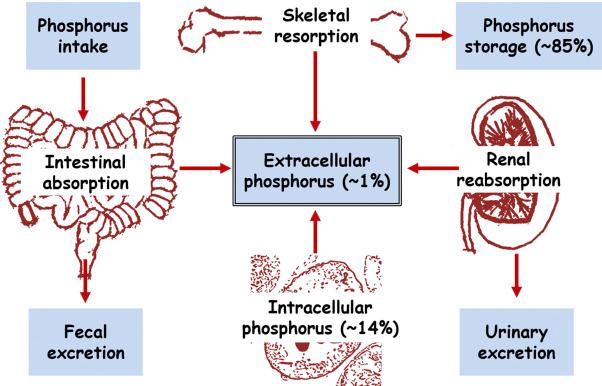
Phosphate from dietary sources is absorbed in the small intestines, mainly through active transport by the type II sodium-dependent phosphate cotransporter, Npt2b (encoded by gene SLC34a2), and absorption is regulated by hormones and dietary conditions. The sodium and phosphorus ions are transported by the Npt2b cotransporters, which involves voltage-dependent and electroneutral mechanisms. In addition to 1,25(OH) 2 D 3 , factors that regulate the expression and function of Npt2b include FGF23, matrix extracellular phosphoglycoprotein (MEPE), epidermal growth factor (EGF), thyroid hormone, estrogens, glucocorticoids, and metabolic acidosis. Moreover, Na + /H + exchange regulatory factor 1 (NHERF1) also interacts with Npt2b in the intestines under conditions of low dietary phosphorus intake. A smaller amount of phosphorus is believed to be absorbed in the intestines by type III sodium-dependent phosphate cotransporters, Pit1 and Pit2, which operate by passive transport and are involved in functions of individual cells. In addition, ingestion of large amounts of dietary phosphate can increase serum phosphate levels through increased absorption via a paracellular route, bypassing hormonal regulation of cotransporters.
Serum phosphate levels are regulated mainly through the reabsorption rate of renal phosphate within the kidney, the major organ that regulates phosphate homeostasis according to the body’s requirements. Renal phosphate reabsorption increases or decreases through the expression of sodium-phosphate cotransporters, Npt2a (SLC34a1) and Npt2c (SLC34a3), located in the renal proximal tubule. Npt2a is electrogenic and transports three Na + ions coupled to phosphorus (HPO 4 2− ) at physiologic pH, while Npt2c is electroneutral and transports two Na + ions for each phosphorus ion. Pit2 phosphate transporters are also located in the renal proximal tubule. It is believed that a low concentration of sodium regulated by Na + , K + -ATPase in the cells of the basolateral membrane facilitates phosphorus transport within the proximal tubule. Cotransporters import phosphate from the proximal tubular lumen, translocated across the apical brush-border membrane (BBM) and exported at the basolateral membrane. Although the molecules involved in phosphate translocation have not been identified, xenotropic and polytropic retrovirus receptor 1 (XPR1) appears to act as a phosphate exporter in mammalian cells, but its role as an exporter in renal cells has not yet been determined.
Hyperphosphaturia and hypophosphatemia occur in Npt2a knockout mice (Npt2a –/– ). Concentrations of serum 1,25(OH) 2 D 3 and urine calcium also increase in Npt2a knockout mice; there is a ∼70% decrease in phosphate transport in the BBM, and reabsorption is supported by increased expression of Npt2c. This evidence indicates that Npt2a in mice is a major transporter in renal phosphate reabsorption. Mutation of Npt2a in humans causes recessive Fanconi syndrome with hypophosphatemic rickets, suggesting that Npt2a plays a role in phosphate handling. In Npt2c knockout mice, (Npt2c –/– ), hypercalciuria and higher serum concentrations of 1,25(OH) 2 D 3 occur, but hypophosphatemia or rickets does not. The situation is different in humans with mutation of Npt2c, causing hereditary hypophosphatemia rickets with hypercalciuria (HHRH), which suggests that Npt2c has a more important role in phosphate homeostasis in humans than in mice.
Bone functions as a reserve for calcium and phosphate from which the body can deposit and withdraw minerals to maintain mineral ion balance. Reabsorption is the process by which bone releases calcium and phosphate from the hydroxyapatite bone matrix. Osteocytes in mature bone assist in bone mineralization and phosphate homeostasis by producing factors such as FGF23 and 1,25(OH) 2 D 3 . Other endocrine regulators that target bone include PTH, calcitonin, sex hormones, and osteocalcin. An endocrine communication network that regulates phosphorus homeostasis is formed between bone, kidneys, intestines, and parathyroid glands ( Fig. 31.1 ).
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here