Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
An adrenal incidentaloma is defined as an adrenal mass identified on imaging performed for an indication other than suspected adrenal disease. With increasing utilization of cross-sectional imaging over the past several decades, 40% of adrenal masses are now discovered incidentally. It is estimated that 1% to 8% of adults have an adrenal incidentaloma, and the incidence increases with age.
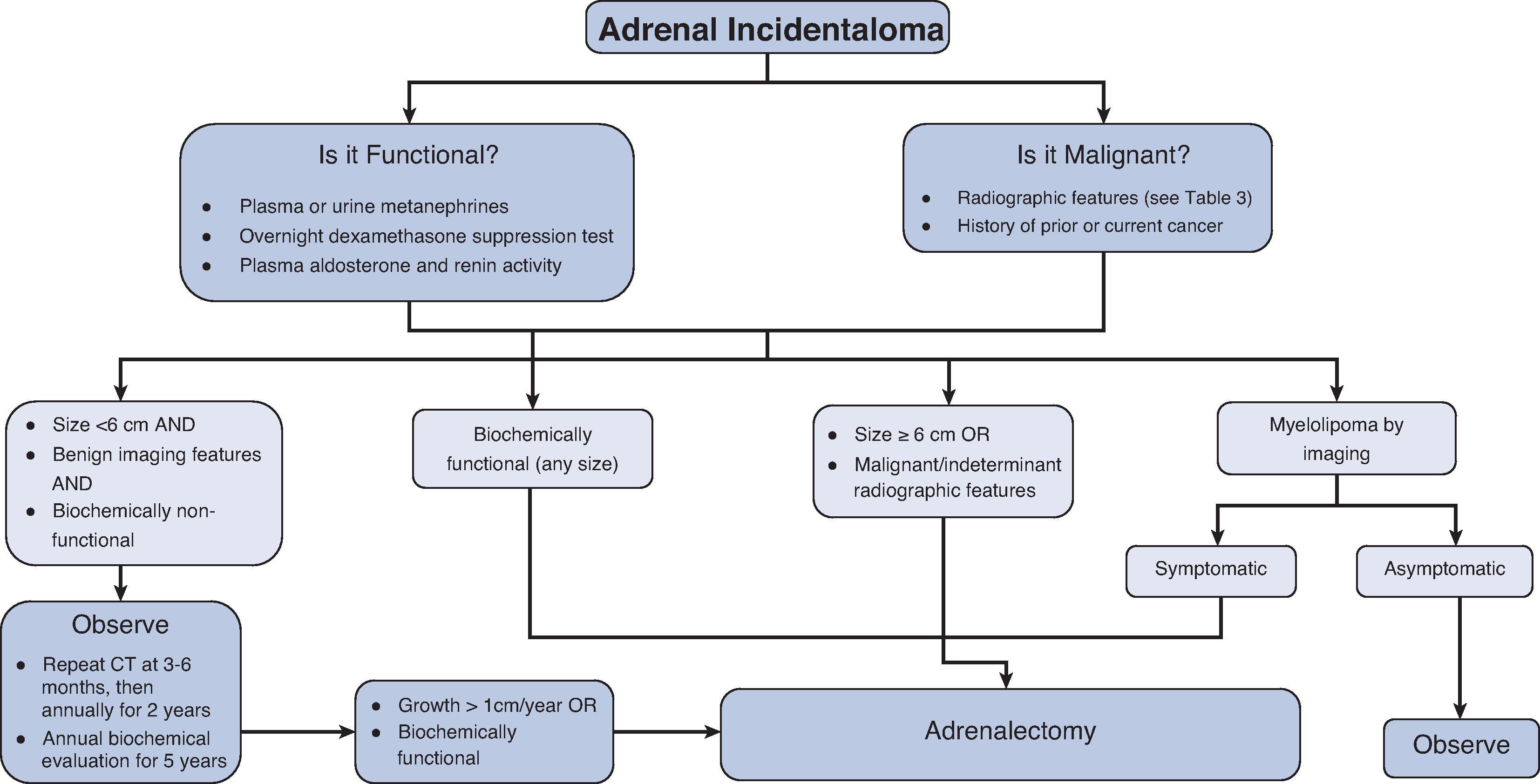
The initial diagnostic evaluation of an adrenal incidentaloma must (1) assess the risk of malignancy and (2) determine whether it is hormonally active. Of the functional lesions, the most common is Cushing’s syndrome followed by pheochromocytoma and aldosteronoma. Once the diagnosis is achieved, if surgery is indicated, the benefits and risks of surgery should be considered and the operative approach determined. Finally, postoperative or nonoperative surveillance must be established.
The initial evaluation of an adrenal incidentaloma consists of: (1) A comprehensive history and physical examination to elicit signs and symptoms of adrenal hormone excess and cancer history, (2) radiologic evaluation, and (3) biochemical evaluation to determine risk of malignancy and functionality ( Fig. 1 ).
The most common signs and symptoms in patients with hypercortisolism include hypertension, obesity, and hyperglycemia/diabetes ( Table 1 ). They can also present with osteoporosis, facial plethora, proximal muscle weakness, easy bruising, weight gain, cutaneous abdominal striae, buffalo hump, and moon facies. Signs and symptoms of catecholamine excess are present in over 60% of patients with a pheochromocytoma and include hypertension, episodes of severe headaches, palpitations, perspiration, anxiety, abdominal pain, and nausea or vomiting. Patients with hyperaldosteronism may present with a history of asymptomatic or symptomatic hypokalemia, refractory hypertension, or fluid retention. In assessing for primary malignancy or metastasis, a personal history of cancer, smoking history, or recent weight loss should be elucidated. Hirsutism or feminization may represent physical signs of sex hormone excess, which can accompany adrenocortical carcinoma. In addition to the aforementioned signs and symptoms, particular attention should paid to the patient’s vitals signs as an elevated systolic and diastolic blood pressure and/or heart rate are consistent with hypercortisolism, hyperaldosteronism, and pheochromocytoma.
| Nonfunctional | Functional | |
|---|---|---|
| Benign |
|
|
| Malignant |
|
|
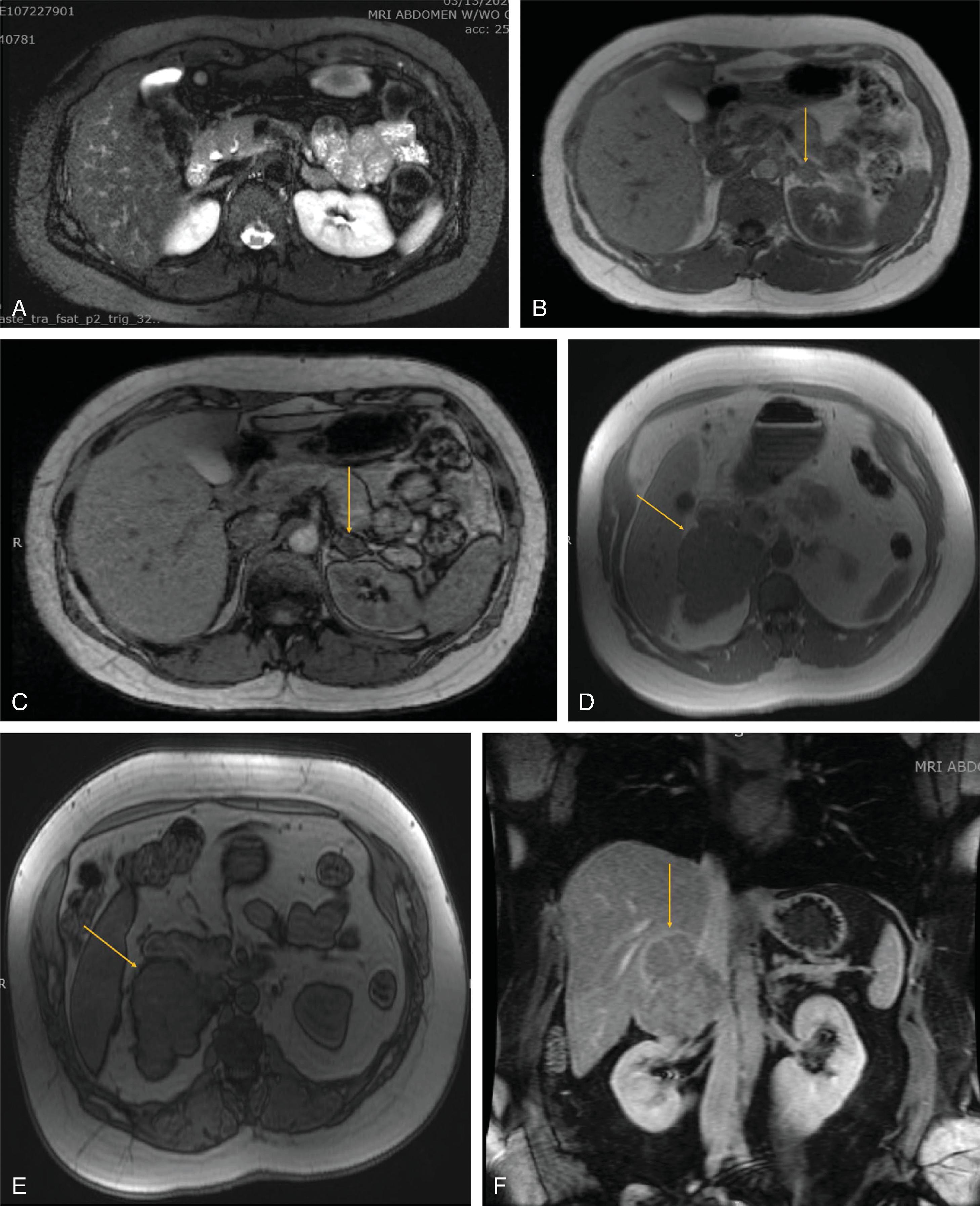
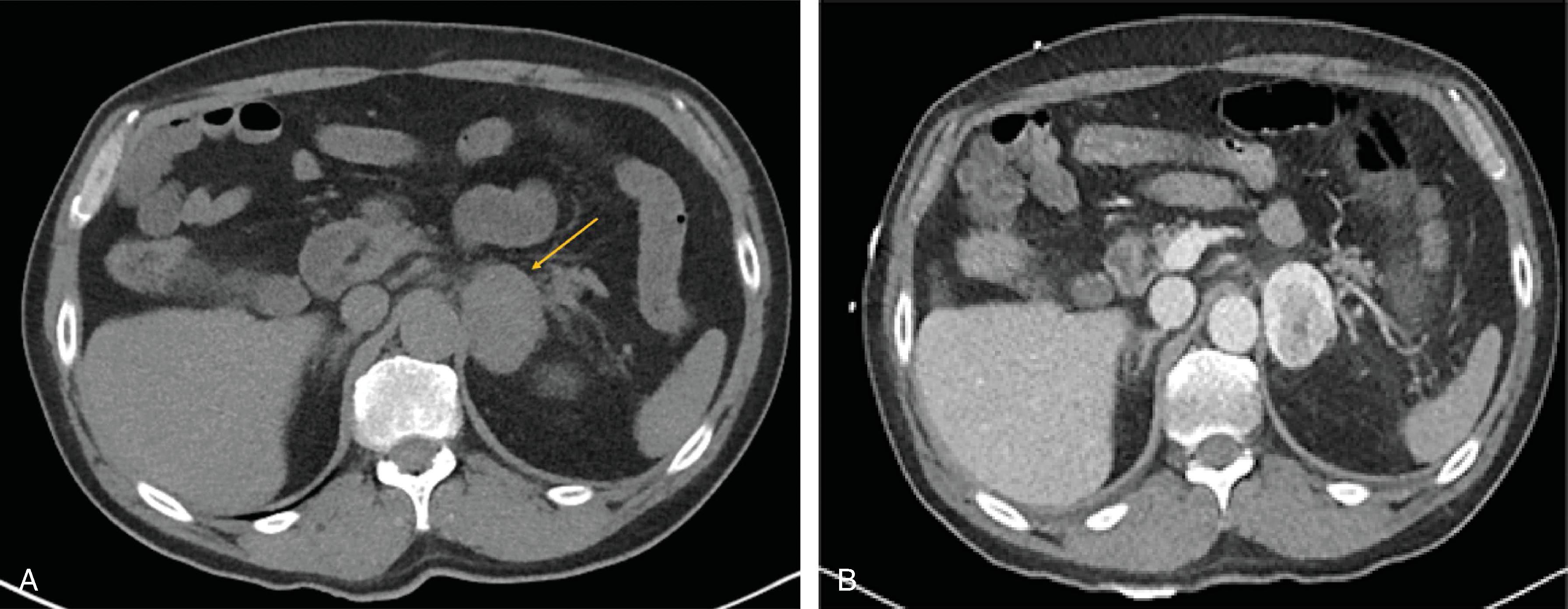
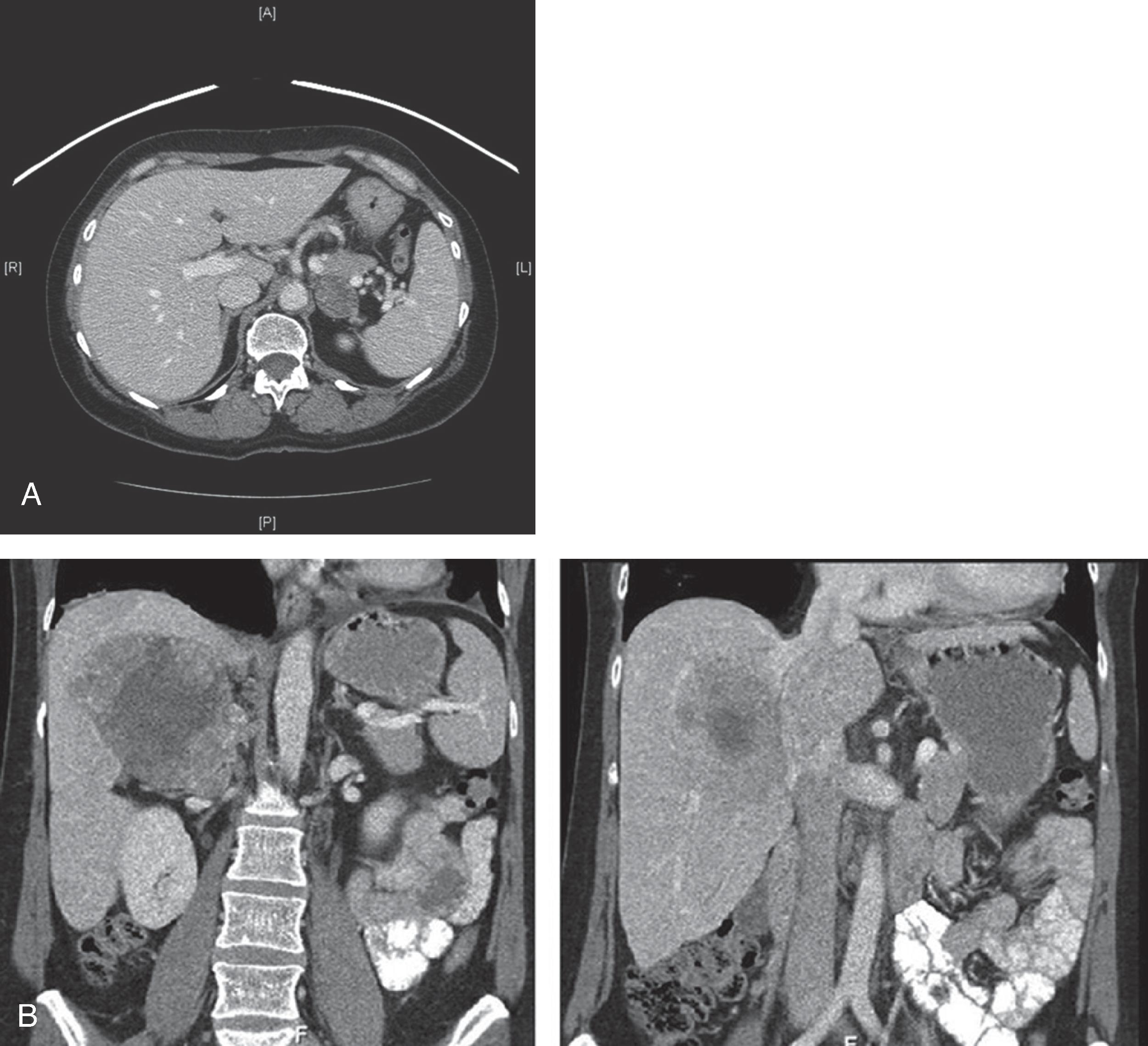
The primary goal of imaging studies is to evaluate for radiographic features that portend a higher risk of malignancy. Although CT is the most commonly used imaging modality, MRI is preferred in children, adolescents, and pregnant women ( Figs. 2 and 3 ). A triple-phase CT scan with and without intravenous contrast including arterial-enhanced, venous-enhanced, and delayed images of the adrenal glands is ideal for initial radiographic assessment as this imaging modality is readily available and provides excellent anatomic guidance should patients require surgery. Several specific radiographic features on CT should be considered to assess for malignancy or need for surgical resection. These are discussed in the following sections.
Adrenal mass size >4 cm has been associated with an increased risk for cancer across several studies. Prior studies have approximated a 2% risk of adrenocortical carcinoma in masses <4 cm, a 6% risk in masses measuring 4 to 6 cm, and a 25% risk in masses >6 cm. However, size must be considered along with additional radiographic features in determining the threshold for surgery. The majority of incidentalomas >6 cm should undergo adrenalectomy. Management of benign-appearing, nonfunctional incidentalomas 4 to 6 cm in size is controversial, and either adrenalectomy or active surveillance can be offered for the appropriately selected patient. Myelolipomas, for example, can present as large adrenal lesions, but they have distinct radiographic features and do not necessitate surgery at >6 cm unless the patient is symptomatic.
CT characteristics that differentiate lesions from one another include margins, shape, heterogeneity, attenuation, and washout. Typical benign features include distinct and smooth margins, round shape, and homogenous composition. Attenuation of <10 Hounsfield units (HU) without contrast and rapid (>60%) absolute washout on 15-minute delayed-phase images is characteristic of lipid-rich benign adenomas. In contrast, lipid-poor lesions, such as malignant tumors, have a density >10 HU without contrast. However, 20% to 30% of benign adenomas can be lipid poor. Contrast enhancement and percentage of contrast washout convey information related to the vascularity of lesions. Lesions with increased vascularity, such as malignant tumors, demonstrate strong arterial enhancement and absolute washout <60% on delayed-phase imaging. Further features suggestive of malignancy include large size, irregular borders, heterogenous composition, central necrosis, and local invasion into surrounding structures. Pheochromocytomas may be difficult to distinguish from malignant lesions on CT as both have a high density, enhance, and have <60% washout. Additionally, central necrosis and calcifications may be present in pheochromocytomas. Pheochromocytomas, however, do not have the same predilection for local invasion as adrenocortical carcinomas and are distinguished by biochemical testing. Myelolipomas are composed of mature adipose tissue and interspersed areas of myeloid tissue. These features provide a characteristic appearance on CT of a well-circumscribed mass with areas of higher attenuation intermixed with areas of predominantly low attenuation, measuring –130 to –23 HU without contrast. Myelolipomas can be diagnosed on the basis of imaging alone.
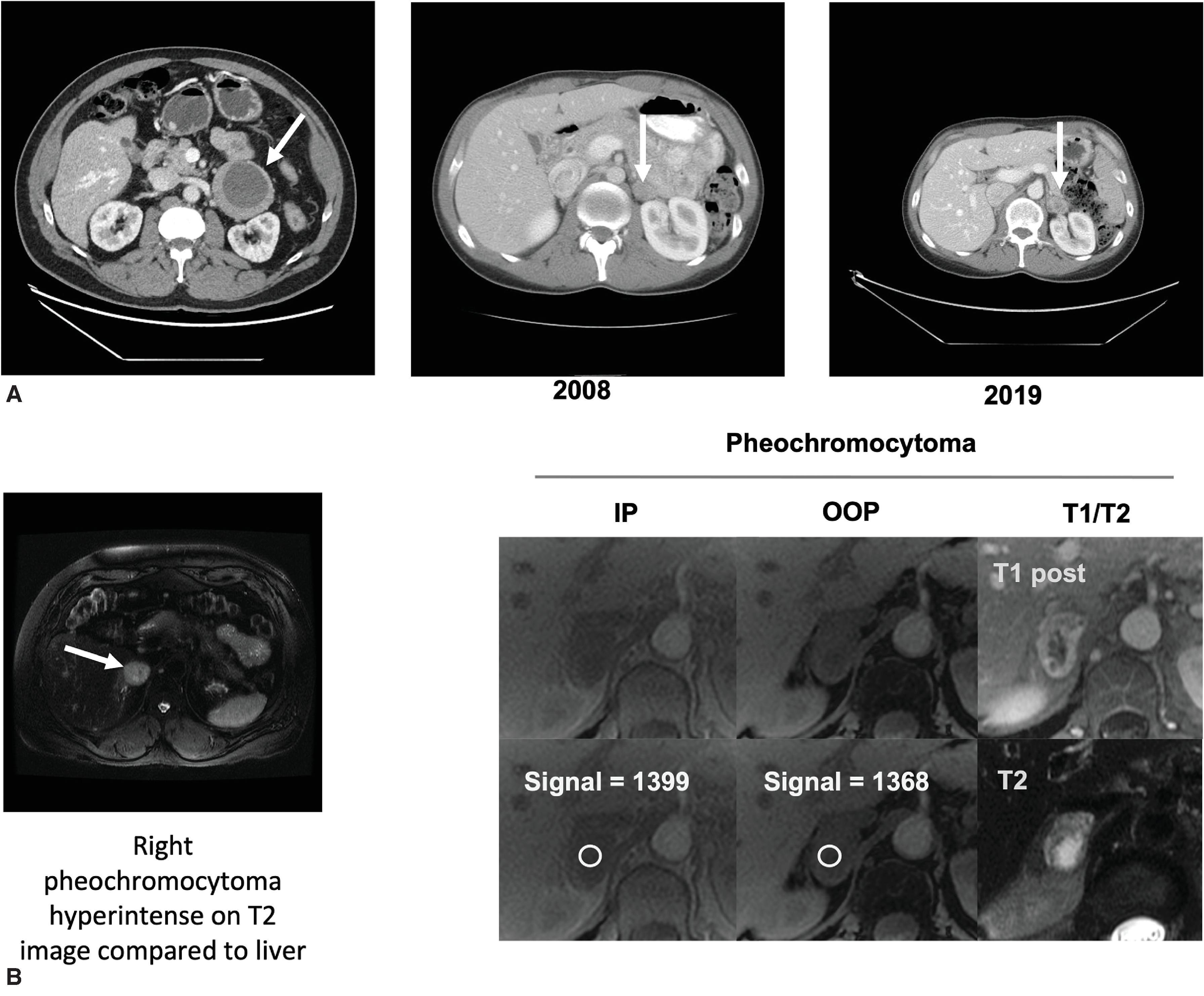
MRI for evaluation of the adrenal glands includes contrast-enhanced and T1- and T2-weighted images of the entire abdomen. In-phase and out-of-phase images of the adrenal glands are also performed to evaluate the fat content of lesions. A loss of signal intensity on out-of-phase images indicates fatty composition, which is compatible with lipid-rich lesions such as adenoma or myelolipoma. A lack of signal drop in out-of-phase images is more suggestive of malignancy or pheochromocytoma. These dense lesions show high signal intensity in T2-weighted images on MRI. Large pheochromocytomas and cancers can demonstrate heterogenous signal intensity that is related to the cystic and necrotic degeneration that may accompany large lesions. A classic “light bulb” T2 signal can be found in 11% of pheochromocytomas. Similar to their appearance on CT, pheochromocytomas and malignant lesions will demonstrate strong contrast enhancement following injection of gadolinium. Myelolipomas appear hyperintense on T1-weighted images (macroscopic fat) with regions of hypointensity caused by the presence of interspersed marrow elements.
[ 123/131 I] Metaiodobenzylguanidine (MIBG) scintigraphy, FDG-PET/CT, and DOTA-PET/CT are molecular imaging tests available for the evaluation of adrenal malignancies and neuroendocrine tumors. The clinical utility of molecular imaging includes staging of malignancies (PET/CT) and localization of pheochromocytomas (MIBG). FDG-PET/CT carries a sensitivity and specificity of 100% and 98%, respectively, in differentiation of malignant and benign lesions. FDG-PET/CT should be used in patients in whom a high clinical suspicion of metastatic adrenal cortical carcinoma or another primary cancer with suspected metastasis to the adrenal gland exists. In the setting of an adrenal incidentaloma, this includes patients with a synchronous malignancy, history of previous cancer, and patients with incidentaloma characteristics highly suggestive of adrenal cortical cancer. MIBG scintigraphy carries a sensitivity of 83% to 100% and a specificity of 95% to 100% for pheochromocytomas. MIBG is useful in the localization of paragangliomas, metastatic pheochromocytomas, and in clinical scenarios in which the diagnosis between a nonfunctional pheochromocytoma and malignancy remains uncertain. MIBG is particularly useful in patients with biochemical findings consistent with pheochromocytoma but without evidence of an adrenal lesion on CT. DOTA-PET, which uses a somatostatin receptor imaging agent, carries a sensitivity of 88% for pheochromocytoma, and it can be used in a similar context as MIBG scintigraphy.
There is limited utility in the use of fine-needle aspiration (FNA) biopsy of adrenal lesions, most importantly because it cannot differentiate between benign and malignant disease. If adrenocortical carcinoma is present, a biopsy can cause peritoneal seeding of tumor along the tumor tract. Finally, in the case of pheochromocytoma, adrenal biopsy can cause a physiologically catastrophic release of catecholamines. Unless there is a clinical concern for metastasis in which pathologic confirmation could affect therapy, biopsy should not be performed.
The biochemical workup of adrenal incidentalomas is critical to assess for functionality, and ultimately for surgical decision making ( Table 2 ). It is imperative to identify or rule out pheochromocytoma, cortisol hypersecretion, or primary hyperaldosteronism before an attempted resection of any adrenal mass as it affects perioperative management, guides subsequent medical therapies, and informs surveillance. Positive screening tests should be followed by confirmatory testing.
| Screening Test | Confirmatory Test | |
|---|---|---|
| Cushing’s syndrome |
|
Random ACTH level (low/suppressed) |
| Pheochromocytoma | Plasma free metanephrines and normetanephrines (≥2–4 × upper limit of normal) | 24-hour urine fractionated metanephrines and normetanephrines (≥2–4 × upper limit of normal) |
| Primary hyperaldosteronism | Serum aldosterone (PA) >15 and serum aldosterone-to-renin ratio (ARR) ≥ 20 |
Saline suppression test (PA ≥ 10 ng/dL) Salt load test (elevated aldosterone) ** Adrenal vein sampling |
* Levels in parentheses indicate a positive result indicative of a functional tumor.
** Adrenal vein sampling is used to lateralize an aldosteronoma. ACTH, Adrenocorticotropic hormone.
All patients with an adrenal incidentaloma should be screened for autonomous cortisol secretion. Screening tests include a low-dose dexamethasone suppression test, midnight salivary cortisol × 2, or urine free cortisol × 2. A 1-mg dexamethasone suppression test involves administering 1 mg dexamethasone at 11:00 pm the night before and drawing blood between 8:00 am and 9:00 am the following morning for cortisol and dexamethasone levels. A cortisol level of >1.8 μg/dL is 90.4% to 100% sensitive and 51.8% to 100% specific for autonomous cortisol secretion. Dexamethasone levels are included to confirm proper administration. If results of the dexamethasone suppression test, 24-hour urine cortisol, or midnight salivary cortisol are abnormal, the patient can undergo further confirmatory testing with an adrenocorticotropic hormone (ACTH) level to differentiate ACTH-dependent versus ACTH-independent hypercortisolism. A suppressed ACTH level confirms the diagnosis of autonomous cortisol secretion from the adrenal gland. Although not a routine step in the diagnostic algorithm, low dehydroepiandrosterone sulfate (DHEA-S) concentrations caused by sustained negative feedback on the hypothalamus and pituitary gland can provide further support for the diagnosis.
Measurement of plasma free metanephrines (normetanephrines and metanephrines) is the most sensitive test tool to rule out pheochromocytoma. Several physiologic states and medications, such as hypertension, exercise, trauma, amphetamines, antidepressants, and β-blockers can elevate metanephrine levels, leading to false positives. However, they do not typically cause levels as high as two times to four times the laboratory upper normal limit, which are considered diagnostic. If plasma metanephrines are equivocal or mildly elevated, a diagnosis can be confirmed by 24-hour collection of fractionated metanephrines, which carry a higher specificity.
Hyperaldosteronism is less commonly diagnosed from an adrenal incidentaloma, as aldosterone-secreting adenomas tend to be smaller. Concurrent diagnosis of hypertension must be present in patients with an adrenal incidentaloma. Only 9% to 37% of patients present with hypokalemia. Screening involves measurement of aldosterone and renin levels, and computation of the aldosterone-to-renin ratio (ARR). An ARR >20 in the absence of mineralocorticoid antagonists such as spironolactone encourages further investigation. The diagnosis of hyperaldosteronism can be confirmed with aldosterone suppression testing including a saline infusion test or a 24-hour urinary aldosterone test while the patient maintains a high-sodium diet. All mineralocorticoid antagonists should be stopped for at least 6 weeks before confirmatory testing. Once confirmed, surgical candidates older than 35 years of age who wish to pursue surgery must undergo adrenal venous sampling (AVS) to localize the hypersecreting gland. AVS can also be obtained routinely in all patients with hyperaldosteronism in the setting of an adrenal nodule.
Screening for sex hormones should only be performed for patients with clinical signs or symptoms of virilization or feminization, or those with masses suspicious for adrenocortical carcinoma. Screening is performed by measuring plasma DHEA-S levels. For men exhibiting features of feminization, estradiol-17 can be measured.
Treatment should be individualized according to the patient’s comorbidities, preferences, and the clinical expertise available. Surgery is indicated for functional adrenal tumors and adrenocortical carcinoma in the absence of distant metastatic disease. Surgery may be indicated for indeterminate nodules and large, benign, or symptomatic tumors as well ( Table 3 ).
| Characteristics | Likely Benign | Likely Malignant |
|---|---|---|
| Size | <4 cm | ≥4 cm |
| Margins | Smooth | Irregular |
| Heterogeneity | Absent | Present |
| Rate of growth | <1 cm/year | ≥1 cm/year |
| CT Density (HU) | ||
| Unenhanced density | <10 HU | ≥10 HU |
| Contrast-enhanced density | Low | High |
| Washout | >60% | <60% |
| Features on MRI | ||
| T2-weighted intensity | Isointense/hypointense | Hyperintense |
| Out-of-phase imaging | Loss of signal intensity | No change |
| FDG avidity on PET | No | Yes |
Adrenalectomy can be performed with an open or minimally invasive technique (laparoscopic versus robotic) and with either a transabdominal or retroperitoneal approach. Open surgery should be performed in cases of suspected adrenocortical carcinoma or large tumor size. An open approach for suspected adrenocortical carcinoma allows for optimal exposure of surrounding structures should the need arise for more extensive dissection or vascular reconstruction. For presumed benign lesions, minimally invasive techniques, such as laparoscopic or robotic, can be used and choice of technique is a matter of surgeon preference, as there is no additional benefit to robotic-assisted adrenalectomy. The adrenal glands can be approached transabdominally in the lateral decubitus position and retroperitoneoscopically in a modified prone position. A retroperitoneal approach can be particularly useful in patients with a history of multiple abdominal surgeries to avoid the risks of extensive adhesiolysis and in those who need a bilateral adrenalectomy.
All adrenalectomy patients should receive chemical deep venous thrombosis prophylaxis. Additionally, each type of functional tumor requires specific considerations in the perioperative period.
Patients with Cushing’s syndrome must undergo optimization of blood pressure and glucose control. They may require stress doses of glucocorticoids well into the postoperative period resulting from adrenal insufficiency of the remaining contralateral gland. Preoperative antihypertensives and diabetic medications can slowly and judiciously be adjusted as the patient’s blood pressure and glucose trend demand. Patients eventually may not require some or even all of their previous medications.
Patients with pheochromocytomas carry an elevated risk of catastrophic perioperative hemodynamic lability. These patients require preoperative nonselective alpha blockade with agents such as doxazosin or phenoxybenzamine to reduce the risk of a hypertensive crisis for approximately 2 weeks. After alpha blockade is established, beta blockade may be added for patients with tachycardia. Anesthesia plays a critical role in the care of patients intraoperatively; therefore excellent communication with the anesthesia team regarding vascular access, hemodynamic monitoring, and critical steps during surgery is key for optimal patient outcomes. All patients with a pheochromocytoma should be offered genetic testing to include NF1, VHL, RET, SDHB, and SDHD mutations. Genetic mutations have been reported in 19% of patients with apparently sporadic pheochromocytomas and a negative family history, and the prevalence is much higher in families with known syndromes. Postoperative hypotension may occur; it is transient and more often seen in those treated with phenoxybenzamine preoperatively because of its residual longer-acting alpha-blocking effects in the setting of sudden excess catecholamine withdrawal. Hypotension can be managed with fluid boluses and vasopressors should it persist despite adequate fluid resuscitation. Rebound hypoglycemia is rare but can occur immediately postoperatively due to preoperative suppression of alpha and beta pancreatic cell function from high catecholamine levels that are no longer present after adrenalectomy.
Patients with aldosteronomas should discontinue any potassium supplements and potassium-sparing diuretics, such as spironolactone, immediately postoperatively. Rebound hyperkalemia is a risk in the postoperative period. Potassium-containing crystalloid solutions and potassium repletion should be avoided. Similar to patients with hypercortisolism, preoperative antihypertensives can be resumed cautiously.
Patients with bilateral pheochromocytomas require bilateral adrenalectomy. The bilateral adrenal glands can be resected during the same anesthetic event. Because pheochromocytomas arise from the adrenal medulla, bilateral cortical-sparing adrenalectomy can be performed to minimize life-long adrenal insufficiency. Ideally, a remnant of adrenal cortical tissue around the adrenal vein should be spared. The management of patients with hypercortisolism is more controversial. Options include: bilateral adrenalectomy, AVS to identify and then excise the more biochemically active gland, or removal of the larger gland. The latter two options require close postoperative surveillance as hypercortisolism can recur requiring contralateral adrenalectomy. Patients with hyperaldosteronism caused by bilateral adrenal hyperplasia are not surgical candidates and should be managed medically.
Both nonoperative and postoperative patients require long-term surveillance, and management varies according to the type of adrenal lesion.
After adrenalectomy for pheochromocytoma, patients require lifelong follow-up as pheochromocytomas can recur. Surveillance entails annual history and physical examination with particular attention paid to blood pressure, heart rate, and resurgence of symptoms of catecholamine excess. Plasma metanephrines can be assessed periodically, and a rising trend should be evaluated further with cross-sectional imaging.
Patients undergoing adrenalectomy for Cushing’s syndrome require short-term glucocorticoid supplementation until normal function of the hypothalamic-pituitary-adrenal (HPA) axis is restored. In the first year after surgery, they should be followed closely by an endocrinologist as they may require frequent medication adjustments because of weight loss, lower blood pressure, and lower glucose levels. Cushing’s syndrome may recur, especially in patients status post–unilateral adrenalectomy in the setting of bilateral nodules, and these patients should continue to have routine long-term follow-up with particular attention paid to resurgence of hyperglycemia, worsening hypertension, and weight gain. Patients undergoing bilateral adrenalectomy will require life-long physiologic glucocorticoid supplementation as well as education regarding stress doses during illness, injury, and so on.
After adrenalectomy for hyperaldosteronism, it may take several weeks or months before patients can be weaned off some or all of their antihypertensive agents. In some cases, especially if hyperaldosteronism went unrecognized for a prolonged period causing permanent renal changes, patients will remain hypertensive and continue to require all preoperative antihypertensives. Patients should continue to be seen and evaluated by a clinician to address these potential changes.
Standardized guidelines for surveillance after resection of adrenocortical carcinoma do not exist; however, patients require close life-long follow up to monitor for local and metastatic recurrences. Physical examinations, hormonal studies, and CT of the chest and abdomen should be performed every 3 months, which can be increased to every 6 months after a few years. Surveillance can be extended to every 1 to 2 years after a 5-year disease-free interval.
Observation with repeat imaging and biochemical testing is an acceptable alternative for patients with indeterminate, nonfunctional nodules with low suspicion of adrenocortical carcinoma. Repeat imaging should be performed at 3 months, 6 months, and then annually for 1 to 2 years. Biochemical testing should be repeated annually for up to 5 years. Should the tumor grow more than 1 cm or become hormonally active, adrenalectomy is indicated.
The adrenal cortex is the main source of corticosteroid hormone synthesis, producing both mineralocorticoids and glucocorticoids. These hormones help regulate inflammation, stress response, metabolism, catabolism, as well as electrolyte and water balance, depending on the anatomic adrenal layer from which the hormone is produced. Tumors arising from the zona glomerulosa, zona fasciculata, or zona reticularis can produce increased levels of hormones from the involved layer (functional), or they may be hormonally inactive (nonfunctional). Although they are important in the discussion of surgical management of adrenal tumors, tumors arising from the adrenal medulla (pheochromocytoma and neuroblastoma) are discussed elsewhere.
The prevalence of adrenal cortical tumors has been reported at about 2% (ranging 1% to 9%) in autopsy studies, with higher prevalence noted in the older population. Because of the increased use of diagnostic imaging over the past 3 decades in conjunction with advances in each imaging modality, the prevalence of adrenal tumors on computed tomography (CT) imaging has been reported at 4% to 10%. The majority of these tumors are found incidentally.
Less than 2% of adrenal tumors are found to be adrenocortical carcinomas (ACCs); however, despite the rarity of this malignancy (annual incidence of 1 to 2 per 1 million), the diagnosis of ACC carries a 5-year disease-specific survival (DSS) of approximately 50%. Therefore prompt identification and treatment are critical in these patients.
The initial workup of a patient with an adrenal tumor includes a detailed personal history (including a history of malignancy to evaluate for recurrent primary or metastatic disease to the adrenal glands) and a physical examination. A careful family history is important to identify potential underlying genetic syndromes that would require further investigation (multiple endocrine neoplasia, familial paraganglioma syndrome, von Hippel-Lindau disease, neurofibromatosis type 1, Carney complex, Li Fraumeni syndrome). The history should focus on the signs and symptoms that may also help identify the specific adrenal hormone(s) being overproduced. A complete biochemical assessment should be performed to evaluate for elevated adrenal hormone levels ( Table 1 ). For adrenal cortical tumors, it is helpful to always send for serum aldosterone, renin, ACTH, and dehydroepiandrosterone sulfate (DHEAS) as well as perform an overnight low-dose dexamethasone suppression test.
| Biochemical Abnormality | Layer | Syndrome | Symptoms | Screening Test | Indications For Further Evaluation |
|---|---|---|---|---|---|
| Hyperaldosteronism | Zona glomerulosa | Conn’s syndrome | Hypertension, weight gain, edema, muscle cramping, weakness, temporary paralysis, headaches, polyuria, polydipsia | Plasma aldosterone, plasma renin activity | Plasma aldosterone to renin activity ratio > 20 |
| Hypercortisolism | Zona fasciculata | Cushing’s syndrome | Hypertension, weight redistribution (central obesity, “buffalo hump,” “moon face”), diabetes, bruising, fatigue, abdominal striae, proximal muscle weakness, hirsutism, amenorrhea, impotence, depression | ACTH, overnight low-dose (1-mg) dexamethasone suppression test | Serum cortisol > 1.8 mcg/dL with suppressed ACTH |
| Increased sex hormones | Zona reticularis | Virilization/ feminization | Hirsutism, menstrual irregularities, precocious puberty, postmenopausal bleeding, feminization (gynecomastia, testicular atrophy) | DHEA-S, +/–androstenedione, 17-hydroxyprogesterone, testosterone (total and free) for women, estradiol for men and postmenopausal women | Abnormally elevated levels with associated clinical symptoms |
Approximately 10% to 15% of adrenal cortical tumors secrete excess hormone and are considered functional adrenal cortical tumors. These are typically referred for adrenalectomy and can be identified via the clinical symptoms and screening tests listed in Table 1 . Nonfunctional tumors with indeterminate initial imaging should have additional adrenal-specific imaging including adrenal protocol computed tomography (CT) with timed contrast and multiphase scanning, magnetic resonance imaging (MRI), and/or fluorodeoxyglucose positron emission tomography (FDG-PET). These imaging modalities can help differentiate between benign adrenal tumors that can be observed and suspicious or malignant tumors that should be resected. ( Table 2 )
| Characteristic | Benign | Indeterminant |
|---|---|---|
| Size | < 4 cm | > 4–6 cm |
| Growth | < 3 mm/year | > 5 mm/year |
| Noncontrast CT (Hounsfield units) | < 10 | > 10 |
| Percentage washout on CT (if homogeneous) | > 50%–60% | < 50%–60% |
| MRI chemical shift | > 50% | < 50% |
| Internal attenuation | Homogeneous | Heterogeneous |
| Calcification | Absent | Present |
| Necrosis | Absent | Present |
| Borders | Regular | Irregular |
| Invasion of adjacent structures | Absent | Present |
| Intravascular tumor thrombus | Absent | Present |
| Lymphadenopathy | Absent | Present |
| FDG PET CT: Tumor SUV | < 3.4 | > 3.4 |
| FDG PET CT: Tumor/liver SUV ratio | < 1.45 | > 1.45 |
Percutaneous fine-needle aspiration (FNA) is not routinely recommended in the preoperative workup of adrenal tumors. Biochemical and imaging findings, in addition to personal/family history and physical examination, are sufficient to make a diagnosis in most cases. Additionally, FNA cytology cannot reliably differentiate a benign adrenal adenoma from adrenal cortical carcinoma (ACC). Finally, adrenal biopsy is associated with multiple risks including malignant seeding of the FNA tract, adrenal hemorrhage that may complicate subsequent adrenalectomy, damage to surrounding organs and vessels, and hypertensive crisis in patients with undiagnosed pheochromocytoma.
There are rare instances in which FNA can be useful, such as for patients in whom surgery is not recommended but a tissue diagnosis will help guide appropriate treatment. For example, tissue diagnosis by FNA may be helpful in the workup of metastatic disease from a non-adrenal primary tumor, an unresectable adrenocortical carcinoma, or for patients who are poor surgical candidates because of multiple medical comorbidities.
In patients with a history of nonadrenal malignancy and a new adrenal lesion, FNA may be useful in determining cell type of origin. However, if the adrenal lesion is the only evident site of metastasis and clinical suspicion is high, adrenalectomy may be performed without adding the risks associated with FNA. Despite the adrenal glands being the fourth most common location for metastatic disease, a series of studies showed that, on average, greater than 50% (range 9%–76%) of adrenal masses found in these patients are benign adrenal adenomas.
Adrenalectomy is recommended if biochemical evaluation and imaging characteristics are consistent with a hormonally active and/or potentially malignant adrenal lesion, and the patient is a reasonable candidate for surgery ( Fig. 1 ). Patients with functional or suspicious-appearing lesions who are not operative candidates may be managed with observation or medical management.
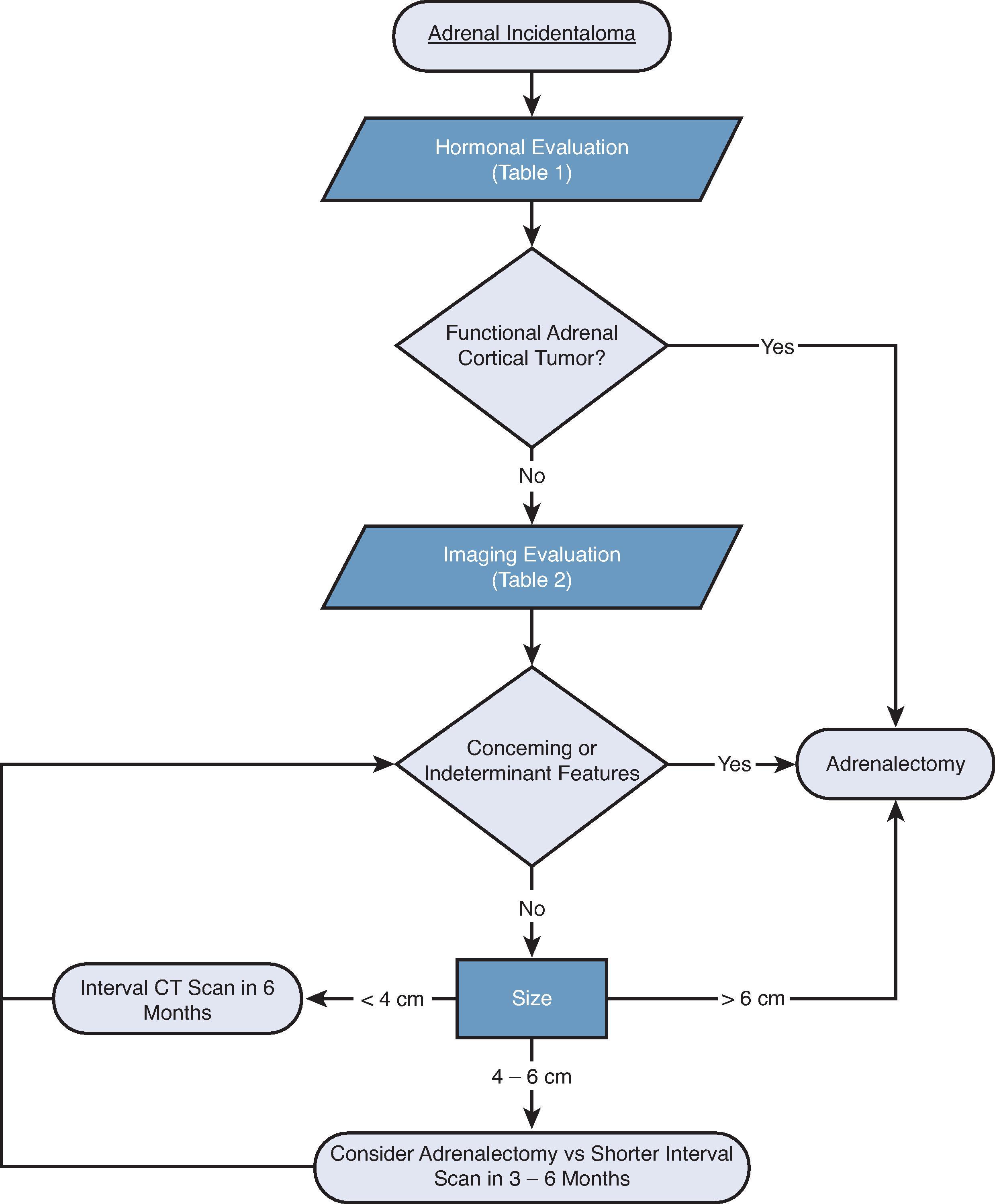
Although tumor size (>4 or 6 cm) has in the past determined the threshold for adrenal surgery in nonfunctional adrenal lesions, imaging characteristics are also necessary to consider in a complete evaluation. Washout of contrast of >50% to 60% on CT or a loss of signal on out-of-phase images on MRI can reliably identify benign adrenal adenomas. The rate of growth in solid adrenal lesions is another important determinant used to help identify benign and malignant tumors; a growth rate of less than 3 mm/year is consistent with an adenoma, compared with malignant tumors that characteristically grow more than 5 mm/year (see Table 2 ). Previous size criteria that guided surgical resection for adrenal tumors 6 cm and greater were determined given the greater risk of a malignancy with larger tumors: > 6 cm carries a 22% risk of malignancy, 4.1 to 6 cm carries a 6% risk of malignancy, and ≤ 4 cm has approximately 2% or less risk of malignancy. On the other hand, benign adrenal masses like myelolipomas and simple adrenal cysts have characteristic imaging features, can grow to large sizes, remain asymptomatic, and do not need to be resected. Benign-appearing tumors can be observed with repeat scans in 3 to 6 months and then annually. Biochemical testing should be repeated annually for 5 years to monitor for any changes in hormonal activity.
Functional adrenal tumors, especially in patients with worsening symptoms, should undergo adrenalectomy to remove the source of excess hormone production. The rate of malignant transformation of functional tumors is unknown, with only a few case reports identifying this rare situation.
Nonfunctional, stable, benign-appearing adenomas do not need surgical resection. Nonfunctional adenomas that are continually growing or with indeterminant radiographic features should be reevaluated for resection with the surveillance schedule mentioned previously. ( Fig. 2A ; see Table 2 ). It is important to note that adenomas are not thought to be premalignant lesions and do not seem to have the potential for malignant transformation.
Complete surgical resection is the only potential curative treatment for ACC. It is typically recommended to be done via an open approach, regardless of size and functional status. As further explained later in this chapter, this is done via en-bloc open resection for a decreased rate of recurrence compared with the laparoscopic approach (see Fig. 2B ).
Lung cancer, breast cancer, melanoma, gastrointestinal malignancies, and renal cell carcinoma are the most common primary malignancies that metastasize to the adrenal glands. Approximately 50% of these tumors metastasize to bilateral adrenal glands. Isolated adrenal metastases should be surgically resected in appropriate patients if feasible and can be done so safely via a laparoscopic approach unless characteristics of more aggressive disease are present (e.g., lymphadenopathy, invasion into surrounding organs or vessels), at which time open adrenalectomy is recommended.
There have been very few studies looking at neoadjuvant systemic therapy for ACC, and although they have suggested a potential benefit in patients with borderline resectable ACC, the studies were not randomized controlled trials, and they lacked power; therefore there is no clear consensus on neoadjuvant therapy from the various society guidelines.
It is critical to avoid capsular rupture when handling an ACC as they have a high propensity to seed the entire field, increasing local recurrence rates. For large or invasive ACCs that involve the inferior vena cava (IVC), liver, diaphragm, spleen, or kidneys, a multidisciplinary team approach at a regional center where there is adequate expertise should be considered, and resection should be planned in conjunction with hepatic, vascular, and/or cardiothoracic surgeons as needed. For tumors with extensive IVC involvement, vascular bypass, IVC resection and repair or replacement may be necessary.
Determining the optimal surgical approach for adrenalectomy should be done on a case-by-case basis, and certain factors may lead to choosing one approach over the other ( Table 3 ). In general, although minimally invasive laparoscopic and retroperitoneoscopic approaches have replaced traditional open adrenalectomy for most presumed benign adrenal tumors, the debate over open versus minimally invasive surgery for ACC continues to evolve. The American Association of Clinical Endocrinologists and the American Association of Endocrine Surgeons currently recommend open adrenalectomy for ACC, regardless of size, per their most recent guidelines (published in 2009). The National Comprehensive Cancer Network (NCCN) agrees with these recommendations in their guidelines (Version 1.2021 for Neuroendocrine and Adrenal Tumors), yet also acknowledges that depending on individual surgeon expertise and tumor characteristics, a minimally invasive approach may be used for small tumors, with a plan for converting to an open procedure if local invasion is noted. This indicates that despite their recommendation for open resection of ACC, there is still debate within the guidelines. In contrast, the European Society of Endocrine Surgeons recommends that laparoscopic adrenalectomy may be attempted for stage I or II ACCs < 10 cm.
| Adrenalectomy | Open | Laparoscopic | Retroperitoneoscopic | Robotic | Single-Port |
|---|---|---|---|---|---|
| Advantages | Tactile feedback | Familiar anatomic orientation to open approach | Avoids peritoneal cavity | Increased dexterity | Excellent cosmesis |
| Better exposure | Decreased postoperative pain | Shorter distance of dissection | Three-dimensional depth perception | Similar results to laparoscopic | |
| Decreased local recurrence of ACC with en bloc resection | Decreased hospital stay | Can do bilateral without patient repositioning | Good for obese patients | ||
| Appropriate for any tumor size | Better cosmesis | Prior abdominal surgery does not hinder this approach | Appropriate for larger tumors (even in RP given dexterity) | ||
| Can utilize a single midline incision for bilateral adrenalectomy | Shorter operative time compared with robotic/single-port | ||||
| Quicker management if catastrophic injury occurs | Appropriate for larger tumors | ||||
| No CO 2 insufflation for preload-dependent patients | Good for obese patients | ||||
| Good for obese patients | |||||
| Disadvantages | Larger incision | Risk of iatrogenic injury to intraabdominal organs | Requires increased insufflation pressure to maintain exposure | Increased cost | Learning curve |
| Increased postoperative pain | Increased manipulation of additional organs | Restricted exposure to retroperitoneum | Longer operative time | ||
| Increased hospital stay | Difficult in patients with prior abdominal surgery | Difficult in obese patients | |||
| Increased risk for hernia | Requires patient repositioning for bilateral adrenalectomy | Difficult with larger tumors | |||
| Risk of iatrogenic injury to intraabdominal organs | Patient must tolerate prone positioning | ||||
| Difficult in patients with prior abdominal surgery |
Benign adrenal masses, both functional and nonfunctional, can be safely approached using a minimally invasive approach, barring any intraoperative concern for malignancy, at which point the procedure should be converted to open. Benign tumors 10 cm or greater can pose technical challenges to minimally invasive resection; however, tumor size alone is not an absolute contraindication to a minimally invasive approach.
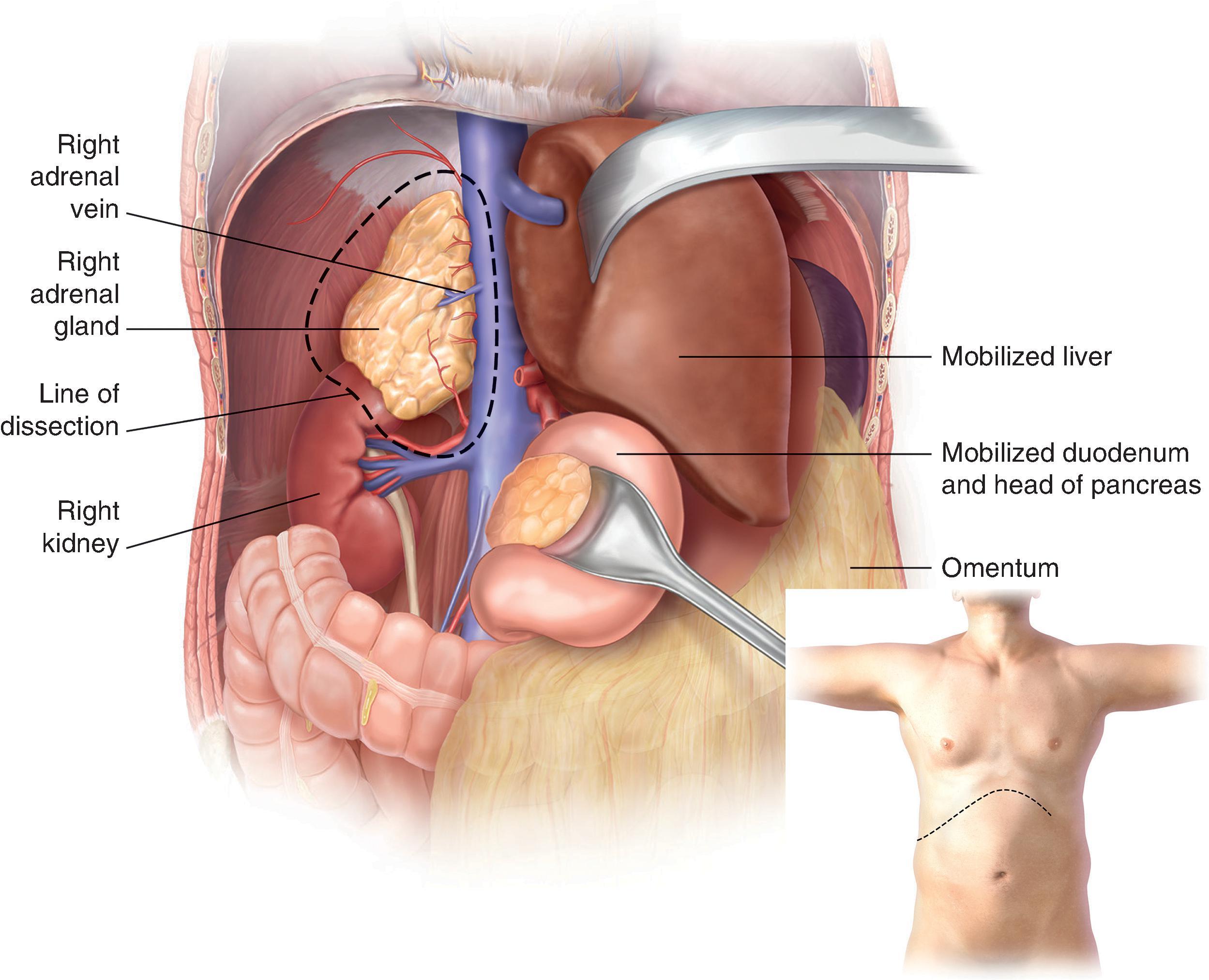
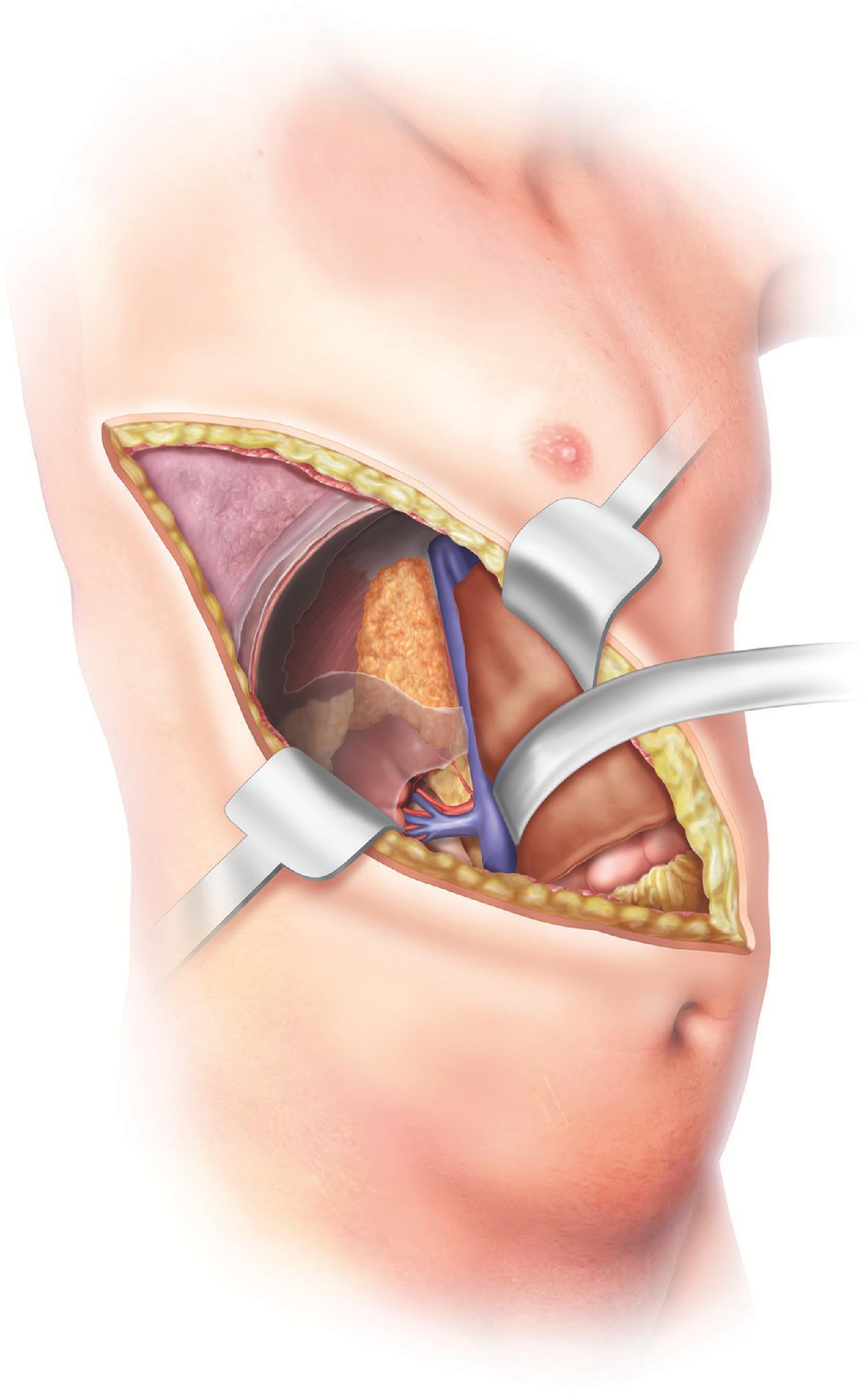
The patient is placed in the supine position with a wedge or bump to elevate and separate the pelvis from the costal margin on the right side. The initial steps involve making a long right subcostal or bilateral subcostal incision and then examining the peritoneal cavity to assess for distant metastases, lymph node involvement, and fixation of the mass to the surrounding tissues. The liver is mobilized by dividing the falciform and right lateral triangular ligaments, allowing for retraction of the liver upward. The hepatic flexure of the colon is mobilized by dividing the lateral gastrocolic ligament and exposing the IVC via the Kocher maneuver. Upon entering the Gerota fascia along the IVC, the adrenal gland can now be examined ( Fig. 3 ). If the adrenal mass does not involve the upper pole of the kidney, the Gerota fascia is opened, and all perinephric fat below the adrenal gland is mobilized upward with the adrenal gland en-bloc to ensure an adequate margin. If invasion into the kidney or renal vessels is found, continuation of the mobilization with en-bloc partial or total resection of the right kidney as needed to ensure capsule integrity and minimize the chance of tumor spillage. If there is IVC invasion, the IVC must be secured superiorly under the diaphragm at the crus as well as distally to the right renal vein inferiorly. If there is invasion into the liver, a limited right hepatectomy may be performed, typically using a multidisciplinary approach. The superior dissection can extend up to the diaphragm, and the inferior dissection should include all perinephric fat over the superior pole of the kidney to ensure an adequate margin. The medial dissection is done very delicately as it is just anterior to the vena cava, and the right adrenal vein is very fragile. The medial attachments are then mobilized and ligated with the right adrenal vein being last, including all fatty tissue medial to the gland and lateral to the IVC. If bleeding does occur after ligation of the right adrenal vein, the surgeon can obtain proximal and distal control of the vena cava and repair the stump primarily via suture ligation.
The patient is positioned in the same way as described earlier for the open right adrenalectomy resection, with a bump/wedge placed under the left side. A left-sided or bilateral subcostal incision is made, and the abdomen is examined in similar fashion. The gastrocolic ligament is divided to mobilize the splenic flexure of the colon distally. The spleen and tail of the pancreas are mobilized medially to expose the kidney and adrenal gland (see Fig. 3 ). For tumors that surround or invade the splenic vessels, a simultaneous splenectomy is performed, dividing the splenic artery at the upper border of the pancreas and the individual gastrosplenic arteries, while preserving the vessels along the greater curvature of the stomach. The tail of the pancreas is usually preserved unless directly invaded. If the left kidney is involved with the adrenal tumor, the kidney is mobilized from lateral to medial from the lower pole and removed en-bloc with the adrenal tumor.
If there is no invasion into surrounding structures, in contrast with a right adrenalectomy, the left adrenal vein is ligated as an earlier step along with the inferior adrenal arteries, to aid in mobilization of the adrenal gland. The superior and inferior arteries are controlled and ligated, and the adrenal gland is dissected along with all surrounding perinephric fat off the superior pole of the kidney to ensure adequate margins.
The patient is placed in the prone position with the patient’s arms lifted over his or her head. An incision is made over the 12th rib, and the rib is resected for exposure. The peritoneum is swept inferiorly and laterally, and the retroperitoneal organs are identified and mobilized, with the kidney being retracted inferiorly. The adrenal gland is identified medially and superior to the renal hilum, and adrenalectomy is completed in a similar fashion to the anterior approach. Open posterior adrenalectomy can be complicated by subcostal nerve injury and lumbar hernias.
The patient is placed in the supine position, with the upper torso rotated laterally approximately 45 degrees with the help of a bump. The incision is made in either the 8th or 9th intercostal space, and the diaphragm is divided peripherally near the costal margin to avoid injury to the phrenic nerve and to allow for easy reconstruction at the end of the case. Once the abdominal cavity is opened, the adrenal glands are exposed and ultimately resected in the same manner as the anterior approach ( Fig. 4 ).
At the end of the case, the diaphragm should be repaired with interrupted nonabsorbable sutures in a figure-of-8 fashion, and a chest tube is placed to water seal.
Patient anatomy, past surgical history, underlying pathology, and the surgeon’s expertise should be considered when determining the appropriate minimally invasive surgical approach to adrenalectomy. A summary of such considerations can be found in Table 3 . For instance, patients who have had extensive abdominal surgery or require bilateral adrenalectomy will likely benefit from a retroperitoneoscopic approach as this approach avoids the peritoneal cavity and does not require repositioning for bilateral procedures, whereas patients with tumors > 6 cm may benefit from a transabdominal laparoscopic approach given the larger working area. Patients with high body mass index (BMI) or extensive retroperitoneal fat (both are particularly difficult in male patients) may be better suited for the laparoscopic approach.
The patient is placed in the left lateral decubitus position, with the bed flexed just above the waist and the kidney rest elevated, to allow for maximum exposure to the flank. The patient is secured in place with a beanbag, and the right arm is supported on an upper arm rest. A gel roll is placed under the left axilla, and the head is supported.
Four ports are typically used for the right-sided approach ( Fig. 5A ). The most medial port, placed in the epigastric/subxiphoid region, is used to retract the liver superiorly. The two other working ports are placed approximately 6 to 10 cm apart along the subcostal margin, with the most lateral in the midaxillary line, to allow for the unobstructed use of the instruments. The final port, for the camera, is placed in the right upper quadrant. These locations should be marked before insufflation as a guide, but after placing the first port (the camera port via an open technique) and insufflating the abdomen to 15 mm Hg with CO 2 , the other ports should be placed under direct visualization. Before placing the most lateral port, the surgeon should examine the hepatic flexure to determine if mobilization is needed (rare). The liver is mobilized by dividing the right triangular ligament, and use of a liver triangular retractor (or fan retractor) through the medial subxiphoid port aids in retracting the liver superiorly and medially. The use of a harmonic scalpel or hook electrocautery allows the opening of the retroperitoneal fascia between the inferior edge of the liver and the superior margin of the adrenal gland. It is helpful to work from medial to lateral and avoid the adrenal vein at the superomedial aspect of the gland. At this point, the surgeon should look for and carefully divide any accessory veins between the adrenal and liver or IVC. In general, for any rotation or movement of the adrenal gland, blunt instruments like a closed grasper or laparoscopic peanut should be used, or, alternatively, grasp the periadrenal fat to avoid disrupting the adrenal capsule. Next, mobilization is performed along the medial edge of the adrenal gland to open the Gerota fascia in a longitudinal fashion from inferior to superior and identify the lateral edge of the IVC. The perinephric fat is dissected off while leaving a small layer on the adrenal gland for margin. The surgeon continues to carefully develop the space between the kidney, adrenal gland, and IVC while working superiorly toward the adrenal vein. The adrenal vein is divided using a harmonic scalpel or clips. The harmonic scalpel is then used to dissect the inferior aspect of the adrenal gland off the superior pole of the kidney, while dividing the small inferior adrenal arteries. At this point, attention is paid to protect any accessory renal artery branches. The dissection continues to remove all periadrenal fat with the gland, working from a medial to lateral fashion until the adrenal gland is taken off the diaphragm posteriorly and laterally. The adrenal gland can then be removed in a retrieval bag via the camera port. The port site can be enlarged to remove the tumor in an intact fashion, and then all port sites are closed in the normal fashion.
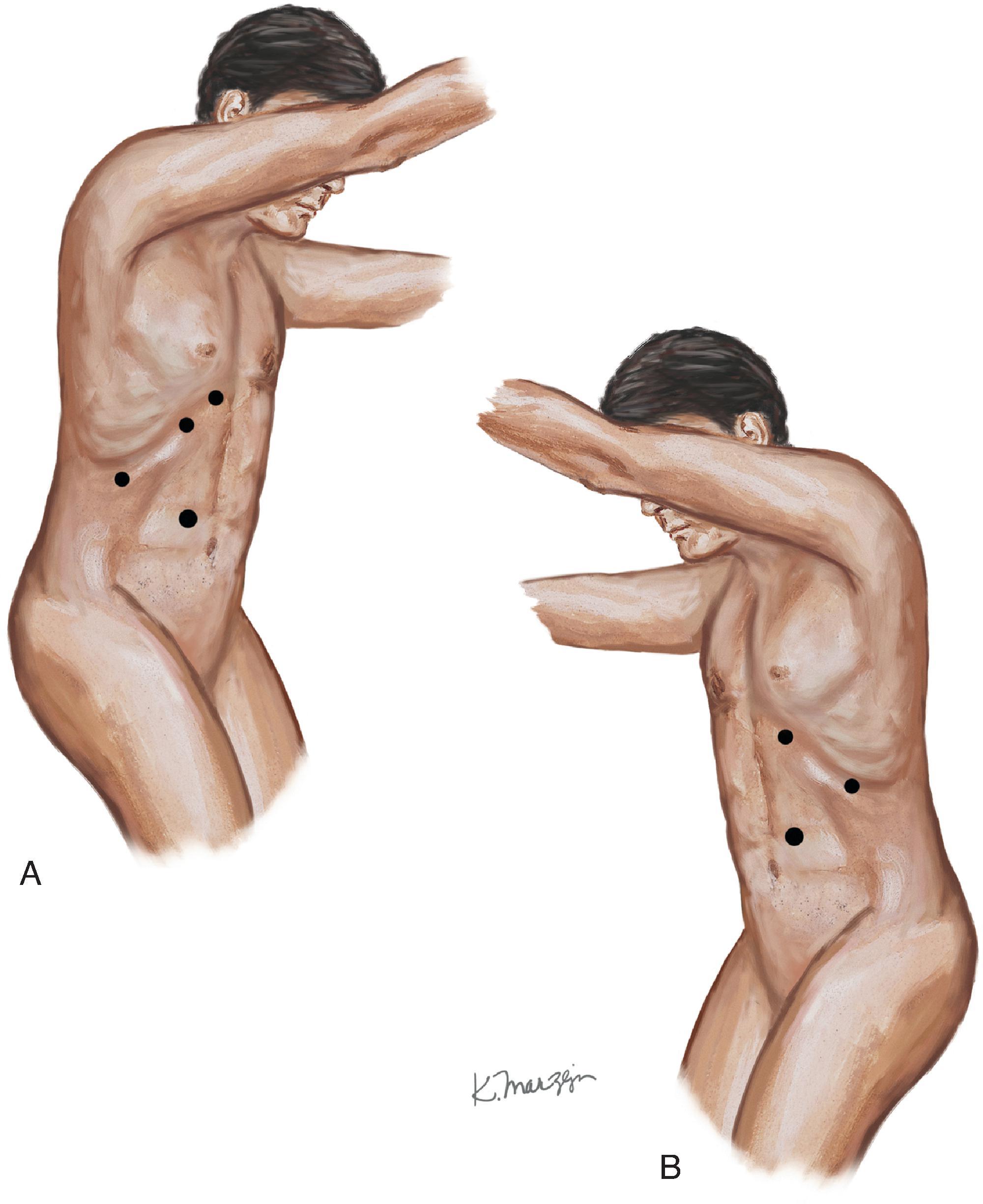
The patient is positioned and port placements are mirrored to those of a laparoscopic right transabdominal adrenalectomy, with the only difference being that the subxiphoid port can be used if needed to rotate the spleen (see Fig. 5B ). The splenocolic ligaments are divided, and the splenic flexure is mobilized while retracting the colon inferiorly. The spleen is then mobilized along its lateral aspect from an inferior to superior dissection, continuing all the way superiorly to the fundus of the stomach. The spleen is then retracted medially with the tail of the pancreas.
The Gerota fascia is then opened medially to identify the kidney and the adrenal gland. The dissection then continues along the superomedial edge of the adrenal gland, sweeping the gland off the diaphragm posteriorly. The phrenic vein is identified and traced down from the diaphragm to the left adrenal vein and then to the location where the left adrenal vein enters the left renal vein. The adrenal vein is then ligated and divided using a harmonic scalpel or clips. The adrenal gland is elevated superiorly, and the small inferior adrenal arteries are then divided while mobilizing all periadrenal fat off the superior medial aspect of the kidney while working from a medial to lateral fashion. The superior attachments are then divided, and the adrenal gland is removed using a retrieval bag placed through the camera port. This port site may be enlarged as necessary to remove the gland intact, and the ports can then be closed in the normal fashion.
The patient is placed in the prone position with the abdomen resting in an open rectangular support, and the hips and knees flexed and supported at 90 degrees ( Fig. 6 ). The retroperitoneal space is accessed via a 1.5-cm incision just under the tip of the 12th rib using sharp and blunt dissection. With a finger inserted into the retroperitoneum and lifting up on the 11th rib, a 5-mm trocar is placed just below the tip of the 11th rib. CO 2 insufflation to 20 to 25 mm Hg is initiated through a blunt trocar with an inflatable balloon. A 10-mm trocar is then placed medially under direct vision to avoid the subcostal nerve, about halfway between the spine and the initial incision. The retroperitoneum is then examined with a 30-degree camera, and the area is bluntly dissected to expose the diaphragm, spine, and top of the kidney. Next, the adrenal gland is mobilized inferiorly first, elevating it superiorly away from the kidney. Definitive identification of the vena cava is critical as the pressure from the insufflation may likely cause compression of this structure. The inferior adrenal arteries are divided along the IVC, working clockwise or lateral to medial, from 3:00 to 9:00 superiorly toward the adrenal vein at the superomedial border. Once the adrenal vein is divided, the remainder of the adrenal attachments are dissected free.
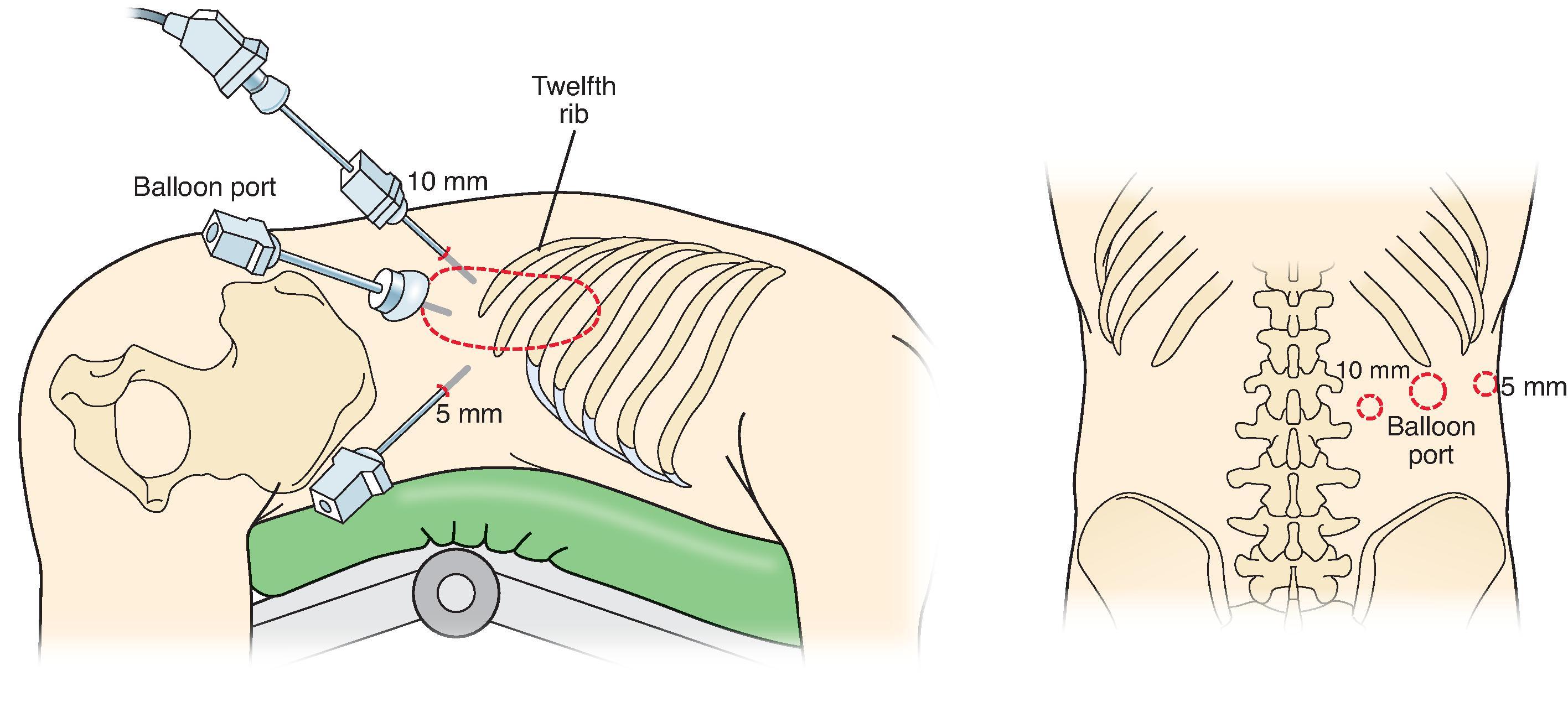
Throughout the dissection, be careful not to enter the peritoneal cavity as the hole will lead to loss of pneumoretroperitoneum and exposure. The adrenal gland is extracted through the central incision using a retrieval bag, and the ports are closed in the normal fashion.
A similar approach is taken for a left retroperitoneoscopic adrenalectomy. Here, the adrenal gland is often anterior and medial to the superior pole of the kidney, requiring more extensive mobilization of the superior pole to expose it. Dissection of the adrenal gland is again approached from its inferior aspect, working counterclockwise or lateral to medial, from 9:00 to 3:00, to expose the adrenal vein medially. The superior attachments are removed last, and the adrenal gland is extracted as described earlier.
The single-port approach is a newer technique that has been gaining more traction over the past decade. The initial studies comparing the single-port and the traditional laparoscopic approaches showed no significant difference in complications, but significantly increased operative times, decreased hospital stay, and decreased postoperative pain. However, newer studies have since shown that with continued refinement, all variables showed no significant differences between the two techniques, proving that a single-port adrenalectomy is a feasible option in the skilled surgeon that results in a single incision with excellent cosmesis.
This approach can be done in either a transabdominal or retroperitoneal fashion. A transabdominal approach utilizes a 2- to 3-cm periumbilical incision, a specialty multiport device, and extra-long instruments to reach the adrenal glands. A right-sided adrenalectomy may also utilize an additional 2-mm needlescopic port for liver retraction.
This technique is essentially the same as the retroperitoneoscopic approach; however, a 2- to 3-cm incision below the 12th rib is used in conjunction with a specialty multiport device allowing for a total of three ports. The space is then insufflated to 30 mm Hg, and using a blunt dissector and a LigaSure, the remainder of the operation is similar as noted earlier. The difficulty with this approach is the minimal maneuverability caused by the smaller retroperitoneal space and the close location of the ports to fit into the single 2- to 3-cm incision. An extended mobilization of the upper pole of the kidney may be required compared with the standard retroperitoneoscopic approach.
Studies have shown that this is a safe and feasible approach when done by an experienced surgeon; however, there is no significant advantage in outcomes compared with the standard laparoscopic approaches other than cosmesis.
A computer-assisted surgical device (robot) can be used in both minimally invasive transabdominal and retroperitoneal approaches. Since the first robotic adrenalectomy was described in 2001, perioperative outcomes and average length of hospitalization are decreased compared with open adrenalectomy and are comparable to laparoscopic adrenalectomy—albeit at an increased cost. Meta-analyses have shown that there are no differences in intraoperative and postoperative morbidity and mortality between the robotic and laparoscopic approaches. Operative times are increased in the robotic approach but appear to decrease as a surgeon’s experience moves past the learning curve. Conversely, multiple studies have shown that robotic adrenalectomies actually have shorter operative times in patients with tumors > 6 cm. The main advantages of the robotic approach are the added dexterity in a small working space and the three-dimensional visualization of the operative field.
In placing the robotic trocars, special consideration is needed to avoid having the robotic arms hit each other throughout the case. The optical trocar is inserted first 2 cm beneath the costal margin at the anterior axillary line under direct visualization, and the abdomen is then insufflated. Two 8-mm trocars are placed a few centimeters away, equidistant, on either side of the optical trocar and 2 cm below the costal margin. For a left adrenalectomy, a single 5-mm trocar is placed a few centimeters medial to the most medial robotic trocar for the bedside assistant to utilize, yet still along the same line connecting the other ports to facilitate a fast and easy conversion to an open procedure if needed. For a right adrenalectomy, the same 5-mm port is placed medially, but an additional 10-mm port can be placed inferiorly along between the mid and anterior axillary lines to utilize a fan liver retractor. The robot is then docked, approaching the patient from behind and over his or her shoulder. The reminder of the procedure is performed similar to the laparoscopic approach described earlier.
As discussed previously, obtaining a family history is crucial in helping to identify a genetic link to adrenocortical tumors. ACCs, for example, are thought to be hereditary in 50% of cases. Some genetic mutations driving sporadic ACC are also found as the driving mutation for some familial tumor syndromes that are characterized by including ACC such as Li Fraumeni syndrome ( TP53 mutation), Beckwith-Wiedemann syndrome ( IGF2 mutation), and Gardner syndrome ( APC mutation). Mutations in the tumor suppressor gene ARMC5 have identified families with an autosomal dominant inheritance type of Cushing’s syndrome (primary bilateral macronodular adrenal hyperplasia), and activating mutations in GNAS1 caused adrenocortical tumors in infants with McCune-Albright syndrome via elevated cAMP signaling. While most benign cortisol-producing tumors are related to elevated cAMP signaling, the drug mitotane has been found to inhibit basal and cAMP-induced cortisol secretion in ACC. Additional genetic syndromes that were associated with adrenal neoplasia or hyperplasia include Carney complex (PRKAR1A) and MEN 1 (Menin) ( Table 4 ). If any of these mutations are identified, further workup is indicated with a referral to the appropriate specialty.
| Condition | Gene | Adrenal Disease | Associated Nonadrenal Disease | Important Next Steps |
|---|---|---|---|---|
| Beckwith-Wiedemann syndrome | IGF2 | ACC, adrenal hyperfunction, and hyperplasia | Embryonal tumors, macroglossia, omphalocele, renal abnormalities, visceromegaly/hemihypertrophy | Genetic testing |
| Carney complex | PRKAR1A | Primary pigmented nodular adrenocortical disease | Heart and skin myxomas, skin hyperpigmentation | Cardiac evaluation |
| Gardner syndrome | APC | Adrenal adenomas | Desmoid tumors, epidermoid cysts, fibromas, gastrointestinal polyps, lipomas, osteomas, periampullary carcinoma, thyroid cancer | Annual thyroid evaluation, colonic screening, genetic testing, family history |
| Li Fraumeni syndrome | TP53 | ACC | Breast cancer, leukemia, sarcoma | Family history, genetic testing, associated cancer screening |
| McCune-Albright syndrome | GNAS1 | Bilateral adrenal hyperplasia, Cushing’s syndrome | Fibrous dysplasia, skin hyperpigmentation (near midline) | Endocrinology consult, orthopedic consult |
| Multiple endocrine neoplasia, type 1 | Menin | Adrenal adenomas, ACC | Pituitary tumors (prolactinomas, growth hormone–secreting), pancreatic tumors (gastrinomas, insulinomas), parathyroid tumors | Endocrinology and endocrine surgery consults |
| Primary bilateral macro-nodular adrenal hyperplasia | ARMC5 | Hyperplasia, Cushing’s syndrome | — | Endocrinology and endocrine surgery consults |
Cortisol-secreting tumors are often associated with diabetes, and blood glucose levels must be monitored closely in the perioperative setting. ACCs commonly secrete cortisol; therefore, like other cortisol-secreting tumors, resection can be associated with perioperative and postoperative adrenal insufficiency (e.g., hypotension, tachycardia, hypoxia, fever). Patients with cortisol-producing tumors often require perioperative stress-dose steroids with a long postoperative taper often managed in conjunction with endocrinology. Additionally, cortisol-producing ACCs have been shown in several studies to be an independent risk factor for poorer prognosis compared with non–cortisol-producing ACCs, and these patients should be closely monitored.
Patients with aldosteronomas are usually managed with different types of hypertension medications such as calcium channel blockers (nifedipine), mineralocorticoid antagonists (spironolactone), ACE inhibitors (lisinopril), and/or angiotensin receptor blockers (losartan). These must be weaned off so as to not cause hypotension in the postoperative period. Spironolactone should be started preoperatively for blood pressure control as it also gives an idea as to the estimated blood pressure the patient will obtain postoperatively. Patients are also generally on potassium supplementation before surgery, and this should be stopped immediately preoperatively.
It is important to note the final pathology for ACC not only to ensure that a complete oncologic resection was achieved, but also to identify the Ki67 Labeling Index (Ki67 LI) status of the tumor. Of all clinicopathologic variables, the Ki67 LI status is the most important prognostic factor in regard to recurrence and survival—even in patients with a complete oncologic resection. Ki67 is a proliferation marker identified in an ACC sample by immunohistochemistry, and tumors that have a lower percent of Ki67 staining (<10%) have decreased risk of recurrence and increased survival compared with patients with higher levels of Ki67. Therefore patients with an ACC that has ≥10% of its cells staining of Ki67, despite an R0 resection, may need additional adjuvant therapies.
After resection of an ACC, adjuvant treatment with mitotane and/or external-beam radiation therapy may be utilized to potentially improve cure rates and increase survival in high-risk patients for local recurrence. Chemotherapy treatment, with or without mitotane, has not been well studied for treating ACC in the adjuvant setting; however, a cisplatin-based regimen has been suggested with mitotane treatment for high-risk patients. The European Society of Endocrinology and the European Network for the Study of Adrenal Tumors (ENS@T) recommend the use of adjuvant mitotane in high-risk patients. Retrospective studies have differed on their results as to if adjuvant mitotane improves overall survival; however, multiple prospective randomized clinical trials are currently in process looking at mitotane with or without additional chemotherapeutic agents in the adjuvant setting, and their results will be crucial for this fatal disease.
Pheochromocytomas and paragangliomas (PPGLs) are rare tumors that arise from chromaffin cells, and they have the potential to produce, accumulate, and/or secrete catecholamines (epinephrine, norepinephrine, dopamine) and metabolites of catecholamines. Approximately 80% to 85% of these tumors arise from the adrenal medulla and are referred to as pheochromocytomas. Conversely, paragangliomas make up the remaining 15% to 20% of chromaffin neoplasms that arise from outside the adrenal gland, but along the path of the embryologic neural crest cells as they migrate in the autonomic ganglia. Because of the cranial, vagal, trunk, and sacral locations of the neural crest cells, patients can develop paraganglioma anywhere from the skull base to the perineum. The incidence of PPGL is about 2 to 8 cases per 1 million, with a prevalence between 1 in 2500 and 1 in 6500. The prevalence is higher between the third and fifth decades of life and among patients with hypertension (0.1%–0.6% in adults and 2%–4.5% in the pediatric population). Although the overall prevalence is low, the incidence and prevalence of PPGL reported at the time of autopsy is higher (about 0.05%–0.1%), which suggests that a good proportion of PPGLs go undiagnosed, and these patients are at risk of premature death resulting from cardiovascular and metabolic complications associated with a functional PPGL. Patients with a functional PPGL may have cardiovascular and metabolic complications such as hypertension, myocarditis, cardiomyopathy, arrhythmias, impaired glucose tolerance, and diabetes. With the increasing use of imaging studies, adrenal incidentalomas are increasingly being discovered. Approximately 10% of all PPGLs are discovered incidentally on imaging performed for other reasons. Recent advancements in genetics and whole-genome sequencing have revealed that about one-third of patients with pheochromocytomas and one-half of patients with paragangliomas have a genetic predisposition to developing these tumors. Although once described as “the rule of 10s tumor” with 10% of tumors described as being familial, bilateral, multiple, extraadrenal, malignant, and in pediatric populations, it is now recognized that the rate of these clinical characteristics is associated with the specific genetic background of PPGL. This information has become particularly important in the workup and management of patients diagnosed with PPGL because knowledge of genetic mutation status has been associated with increased overall disease-free survival and optimal treatment and surveillance. Patients with a genetic predisposition to PPGL have an increased likelihood of developing multiple, bilateral adrenal and/or extraadrenal tumors. This information is essential in determining the optimal surgical treatment for such patients because the preoperative identification of these genes has been shown to affect the surgical approach and the extent of adrenalectomy. For example, patients diagnosed with PPGL who are found to have a mutation in RET, NF1, or VHL have a low risk of metastatic disease and are therefore good candidates for cortical-sparing adrenalectomy, especially if the tumor is small (<2–3 cm) and there is no family history of metastatic disease. On the other hand, patients with a mutation in the SDHB, FH, or MAX genes and a large primary tumor are at a much higher risk of metastatic disease and may therefore be better treated with a total adrenalectomy and regional lymphadectomy. Similarly, improvements in functional imaging and tumor localizing studies have made it easier to accurately define the extent of disease in patients with PPGL and has important implications in preoperative and intraoperative decision making.
In this chapter, we highlight the important clinical features, diagnostic tests, syndromic features and susceptibility genes, imaging studies, and key perioperative measures that are essential for the optimal surgical management of patients with PPGL.
Some patients with functional PPGL tumors may have a triad of symptoms: paroxysmal headaches, excessive sweating, and palpitations. These symptoms may be provoked by a variety of stimuli including postural changes, exercise, anxiety, trauma, pain, or certain medications. Hypertension among PPGL patients can be sustained, or it can be paroxysmal, with occasional orthostatic hypotension and hypertensive episodes occurring daily, weekly, or even every few months. Patients may also experience other symptoms such as warmth, pallor, tremors, paresthesia, weight loss, anxiety and fear of death, vision changes, dyspnea, chest pain, abdominal pain, nausea, vomiting, and grand mal seizures. Patients who present with these symptoms as well as asymptomatic patients with an incidental finding of an adrenal mass with or without hypertension, those who experience hypertensive episodes during surgery or anesthesia without a previous diagnosis of pheochromocytoma, and those with a genetic predisposition or history of PPGL should all undergo screening for PPGL.
Patients with hereditary forms of PPGL commonly have a familial history, multiple and bilateral lesions, younger age at presentation, and extraadrenal disease. However, patients with apparently sporadic PPGL can have an inherited predisposition in nearly one-third of cases. Von Hippel-Lindau (VHL) syndrome, multiple endocrine neoplasia type 2 (MEN2), neurofibromatosis type 1 (NF1), and familial paraganglioma/pheochromocytoma syndrome (caused by mutations in succinate dehydrogenase A [SDHA], B [SDHB], C [SDHC], D [SDHD] and AF2 [SDHAF2] subunits) account for most hereditary PPGL syndromes. Over 15 genes with germline or somatic mutations have been implicated in hereditary PPGL with varying likelihood of disease susceptibility ( Table 1 ), so all patients diagnosed with PPGL should have genetic counseling and testing. This information is also extremely important to enable the physician to identify syndromic disease (other diseases that may be present), for screening and surveillance of the patient and other at-risk family members, and for surgeons to select the optimal management strategy, surgical approach, and treatment ( Table 2 ).
| Gene | SDHD | SDHC | SDHB | SDHA | SDHAF2 | VHL | RET | NF1 | TMEM127 | MAX | HIF2α † | PHD1 † | PHD2 † | FH |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Single PCC | + | +/− | ++ | − | − | ++ | ++ | + | +++ | ++ | ++ | + | + | + |
| Chest/abdomen/pelvic PGL | ++ | + | +++ | + | − | + | − | − | +/− | − | ++ | +/− | +/− | +/− |
| Head and neck PGL | +++ | ++ | ++ | + | +++ | +/− | − | − | +/− | − | +/− | − | − | − |
| Multiple PGL | +++ | + | ++ | − | ++ | + | − | − | +/− | − | + | ++ | ++ | ++ |
| Bilateral PCC | + | − | + | − | − | +++ | ++ | − | ++ | ++ | ++ | + | + | + |
| Malignant PGL/PCC | + | +/− | +++ | +/− | +/− | + | − | + | +/− | + | + | +/− | +/− | + |
* Patients who have bilateral or multiple pheochromocytomas/paragangliomas, extra-adrenal paragangliomas, younger than 40 years of age at presentation, or with a family history pheochromocytoma/paraganglioma should undergo genetic testing for SDHx , VHL and RET . Patients with a history of any of the tumors present in MEN2 (medullary thyroid cancer, hyperparathyroidism) and VHL (central nervous system and retinal hemangioblastomas, renal cell carcinoma and cysts, pancreatic tumors and cysts, and epididymal cystadenomas) should also undergo genetic testing.
† Occur as a somatic mutation (or mosaic) and have been associated with polycythemia and require aggressive hydration perioperatively, and most of these patients are on aspirin therapy. Patients with HIF2α mutation are also at risk of developing neuroendocrine tumors such as somatostatinomas. +/−, Not enough data; FH, Fumarate Hydratase gene; HIF2α, hypoxia-inducible factor 2-alpha gene; MAX, MYC Associated Factor X gene; NF1, Neurofibromin 1 gene; PCC, pheochromocytoma; PGL, paraganglioma; PHD1-2, Prolyl hydroxylase domain 1-2 gene; RET, Ret proto-oncogene; SDH(A-AF2), succinate dehydrogenase (A–AF2); TMEM127, transmembrane protein 127; VHL, Von Hippel-Lindau tumor suppressor gene.
| Syndrome | Susceptibility Genes | Clinical Features | Surgical Management Considerations |
|---|---|---|---|
| Carney-Stratakis syndrome | SDHB, SDHC, SDHD | Dyad of PPGL and GISTs, with risk of RCC | Patients with large tumors and SDHB mutation have a high risk of malignant tumors, so consider open resection and prophylactic lymphadenectomy. |
| Hereditary PPGL syndrome | SDHA, SDHB, SDHC, SDHD, SDHAF2 | Most common germline mutations found in hereditary PPGL SDHB related with higher morbidity and mortality and higher risk of malignancy GISTs, renal cell carcinoma, pituitary adenomas |
Patients with large tumors and SDHB mutation have a high risk of malignant tumors, so consider open resection and prophylactic lymphadenectomy. |
| HIF-related PPGL | HIF2α/EPAS1 | PPGL, polycythemia | High-risk for multiple tumors that should be localized preoperatively to avoid persistent disease |
| MAX-related PPGL | MAX | PPGL, renal cell carcinoma, renal oncocytoma | High-risk of malignant tumors; should consider open resection for large tumors and include prophylactic lymphadenectomy |
| MEN2A/B | RET | Recurrent and bilateral PPGL with a relatively low risk of malignancy Pheochromocytoma >>> Paraganglioma
|
Consider partial adrenalectomy to avoid need for steroid replacement as low risk of malignant tumors |
| TMEM-related PPGL | TMEM127 | Pheochromocytoma > Paraganglioma RCC |
|
| VHL | VHL | Pheochromocytoma > Paraganglioma Low risk of malignancy Clear-cell renal cell carcinoma, pancreatic islet cell tumors, lymphatic sac tumors, hemangioblastoma of the kidney, pancreas, cerebellum and retina |
Consider partial adrenalectomy to avoid need for steroid replacement as low risk of malignant tumors. |
| Von Recklinghausen’s disease | NF1 | Relatively later age of presentation Pheochromocytoma > Paraganglioma Neurofibromas, dermal café-au-lait spots, Lisch nodules, bone dysplasia, skinfold freckling, optic gliomas |
Consider partial adrenalectomy to avoid need for steroid replacement as low risk of malignant tumors. |
* Mutations in the following genes have also been described, but are either very rare, have not been well-studied, or have not been associated with any known genetic syndromes: FH, SLC25A11, GOT2, MDH2, PHD2, KIF1β, MERTK, MET, and H3F3A . GIST, Gastrointestinal stromal tumor; HIF, hypoxia-inducible factor; MEN, multiple endocrine neoplasia; PPGL, pheochromocytoma/paraganglioma; RCC, renal cell carcinoma; VHL, von Hippel-Lindau.
Patients with a suspected catecholamine-secreting tumor should undergo biochemical testing. Confirmation of the presence of pheochromocytoma and functional paraganglioma with biochemical testing is then followed by imaging studies to localize the tumor. The initial biochemical screening tests include 24-hour urinary fractionated metanephrine and normetanephrine as well as plasma-free fractionated metanephrine and normetanephrine. The sensitivities and specificities of these biochemical tests are dependent on the cutoff value used (> two- to fourfold of the upper limit of normal) and whether the patient has a hereditary predisposition for pheochromocytoma. In patients with a hereditary predisposition for pheochromocytoma, plasma metanephrine testing should be used for screening because it has an overall higher sensitivity compared with 24-hour urinary fractionated testing. However, its specificity is lower, and it is associated with a false-positive rate as high as 10%. Plasma metanephrine should therefore be drawn in the supine position and after an overnight fast to reduce the likelihood of false-positive results resulting from increased levels caused by upright posture, caffeine use, or nicotine use. In patients with borderline elevation in metanephrine and normetanephrine levels, checking chromogranin A and urinary-fractionated metanephrine levels in combination or performing a clonidine suppression test have also been proposed to reduce the likelihood of false-positive results. In addition to many food products, several medications, including acetaminophen, labetalol, sotalol, alpha-methyldopa, tricycle antidepressants, buspirone, phenoxybenzamine, monoamine oxidase inhibitors (MAOIs), sympathomimetics, sulphasalazine, and levodopa, can also cause falsely elevated levels of plasma and urinary metanephrine and normetanephrine. Paragangliomas (by virtue of their extraadrenal location) do not secrete epinephrine because of the lack of phenylethanolamine N- methyltransferase (PNMT), except when located in the organ of Zuckerkandl.
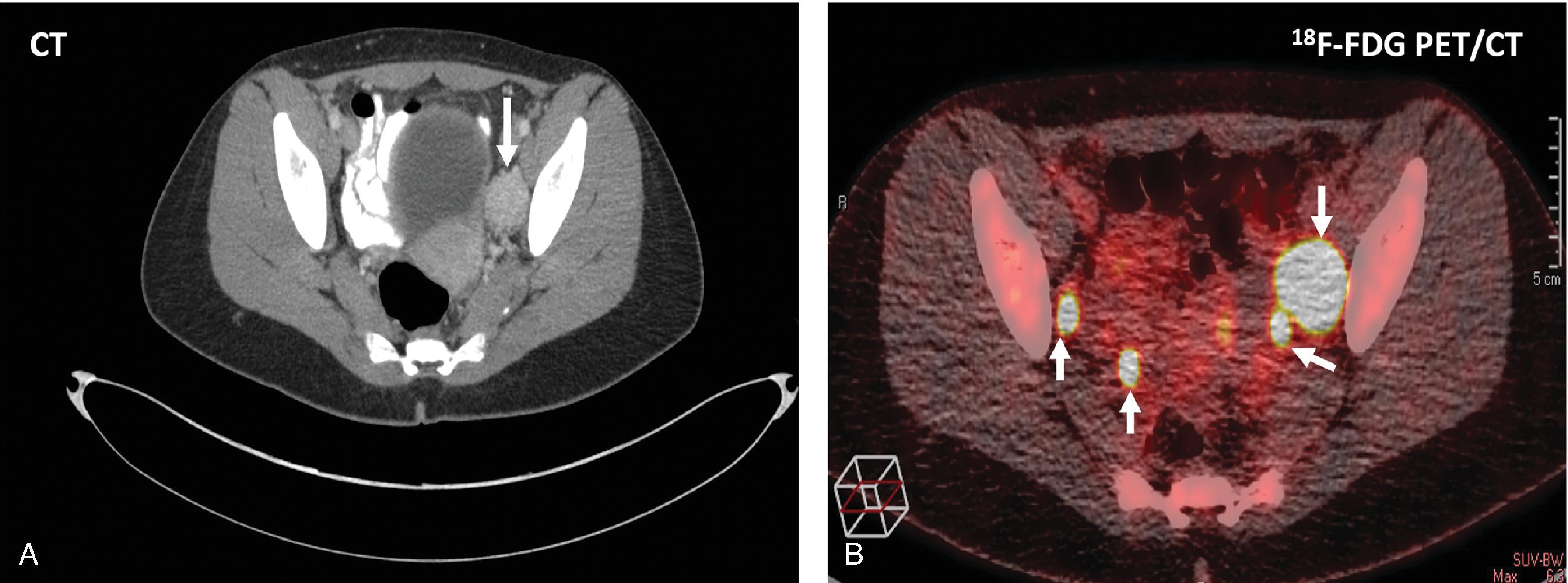
Once biochemical evidence of PPGL is confirmed, the precise location of the tumor(s) must be identified. Computed tomography (CT) and magnetic resonance imaging (MRI) are the most commonly used imaging modalities ( Fig. 1 ). Pheochromocytomas are better visualized on adrenal protocol CT scans that use thin-cut and timed intravenous (IV) contrast images to evaluate for tumor enhancement and washout. CT has excellent sensitivity (ranging from 90%–100%) for the detection of both PPGLs. CT findings consistent with a pheochromocytoma include Hounsfield units (HUs) greater than 10 and vigorous tumor enhancement in the early contrast phase, with less than 60% of washout in the venous phase (see Fig. 1A ). On noncontrast CT scans, pheochromocytomas commonly have HUs in the range of 40 to 50. PPGLs on CT scan may appear homogenous or heterogeneous, necrotic with some calcification, solid, or cystic. Smaller tumors tend to be more homogenous, while larger tumors are typically more heterogenous and more likely to have necrotic, calcified, and cystic components. CT scans of the neck, chest, abdomen, and pelvis, with and without contrast, should be performed to exclude extraadrenal paragangliomas, using a section thickness of 2 to 2.5 mm in the neck and 5 mm through the chest, abdomen, and pelvis, especially in high-risk patients: young patients (< 40 years) and patients with germline SDHx mutations or a family history of PPGLs (see Table 1 ).
| Imaging Modality | MRI | CT | 18 F-FDG | 68 Ga-DOTA Peptide | 18 F-FDA | 18 F-DOPA | MIBG |
|---|---|---|---|---|---|---|---|
| Advantages | Anatomic detail | Anatomic detail | Widely available, preferred functional imaging test | Most sensitive functional imaging study for all sites of disease, potential for therapy with avid disease | Sensitive functional imaging for primary tumor (except skull base and neck) | Most sensitive functional imaging study for skull base and neck paragangliomas | Potential for therapy if disease avid tumor is not resectable |
| Disadvantages | Lower specificity | Lower specificity | Lower sensitivity and higher false-positive rate compared with other functional imaging studies | Not widely available | Not widely available | Not widely available | Lower sensitivity than other functioning imaging studies |
| Best clinical setting to use imaging studies | Initial localization of tumor | Initial localization of tumor | Rule out metastatic disease or multiple primary | Rule out metastatic disease or multiple primary; SDHx mutation carriers; when planning treatment with peptide receptor radionuclide therapy | Rule out metastatic disease or multiple primary | Patients with primary skull base and neck paragangliomas; multiple and metastatic disease | When planning treatment with 131 I-MIBG |
MRI is the imaging test of choice in pediatric and pregnant or lactating patients. Other patient groups that benefit from this modality include patients with documented allergies to CT contrast agents and patients who refuse radiation exposure. Because of their increased vascularity and high water content, PPGLs may have a pathognomonic high signal intensity on T2-weighted MRI (see Fig. 1B ). However, this is not always the case, and PPGLs can appear homogenous with intermediate intensity, diffusely heterogeneous with swirl-like areas of high and low signal intensity, and heterogeneous with multiple pockets of high signal intensity (cystic lesions) (see Fig. 1B ). MRI has a higher sensitivity for the detection of extraadrenal paraganglioma compared with CT scans, with some studies reporting a sensitivity as high as 100% with MRI.
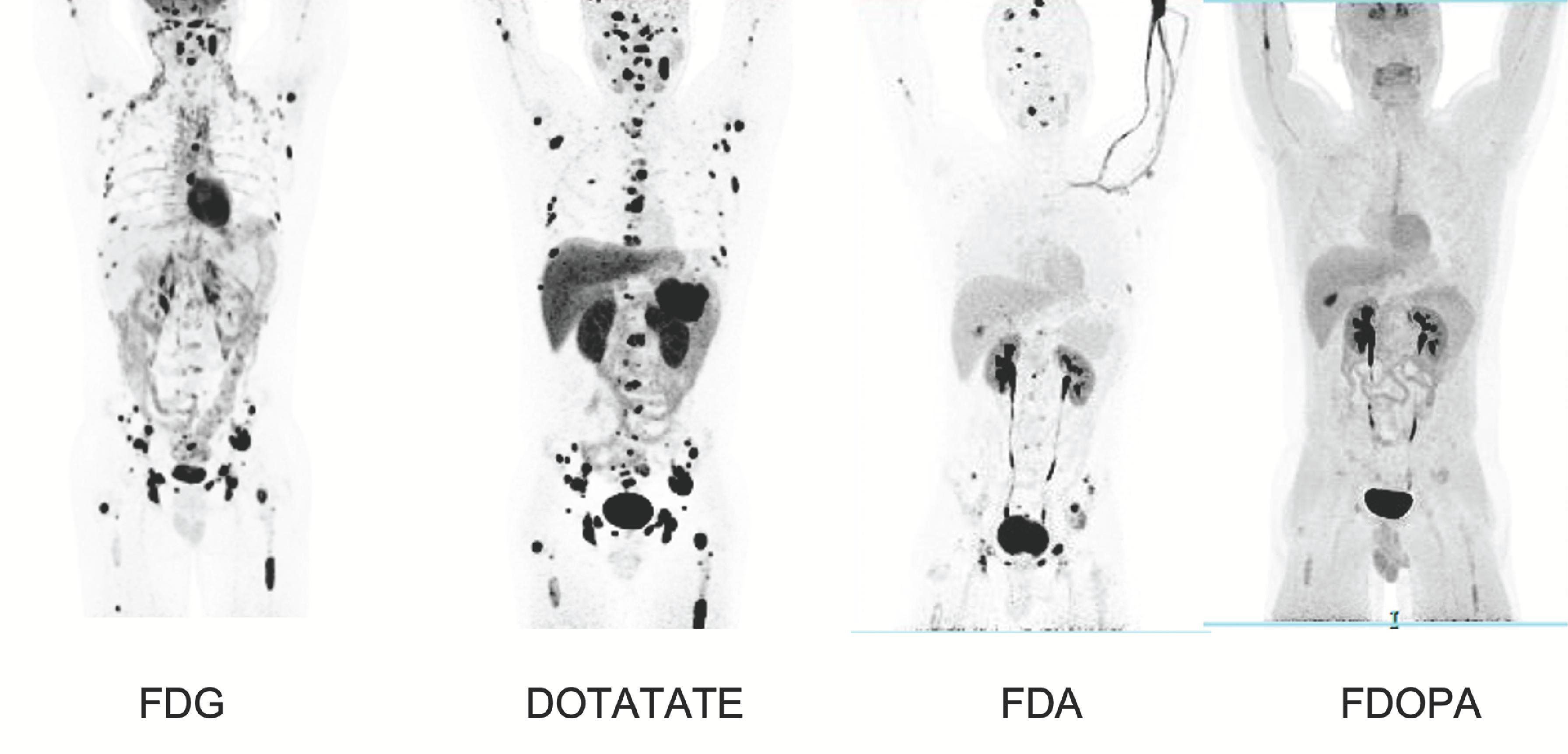
Although anatomic imaging modalities like CT and MRI have excellent sensitivity, they have very poor specificity. Functional imaging studies, such as nuclear scintigraphy with 123 I-metaiodobenzylguanidine (MIBG), 18 F-fluorodeoxyglucose (FDG), 18 F-3,4-dihydroxyphenylalanine (DOPA), 18 F-fluorodopamine (FDA), and 68 Ga-DOTA (DOTATATE, DOTATOC, DOTANOC) peptide positron emission tomography (PET)/CT, can be helpful in localizing previously undetected tumors on CT/MRI and for identifying additional tumor sites ( Figs. 2 and 3 ). For ruling out metastatic disease and localizing the primary tumor, 18 F-FDG PET/CT and 68 Ga-DOTA peptide PET/CT are most accurate (see Figs. 2 and 3 ). 123 I-MIBG scanning is not as sensitive as the other functional imaging modalities and should only be used when planning radiotherapy with 131 I-MIBG for patients with metastatic disease. Table 3 summarizes the anatomic and functional imaging studies that can be used to localize PPGLs, with their advantages and disadvantages, and the clinical settings in which their use can facilitate the selection of the optimal surgical management strategy.
| Diagnostic Category | Risk of Malignancy if NIFTP ≠ CA (%) | Risk of Malignancy if NIFTP = CA (%) | Usual Management † |
|---|---|---|---|
| Nondiagnostic or unsatisfactory | 5–10 | 5–10 | Repeat FNA with ultrasound guidance |
| Benign | 0–3 | 0–3 | Clinical and sonographic follow-up |
| Atypia of undetermined significance or follicular lesion of undetermined significance | 6–18 | ∼10–30 | Repeat FNA, molecular testing, or lobectomy |
| Follicular neoplasm or suspicious for a follicular neoplasm | 10–40 | 25–40 | Molecular testing, lobectomy |
| Suspicious for malignancy | 45–60 | 50–75 | Near-total thyroidectomy or lobectomy ‡ , § |
| Malignant | 94–96 | 97–99 | Near-total thyroidectomy or lobectomy § |
† Actual management may depend on other factors (eg, clinical, sonographic) besides the FNA interpretation.
‡ Estimate extrapolated from histopathologic data from patients with “repeated atypicals.”
§ In the case of “Suspicious for malignancy” or a “Malignant” interpretation indicating metastatic tumor rather than a primary thyroid malignancy, surgery may not be indicated. From Ali S, Cibas E. The Bethesda System for Reporting Thyroid Cytopathology: Definitions, Criteria and Explanatory Notes . Second edition. Springer; 2018.
| Signs | Symptoms | |
|---|---|---|
| General | Weight loss | Increased appetite; heat intolerance; sweating |
| Cardiovascular | Tachycardia, hypertension, arrhythmia (atrial fibrillation) | Palpitations |
| Pulmonary | Tachypnea | Dyspnea |
| Gastrointestinal | Diarrhea, nausea, vomiting | |
| Reproductive | Changes in menstrual cycle | |
| Ocular | Proptosis, eyelid retraction, periorbital edema | Diplopia, blurry vision, retro-orbital pain, irritated eyes |
| Dermatologic | Warm and moist ("clammy") skin | |
| Neuromuscular | Tremors, hyperactivity, hyperreflexia | Nervousness, anxiety, fatigue, weakness, disturbed sleep, poor concentration |
The preoperative management of patients with PPGL should focus on the optimization of blood pressure and treatment for tachyarrhythmias. Hypertension and elevated levels of circulating catecholamines predispose patients with PPGL to compromised cardiovascular function, which is commonly manifested as a tachyarrhythmia. These patients may have concomitant, long-standing hypertension, which predisposes them to cardiomyopathy and aseptic myocarditis. A preoperative cardiac evaluation is therefore prudent, regardless of blood pressure levels, to assess cardiac function, optimize perioperative management, and risk-stratify patients for perioperative cardiovascular events.
Preoperative antihypertensive medications to reduce the risk of intraoperative hemodynamic instability are required for all patients with catecholamine-producing PPGLs. Alpha-adrenergic antagonists are usually the first agents of choice because of their ability to lower blood pressure, reverse volume depletion, and reduce peripheral vascular resistance. Patients who are normotensive should be started on alpha blockade to counter any potential activity from dormant tumors that may release catecholamines during intraoperative tumor manipulation. Adrenergic blockade should start 7 to 14 days preoperatively to normalize blood volume, blood pressure, and heart rate. We normally initially place patients on phenoxybenzamine (10 mg twice a day). Other alternative agents include a selective alpha antagonist such as doxazosin (2 mg once a day), prazosin (2 mg twice a day), or terazosin (2 mg once a day). The goal should be to achieve a preoperative blood pressure of 130/80 mm Hg or less while sitting and approximately 100 mm Hg while standing. If the patient continues to have high blood pressure, the dose is increased in increments of 10 to 20 mg every 2 to 3 days. In addition, medications are titrated for a heart rate of about 60 to 70 beats per minute (bpm) while sitting and 70 to 80 bpm while standing.
Many patients with PPGL have chronic vasoconstriction secondary to high levels of catecholamines. Preoperative normalization of blood volume reduces the risk of hypotension during or after removal of the tumor. A high-salt diet 3 days after initiation of alpha blockade and 1 to 2 L of normal saline starting the evening before surgery can normalize intravascular volume and reduce the risk of hypotension after removal of the tumor. With appropriate alpha-adrenergic blockade, patients may experience tachyarrhythmias. A beta blocker should be added to control the heart rate 2 to 3 days after initiation of alpha blockade. Most importantly, a beta blocker should never be used in the absence of adequate alpha blockade. The utilization of a beta blocker without alpha blockade leads to unopposed alpha-adrenergic catecholamine stimulation, which will exacerbate vasoconstriction. Beta-selective drugs such as atenolol or metoprolol are started for a target heart rate of 60 to 80 bpm. Labetalol, a nonselective beta blocker, should not be used as first-line therapy because the beta effect is seven times more potent than the alpha effect. For patients with persistent hypertension despite optimal alpha and beta blockade, a calcium channel blocker (30 to 90 mg of nifedipine a day) or metyrosine (1 to 4 grams a day) may be used, although it is important to note that metyrosine has very significant side effects including sedation, fatigue, sexual dysfunction, extrapyramidal symptoms, headache, diarrhea, nausea, and vomiting. Its use should therefore be limited to patients with refractory hypertension and not as a first-line agent.
A diagnosis of PPGL during pregnancy is associated with significant rates of maternal mortality (up to 40%) and fetal mortality (over 50%), resulting largely from the challenges related to preoperative management. Alpha-adrenergic antagonists should be initiated immediately at the time of diagnosis, and plans should be made for an elective third-trimester caesarean delivery. Vaginal delivery is contraindicated in patients with PPGL because of the potential for catastrophic, massive catecholamine release during labor and at the time of delivery. Overall, a combined adrenalectomy at the time of caesarean section versus early postpartum resection is generally preferred. Tumor removal should be avoided during the first trimester because of the high risk of miscarriage. However, adrenalectomy in the second trimester has been reported and may be considered if necessary.
Successful intraoperative management of PPGL requires collaborative preparation and good communication between the surgery and anesthesiology teams. Despite being adequately blocked by preoperative adrenergic blockade, patients with PPGL may still have hypertensive crises or tachyarrhythmias during manipulation of the tumor. Therefore intraarterial monitoring is required and appropriate venous access, including a central venous catheter for monitoring the volume status, is imperative. Patients may have fluctuations in blood pressure during the operation, so multiple vasoactive medications should be premixed and ready to be administered in anticipation of these labile blood pressures. Hypertensive episodes can be treated with esmolol (500 µg/kg over 1 minute loading dose, followed by a 4-minute maintenance infusion of 50 µg/kg/min) or sodium nitroprusside (0.5 to 3 µg/kg/min), which reduces cardiac preload. Excellent communication between the surgeon and anesthesiology team is required just before ligation of the adrenal vein because this maneuver can lead to abrupt hypotension, which should initially be treated with crystalloids. Once adequate volume resuscitation has been achieved, treatment of persistent hypotension with vasopressors such as norepinephrine, phenylephrine, or vasopressin can be initiated.
Options for the surgical approach in the resection of PPGL include open and minimally invasive approaches (laparoscopic, robotic), which can all be performed using either a transabdominal or retroperitoneal approach. Minimally invasive surgery for the removal of PPGL is the procedure of choice in most cases and has been shown to be associated with significant advantages compared with open resection: decreased blood loss, decreased blood transfusion requirement, shorter intensive care unit and postoperative hospitalization, decreased analgesic requirements, and faster return to normal patient activity. Furthermore, minimally invasive approaches are associated with fewer catecholamine surges during tumor manipulation and therefore fewer episodes of blood pressure lability. A direct open approach is typically reserved for larger, locally advanced or malignant tumors. Conversion to an open procedure may also be required because of intraoperative suspicion of malignancy or metastatic disease. Robotic-assisted removal of PPGL is also safe and feasible, but it does not appear to afford any significant advantages over laparoscopic resection using either the transabdominal or retroperitoneal approach, especially in light of its higher financial cost.
The surgical approach and extent of resection for pheochromocytoma we use depends on (1) whether the tumor is unilateral or bilateral, (2) whether there are any additional paragangliomas or possible sites of metastatic disease, (3) tumor size, (4) patient body habitus and body mass index, (5) whether the tumor is sporadic or familial relative to the risk of malignancy, and (6) the risk of developing metachronous pheochromocytoma in the contralateral and remnant adrenal gland. We most commonly use a transabdominal laparoscopic approach in patients who have unilateral adrenal tumors and sporadic disease and those who have no evidence of extraadrenal tumors. In patients with bilateral tumors and familial disease with a low risk of malignant pheochromocytoma, such as the MEN2 or VHL syndromes, we perform a cortical-sparing adrenalectomy using a transabdominal or retroperitoneal laparoscopic approach. When patients have unilateral or recurrent tumors, possible site(s) of lymph node metastasis/local invasion, and an SDHB germline mutation, we perform a total adrenalectomy and retroperitoneal lymph node dissection with an open approach. Even when the tumor is large, we usually begin the operation using a laparoscopic approach until we encounter evidence of local invasion or features suspicious for malignancy. We believe that a complete resection is most important and, whatever the surgical approach, there should be no fracturing of the tumor or violating the tumor capsule, as there have been reports of pheochromocytomatosis with evidence of higher rates of peritoneal implants, locoregional recurrence, and metastatic disease among patients who underwent laparoscopic resection. For this reason, clinical guidelines recommend open resection for large tumors (>6 cm) and locally invasive PPGLs. Minimally invasive approaches may be used, however, for small, noninvasive paragangliomas in safe and favorable locations.
Transabdominal laparoscopic adrenalectomy is the most common laparoscopic approach used by most surgeons. The main advantage of this approach is a larger working space than that afforded by the retroperitoneal approach. The procedure is performed by placing the patient in the lateral decubitus position, with the operating table flexed. An incision is made one fingerbreadth below the costal margin in the midclavicular line. We use a 0-degree videoscope to enter the peritoneal cavity under direct visualization. Then we use a 30-degree videoscope to place additional trocars and for the dissection. The tumor should not be manipulated, or manipulation should be kept to a minimum, until the venous drainage is divided, as severe hypertension can result otherwise. Clear communication with the anesthesia team is important to anticipate and treat hemodynamic instability during manipulation of the tumor and division of the adrenal vein. The key steps and intraoperative images of a transabdominal laparoscopic adrenalectomy (right and left) are summarized in Figure 4 .
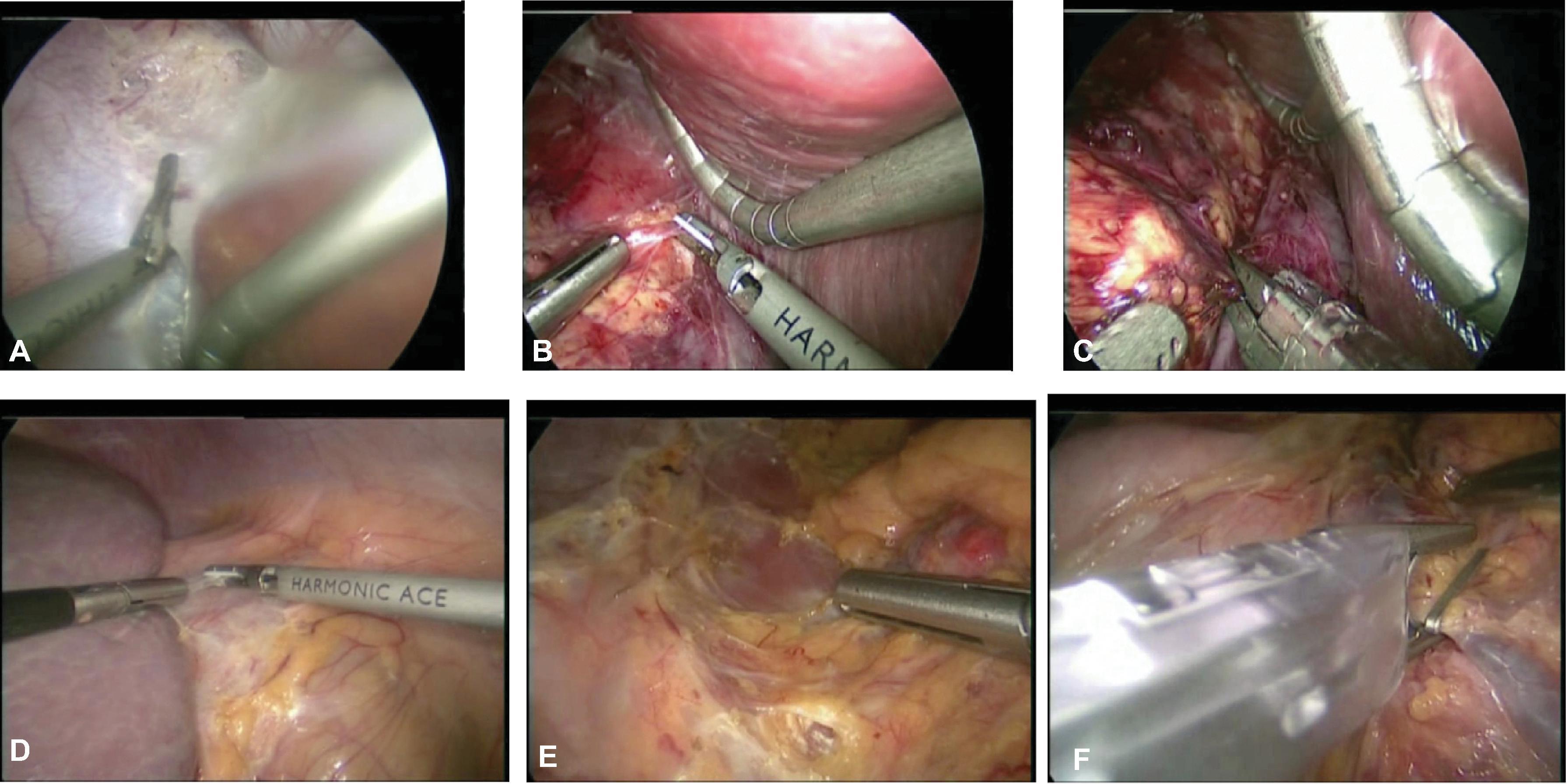
For a right adrenalectomy, we use four trocars, from the mid-clavicular line to the mid-axillary line, with a distance of approximately two fingerbreadths between the trocars. The initial dissection consists of dividing the triangular ligament of the liver to mobilize the right lobe of the liver medially and expose the right adrenal gland. Once this has been completed, a fan, paddle, or snake retractor is placed on the right lobe of the liver to maintain the working space. The dissection from the triangular ligament is extended down inferiorly, dividing the peritoneum and Gerota’s fascia along the liver down to the infrahepatic inferior vena cava. Commonly, the adrenal vein is encountered during this top-down dissection and is circumferentially dissected, clipped, and divided. Any accessory veins and the three adrenal arteries, which are usually more prominent in patients with pheochromocytoma, are divided by an energy device along this medial dissection. The inferior pole of the gland is then mobilized free from the renal hilum, taking care not to injure the renal artery and vein. The attachment to the superior pole of the kidney and posterior musculature is then divided, and the dissection is extended up to the diaphragm, removing the periadrenal fat along with the gland. We usually detach the attachment to the diaphragm last as it keeps the adrenal gland in place, allowing good visualization of the adrenal hilum and inferior limb of the gland.
For a left adrenalectomy, we usually use three trocars because the spleen and the tail of the pancreas are retracted by gravity. The first subcostal trocar is placed in the midclavicular line at the Palmer point, and the lateral two are placed approximately two fingerbreadths apart and one fingerbreadth from the costal margin. The lateral attachment of the spleen (approximately 1 cm from the lateral edge of the spleen) and the splenic flexure of the colon are released up to the diaphragm until visualization of the cardia of the stomach. This dissection allows the spleen and the tail of the pancreas to be retracted medially by gravity. At the medial aspect, the adrenal gland is dissected top-down along with the periadrenal fat and the adrenal arteries divided with an energy device. Often the inferior phrenic vein is encountered during this medial dissection and can be traced to the left adrenal vein. The adrenal vein is then clipped and divided above the renal vein into which it drains. The inferior pole is carefully dissected free from the renal hilum without injury to the renal vein and artery, and then free from the superior pole of the kidney and from the posterior musculature.
We commonly use the posterior retroperitoneal approach in patients who require bilateral total or partial adrenalectomy and in patients who have undergone previous abdominal operations and have significant intraabdominal adhesions. The key steps of a posterior retroperitoneal adrenalectomy (left and right), including intraoperative images, are outlined in Figure 5 . Once there is adequate general anesthesia and intravenous and intraarterial lines are placed and secured, the patient is placed in the prone jackknife position. The hips and knees are flexed at 90 degrees, and all pressure points are padded. Cushions are used to support the hips and lower chest, allowing the anterior abdominal wall to hang unconstrained between the cushions. This helps to expand the retroperitoneal space. The tip of the 12th rib is palpated, and a 1.5-cm transverse incision is made below the tip of the rib and extended into and through the posterior musculature and fascia to enter the retroperitoneum. A working space is initially created by blunt digital separation of the retroperitoneal fat. A lateral 5-mm trocar is inserted 4 to 5 cm from the initial incision, beneath the 11th rib, under finger guidance. Next, a medial 10-mm trocar is inserted 4 to 5 cm medially from the initial incision. Finally, a 10-mm, blunt-tip trocar with an inflatable balloon is inserted and secured in the initial incision, followed by insufflation with carbon dioxide. The pressure should be maintained around 20 mm Hg. The retroperitoneal fat is pushed down and away medially from the posterior musculature and diaphragm. The retroperitoneal fat and the adrenal gland are dissected free from the diaphragm superiorly. The superior pole of the kidney is retracted, and the adrenal gland is mobilized free medially and inferiorly: on the right side, the adrenal vein is identified during this dissection draining into the inferior vena cava; on the left side, it is identified medially and inferiorly along with the phrenic vein in most cases. Once the adrenal vein is clipped and divided, the gland, along with the retroperitoneal fat, is lifted and retracted laterally and superiorly to divide the remaining small vessels and attachments. Hemostasis should be checked after the carbon dioxide pressure is reduced.
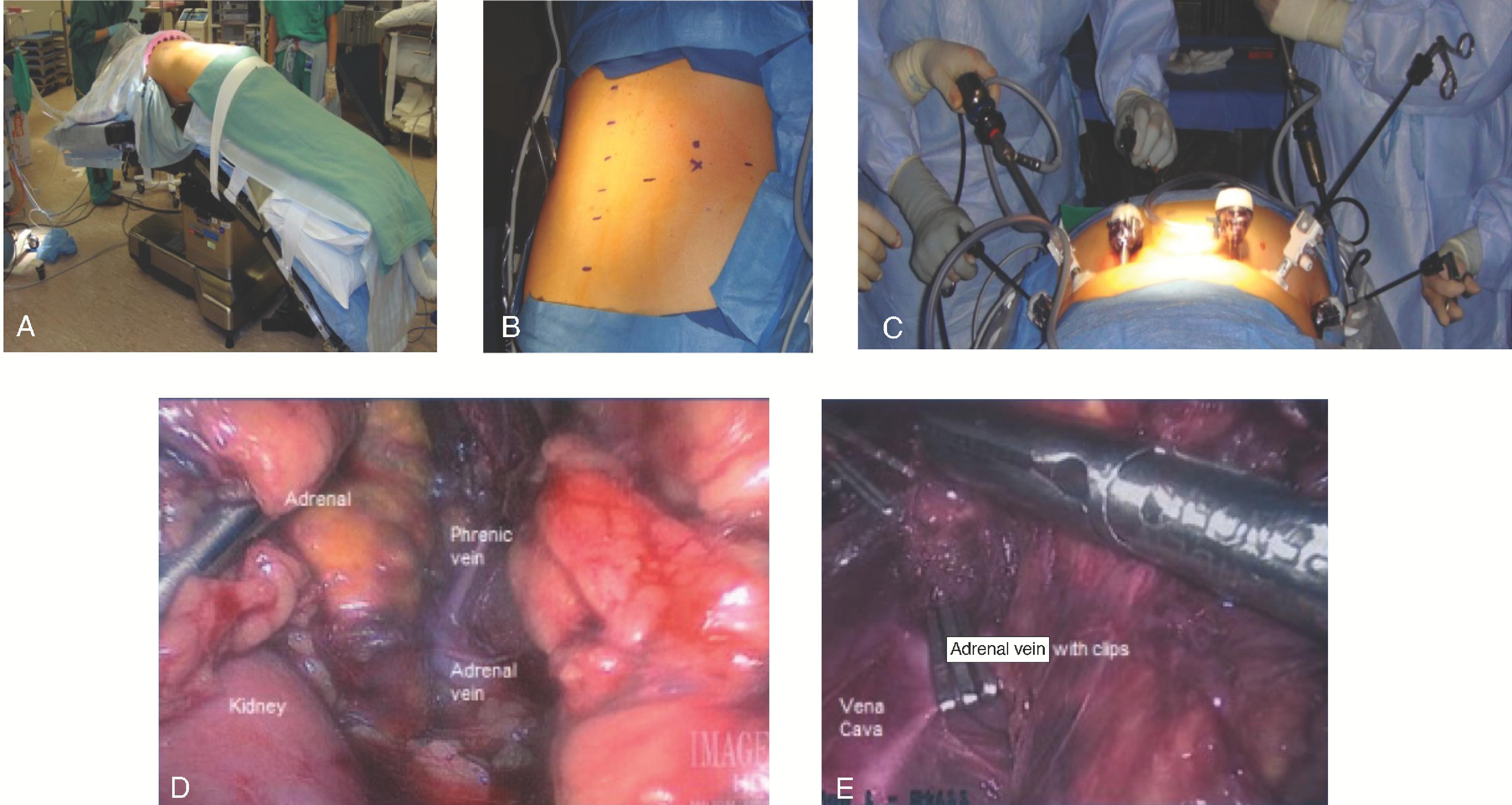
In patients who have MEN2 and VHL and who also have bilateral pheochromocytomas, there is an opportunity to perform cortex-sparing adrenalectomy to avoid the need for long-term steroid replacement and the risk of an Addisonian crisis if a bilateral total adrenalectomy were to be performed. Furthermore, patients with MEN2 and VHL who have unilateral pheochromocytoma may also need to have cortical-sparing adrenalectomy because of the risk of developing a contralateral pheochromocytoma, which would require an adrenalectomy. Laparoscopic cortex-sparing adrenalectomy can be performed in a similar fashion to laparoscopic adrenalectomy in terms of port placement and mobilization of surrounding organs. Intraoperative ultrasound is beneficial in identifying smaller tumors and helping the surgeon identify the margins of resection. The adrenal gland parenchyma can be transected using electrocautery or Harmonic and LigaSure scissors. The remnant adrenal gland is left in situ and not dissected. This is critical, especially if the adrenal vein cannot be spared to maintain adequate blood flow and drainage of the remnant adrenal gland.
We use an open adrenalectomy approach in patients who may have malignant tumors, a diagnosis based on a sufficiently large tumor size, the presence of local invasion, or presentation with locoregional recurrence—and for those who have a positive germline SDHB mutation or other less-common susceptibility gene mutations, such as FH and HIF2α, which are associated with a high rate of metastatic disease. We use a subcostal incision for our open adrenalectomies, but a thoracoabdominal incision may be necessary in patients with large, locally invasive tumors. In addition to resecting the primary tumors or site of recurrent disease, we routinely perform a prophylactic retroperitoneal locoregional lymph node dissection. There are several key steps in an open right adrenalectomy for malignant or potentially malignant PPGLs. An extended Kocher maneuver is essential for adequate exposure and to allow vascular control of the infrahepatic inferior vena cava ( Fig. 6 ). The triangular ligament should be routinely divided, and the right lobe of the liver should be fully mobilized medially. When resecting locally invasive tumors, it is essential to gain vascular control over the suprahepatic and infrahepatic inferior vena cava before dissecting the tumor free from the vena cava or dissecting the right adrenal vein. Depending on the extent of local disease, adjacent organs may need to be resected (nephrectomy/nonanatomic liver resection/right hepatic lobectomy). For a left open adrenalectomy, division of the gastrocolic ligament and mobilization of the pancreas may be necessary if an en bloc resection of the tail of the pancreas and spleen is planned. If not, the left adrenal gland is exposed by medial rotation of the spleen, pancreas, and left colon. Vascular control of the renal vessels is important for a right or a left open adrenalectomy when removing a locally invasive tumor. For negative margins, a cuff of the posterior musculature or diaphragm, or a full-thickness resection of the diaphragm may be necessary and should be reconstructed primarily or with a Gore-Tex patch graft and an angled chest tube left above the diaphragm. Although it is rare for a patient to require such aggressive surgical treatment for locally advanced PPGLs, most patients who have cardiovascular complications associated with their tumors benefit from such an approach to functioning tumors, especially when an R0 or R1 resection can be performed.
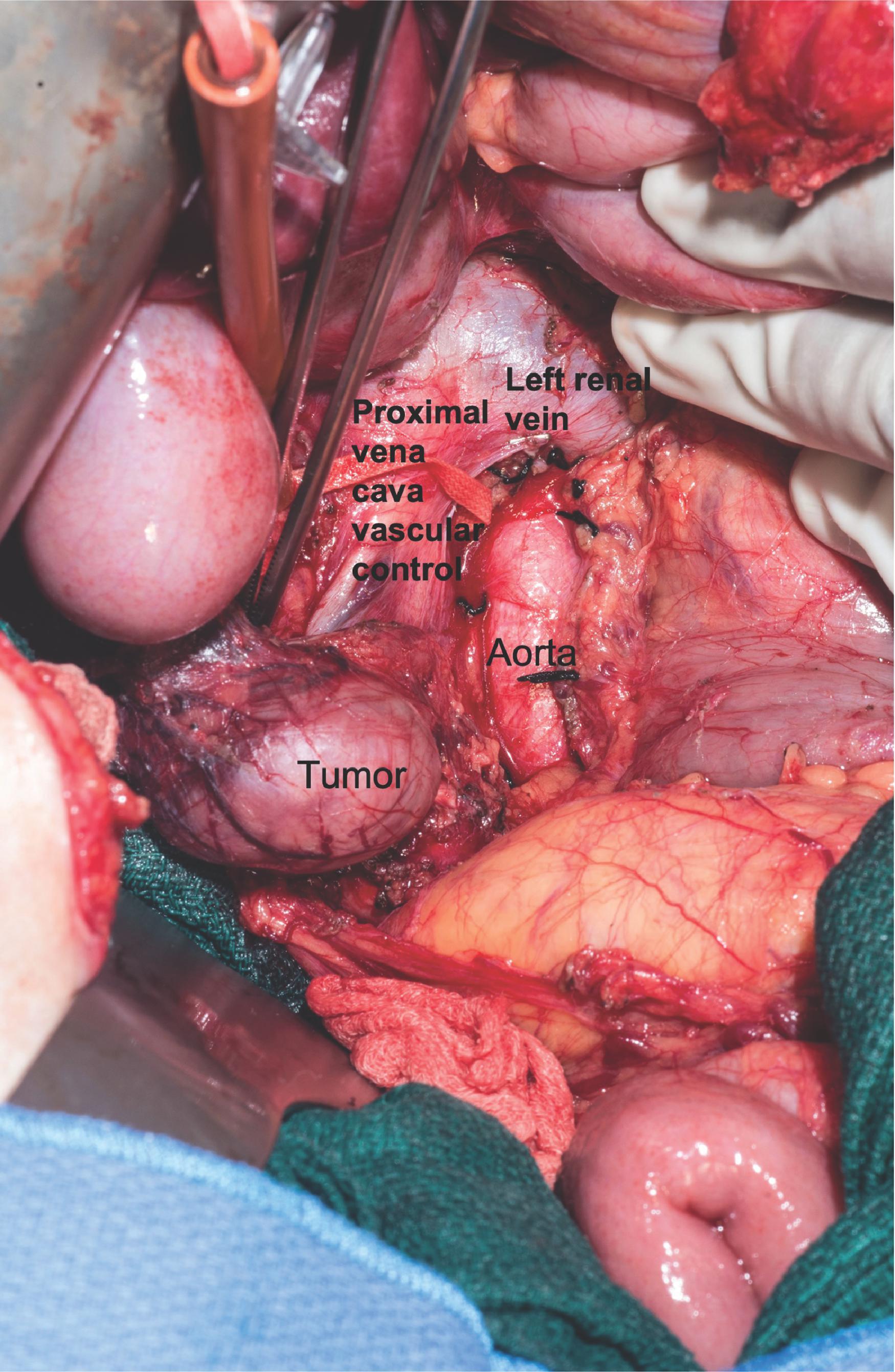
Postoperatively, patients should be monitored for blood pressure fluctuations in the intensive care unit or a step-down unit for the first 24 hours. Patients may have hypotension caused by the continued effects of alpha-adrenergic blockade or withdrawal of catecholamine and metabolites, and they may require vigorous infusion of intravenous fluids. There have been reports of hypoglycemia caused by rebound secretion of insulin after removal of a pheochromocytoma; therefore blood sugars should be monitored. Plasma metanephrine levels should be evaluated at 6 weeks and at 6 months to confirm the patient’s biochemical response.
Currently, there are no histologic, biochemical, or molecular markers that can differentiate patients with benign and malignant PPGLs in the absence of locoregional invasion, distant metastasis, or lymph node metastasis. A metastatic PPGL may be present at the initial time of diagnosis or (commonly) become evident within 5 years of diagnosis. However, there are reports in the literature of recurrences up to 42 years after pheochromocytoma resection, which emphasizes the importance of annual biochemical screening in both sporadic and hereditary PPGLs and imaging surveillance in patients without a functional tumor. Features that have been associated with metastatic PPGLs include a tumor size greater than 5 cm and germline SDHB gene mutation.
In patients with metastatic PPGLs, surgical resection of recurrent or metastatic disease is the best treatment option. This is especially true in patients for whom an R0 or R1 resection can be achieved. However, some patients who have widely metastatic disease with elevated catecholamine and metabolites may still benefit from cytoreductive surgery to palliate symptoms or control cardiovascular events associated with the presence of excess catecholamine and their metabolites. The prognosis of patients with metastatic PPGLs is variable, with some patients developing stable disease and others developing rapidly progressive disease, including debilitating symptoms and difficult-to-control cardiovascular complications from the presence of excess catecholamines and metabolites. Overall, the median survival for patients with malignant PPGLs is 7 years, with less than 50% of patients surviving past 5 years.
Patients with unresectable disease may benefit from cytotoxic chemotherapy, such as treatment with a combination of cyclophosphamide, vincristine, and dacarbazine or 131 I-MIBG therapy. Unfortunately, only two-thirds of metastases are avid upon MIBG scanning, which can limit the efficacy of this therapy. Up to two-thirds of patients can have an objective response, with complete response, partial response, or stable disease with 131 I-MIBG therapy. PPGLs have been localized using newer imaging modalities such as 68 Ga-DOTA peptide PET/CT, which shows superior metastatic disease detection rates compared with other anatomic and functional imaging studies. For tumors that are 68 Ga-DOTA peptide–avid, peptide receptor radionuclide therapy with 177 Lu or 90 Y may result in a partial or complete response and symptomatic relief and is currently being studied in clinical trials.
PPGLs are rare tumors that can occur throughout the body, and they are associated with an increasingly recognized number of genetic mutations. Initial diagnosis of functional PPGLs is by biochemical testing with 24-hour urinary or plasma metanephrines, followed by tumor localization with CT/MRI. Functional imaging studies with 18 F-fluorodeoxyglucose (FDG) and 68 Ga-DOTA peptide PET/CT are helpful for the evaluation of additional primary tumors and metastatic disease. Genetic testing should be performed in all patients with PPGL. Surgical management of PPGL requires close collaboration and communication with the anesthesiologist. Preoperative alpha blockade and beta blockade is imperative to optimize blood pressure, treat tachyarrhythmia, and restore normal circulating blood volume. Surgical removal is the mainstay of treatment and can be performed via minimally invasive or open approaches with excellent results.
There has been a substantial increase in the detection of thyroid nodules and thyroid cancer over the past decades in the United States and other countries, which is attributed to the availability of neck ultrasound. However, deaths from thyroid cancer have remained rare yet relatively constant. Due to increased detection, not accompanied by increased mortality, some are concerned about the overtreatment of thyroid nodules and cancer, and this controversy has been popularized in the mainstream media. It is very important to risk-stratify patients with thyroid nodules to avoid the morbidities of overtreatment that can substantially affect quality of life, while still providing appropriate treatment to achieve optimal outcomes.
Thyroid nodules are very common, affecting as many as half of the population by age 50. Most thyroid nodules are asymptomatic and identified incidentally from imaging studies. Some patients have a palpable neck mass or goiter. The vast majority of thyroid nodules are not cancerous. The rate of cancer, however, is higher in patients with risk factors such as exposure to ionizing radiation at a young age or having a first-degree relative with thyroid cancer. In contrast, thyroid nodules causing hyperthyroidism are rarely cancerous. Patients with suppressed thyroid-stimulating hormone (TSH) (hyperthyroid) and a thyroid nodule are usually first evaluated by scintigraphy to establish the diagnosis (e.g., solitary or multiple hyperfunctioning nodules [Plummer’s disease] or hypofunctioning nodules in Graves’ disease).
Ultrasonography is safe and the most efficient first-line evaluation for thyroid nodules. Multiple large cohort studies have identified ultrasound characteristics of thyroid nodules that are associated with risk of cancer. The high-risk characteristics include nodules that are large, hypoechoic, contain microcalcifications, have irregular borders, or are taller than they are wide. Ultrasound characteristics associated with a low risk of thyroid cancer include nodules with more cystic components and those that have clear borders or are spongiform. Ultrasound characteristics help to stratify the risk of thyroid cancer and are used to determine when fine-needle aspiration (FNA) biopsy is indicated. The 2015 American Thyroid Association guidelines recommend FNA for thyroid nodules larger than 1 to 2 cm depending on their ultrasound characteristics: 1 cm for those classified at highest risk by ultrasound and 2 cm for those classified at the lowest risk ( Fig. 1 ). The American College of Radiology has developed the Thyroid Imaging Reporting and Data Systems (TI-RADS) to provide a point scale option to “quantify” the level of risk for each nodule in addition to guidance for size threshold of when to biopsy based on TI-RADS score ( Fig. 2 ).
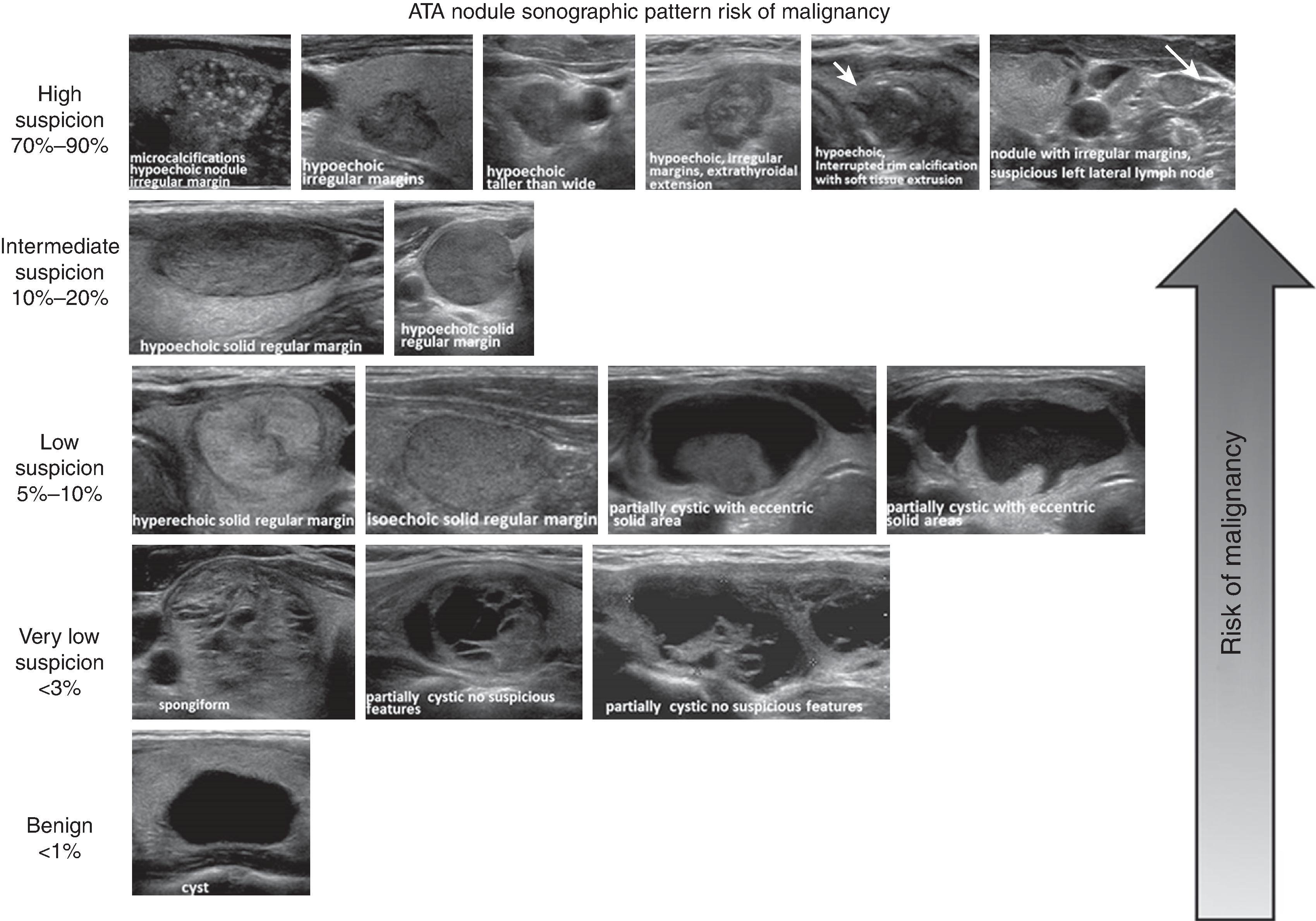
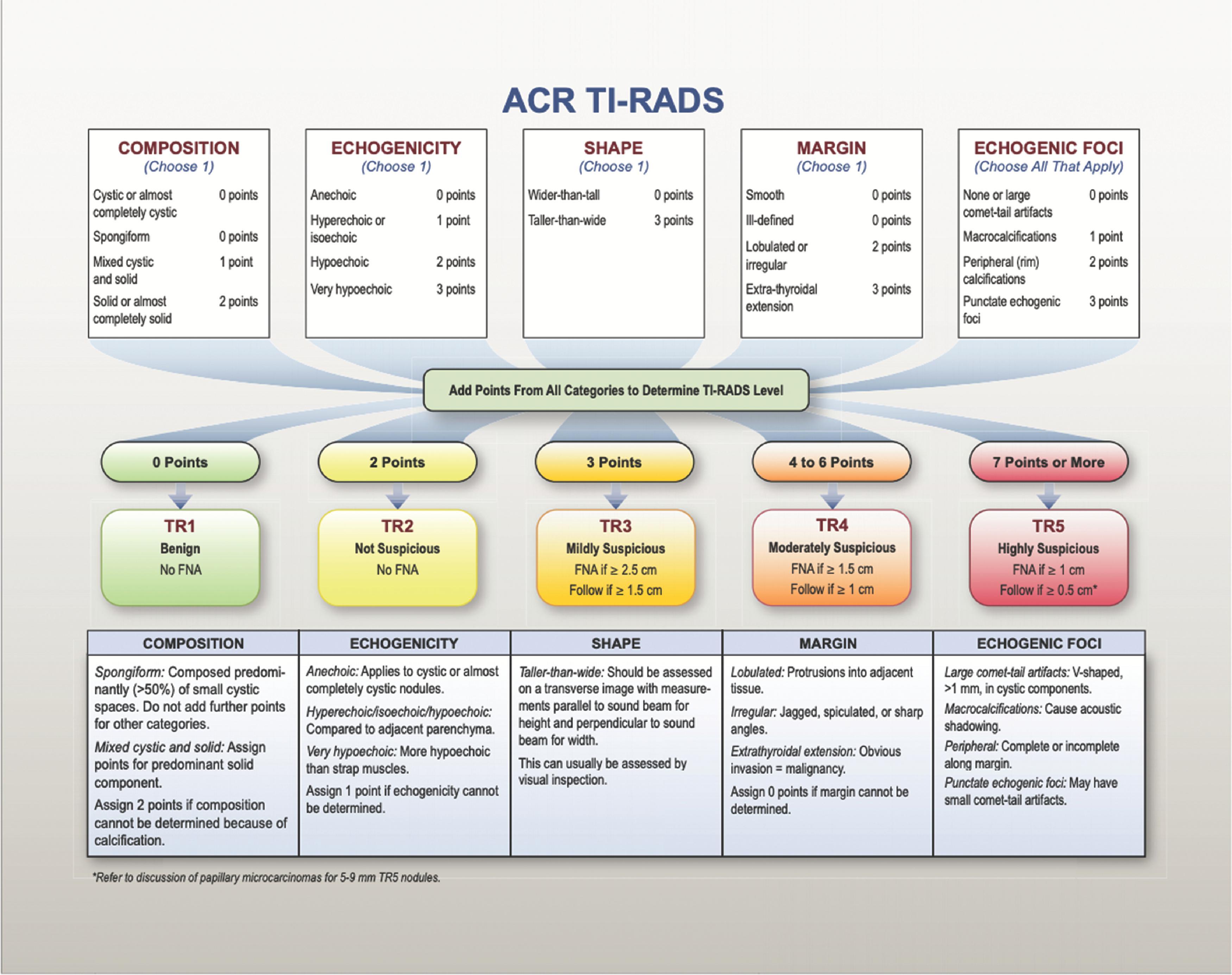
Occasionally, thyroid nodules are found on imaging modalities other than ultrasound, such as computed tomography scans and magnetic resonance imaging. These still should be investigated by ultrasound, and further workup should proceed accordingly. Hypermetabolic thyroid nodules incidentally found on fluorodeoxyglucose-positron emission tomography (PET) scan are at higher risk for cancer, thus FNA is recommended at the lower threshold of more than 1 cm.
Serum levels of thyroglobulin are useful in the follow-up of patients with differentiated thyroid cancer but not in the diagnosis of whether a thyroid nodule is cancerous, because serum thyroglobulin can be elevated in many patients with benign thyroid disease. Although serum calcitonin measurement has been advocated in some European countries as a screening test for medullary thyroid cancer in patients with thyroid nodules, it is not done routinely in the United States. Furthermore, of studies showing that calcitonin screening is helpful, most used pentagastrin stimulation, which is not available in the United States or Canada. Without a known risk factor for medullary thyroid cancer (such as a family history or other manifestation of multiple endocrine neoplasia type 2), routine measurement of serum calcitonin has a low yield, a high false-positive risk, and is not cost effective.
Current guidelines generally recommend further evaluation only for thyroid nodules larger than 1 cm. Nodules smaller than 1 cm in patients with previous radiation exposure, family history of thyroid cancer, or other high-risk clinical findings, such as hoarseness or rapid growth, and those associated with lateral lymphadenopathy may warrant biopsy. Evaluation of multinodular goiters follows similar criteria for biopsy as for solitary thyroid nodules. Rapidly growing goiters or those causing compressive symptoms, such as dysphagia, dysphonia, or dyspnea, have independent indications for thyroidectomy and may not need biopsy unless the results will change the operative plan.
FNA biopsy can be done by palpation, but ultrasound-guided biopsy is associated with fewer nondiagnostic aspirates. To address variability in the reporting of FNA results for thyroid nodules, the National Cancer Institute developed the Bethesda Classification of thyroid cytopathology in 2009, which was updated in 2017. In this system, there are six categories that represent a spectrum for the risk of thyroid cancer. Category I is nondiagnostic, and categories II and VI are benign and malignant, respectively. Benign FNA cytology (category II) has a malignancy rate of up to 3%. A small subset of thyroid nodules with very high-risk ultrasound characteristics will likely have a much higher risk of malignancy even with a benign FNA cytology. Malignant biopsies (category VI) have an accuracy rate of 97% to 99%. Categories III through V nodules have a spectrum of risk that varies by center and depends also on the malignancy rates of the population (“pretest probability”). Category III nodules include nodule with atypia of undetermined significance (AUS) and follicular lesion of undetermined significance (FLUS). Category IV nodules include follicular neoplasm (FN)/suspicious for follicular neoplasm. Hurthle cell neoplasms are included in this category. Category V nodules are those suspicious for malignancy. The reported rates of cancer in these categories are 10% to 30% for AUS/FLUS, 25% to 40% for FN, and 50% to 75% for suspicious for malignancy lesions ( Table 1 ).
| Clinical Situations | RAI | ATD | Surgery |
|---|---|---|---|
| Pregnancy a | — | Preferred, use caution | Acceptable, use caution |
| Comorbidities with increased surgical risk and/or limited life expectancy | Preferred | Acceptable | Contraindicated |
| Inactive GO | Acceptable | Acceptable | Acceptable |
| Active GO | c | Preferred | Preferred |
| Liver disease | Preferred | Use caution | Acceptable |
| Major adverse reactions to ATDs | Preferred | Contraindicated | Acceptable |
| Patients with previously operated or externally irradiated necks | Preferred | Acceptable | Use caution |
| Lack of access to a high-volume thyroid surgeon | Preferred | Acceptable | Use caution |
| Patients with high likelihood of remission (especially women, with mild disease, small goiters, and negative or low-titer TRAB) | Acceptable | Preferred | Acceptable |
| Patients with periodic paralysis | Preferred | Acceptable | Preferred |
| Patients with pulmonary hypertension or congestive heart failure | Preferred | Acceptable | Use caution |
| Elderly with comorbidities | Acceptable | Acceptable | Use caution |
| Thyroid malignancy confirmed or suspected | Contraindicated | b | Preferred |
| One or more large thyroid nodules | b | Acceptable | Preferred |
| Coexisting primary hyperparathyroidism requiring surgery | b | b | Preferred |
a Women considering a pregnancy within 6 months.
b Not first-line therapy but may be acceptable depending on the clinical circumstances.
c RAI is not recommended for those with active, moderate/severe GO, but can be considered with or without glucocorticoids in patients with active, mild GO. ATD , antithyroid drugs; GO , Graves’ orbitopathy; RAI , radioactive iodine From Ross DS, Burch HB, Cooper DS, et al. 2016 American Thyroid Association guidelines for diagnosis and management of hyperthyroidism and other causes of thyrotoxicosis. Thyroid. 2016; 26:1343–1421.
Since the release of the 2015 American Thyroid Association Guidelines, an additional pathologic classification for some thyroid nodules has been created. Noninvasive follicular thyroid neoplasm with papillary-like nuclear features (NIFT-P) represents a subset of thyroid nodules with almost no risk of metastatic spread and is therefore no longer considered a malignancy. The diagnosis requires a complete histopathologic review of the entire nodular capsule to ensure no capsular invasion. NIFT-P is indistinguishable from other follicular lesions on FNA, and therefore surgical removal is required to differentiate these lesions from well-differentiated thyroid cancers. As it is no longer considered malignant, Table 1 demonstrates probabilities of malignancy that are lower because a subset of tumors previously defined as malignant are now considered benign.
For patients with benign or malignant nodules, there is usually a clear treatment plan. For FNA cytology of indeterminate nodules, Bethesda categories III and IV, the decision for operation versus observation is more complex because of the uncertainty for risk of malignancy. Over the past decade, there has been an explosion of research into molecular analysis of these thyroid nodules with indeterminate cytology. Knowing when and whether to use which molecular tests and how to interpret their results has become an important prerequisite for a modern thyroid surgeon.
Molecular testing may be used for patients with indeterminate nodules to help with the decision to operate (or to perform a more aggressive operation) or to observe. It is important to know that molecular testing may not give a simple yes or no answer but usually returns a probability for risk of cancer for the nodule tested.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here