Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
At the time of writing this chapter, COVID-19 is the pandemic of the century, globally affecting macro and micro-economies. Recognition of the economic disruption from outbreaks on the regional level (Ebola viral disease) and global level (COVID-19) has led to justifying novel financial mechanisms to address the macroeconomic impact of uncontrolled communicable diseases. New national and international mechanisms of global collaborations have been established to mitigate the impact of uncontrolled communicable diseases through vaccines in unprecedented time frames, perhaps forever changing how vaccines are valued.
This chapter in past editions covered the components of vaccine economics and analyses to support novel vaccines and strategies from a limited public health perspective. The current COVID-19 outbreak and response additionally demonstrate the dynamic perceptions of vaccine utility in the context of the disruption of all national economies, impacting labor, travel, supply chains, exports, among many other factors measured in the trillions of dollars. Consequently, historical funding models for vaccine discovery, development, and utilization have been upended. Global cooperation has increased exponentially with the recognition of the interdependence of public health and economics when it comes to rapidly transmissible deadly viral agents. A pathogen transmitted from human to human in any part of the world is a potential risk to all parts of the world.
The world had a regional foreshadowing event during the uncontrolled but regionally limited Ebola outbreak in Africa from 2014 to 2016 with a few spillover cases in the US and Europe. Macroeconomic interests of individual nations coalesced toward multinational interests to enhance public-private research collaborations and the acceleration of the development/implementation of novel vaccines with the creation of new funding models. The Coalition for Epidemic Preparedness Innovations (CEPI) evolved as a funding mechanism to catalyze the development of vaccines against emerging communicable agents and teamed with Gavi, the Vaccine, Alliance, and the WHO in the creation of the COVAX facility to address global distribution of SARS-CoV-2 vaccines.
Over the past decade, the threats of pandemics were no longer the purview of ministries of health, but elevated to a transsectoral perspective at the head-of-state level. This transition occurred at a modest pace with SARS-CoV-1, Middle Eastern Respiratory Syndrome (MERS), pandemic influenza, Ebola, and arboviruses such as Zika, Chikungunya, and dengue. But with Covid-19, suddenly there was a global awareness that this was not merely a communicable disease health issue, monitored by ministries of health. Instead, almost all heads of state devoted themselves to addressing Covid-19 at the domestic and international levels. Vaccine diplomacy is providing immunization through international collaborative efforts on an unprecedented scale.
In May 2021, the International Monetary Fund presented a proposal for a $50 billion investment through the support of the access to Covid-19 Tools Accelerator, which includes vaccinating at least 40% of the population in all countries by the end of 2021 and at least 60% by the first half of 2022, among other public health measures. This would deliver substantial potential benefits by hastening an end of the pandemic and restoring economic activity, estimated at a value of $9 trillion. Advanced economies would stand to gain around $1 trillion in additional tax revenues, justifying support as the highest-return public investment ever.
Unlike pandemics, vaccines for endemic diseases have required longer economic deliberations. Vaccination programs have greatly reduced morbidity and mortality worldwide. While new vaccines and the strategic use of existing vaccines offer the prospect of further reductions in disease, policy makers are often constrained by limited public health resources *
* Although private consumers individually judge the value of prevention and manufacturers assess commercial viability, for the purposes of this chapter, we only focus on the public-sector and private philanthropy.
and require assessments of a vaccine’s value to justify and optimize its use. Newer vaccines tend to cost more than older products and those not under patent protection. Studies involving the integration of epidemiologic and economic data are critical to identify the most efficient use of public health resources, to optimize health benefits and to help define research agendas.
Different quantitative techniques have been used to analyze policy decisions related to health outcomes, including those related to vaccine-preventable diseases. Economic analyses, such as cost analysis (CA), cost-benefit analysis (CBA), cost-effectiveness analysis (CEA), and cost-utility analysis (CUA) ( Table 79.1 ) estimate the potential financial requirements and program impact for direct comparisons among vaccination strategies. A systematic assessment of policy options helps to create policies that are based on a set of established criteria and provides transparent accountability for decisions.
| Definition | |
|---|---|
| Cost analysis (CA) | Summation of vaccine program costs including vaccine, administration, adverse events, public health marketing |
| Cost-benefit analysis (CBA) | Computed as a benefit to cost (B:C ratio) equal to cost of prevented disease/costs of program or net benefit equal to cost of prevented disease minus costs of program where prevented disease equals the difference in costs of disease (direct, indirect, and intangibles) without and with vaccine and costs of program equals the cost analysis |
| Cost-effectiveness analysis (CEA) | (Cost of program minus cost of prevented disease) divided by prevented health outcome |
| Cost-utility analysis (CUA) | (Cost of program minus cost of prevented disease) divided by standardized health metric |
These approaches share the characteristics of explicit delineation of possible alternatives, estimation of probabilities and costs associated with each of these alternatives, various health outcome metrics and values, and development of a summary statement of the implications of alternative courses of action. They also involve sensitivity analyses, in which estimated probabilities or costs are varied to determine how sensitive the conclusions are to specified variables.
CA is the basis of all economic studies because it quantifies the costs associated with a given intervention. The costs of the program may include vaccines, vaccine administration, public health surveillance, efforts to address adverse events, and public and vaccine provider education. CBA is an extension of CA that further accounts for the monetary benefits from a policy or program. Benefits are usually calculated as a function of the total cost of a disease in the presence and absence of an intervention. In a CBA, results are usually presented as ratios of the benefits from the intervention to the costs of conducting the program (B:C ratio) or the differences between the two, expressed as a net benefit (cost). If the B:C ratio is greater than 1.0, the intervention is considered cost saving.
Because of the difficulties of assigning economic values for certain outcomes, such as averted deaths and the overall disruption from outbreaks in transportation, labor, education, exports, etc., a full accounting for a CBA is challenging. However, because it can give a summary statement about whether the benefits of a program exceed its costs, irrespective of the character of the outcomes, it can be used to compare health programs with nonhealth programs. CEA and CUA are more useful for comparing different health programs and for avoiding the subjective valuation of intangibles such as death.
In CEA, results are presented in terms of the cost required to achieve a particular health outcome ( Table 79.2 ). Usually no attempt is made to assign an economic value to a prevented death. In general, interventions are considered highly cost effective (good buys) by financial authorities such as the World Bank if the cost per year of life saved is less than three times the annual per capita gross domestic product (GDP). The relative cost effectiveness of a death prevented can therefore depend on the age of the individual, life expectancy, and the consequent number of years of life saved. Ethical considerations are critical, because society must decide on the value of life-years saved at different ages or in populations with different economic means.
| Outcome Metric | Definition |
|---|---|
| Cases | An outcome may be stated as the number of cases that occur or are prevented by an intervention. It does not necessarily account for the various severity states of sequelae. One could further stratify the total number of cases by minor illness, hospitalizations, and long-term sequelae. |
| Deaths | Death may be easily quantified but defies an economic valuation. For this reason, CEAs are often used in health outcome assessments with deaths (or some variation) in the denominator. |
| Years of potential life | YPL is a refinement of the death metric performed by quantifying the total years of life lost or prevented from being lost. It integrates the difference in an expected life span of each person and the age at which a death occurs. YPL may be adjusted by discount and age weights to account for societal preference of time and years of productivity; however, there are considerable ethical considerations in the choice of a weighting scheme. |
| Quality-adjusted life-year (QALY) or disability-adjusted life-year (DALY) | QALY and DALY are further refinements of the YPL metric that integrate mortality and morbidity states. Various health metrics have been developed to help quantify disease burden states to compare disparate health outcomes. QALYs and DALYs integrate mortality, the YPL, and a valuation of the disability from morbidity. They are calculated by adjusting the YPL by the time spent with various disabilities. These units may be discounted and contain an age-weighting factor. The weights are systematically defined and then used as standards for subsequent studies. They offer a common metric to compare many health outcomes. |
CEA is particularly practical when incorporated into a decision analysis of several options to achieve a common outcome (e.g., delivering a second dose of measles vaccine as part of a campaign or as part of a routine schedule to prevent outbreaks). CEA is not suitable to compare investments in alternative health programs with disparate outcomes, for example the cost to prevent a case of measles versus that of hepatitis B. In this situation, CUA is much more useful, because the units of measurement are standardized, albeit with subjective and therefore sometimes controversial, metrics.
CUA is a specific form of CEA in which health outcomes are reduced to a common denominator such as a quality-adjusted life-year (QALY) or a disability adjusted life-year (DALY). The units of each of these outcomes are standardized through a process by which panels of ethicists, researchers and decision makers weigh the value of different morbid states relative to full health for use in studies using either of these metrics. For example, a consensus group might find that two-thirds of a year living with full sight is equivalent to one year living with blindness, effectively equating the quality of the time spent with blindness as 67% of the quality of the time with full sight. The relative proportion of time preference with and without a specific condition allows the formulation of a common health metric unit that can offer direct comparisons of a variety of morbid conditions. There are more than 30 such health outcome metrics that integrate morbidity and mortality. Examples of these are provided in Table 79.2 . A common metric calculated through CUA permits comparisons to be made among interventions aimed at diseases with diverse outcomes, such as death, acute illness, and prolonged disability. Although common outcome metrics facilitate comparisons of many health interventions, numerous subjective, and ethical assumptions underlie their construct, and therefore they are not always easily interpreted.
There are many decisions to be made in designing a quantitative policy analysis. Widespread variability in choices of parameters or metrics has made it difficult to make comparisons among many of the published studies. This variability has previously been criticized. With the active support of philanthropy, there has been active engagement to compile many analyses which offer a compendium of the benefits of individual vaccines and vaccination programs. In the 1990s, a task force in the United States drew up suggested guidelines for carrying out cost-effectiveness studies of preventive services. The journal Vaccine has published an editorial statement on the submission of economic evaluations of vaccines. The US Centers for Disease Control and Prevention (CDC) has published guidelines that are required to be followed to present any economic analysis to the CDC Advisory Committee on Immunization Practices (ACIP). Additionally, the World Health Organization (WHO) has published a guide for the standardization of economic evaluations of immunization programs. , Guidelines help to ensure high quality studies to support the potential advocacy for vaccines and programs.
Economic models also may consider the indirect effects of immunization from the decrease of transmission (herd immunity). As an example, the impact of immunization of US infants with pneumococcal conjugate vaccine on the occurrence of pneumococcal disease in older, unvaccinated populations has been quite large. Additionally, there has been increasing interest in providing different modelers with common data to assess the divergence and convergence of results and identify important nuances for consideration. This approach has been taken with respect to research focusing on rotavirus and human papillomavirus (HPV) vaccines and measles eradication.
As a result of the increased levels of attention being given to the evidence base for new vaccine use, analytical tools and initiatives to assess new vaccine cost effectiveness have been developed. The ProVac Initiative of the Pan American Health Organization (PAHO) is a notable example. The Strategic Multi-Attribute Ranking Tool for Vaccines (SMART-Vaccines) has been developed to capture both quantitative and qualitative explicitly stated values to help make transparent decisions related to vaccine policy. Many attributes, such as impacts on societal disruptions, macroeconomic considerations and development, may be considered. These tools can help National Immunization Technical Advisory Groups (NITAGs) assess available information and make a country- and evidence-based decision on quantitative issues related to vaccines and immunization programs.
Individuals, health care systems, commercial vaccine manufacturers, and society at large all benefit from immunizations. Although the societal perspective theoretically captures all costs and benefits, the costs and benefits of a vaccination program clearly accrue to different budget lines. Results from any analysis would vary from each perspective , so should be explicitly stated.
Vaccines protect against diseases that would otherwise occur at a variable future time. While measles vaccine protects against childhood disease, hepatitis B or human papilloma virus vaccines can prevent liver and cervical cancer, respectively, in adults. As a vaccine investment is made in a time frame different from the eventual benefit, economic analyses frequently discount future effects to take into account the implicit valuation that society has for health and financial costs and benefits over time. Even after accounting for inflation (which affects both costs and potential savings), there is an implicit difference in the value of an event that occurs today versus one that occurs in the future. For example, it is generally felt that 100 infant deaths prevented today is worth more than 100 deaths prevented 1 year from today. While this is an ethical and subjective valuation, there is general agreement among economists to discount both costs and benefits at the same rate, typically from 3% to 10% per year, most often at the lower end of the range. Mathematically, for the example above at a 3% discount rate, 100 deaths prevented today is equivalent to 103 prevented 1 year from now. Study results may be presented as both discounted and undiscounted to see the influence of this subjective assumption.
Estimates of the potential impact of a vaccine on disease may be derived from surveillance data, extrapolations from other representative populations, clinical trials (e.g., vaccine probe studies ), or the results of mathematical models that simulate the natural history of microbes, infections, degrees of severity and population mixing, among other variables. Analyses might need to be specific for age group, population and occupational group, risk of exposure (e.g., health care workers), and outcome, including the timing of the outcome (e.g., death in the next year vs death 50 years in the future). An understanding of the different rates of transmission of microbial agents, given by generation time, and the reproductive rate, is essential to appreciate the direct and indirect impact that vaccines can have on populations. Disease burden is frequently underestimated. Recent publications have emphasized the severe limitations of estimating public health impact of vaccines which have considered only etiologically confirmed outcomes. Outcome sensitivity may be reduced by study enrollment case definitions, study analytic population definitions, limited test sensitivity, and the requirement for patients to have a biologic specimen obtained. Use of clinically defined outcomes—such as all-cause pneumonia for Hib and pneumococcal vaccines, all-cause acute gastroenteritis for rotavirus vaccines, and fever for dengue vaccines—overcome many of these limitations and often identify rate reductions several fold-higher than documented with etiologically confirmed outcomes. ,
Outcomes ( Table 79.2 ) may be stated as the number of cases or deaths that occur or are prevented by an intervention. Years of potential life (YPL) represents a refinement of the death metric that quantifies the total years of life lost or prevented from being lost. It integrates the difference in an expected lifespan of each individual and the age at which death occurs. QALYs, which measure the number of years of healthy life lived, or the currently used WHO metric, DALYs, which measure the years of healthy life lost, are further refinements of YPL that integrate a valuation of various morbid states.
QALYs and DALYs are complementary concepts. Both metrics are derived from the product of the number of years lived and by the quality of those years. QALYs use utility weights of health states; DALYs use disability weights to reflect the burden of the same states. For example, if the utility of blindness is 0.67, the disability weight of blindness is 1–0.67 = 0.33. Disregarding age weighting and discounting, all other things being equal, a person who is blind for 50 years has 0.67 × 50 = 33.5 QALYs compared to 50 QALYs for a person with full sight. The condition of blindness contributed to 50–33.5 = 16.5 DALYs. These metrics defy simplicity, are derived from many subjective inputs, and might not be readily understood by some policymakers; however, they offer a common metric to compare disparate health outcomes.
Direct and indirect costs and benefits are usually counted. Direct costs include those related to medical treatment and to administering the vaccine, including ancillary costs such as screening for target groups. Indirect costs include lost wages of those ill and/or their caregivers. Intangible costs, such as pain and suffering or death, are difficult to quantify; however, they may be implicitly valued as the denominator of a CEA or CUA (e.g., as cost per death prevented), allowing the reader to apply his or her own value to the stated health outcome. An alternative valuation tool is the concept of willingness to pay, where individuals are asked what they would be willing to pay to achieve alternative health outcomes given different economic circumstances and probabilities. These collective responses are integrated with actual probabilities of risk to gauge individual or societal valuation of prevention.
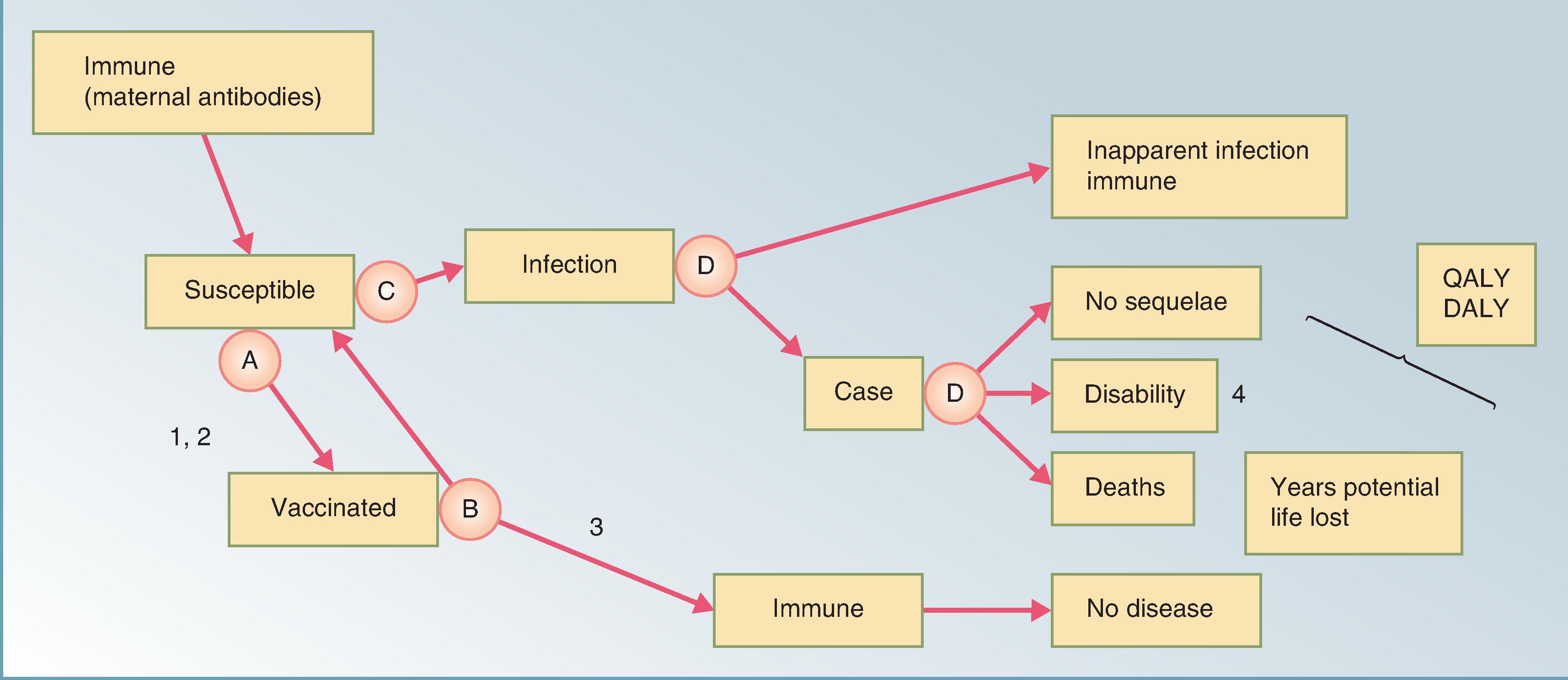
Vaccine effectiveness, coverage, adverse effects, and the potential for indirect benefits to accrue to unvaccinated persons owing to the reduction of transmission are all program characteristics that should be accounted when evaluating the cost-effectiveness of vaccine initiatives. A schematic model of a vaccine program is shown in Fig. 79.1 . Each component has, an associated cost or a benefit exists.
Quantitative policy analysis provides the opportunity to evaluate the likely outcomes from a range of plausible epidemiologic and economic assumptions from those postulated in the base case analysis. This is particularly relevant when the value of a parameter used in an analysis is uncertain and must be estimated. Testing a range of values for one or more uncertain assumptions can help identify the variables to which the conclusion is most sensitive. In turn, defining those variables’ values can be the focus of further research to help guide a policy.
In general, economic studies have shown that immunization is an excellent investment. For example routine childhood immunization of the 2009 US birth cohort would prevent ∼42,000 early deaths and 20 million cases of disease, and save $13.5 billion in direct costs and $68.8 billion in total societal costs, respectively, a societal benefit-cost ratio of 10.1. Consideration of particular strategies in certain populations can show considerable variation, as demonstrated in Table 79.3 , which summarizes selected studies of different vaccines. With the advancement of computational capabilities, modeling software, the maturation of health economics applications to public sector health programs, and the expansion of the repertoire of new vaccines in recent years, the number of economic studies to evaluate vaccines has greatly expanded. Given that there have been more than 1000 economic analyses published since the 1980s, we only present results of selected studies to highlight the relevant issues for consideration. Unless otherwise indicated, all costs are in U.S. dollars. A summary table of cost-effectiveness studies of other selected health interventions is included for reference ( Table 79.4 ). ,
| Reference | Type | Scope | Result | Comment |
|---|---|---|---|---|
| General | ||||
| Brenzel (2006) | CEA | Global | Reviewed 30 studies; average cost to fully immunize a child with DPT, BCG, polio and measles vaccines $22, ranging from $8 (Tanzania, fixed site) to $33 (Cameroon, national campaigns) | All costs adjusted to 1992 dollars (equivalents) |
| Miller (2000) | CEA | Global | Country-specific analysis for vaccination against hepatitis B, Haemophilus influenzae type b, Streptococcus pneumoniae , and rotavirus. The global cost per life-year saved ranged from $29 to $150, with great variation by income and economic groups | Costs of vaccine were based on relative wealth of individual countries |
| Zhou (2005) | CBA | United States | Benefit-to-cost ratios of seven routine childhood immunization results in direct and societal benefit-to-cost ratios of 5.3 and 16.5, respectively | |
| Bacille Calmette-Guérin | ||||
| Trunz (2006) | CEA | Global | At $2–3 per dose, BCG vaccination costs $206 ($150–$272) per year of healthy life gained | |
| Cholera | ||||
| Schaetti et al. (2012) | CEA | Zanzibar | During a mass vaccination campaign, the incremental CE of oral cholera vaccine compared to no vaccine was US$750,000 per death, US$6000 per case, and US$30,000 per DALY averted | Static model used |
| Dengue | ||||
| Durham (2013) | CEA | Brazil | Dengue vaccine estimated to be CE at vaccination costs up to US$534 and cost saving at costs up to $207 | Base-case assumption of 70% vaccine efficacy against all dengue combined |
| Diphtheria–Tetanus–acellular Pertussis | ||||
| Ekwueme (2000) | CBA | United States | Benefit-to-cost ratios for acellular pertussis vaccine from a societal and health care system perspective were 27:1 and 9:1, respectively, but were marginally more expensive than whole-cell vaccine | Additional benefit of acellular vaccine is increased consumer confidence |
| Group B Streptococcus | ||||
| Oster et al. (2014) | CEA | United States | Incremental CE of routine maternal immunization added to screening and intrapartum antibiotic prophylaxis would be US$91,321 per QALY gained. | Base case assumed 85% coverage with vaccine cost of US$100 per person |
| Hepatitis A and B | ||||
| CDC (2006) | CEA | United States | Cost effectiveness was estimated at $173,000 per life-year gained and $24,000 per QALY gained | Substantially more cost effective when decreased transmission through herd effects are included |
| Human papillomavirus | ||||
| Jit et al. (2015) | CEA | Global | HPV vaccine very cost effective in 158 of 179 countries; vaccinating 58 million 12year-old girls would prevent 690,000 cases of cervical cancer at a cost of US$4 billion | Model: Papillomavirus Rapid Interface for Modeling and Economics (PRIME). Assumed lifelong protection, vaccination presexual debut |
| Graham et al. (2015) | CEA | Canada | US$94.49 saved and 0.04 QALYs gained per vaccinated male age 12 years | Base case vaccine efficacy of 84%, coverage 50% |
| Laprise et al. (2014) | CEA | Canada | All 2- and 3-dose girls only and girls+boys strategies had a CE ratio <$40,000/QALY gained. 2-dose girls only had the lowest cost/QALY gained at $7900-$10,400 | Incremental value of 3-dose girls only and 2-dose girls+boys varied depending on parameters. 3-dose girls+boys greater than US$100,000/QALY gained under most scenarios. |
| Influenza | ||||
| Skedgel et al. (2011) | CEA | Canada | Compared to vaccinating only pregnant women with comorbidities, the incremental cost effectiveness of vaccinating all pregnant women was <UW$40,000 per QALY gained. | Net cost of universal vaccination was <US$10 per pregnant woman. |
| Malaria | ||||
| Seo et al. (2014) | CEA | Malawi | The incremental CE of adding RTS,S malaria vaccine to existing strategies (primarily bednets) was US$145 per DALY averted. | The incremental CE was sensitive to vaccine’s duration of protection |
| Measles-Mumps-Rubella | ||||
| Hatziandreu (1994) | CBA | United States | B:C ratio 16.3 for direct costs; 21.3 for total costs | Also considered B:C of individual components: measles 17.2, 17.2 for direct, total costs; mumps 6.1, 13.0; rubella 4.5, 11.1 |
| Measles | ||||
| Shepard (1994) | CUA | Global | Vaccination results in $17 per DALY, one of the most cost-effective health interventions in developing countries | |
| Neisseria meningitidis | ||||
| Trotter et al. (2002) | CEA | England and Wales | The cost per life-year saved for a vaccination campaign was 6259 pound sterling; school-based campaigns were more cost effective than vaccine delivered through physician practices | From health care provider perspective |
| Tu HA et al. (2014) | CEA | Canada | Over the lifetime of an Ontario birth cohort, vaccine would cost C$4.8 million per QALY gained | Baseline with 10-year duration of protection, vaccine cost of C$75/dose |
| Pneumococcal (conjugate) | ||||
| Ray (2002) | CEA | Australia, Europe, United States | Break-even vaccine cost ranged from the health care system perspective averaged $17 (range, $4–37). | Reviewed multiple studies and scenarios with consideration of multiple outcomes and herd effects. |
| Respiratory syncytial virus | ||||
| Meijboom (2012) | CEA | Netherlands | RSV immunization with a three-dose infant schedule would cost 34,142 euros per QALY gained | Over a full year and with 91% coverage, vaccination would prevent 1.5 deaths and 544 hospitalizations |
| Typhoid (polysaccharide) | ||||
| Carias (2015) | CEA | Uganda | Over 3 years following a one-time vaccination campaign, typhoid vaccine would cost US$484 per DALY and US$341 per case averted | Results sensitive primarily to preventable disease burden and vaccine cost |
| Varicella and Zoster | ||||
| Damm (2015) | CE, CBA | Various | Review of multiple studies one-dose varicella vaccination of young children would save 1.61 to 19.33 per unit spent; Zoster vaccine mostly CE | Societal perspective; results dependent on importance of exogenous boosting |
| Intervention | Median Cost/QALY (US$) |
|---|---|
| Immunizations and chemoprophylaxis | |
| Immunizations and vaccinations | 1500 |
| Pharmaceuticals for asymptomatic persons (e.g., hormone replacement) | 13,000 |
| Screening tests | |
| Cardiovascular disease | 3300 |
| Neoplasms | 18,500 |
| Osteoporosis screening and treatment | 13,000 |
| Screening blood donors against pathogens | 355,000 |
| Other disease screenings | 11,500 |
| Counseling | |
| HIV-risk behavior | 1200 |
| Cardiovascular disease risk | 74,000 |
| Presurgical autologous blood donation | 730,000 |
Economic models are becoming more important to decision-making. A survey of NITAGs published during 2010 found that over 90% considered financial aspects when issuing recommendations. The UK and Australia NITAGs use the results of economic analysis as a criteria for public vaccine funding. Vaccine recommendations by the World Health Organization now routinely include data on economic impact. Gavi, the Vaccine Alliance uses four categories of criteria and indicators to determine which vaccines it will support, one of which is cost and value as measured by total procurement costs, incremental in-country operational cost per vaccinated person, and procurement cost per death or case averted. From another perspective, vaccine manufacturers may consider the results of economic analyses when setting vaccine prices.
A single study evaluated the economic impact of the routine US childhood immunization schedule, including DTaP (diphtheria and tetanus toxoids and acellular pertussis vaccine), Hib ( Haemophilus influenzae type b) conjugate vaccine, IPV (inactivated poliovirus vaccine), MMR (measles, mumps, rubella) vaccine, hepatitis B vaccine, and varicella vaccine. Routine childhood immunization with these seven vaccines was evaluated as cost-saving, with a societal B:C ratio of 16.5.
Many studies have addressed immunization in developing countries using the vaccines against six diseases initially used in the WHO’s Expanded Programme on Immunization (EPI): Bacillus Calmette-Guérin (BCG) for tuberculosis (TB), DTP (diphtheria and tetanus toxoids and whole cell pertussis vaccine), OPV (oral poliovirus vaccine), and measles. They generally have demonstrated low costs per immunized child and favorable B:C ratios. Studies published in 2000 in Ethiopia, Morocco, Bangladesh, and Côte d’Ivoire indicated a total cost of less than $25 to fully immunize a child with basic EPI vaccines. ,
The Disease Control Priorities Project has estimated that increased coverage from 70% to 79% with the traditional EPI program in South Asia would cost $8 per DALY averted and that a second opportunity for measles vaccination would cost $1-$5 per DALY averted in sub-Saharan Africa. ,
BCG vaccine primarily prevents disseminated TB in infants and young children but has little, if any, impact on disease transmission. Development of highly cost-effective strategies in industrialized countries based on case-finding and treatment have made BCG vaccination less competitive as an effective intervention against TB. Because BCG vaccination complicates identification of TB exposure, many countries have discontinued routine BCG vaccination. A B:C ratio of 0.13 for revaccination of schoolchildren was calculated in Japan and represented a very low level of cost effectiveness. It was subsequently found that the cost of treating a case of pediatric TB in Japan was much lower than the cost of preventing cases through vaccination. ,
In developing countries with high rates of childhood TB, BCG vaccination of infants might represent a better investment. In 2006, a meta-analysis and assessment of the cost effectiveness of BCG vaccination to prevent childhood tuberculous meningitis and miliary TB concluded that BCG vaccination of infants in Southeast Asia, sub-Saharan Africa, and the western Pacific would cost $206 per year of healthy life gained.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here