Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Diuretics increase the rate of urine volume output, as the name implies. Most diuretics also increase the urinary excretion of solutes, especially sodium and chloride. In fact, most diuretics that are used clinically act by decreasing renal tubular sodium reabsorption, which causes natriuresis (increased sodium output), in turn causing diuresis (increased water output). That is, in most cases, increased water excretion occurs secondary to inhibition of tubular sodium reabsorption because sodium remaining in the tubules acts osmotically to decrease water reabsorption. Because renal tubular reabsorption of many solutes, such as potassium, chloride, magnesium, and calcium, is also influenced secondarily by sodium reabsorption, many diuretics raise the renal excretion of these solutes as well.
The most common clinical use of diuretics is to reduce extracellular fluid volume, especially in diseases associated with edema and in hypertension. As discussed in Chapter 25 , loss of sodium from the body mainly decreases extracellular fluid volume; therefore, diuretics are usually administered in clinical conditions in which extracellular fluid volume is expanded.
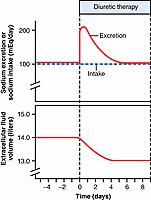
Some diuretics can increase urine output more than 20-fold within a few minutes after they are administered. However, the effect of most diuretics on renal output of salt and water subsides within a few days due to activation of compensatory mechanisms initiated by decreased extracellular fluid volume ( Figure 32-1 ). For example, a decrease in extracellular fluid volume may reduce arterial pressure and glomerular filtration rate (GFR) and increase renin secretion and angiotensin II (Ang II) formation; all these responses together eventually override the chronic effects of the diuretic on urine output. Thus, in the steady state, urine output becomes equal to intake, but only after reductions in arterial pressure and extracellular fluid volume have occurred, relieving the hypertension or edema that prompted the use of diuretics in the first place.
The many diuretics available for clinical use have different mechanisms of action and, therefore, inhibit tubular reabsorption at different sites along the renal nephron. The general classes of diuretics, their mechanisms of action, and their tubular sites of action are shown in Table 32-1 .
| Class of Diuretic (examples) | Mechanism of Action | Tubular Site of Action |
|---|---|---|
| Osmotic diuretics (mannitol) | Inhibit water and solute reabsorption by increasing osmolarity of tubular fluid | Mainly proximal tubules |
| Loop diuretics (furosemide, bumetanide) | Inhibit Na + -K + -Cl − co-transport in luminal membrane | Thick ascending loop of Henle |
| Thiazide diuretics (hydrochloro-thiazide, chlorthalidone) | Inhibit Na + -Cl − co-transport in luminal membrane | Early distal tubules |
| Carbonic anhydrase inhibitors (acetazolamide) | Inhibit H + secretion and HCO 3 − reabsorption, which reduces Na + reabsorption | Mainly proximal tubules |
| Aldosterone antagonists (spironolactone, eplerenone) | Inhibit action of aldosterone on tubular receptor, decrease Na + reabsorption, decrease K + secretion | Collecting tubules |
| Sodium channel blockers (triamterene, amiloride) | Block entry of Na + into Na + channels of luminal membrane, decrease Na + reabsorption, decrease K + secretion | Collecting tubules |
Injection of substances into the blood stream that are filtered by the glomerular capillaries but are not easily reabsorbed by the renal tubules, such as urea, mannitol, and sucrose, causes a marked increase in the concentration of osmotically active molecules in the tubules. The osmotic pressure of these solutes then reduces water reabsorption, flushing large amounts of tubular fluid into the urine.
Large volumes of urine are also formed in certain diseases associated with excess solutes that fail to be reabsorbed from the tubular fluid. For example, when blood glucose concentration rises to high levels in diabetes mellitus, the increased filtered load of glucose into the tubules exceeds their capacity to reabsorb glucose (i.e., exceeds their transport maximum for glucose). Above a plasma glucose concentration of about 250 mg/dl, little of the extra glucose is reabsorbed by the tubules; instead, the excess glucose remains in the tubules, acts as an osmotic diuretic, and increases urine flow rate. Therefore, one of the hallmarks of uncontrolled diabetes mellitus is polyuria (frequent urination), which is balanced by a high level of fluid intake (polydipsia) secondary to dehydration, increased extracellular fluid osmolarity, and activation of the thirst mechanism.
Furosemide, ethacrynic acid, and bumetanide are powerful diuretics that decrease reabsorption in the thick ascending limb of the loop of Henle by blocking the 1-sodium, 2-chloride, 1-potassium co-transporter located in the luminal membrane of the epithelial cells (see Figure 28-9 ). These loop diuretics are among the most powerful of the clinically used diuretics.
By blocking sodium-chloride-potassium co-transport in the luminal membrane of the loop of Henle, the loop diuretics increase urine output of sodium, chloride, potassium, and other electrolytes, as well as water, for two reasons: (1) they greatly increase the quantities of solutes delivered to the distal parts of the nephrons, and these solutes act as osmotic agents to prevent water reabsorption; and (2) they disrupt the countercurrent multiplier system by decreasing absorption of ions from the loop of Henle into the medullary interstitium, thereby decreasing the osmolarity of the medullary interstitial fluid. Therefore, loop diuretics impair the ability of the kidneys to concentrate or dilute the urine. Urinary dilution is impaired because the inhibition of sodium and chloride reabsorption in the loop of Henle causes more of these ions to be excreted, along with increased water excretion. Urine concentrating ability is impaired because the renal medullary interstitial fluid concentration of these ions, and therefore renal medullary osmolarity, is reduced. Consequently, reabsorption of fluid from the collecting ducts is decreased, so the maximal concentrating ability of the kidneys is also greatly reduced. In addition, decreased renal medullary interstitial fluid osmolarity reduces reabsorption of water from the descending loop of Henle. Because of these multiple effects, 20% to 30% of the glomerular filtrate may be delivered into the urine, causing urine output, under acute conditions, to be as great as 25 times normal for at least a few minutes.
The thiazide derivatives, such as chlorothiazide, act mainly on the early distal tubules to block the sodium-chloride co-transporter in the luminal membrane of the tubular cells (see Figure 28-10 ). Under favorable conditions, these agents may cause a maximum of 5% to 10% of the glomerular filtrate to pass into the urine, which is about the same amount of sodium normally reabsorbed by the distal tubules.
Acetazolamide inhibits the enzyme carbonic anhydrase , which is critical for reabsorption of bicarbonate (HCO 3 − ) in the renal tubules, as discussed in Chapter 31 . Carbonic anhydrase is especially abundant in the proximal tubule, the primary site of action of carbonic anhydrase inhibitors. Some carbonic anhydrase is also present in other tubular cells, such as in the intercalated cells of the collecting tubule.
Because hydrogen ion (H + ) secretion and HCO 3 − reabsorption in the proximal tubules are coupled to sodium reabsorption through the sodium-hydrogen ion counter-transport mechanism in the luminal membrane, decreasing HCO 3 − reabsorption also reduces sodium reabsorption. The blockage of sodium and HCO 3 − reabsorption from the tubular fluid causes these ions to remain in the tubules and act as an osmotic diuretic. Predictably, a disadvantage of the carbonic anhydrase inhibitors is that they cause some degree of acidosis because of the excessive loss of HCO 3 − in the urine.
Spironolactone and eplerenone are mineralocorticoid receptor antagonists that compete with aldosterone for receptor-binding sites in the collecting tubule and collecting duct epithelial cells and, therefore, can decrease the reabsorption of sodium and secretion of potassium in these tubular segments (see Figure 28-12 ). As a consequence, sodium remains in the tubules and acts as an osmotic diuretic, causing increased excretion of water, as well as sodium. Because these drugs also block the effect of aldosterone to promote potassium secretion in the tubules, they decrease the excretion of potassium.
Mineralocorticoid receptor antagonists also cause movement of potassium from the cells to the extracellular fluid. In some cases, this movement causes extracellular fluid potassium concentration to increase excessively. For this reason, spironolactone and other mineralocorticoid receptor antagonists are referred to as potassium-sparing diuretics . Many of the other diuretics cause loss of potassium in the urine, in contrast to the mineralocorticoid receptor antagonists, which spare the loss of potassium.
Amiloride and triamterene also inhibit sodium reabsorption and potassium secretion in the collecting tubules, similar to the effects of spironolactone. However, at the cellular level, these drugs act directly to block the entry of sodium into the sodium channels of the luminal membrane of the collecting tubule epithelial cells (see Figure 28-12 ). Because of this decreased sodium entry into the epithelial cells, there is also decreased sodium transport across the cells’ basolateral membranes and, therefore, decreased activity of the sodium-potassium–adenosine triphosphatase pump (Na + -K + ATPase pump). This decreased activity reduces the transport of potassium into the cells and ultimately decreases the secretion of potassium into the tubular fluid. For this reason, the sodium channel blockers are also potassium-sparing diuretics and decrease the urinary excretion rate of potassium.
Diseases of the kidneys are among the most important causes of death and disability in many countries throughout the world. For example, in 2018, more than 14% of adults in the United States, or more than 30 million people, were estimated to have chronic kidney disease, and many more millions have acute renal injury or less severe forms of kidney dysfunction.
Severe kidney diseases can be divided into two main categories:
Acute kidney injury (AKI), in which there is an abrupt loss of kidney function within a few days. The term acute renal failure is usually reserved for severe acute kidney injury, in which the kidneys may abruptly stop working entirely or almost entirely, necessitating renal replacement therapy such as dialysis, as discussed later in this chapter. In some cases, patients with AKI may eventually recover nearly normal kidney function.
Chronic kidney disease (CKD), in which there is progressive loss of function of more and more nephrons that gradually decreases overall kidney function.
Within these two general categories, there are many specific kidney diseases that can affect the kidney blood vessels, glomeruli, tubules, renal interstitium, and parts of the urinary tract outside the kidney, including the ureters and bladder. In this chapter, we discuss specific physiologic abnormalities that occur in a few of the more important types of kidney diseases.
The causes of AKI are often divided into three main categories:
AKI resulting from decreased blood supply to the kidneys. This condition is often referred to as prerenal AKI to reflect an abnormality originating outside the kidneys. For example, prerenal AKI can be a consequence of heart failure with reduced cardiac output and low blood pressure or conditions associated with diminished blood volume and low blood pressure, such as severe hemorrhage.
Intrarenal AKI resulting from abnormalities within the kidney itself, including those that affect the blood vessels, glomeruli, or tubules.
Postrenal AKI , resulting from obstruction of the urinary collecting system anywhere from the calyces to the outflow from the bladder. The most common causes of obstruction of the urinary tract outside the kidney are kidney stones, caused by precipitation of calcium, urate, or cystine.
In some important causes of AKI, such as sepsis, prerenal (e.g., reduced blood pressure) and intrarenal (endothelial and tubular injury) abnormalities may occur simultaneously.
The kidneys normally receive an abundant blood supply of about 1100 ml/min, or about 20% to 25% of the cardiac output. The main purpose of this high blood flow to the kidneys is to provide enough plasma for the high rates of glomerular filtration needed for effective regulation of body fluid volumes and solute concentrations. Therefore, decreased renal blood flow is usually accompanied by decreased GFR and decreased urine output of water and solutes. Consequently, conditions that acutely diminish blood flow to the kidneys usually cause oliguria , which refers to diminished urine output below the level of intake of water and solutes. This condition causes accumulation of water and solutes in the body fluids. If renal blood flow is markedly reduced, total cessation of urine output can occur, a condition referred to as anuria .
As long as renal blood flow does not fall below about 20% to 25% of normal, AKI can usually be reversed if the cause of the ischemia is corrected before damage to the renal cells has occurred. Unlike some tissues, the kidney can endure a relatively large reduction in blood flow before there is major damage to the renal cells. The reason for this phenomenon is that as renal blood flow is reduced, the GFR and amount of sodium chloride filtered by the glomeruli (as well as the filtration rate of water and other electrolytes) are reduced. This decreases the amount of sodium chloride that must be reabsorbed by the tubules, which use most of the energy and oxygen consumed by the normal kidney. Therefore, as renal blood flow and GFR fall, renal oxygen consumption is also reduced. As the GFR approaches zero, oxygen consumption of the kidney approaches the rate that is required to keep the renal tubular cells alive when they are not reabsorbing sodium. When blood flow is reduced below this basal requirement, which is usually less than 20% to 25% of the normal renal blood flow, the renal cells become hypoxic, and further decreases in renal blood flow, if prolonged, will cause damage or even death of the renal cells, especially the tubular epithelial cells.
If the cause of prerenal AKI is not corrected, and ischemia of the kidney persists longer than a few hours, this type of renal failure can evolve into intrarenal AKI, as discussed later. Acute reduction of renal blood flow is a common cause of AKI in hospitalized patients, especially those who have sustained severe injuries. Table 32-2 shows some of the common causes of decreased renal blood flow and prerenal AKI.
| Intravascular Volume Depletion |
| Hemorrhage (e.g., trauma, surgery, postpartum, gastrointestinal) |
| Diarrhea or vomiting |
| Burns |
| Cardiac Failure |
| Myocardial infarction |
| Valvular damage |
| Peripheral vasodilation and resultant hypotension |
| Anaphylactic shock |
| Anesthesia |
| Sepsis, severe infections |
| Primary renal hemodynamic abnormalities |
| Renal artery stenosis, embolism, or thrombosis of renal artery or vein |
Abnormalities that originate in the kidney and that abruptly diminish urine output fall into the general category of intrarenal AKI . This category of AKI can be further divided into the following: (1) conditions that injure the glomerular capillaries or other small renal vessels; (2) conditions that damage the renal tubular epithelium; and (3) conditions that cause damage to the renal interstitium. This type of classification refers to the primary site of injury, but because the renal vasculature and tubular system are functionally interdependent, damage to the renal blood vessels can lead to tubular damage, and primary tubular damage can lead to damage of the renal blood vessels. Some causes of intrarenal acute kidney injury are listed in Table 32-3 .
| Small vessel and/or glomerular injury |
| Vasculitis (polyarteritis nodosa) |
| Cholesterol emboli |
| Malignant hypertension |
| Acute glomerulonephritis |
| Tubular epithelial injury (tubular necrosis) |
| Acute tubular necrosis due to ischemia |
| Acute tubular necrosis due to toxins (e.g., heavy metals, ethylene glycol, insecticides, poison mushrooms, carbon tetrachloride) |
| Renal interstitial injury |
| Acute pyelonephritis |
| Acute allergic interstitial nephritis |
Acute glomerulonephritis is a type of intrarenal AKI usually caused by an abnormal immune reaction that damages the glomeruli. In about 95% of patients with this disease, damage to the glomeruli occurs 1 to 3 weeks after an infection elsewhere in the body, often caused by certain types of group A beta streptococci. The infection may have been a streptococcal sore throat, streptococcal tonsillitis, or even streptococcal infection of the skin. It is not the infection itself that damages the kidneys. Instead, over a few weeks, as antibodies develop against the streptococcal antigen, the antibodies and antigen react with each other to form an insoluble immune complex that becomes entrapped in the glomeruli, especially in the basement membrane portion of the glomeruli.
Once the immune complex has been deposited in the glomeruli, many of the glomerular cells begin to proliferate, but mainly the mesangial cells that lie between the endothelium and epithelium. In addition, large numbers of white blood cells become entrapped in the glomeruli. Many of the glomeruli become blocked by this inflammatory reaction, and those that are not blocked usually become excessively permeable, allowing protein and red blood cells to leak from the blood of the glomerular capillaries into the glomerular filtrate. In severe cases, total or almost complete renal shutdown occurs.
The acute inflammation of the glomeruli usually subsides in about 2 weeks and, in most patients, the kidneys return to almost normal function within the next few weeks to few months. Sometimes, however, many of the glomeruli are destroyed beyond repair and, in a small percentage of patients, progressive renal deterioration continues indefinitely, leading to CKD, as described in a subsequent section of this chapter.
Another cause of intrarenal acute renal failure is tubular necrosis , which means destruction of epithelial cells in the tubules. Some common causes of tubular necrosis are as follows: (1) severe ischemia and inadequate supply of oxygen and nutrients to the tubular epithelial cells; and (2) poisons, toxins, or medications that destroy the tubular epithelial cells.
Severe ischemia of the kidney can result from circulatory shock or other disturbances that severely impair the blood supply to the kidneys. If the ischemia is severe enough to seriously impair the delivery of nutrients and oxygen to the renal tubular epithelial cells, and if the insult is prolonged, damage or eventual destruction of the epithelial cells can occur. When this damage occurs, tubular cells slough off and plug many of the nephrons so that there is no urine output from the blocked nephrons; the affected nephrons often fail to excrete urine, even when renal blood flow is restored to normal, as long as the tubules remain plugged. The most common causes of ischemic damage to the tubular epithelium are the prerenal causes of AKI associated with circulatory shock, as discussed earlier in this chapter.
There is a long list of renal poisons and medications that can damage the tubular epithelium and cause AKI. Some of these substances are carbon tetrachloride, heavy metals (e.g., mercury and lead), ethylene glycol (which is a major component in antifreeze), various insecticides , some medications (e.g., tetracyclines) used as antibiotics, and cis-platinum , used in treating certain cancers. Each of these substances has a specific toxic action on the renal tubular epithelial cells, causing death of many of them. As a result, the epithelial cells slough away from the basement membrane and plug the tubules. In some cases, the basement membrane also is destroyed. If the basement membrane remains intact, new tubular epithelial cells can grow along the surface of the membrane, so the tubule may repair itself within 10 to 20 days.
Multiple abnormalities in the lower urinary tract can block or partially block urine flow and therefore lead to AKI, even when the kidneys’ blood supply and other functions are initially normal. If the urine output of only one kidney is diminished, no major change in body fluid composition will occur because the contralateral kidney can increase its urine output sufficiently to maintain relatively normal levels of extracellular electrolytes and solutes, as well as normal extracellular fluid volume. With this type of renal injury, normal kidney function can be restored if the basic cause of the problem is corrected within a few hours. However, chronic obstruction of the urinary tract that lasts for several days or weeks can lead to irreversible kidney damage. Some of the causes of postrenal AKI include the following: (1) bilateral obstruction of the ureters or renal pelvises caused by large stones or blood clots; (2) bladder obstruction; and (3) obstruction of the urethra.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here