Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
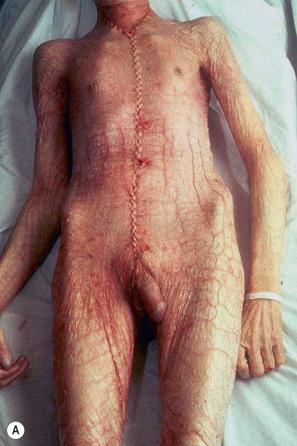
The term ichthyosis (Gr. ichthys , fish) is applied to a number of heterogeneous genetic disorders characterized by permanent and generalized abnormal keratinization. The clinical features range from mild involvement, often passed off as ‘dry skin’ (xerosis), through to severe widespread scaly lesions causing much discomfort and social embarrassment ( Fig. 3.1 ). The scales are shed as clusters rather than as single cells as is the norm. The pathogenesis of the ichthyoses is heterogeneous and ultimately depends upon abnormal differentiation resulting in impaired desquamation of corneocytes (retention hyperkeratosis) or increased corneocyte production (hyperproliferative hyperkeratosis). In some ichthyotic diseases these alterations cause hypohidrosis with dysregulation of the body temperature, abnormal epidermal barrier function, or skin infections.
Originally, ichthyotic skin disorders were classified into noncongenital ichthyoses developing 4 weeks after birth and congenital ichthyoses presenting with a collodion membrane or ichthyosiform erythroderma at birth or within 4 weeks later. As too many exceptions and variations had to be notified a new classification appeared mandatory. A consensus conference on ichthyosis in Sorèz 2009 proposed a differentiation between common and rare ichthyoses, and between syndromic and nonsyndromic forms ( Tables 3.1 and 3.2 ).
| Common ichthyoses | ||
| Ichthyosis vulgaris | semi-dominant | FLG |
| Nonsyndromic recessive X-linked ichthyosis (RXLI) | XR | STS |
| Autosomal recessive congenital ichthyosis (ARCI) | ||
| Lamellar ichthyosis (LI) | AR | TGM1 / NIPAL4 / ALOX12B / ABCA12 / PNPLA1 / CERS3 / LIPH |
| Congenital ichthyosiform erythroderma (CIE) | AR | ALOXE3 / ALOX12B / ABCA12 / CYP4F22 / NIPAL4 / TGM1 / PNPLA1 / CERS3 / LIPH |
| Self-healing collodion baby (SICI) | AR | TGM1 / ALOXE3 / ALOX12B |
| Acral self-healing collodion baby | AR | TGM1 |
| Bathing suit ichthyosis | AR | TGM1 |
| Harlequin ichthyosis | AR | ABCA12 |
| Keratinopathic ichthyosis (KPI) | ||
| Epidermolytic ichthyosis (EI) | AD | KRT1 / KRT10 |
| Superficial epidermolytic ichthyosis (SEI) | AD | KRT2 |
| Annular epidermolytic ichthyosis | AD | KRT1 / KRT10 |
| Congenital reticular ichthyosiform erythroderma (CRIE) | AD | KRT10 (KRT 1) |
| Ichthyosis Curth-Macklin | AD | KRT1 |
| Autosomal recessive epidermolytic ichthyosis | AR | KRT10 |
| Other nonsyndromic forms | ||
| Loricrin keratoderma | AD | LOR |
| Erythrokeratodermia variabilis | AD (AR) | GJB3 / GJB4 |
| Inflammatory peeling skin disease (PSS type B) | AR | CDSN |
| Exfoliative ichthyosis | AR | CSTA |
| Keratosis linearis-ichthyosis congenita-keratoderma (KLICK) | AR | POMP |
| Mode of inheritance | Gene | |
|---|---|---|
| X-linked ichthyosis syndromes | ||
| Recessive X-linked ichthyosis | XR | STS (and others) |
| Ichthyosis follicularis alopecia photophobia | XR | MBTPS2 |
| Conradi-Hünermann-Happle syndrome (CDPX2) | XD | EBP |
| Autosomal ichthyosis syndromes with prominent hair abnormalities | ||
| Netherton syndrome | AR | SPINK5 |
| Severe dermatitis-multiple allergies-metabolic wasting (SAM) | AR | DSG1 |
| Ichthyosis with hypotrichosis | AR | ST14 |
| Neonatal ichthyosis-sclerosing cholangitis (NISCH) | AR | CLDN1 |
| Autosomal ichthyosis syndromes with prominent neurologic signs | ||
| Refsum syndrome | AR | PHYH / PEX7 |
| Multiple sulfatase deficiency | AR | SUMF1 |
| Gaucher syndrome type 2 | AR | GBA |
| Sjögren-Larsson syndrome | AR | ALDH3A2 |
| Dorfman-Chanarin syndrome | AR | ABHD5 |
| Trichothiodystrophy | AR | C7ORF11 ERCC2 / XPD ERCC3 / XPB GTF2H5 / TTDA |
| Cerebral dysgenesis-neuropathy-ichthyosis-palmoplantar keratoderma (CEDNIK) | AR | SNAP29 |
| Arthrogryposis-renal dysfunction-cholestasis | AR | VPS33B |
| Autosomal ichthyosis syndromes with deafness | ||
| Keratitis ichthyosis deafness (KID) | AD | GJB2 (GJB6) |
| ELOVL4 deficiency | AR | ELOVL4 |
| Mental retardation-enteropathy-deafness-neuropathy-ichthyosis-keratoderma (MEDNIK) | AR | AP1S1 |
| Autosomal ichthyosis syndromes with transient neonatal respiratory distress | ||
| Ichthyosis prematurity syndrome | AR | SLC27A4 |
Finally, hereditary ichthyoses should be differentiated from acquired ichthyosis or ichthyosiform skin conditions that do not have a genetic basis and can be caused by different underlying diseases .
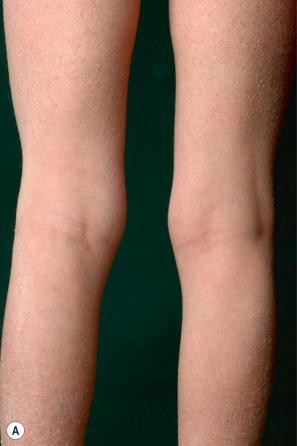
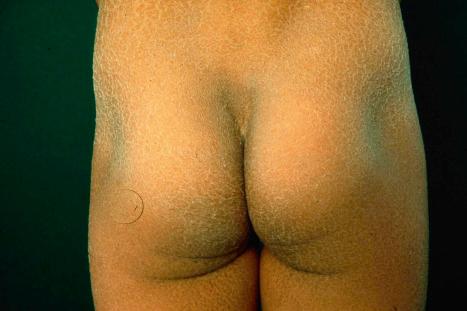
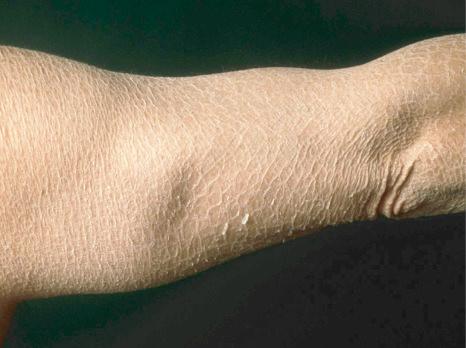
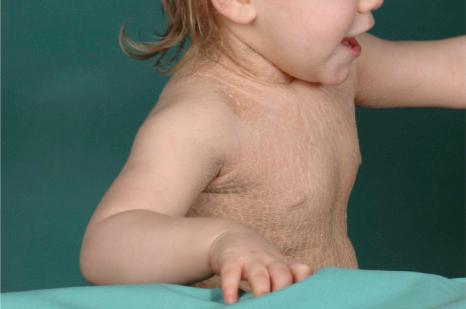
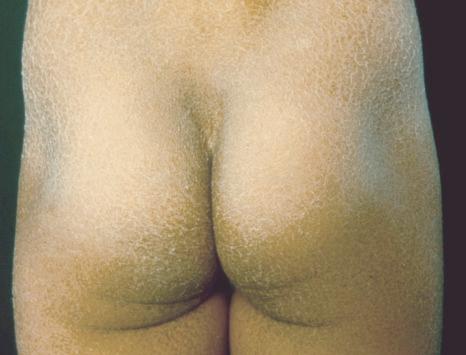
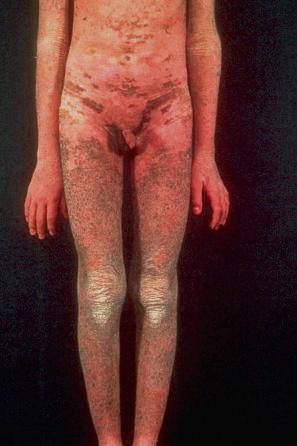
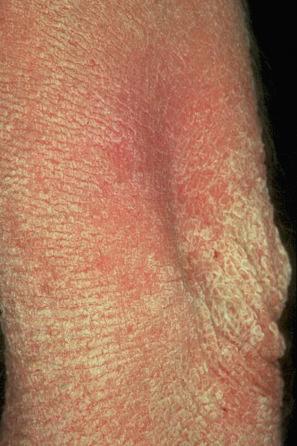
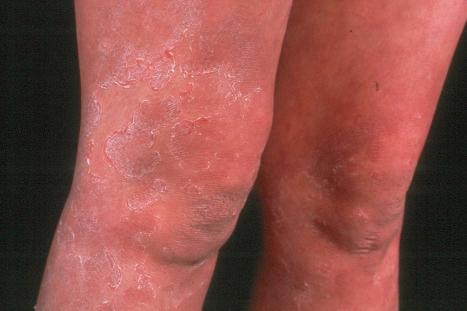
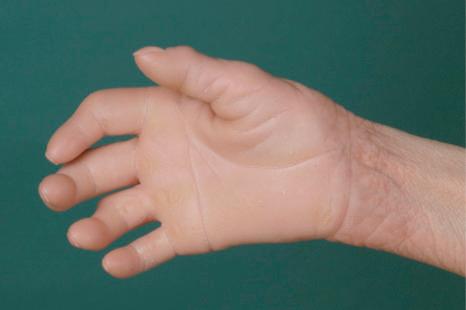
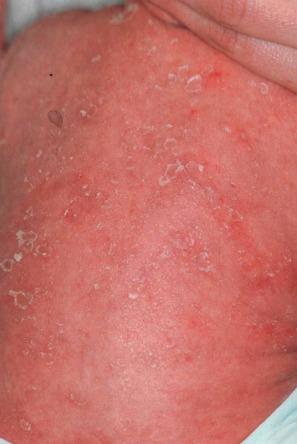
This relatively common disorder (incidence of 1 : 250 to 1 : 1000 births) has an autosomal semidominant mode of inheritance. The disease is usually fairly mild and becomes apparent within the first few months or years of life. It affects the sexes equally and presents as dryness (xerosis) and slight to moderate fine scaling, particularly involving the extensor surfaces of arms and legs and may spare the flexures ( Fig. 3.2A ). The light-gray scales vary in quality from thick adherent shiny plates to simply dusty accumulations which, when scratched, leave a mark just as when one touches a dusty surface. The truncal lesions tend to be thicker than those on the face and scalp. The rims of the ears are often scaly. There is a mild seasonal variation, with improvement of the condition in humid climates. The palms and soles show increased palmar and plantar markings (hyperlinearity), in contrast to pure X-linked ichthyosis ( Fig. 3.2B ). Affected patients often present keratosis pilaris (follicular hyperkeratosis) on the arms, buttocks and thighs and suffer from hypohidrosis. An association with keratosis punctata of the palms and soles has also been documented.
Ichthyosis vulgaris is caused by loss-of-function mutations of the FLG gene that encodes for filaggrin, the major constituent of the keratohyalin granules. Patients may have one or two FLG mutations. The mutation status correlates with disease severity and ultrastructure, i.e., in line with the decreased amount of filaggrin, keratohyalin granules may be reduced, spongy or slightly crumbly. Filaggrin aggregates keratin intermediate filaments in the lower stratum corneum and is subsequently proteolyzed to form free amino acids, including urocanic and pyrrolidone carboxylic acids, which are critical as water-binding compounds in the stratum corneum. The clinical severity of ichthyosis vulgaris correlates with the reduction of keratohyalin granules. Reduced immunostaining for filaggrin correlates with the severity of the defect. Parents with one heterozygous filaggrin mutation may be asymptomatic, whereas affected offspring with two mutations often show classic ichthyosis vulgaris. FLG mutations are a major predisposing factor for atopic dermatitis (AD) and related allergies.
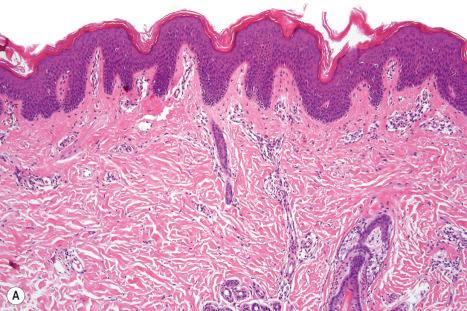
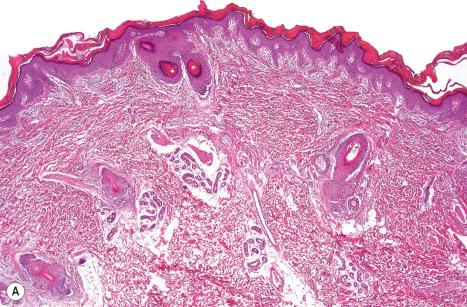
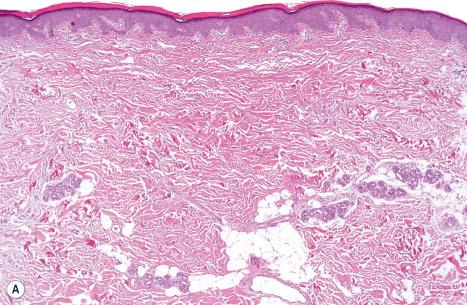
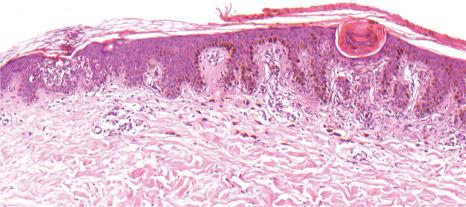
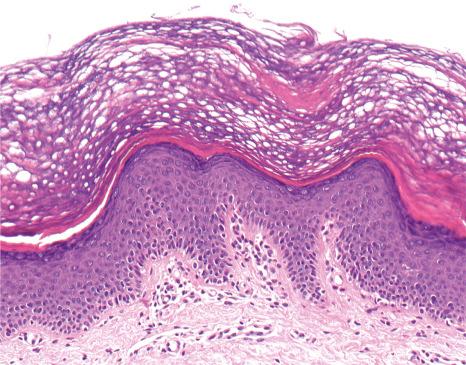
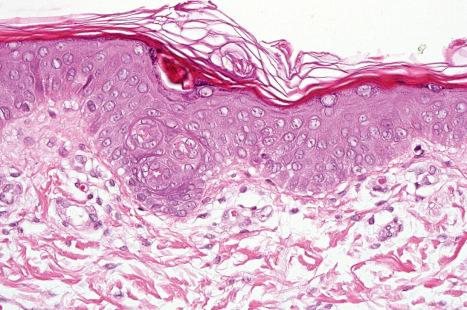
Ichthyosis vulgaris is characterized by mild to moderate orthohyperkeratosis associated with an acanthotic, atrophic or normal epidermis. The key feature is a thin or absent granular cell layer ( Fig. 3.3 ). Regional variation in the thickness and/or presence of the granular cell layer may occur and therefore it is best to take the biopsy from a site of maximal scaling. The lesions of keratosis pilaris show dilated follicles containing large keratin plugs. In the upper dermis a mild perivascular lymphocytic infiltrate may be present. When ichthyosis vulgaris is associated with atopic dermatitis, parakeratosis and other signs of a spongiotic dermatitis can be found.
The histologic differential diagnosis includes other diseases characterized by orthohyperkeratosis and a reduced or absent stratum granulosum ( Table 3.3 ).
| Orthohyperkeratosis and stratum granulosum reduced or absent |
|
| Orthohyperkeratosis and stratum granulosum well developed |
|
| Hyperkeratosis with ortho- and parakeratosis and prominent stratum granulosum |
|
| Epidermolytic hyperkeratosis |
|
| Perinuclear vacuoles and binucleated keratinocytes |
| With parakeratosis: |
| Congenital reticular ichthyosiform erythroderma |
| With orthokeratosis: |
|
| Follicular hyperkeratosis |
|
| Psoriasis-like features |
|
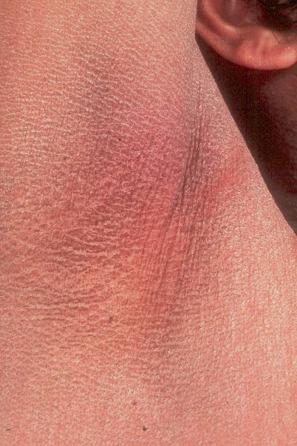
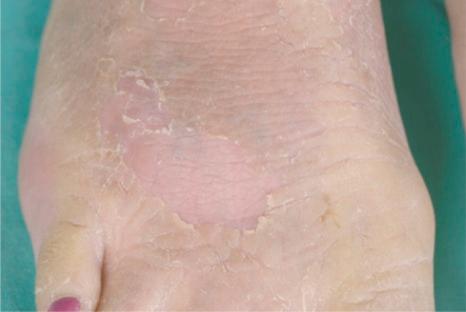
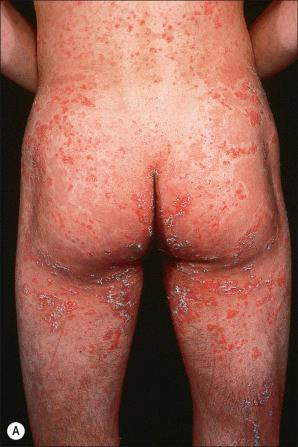
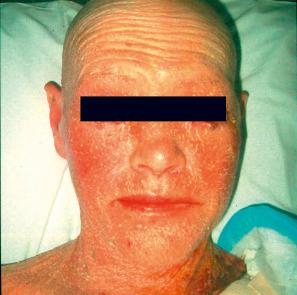
Also known as steroid sulfatase (STS) deficiency and ichthyosis nigricans, this X-linked, recessively inherited disorder has an incidence of 1 : 6000 male births. The disease is exceedingly rarely expressed in females. The disease may present at birth with dramatic but transient scaling of the skin, and this is sometimes misdiagnosed as autosomal recessive congenital ichthyosis. At the age of 2 to 6 months large and dark scales develop on the trunk, the extensor surface of the extremities, the scalp, the preauricular region, and the neck ( Fig. 3.4 ). Mild involvement of the flexures is present ( Fig. 3.5 ), but visible scaling may spare humid flexural regions of the skin. Differentiation from ichthyosis vulgaris can be difficult as one-third of patients present with fine, light scales and the flexures may be spared in both diseases ( Fig. 3.6 ). The palms and soles are usually unaffected and keratosis pilaris is not a feature, except if there is a concomitant deficiency of filaggrin and STS. Involvement of the trunk and neck often gives the skin a dirty appearance. Lesions may improve or disappear in warm weather. The hair, nails, and teeth are not affected.
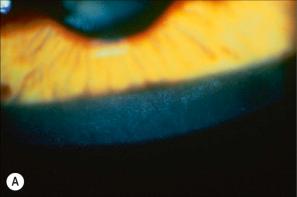
Corneal opacities due to comma-shaped deposits in the posterior capsule of Descemet membrane or corneal stroma without visual impairment ( Fig. 3.7 ), are characteristic and may be detected in female carriers. Inadequate cervical dilatation may lead to prolonged delivery of affected male newborns. Undescended testes and hypogonadism can be a feature in as many as 25% of affected patients. Rarely, testicular cancer has been documented. Interestingly, studies on STS deficiency have revealed an association with attention-deficit hyperactivity disorder (ADHD), and disorders within the spectrum of autism seem to occur more frequently.
The disease is associated with a deficiency of the microsomal enzyme steroid sulfatase/STS (sterol sulfate sulfohydrolase/arylsulphatase C). Absence of this enzyme is associated with persistence of the sulfate moiety on a number of sulfated steroid hormones and cholesterol sulfate.
X-linked recessive ichthyosis is characterized by a raised serum cholesterol sulfate. The corneocytes contain excess cholesterol 3-sulfate and diminished free sterol, This may lead to persistence of the lipid contents of the membrane-coating granules and hence increased or persistent adhesion between adjacent keratin plates in the stratum corneum. Beyond that, increased amounts of cholesterol sulfate may inhibit the epidermal serine protease activity, which results in retention of corneodesmosomes leading to less shedding of scales and retention hyperkeratosis. Steroid sulfatase deficiency can be detected using the patient's peripheral leukocytes and cultured skin fibroblasts. Diagnosis may also be confirmed by lipoprotein electrophoresis, which shows increased mobility of low-density and very low-density beta-lipoproteins in addition to the steroid sulfatase deficiency.
Technologies such as comparative genomic hybridization/comparative microarray analysis (CMA) allow rapid diagnosis in cases with large deletions. Carrier status can also be confirmed by fluorescent in situ hybridization (FISH) analysis. Standard sequencing techniques are used to identify a point mutation that is the cause of the disease in approximately 10% of patients. Severity of this form of ichthyosis may be aggravated by a concomitant filaggrin mutation.
Lesional skin shows compact orthohyperkeratosis and slight acanthosis associated with a granular cell layer, which may be normal or increased in thickness ( Fig. 3.8 ). Keratohyalin granules show no abnormality. Follicular plugging is not a feature. Paradoxically, biopsies of thicker scales can show massive orthohyperkeratosis with reduction of the granular layer and a thin epidermis, causing confusion with ichthyosis vulgaris. A discrete lymphocytic perivascular inflammatory cell infiltrate may be evident.
Ultrastructural features include a high number of transitional cells and an abnormal persistence of desmosomal disks in the horny layer while keratohyalin granules are normal. An increased melanosome transfer accounts for the dark appearance of the scales.
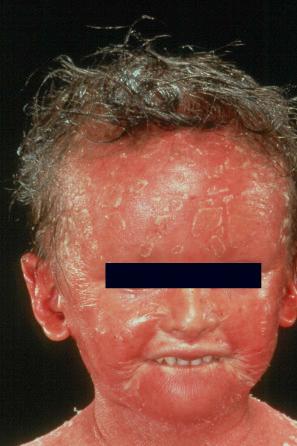
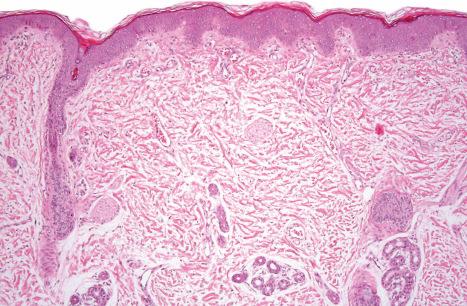
Autosomal recessive congenital ichthyoses (ARCI) are a group of monogenetic, nonsyndromic disorders presenting at birth with generalized hyperkeratosis and scaling (ichthyosis congenita) (see Table 3.1 ). ARCI include lamellar ichthyosis (LI), congenital ichthyosiform erythroderma (CIE), bathing suit ichthyosis, self-improving congenital ichthyosis (SICI) or self-healing collodion baby (SHCB), and the most severe form, harlequin ichthyosis. In Europe ARCI have an estimated prevalence of 1.6 : 100 000.
At birth, most patients with ARCI are encased in a thick ‘collodion’, a platelike shell of keratosis ( Figs 3.9 and 3.10 ) and suffer from ectropion and everted lips. The shiny parchment-like membrane cracks after birth and usually peels off within the first 4 weeks of life. While the term ‘collodion baby’ is most often applied to cases of ARCI, similar appearances are sometimes found in a number of other disorders, such as Gaucher syndrome type 2, trichothiodystrophy, Conradi-Hünermann-Happle syndrome, Sjögren-Larsson syndrome and sometimes Netherton syndrome. Hence, collodion baby is a clinical description but not a disease. Still, the majority of these conditions develop into one of the ARCI subtypes, including SICI or SHCB.
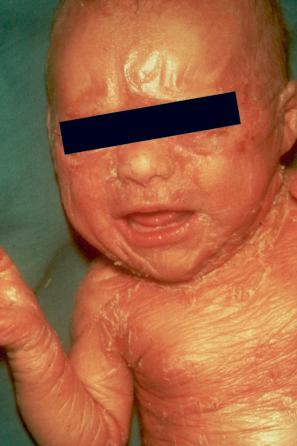
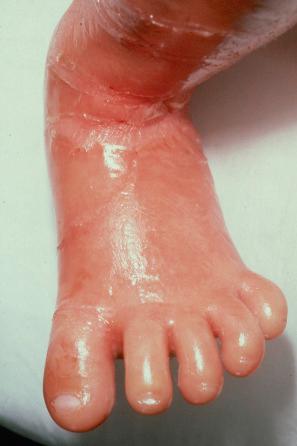
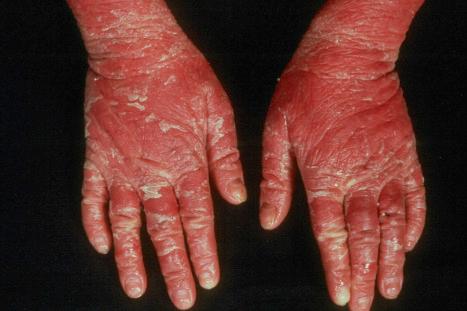
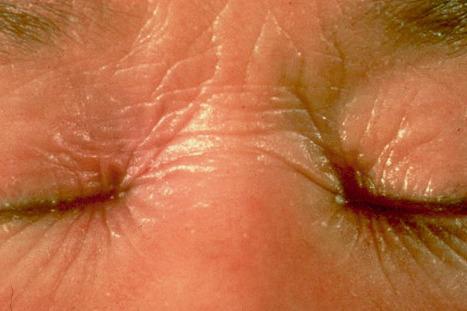
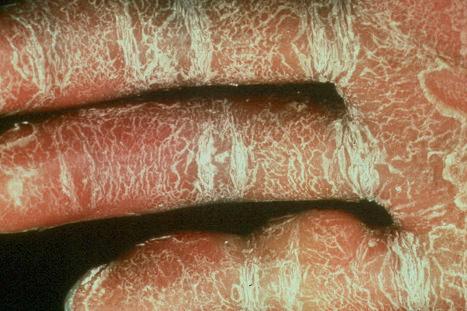
In the nonerythrodermic phenotype of congenital ichthyosis, the so-called ‘lamellar ichthyosis’, the scales are large, dark, and platelike, and cover the entire body, including the palms, soles, scalp, and flexures ( Fig. 3.11 ). Fissuring of the hands and feet may occur and the skin around the joints may become verrucous. Many patients suffer from severe hypo- or anhidrosis with risk of hyperthermia, an important clinical symptom with great impact on the daily life of the patient. Nail dystrophy, hair involvement (scarring alopecia), severe ectropion (up to 80% of patients), and eclabium are characteristic ( Fig. 3.12 ). The ectropion is of the cicatricial type and develops as a consequence of excessive dryness and associated contracture of the anterior lamella of the eyelid. Complications include corneal ulceration, vascularization, and corneal scarring with eventual blindness. Primary conjunctival lesions have been described including keratinization, hyper- and parakeratosis, and papilla development. The teeth are not affected.
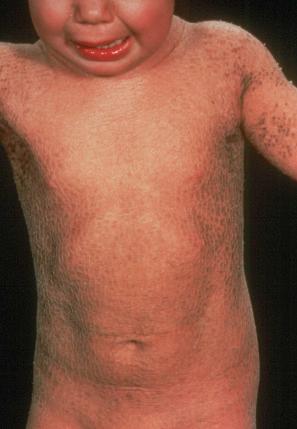
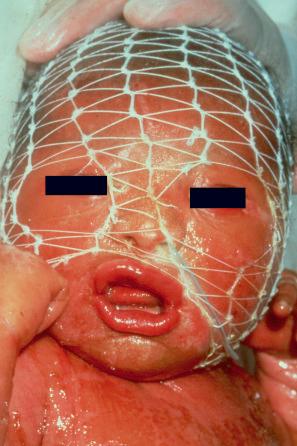
In contrast, other individuals show a more pronounced erythroderma with fine white scaling (congenital ichthyosiform erythroderma, CIE). While platelike scales may be seen on the extensor surfaces of the legs, the scalp, face, upper extremities, and trunk are covered with fine white scaling ( Figs 3.13–3.16 ). Mild ectropion and eclabium may be present. Some variants are associated with severe palmoplantar keratoderma (PPK), e g., yellowish focal palmoplantar keratoderma in ichthyin deficiency. Exceptionally, CIE has been associated with retinitis pigmentosa. There is an increased risk of developing skin cancer including basal and squamous cell carcinoma.
The most common cause of LI is transglutaminase-1 (TG-1) deficiency, which accounts for 30–40% of cases. TG-1 plays a role in the assembly of the cornified envelope by catalyzing calcium-dependent cross-linking of proteins, such as involucrin, loricrin, and proline-rich proteins and by binding omega-hydroxy ceramides to proteins such as involucrin, a major step in connecting the lipid envelope with the cornified envelope. Mutations in the gene of TG-1 ( TGM1 ) result in markedly diminished or lost enzyme activity and/or protein. Since conventional enzyme assays and mutational analyzes are tedious, an assay for the rapid screening of TG activity using covalent incorporation of biotinylated substrate peptides into skin cryostat sections has been developed. Coupled with immunohistochemical assays using TG-1 antibodies, this allows valid identification of TG-1 deficiency.
Patients with TGM1 mutations mostly present with classical LI including ectropion, alopecia, or small and deformed ears. Some variants of TGM1 mutations are associated with distinct clinical features, e.g., in self-healing collodion baby the TG-1 enzyme is pressure-sensitive so that while in utero the enzyme cannot function properly, it resumes normal function after birth. About 10% of collodion babies fall into this group. In bathing suit ichthyosis the mutations of TG-1 appear to be temperature sensitive so that the face and extremities are almost completely spared apart from skin areas overlying blood vessels ( Fig. 3.17 ). Digital thermography has validated a striking correlation between warmer body areas and the presence of scaling, suggesting a decisive influence of skin temperature. In situ TG-1 testing in skin of bathing suit ichthyosis patients has also demonstrated a marked decrease of enzyme activity when the temperature is increased from 25 to 37°C.
Two epidermal lipoxygenases associated with the genes ALOXE3 and ALOX12B play a crucial role in the hepoxilin pathway. The defect is believed to have an impact on secretion of lamellar bodies (LBs) resulting in impaired formation of intercellular lipids in the stratum corneum. Lipoxigenase mutations are often associated with the CIE phenotype, and show a striking palmoplantar hyperlinearity that should not be confused with that of ichthyosis vulgaris. Other features are pruritus and reduced sweating. Biochemical measurement of lipoxygenase activity is available only in specialized research laboratories.
Another common genetic subtype of ARCI affects the NIPAL4 gene that encodes for ichthyin. This protein has been described to localize to desmosomes and keratins and to interact with fatty acid transporter protein 4, which is defective in ichthyosis prematurity syndrome. NIPAL4 mutations may lead to a CIE/LI overlapping phenotype with mild ectropion, clubbing of the nails and – most characteristically – a focal yellowish palmoplantar keratoderma developing in childhood.
Mutations in ARCI of late onset involve the acid lipase gene LIPN that plays a role in triglyceride metabolism of the epidermis. LIPN mutations seem to cause a late-onset form of ichthyosis at the age of 5 years, so that it is not formally a congenital ichthyosis.
Other mutations in ARCI been detected in the CYP4F2 gene encoding a cytochrome P450 polypeptide, CERS3 gene encoding a ceramide synthase, and PNPLA1 , a member of the patatin-like phospholipase family. Moreover, less severe missense mutations in the ABCA12 gene are associated with LI or CIE. Of note, nonfunctional severe nonsense mutations of the same gene are responsible for the far more severe ichthyosis form of harlequin ichthyosis (see below).
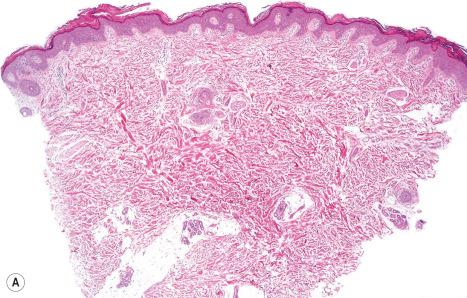
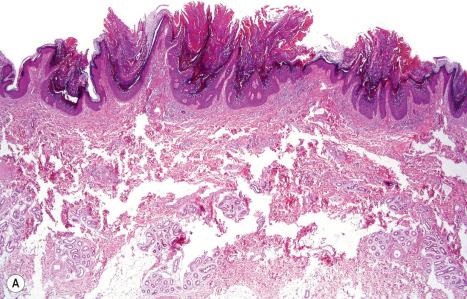
Histologically, the epidermis in ARCI shows marked orthohyperkeratosis, mostly of the compact type (which may be extreme in the collodion baby) and mild acanthosis with a normal or thickened granular cell layer ( Fig. 3.18 ). The hyperkeratosis is much less marked in erythrodermic than in nonerythrodermic forms. Epidermal papillomatosis associated with a psoriasiform appearance has also been documented. A perivascular lymphocytic infiltrate is occasionally a feature. Dilatation and tortuosity of the dermal capillaries is sometimes evident. Follicular hyperkeratosis may rarely be seen. The histologic changes cannot be correlated with the underlying genetic defect.
Ultrastructural studies show a variety of features, including defective development of the cornified cell envelopes and electron-dense debris adjacent to the plasma membranes, lipid vacuoles, increased numbers of small and dysmorphic LBs, or membrane packages. Cholesterol clefts are found in TG-1 defects. Abnormal LBs and elongated membranes in the stratum granulosum indicate NIPAL4 deficiency classified as ARCI electron microscopy (EM) type III. However, other ARCI subtypes lack specific ultrastructural features. Special techniques with frozen sections or osmium tetroxide and ruthenium tetroxide postfixation allow for an advanced EM diagnostic of ARCI. Prenatal diagnosis of LI can be achieved by fetoscopy and biopsy.
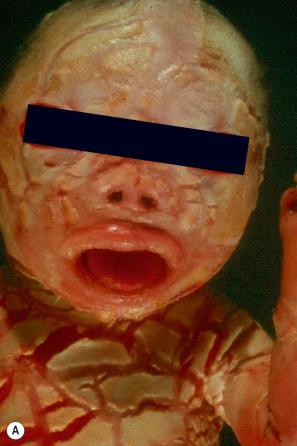
Harlequin ichthyosis (harlequin fetus, ichthyosis fetalis, ichthyosis congenita gravis) is a distinct severe subtype of autosomal recessive congenital ichthyosis, where babies are born with a fissured ‘armor-plated’ skin ( Fig. 3.19 ). Characteristically, ectropion and eclabium are very severe. Constricting bands of digits can lead to autoamputation; ears and nose may be malformed. The harlequin fetus still has a high mortality due to infections, and respiratory and feeding difficulties accompanied by excessive fluid loss. However, treatment with retinoids and improved intensive care have certainly improved the prognosis and quality of life. Surviving neonates develop a severe erythroderma reminiscent of CIE, and there are intermediate cases that show considerable improvement of the skin compared with the initial dramatic presentation. Antenatal diagnosis is possible by genetic analysis as well as electron microscopy of fetal skin biopsy (fetoscopy).
This very rare form of ichthyosis is due to a dramatic loss of function of the LBs, which results from nonsense mutations in the ABCA12 gene. Less severe missense mutations are associated with LI or CIE. Intermediate forms are possible. The ABCA12 gene product belongs to the ATP-binding cassette (ABC) transporter family that encompass a variety of membrane proteins involved in the energy-dependent transport across membranes. In the epidermis, ABCA12 plays an important role in the LB function. As such it is responsible for the transfer of glucosylceramides, which are essential lipids for epidermal barrier formation. Moreover, it transports proteases such as kallikrein 5, 7, or 14 and secretes them into the intercellular space in the stratum corneum. These proteases play an important role in desquamation by degrading corneodesmosomes, thus leading to retention hyperkeratosis.
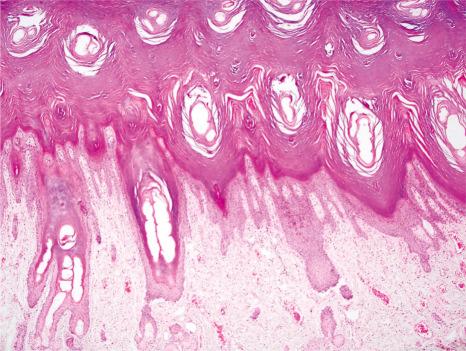
Histologically, the epidermis is pale and characterized by massive hyperkeratosis (sometimes with lipid deposits) associated with a normal or absent granular cell layer ( Fig. 3.20 ). The hair follicles are usually affected first, during the second trimester. Parakeratosis may also sometimes be evident. Acanthosis is often marked and papillomatosis is sometimes a feature. A sparse mixed inflammatory cell infiltrate can be present in the superficial dermis.
Ultrastructurally, the harlequin fetus is associated with deficient or morphologically abnormal LBs (including concentrically lamellated forms) and deficient intercellular lipid lamellae within the stratum corneum. Small vesicles, devoid of internal lamellation, may be present in the granular cell layer (and retained in the stratum corneum), but show no association with the keratinocyte cell membranes as is typical of normal LBs.
Immunohistochemical evidence suggests that these vesicles represent abnormal LBs characterized by an inability to discharge their lipid contents into the intercellular space. Keratin and filaggrin expression have also been shown to be defective. In the harlequin fetus, the keratinocytes may display the hyperproliferative keratins K6 and K16 and show an inability to convert profilaggrin to filaggrin.
Autosomal dominant LI is characterized by generalized scaling with palmoplantar keratoderma. Patients may present as a collodion baby. They are later covered by diffuse dark-gray scales that involve all areas of the body, but are most prominent on the extensor surfaces ( Fig. 3.21 ). Backs of the hands and feet are characterized by lichenification. There may be massive plantar hyperkeratosis with thick, yellow scales. The palms are usually only minimally involved and show accentuated markings.
This disorder appears to be genetically and clinically heterogeneous and of variable penetrance. Its genetic defect has not been identified. Biochemically, an abnormal lipid profile has been detected in the scales.
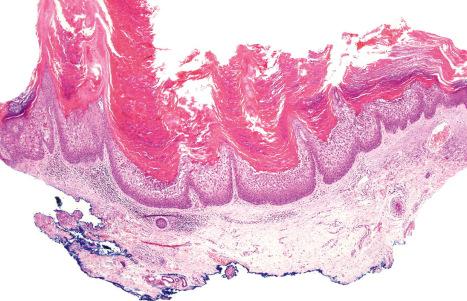
Histologically, there is acanthosis, papillomatosis, and compact orthohyperkeratosis with focal parakeratosis that, paradoxically, is associated with a thickened stratum granulosum ( Fig. 3.22 ).
Electron microscopy shows a high number of transitional cells and a spongy appearance of the keratohyaline granules.
The differential diagnosis includes lichen simplex chronicus which, however, differs by the presence of inflammatory changes and fibrosis of the papillary dermis (see Table 3.3 ).
According to the Sorèze Consensus conference in 2009, keratinopathic ichthyoses have been defined as a group of rare ichthyoses, which are caused by mutations in one of the keratin genes, namely keratin 1 ( KRT1 ), keratin 10 ( KRT10 ) or keratin 2 ( KRT2 ). The old term ‘bullous congenital ichthyosiform erythroderma (of Brocq)’ has been renamed epidermolytic ichthyosis (EI); the term superficial epidermolytic ichthyosis (SEI) refers to the KRT2 associated subtype formerly known as ‘ichthyosis bullosa of Siemens’. Moreover, there are distinct keratinopathic ichthyosis forms, such as congenital reticular ichthyosiform erythroderma (CRIE) (also known as ichthyosis en confettis), ichthyosis Curth-Macklin, autosomal recessive EI, and annular EI (see Table 3.1 ).
In the skin, basal keratinocytes predominantly express keratin 5 and 14 while suprabasal cells switch to the expression of keratin 1 and 10. Keratin 2e is only expressed in the subcorneal layer. Keratin monomers form obligate heterodimers in pairs of acidic (type I) and basic (type II) keratins, which assemble into keratin intermediate filaments building a cytoskeleton for the structural stability and flexibility of epidermal cells. Mutations in the higher molecular weight keratins 1 or 10 lead to collapse of the keratin skeleton in suprabasal keratinocytes while mutations in keratin 2 affect the upper layers. As a result the keratinocytes appear pale and show perinuclear shell formation or eosinophilic clumps, the latter changes known as epidermolytic hyperkeratosis. In these genodermatoses aggregation of the keratins is not permanent, but is inducible by pressure, high temperature, fever or skin infections. Epidermal sensitivity to hyperosmotic stress may be reduced by the chemical chaperone trimethylamine-N-oxide. Keratin aggregates also induce inflammation via interaction with the ubiquitin-proteasome system, activated MAP kinases, and chaperones, such as HSP70. Retinoids may interfere with collapse of the keratin skeleton in heat stressed keratinocytes with KRT10 mutation.
Epidermolytic hyperkeratosis as seen in some types of epidermal nevus is the result of somatic mutations in KRT1 or KRT10 . A specific histologic diagnosis of this postzygotic mosaicism is mandatory as the patients risk developing full-blown keratinopathic ichthyosis.
Epidermolytic hyperkeratosis is a histopathologic pattern that is seen in many conditions, including keratinopathic ichthyoses, palmoplantar keratoderma of the epidermolytic type epidermal nevus (see epidermolytic epidermal nevus ), epidermolytic acanthoma, and epidermolytic leukoplakia (see Table 3.3 ). Epidermolytic hyperkeratosis has been shown to be one of the many patterns in Grover disease. It may also represent an incidental finding in normal, inflamed, and neoplastic skin (see incidental epidermolytic hyperkeratosis ). Accurate clinical information is necessary to avoid diagnostic confusion.
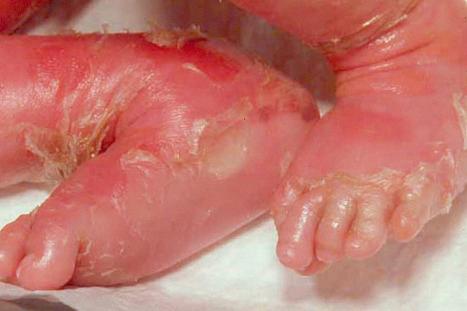
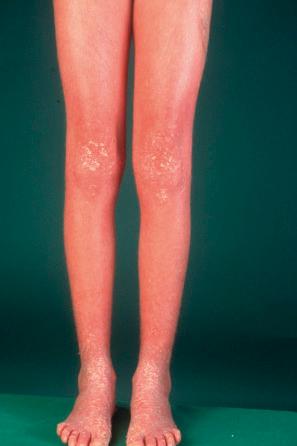
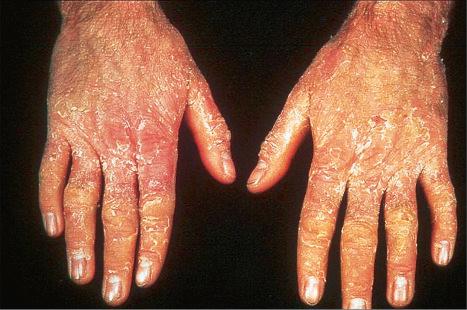
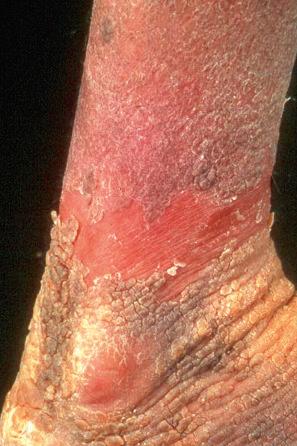
Epidermolytic ichthyosis (EI) is a rare autosomal dominant disease (incidence of 1 : 300 000 births), but it often arises by a spontaneous mutation. At birth infants may show marked erythroderma and develop widespread blistering ( Fig. 3.23 ). Therefore, a major differential diagnosis is epidermolysis bullosa. As the patient becomes older, the erythema and blistering become less apparent and, later, the disease is complicated by the development of verrucous hyperkeratosis, especially in the flexures ( Figs 3.24–3.28 ). The descriptive word ‘hystrix’ (porcupine) for this cobblestone-like appearance of the skin has been abandoned. The nape, axilla, groin, and flexural folds are sites of predilection. Severe involvement of the scalp may simulate tinea capitis. Occasional blisters still arise at sites of mechanical stress, often in summer or when the patients have fever and skin infections. Many patients suffer from an offensive body odor. Neonates with EI have an increased risk of sepsis, fluid loss, and electrolyte imbalance. Patients with KRT1 mutation often develop severe, sometimes painful palmoplantar keratoderma. Nail dystrophy may also occur. Therapeutic administration of systemic retinoids may aggravate the complications, especially in patients with KRT1 mutation.
First, it was shown that EI shows linkage to the keratin gene cluster either on chromosome 12q11–13 (type II keratin) or chromosome 17q21-q22 (type I keratin) and that transgenic mice expressed a mutant KRT10 gene. This was followed by detection by direct DNA sequencing of a point mutation in KRT1 or KRT10 in a number of affected families. Most mutations are missense mutations and clustered at the ends of the central helical rod domains. KRT1 mutations typically affect palmoplantar skin. This region is most often spared in EI with KRT10 mutation, which can be explained by its physiological substitution of keratin 9 on palmoplantar skin.
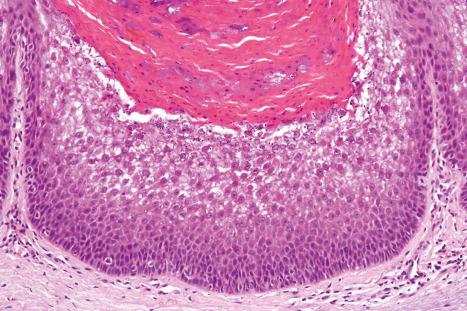
EI shows the typical features of epidermolytic hyperkeratosis. Suprabasal keratinocytes appear vacuolated and typically contain distinct eosinophilic intracytoplasmic inclusions. The cell borders are ill defined and intraepidermal blister formation may be present. There is massive orthohyperkeratosis, papillomatosis, and acanthosis. The granular cell layer is prominent and contains coarse and irregular keratoyhaline granules ( Fig. 3.29 ).
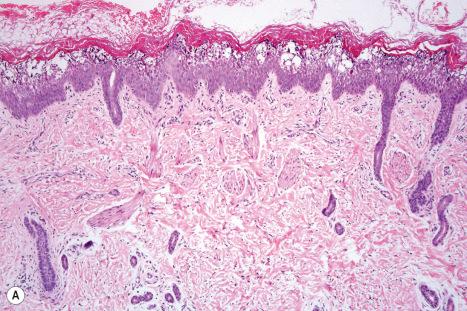
By immunohistochemistry, epidermolytic hyperkeratosis shows a normal distribution pattern of keratins 5/14 and 1/10, but in addition, there is overexpression of keratin 14 in the suprabasal epithelium accompanied by quite marked labeling of the upper epithelial layers by keratin 16, as would be expected in a hyperproliferative state.
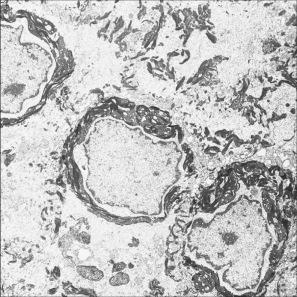
Ultrastructural studies have shown that the intracytoplasmic inclusions seen on light microscopy are composed of abnormally aggregated keratin filaments. Since large areas of the cytoplasm lack a regular keratin skeleton, the suprabasal keratinocytes appear vacuolated and contain irregular keratoyhaline granules. Impairment of desmosome-keratin complexes accounts for the fragility of the epidermis ( Fig. 3.30 ). Immunoelectron microscopy has identified that the keratin clumps are composed of keratins 1 and 10. These ultrastructural changes may form the basis of prenatal diagnosis including amniotic fluid squame analysis.
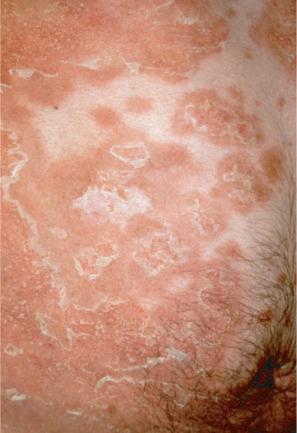
Annular EI is a rare but clinically distinct form of keratinopathic ichthyosis related to a novel dinucleotide mutation in KRT10 . Patients have mild erythroderma and blisters at birth, but the characteristic feature is the presence of migrating annular and polycyclic, gray hyperkeratotic plaques with a peripheral erythematous border on the trunk and on the extremities ( Fig. 3.31 ). High temperature in the summer, fever or pregnancy can induce recurrences.
Histologically, epidermolytic hyperkeratosis is the dominant pattern, however a biopsy from the inflammatory border shows features of a lymphocytic, psoriasiform dermatitis with parakeratosis.
Superficial epidermolytic ichthyosis (SEI), formerly termed ichthyosis bullosa Siemens, is a keratinopathic ichthyosis, which is milder than EI. It presents at birth with blistering subsequently replaced by dark lichenified hyperkeratosis of the limbs, predominantly affecting the flexures and shins ( Fig. 3.32 ). The skin remains fragile and blisters on mild trauma, giving rise to characteristic superficial peeling with a molting-like appearance (Mauserung phenomenon) ( Fig. 3.33 ). Symptoms usually improve with age. Erythroderma is typically absent. Rarely, pustulation and hypertrichosis may be additional features. There is considerable clinical overlap between SEI and EI, and their distinction can best be achieved by molecular genetic analysis.
SEI is associated with a point mutation in the keratin 2 gene ( KRT2 ) located on chromosome 12q11-q13. Since this keratin is not expressed on volar skin, palmoplantar keratoderma does not develop.
Histologically and by electron microscopy, the features are indistinguishable from EI except that they are milder and the vacuolation of the keratinocytes and cytoplasmic inclusions are restricted to the more superficial spinous and granular cell layers. Subcorneal separation may be evident.
A nevoid variant of EI is the result of somatic mutations in KRT1 or KRT10 . These nevi follow one or more Blaschko lines and are characterized by a prominent hyperkeratosis. The dirty-appearing verruciform aspect should raise the diagnostic suspicion of a keratin mutation (linear verrucous epidermal nevus). A biopsy can easily identify the histologic pattern of epidermolytic hyperkeratosis which allows distinction from other types of epidermal nevi ( Fig. 3.34 ). Since children of affected patients may develop a form of generalized EI, genetic counseling should be offered.
Epidermolytic acanthoma is an acquired lesion that presents as a verrucous papule or plaque approximately 1.0 cm in diameter and sometimes resembles a viral wart, nevus or seborrheic keratosis. Caucasians and the Japanese are predominantly affected. Lesions may present at any site, but the scrotum, head, neck, and lower limbs are particularly affected. Although usually solitary, occasional patients may present with multiple localized or disseminated lesions. In Japanese patients multiple depigmented flat keratotic papules following sun exposure on the shoulders and back were described as persistent actinic epidermolytic hyperkeratosis. Variants affecting the mucosae of the oral cavity and female genital tract have also been documented.
Although it has been assumed that epidermolytic acanthoma develops as a consequence of keratin 1 and 10 gene mutation, two recent genetic studies failed to show any mutations in these keratins. Still, trauma or other external triggers may interfere with the transcription or translation of the suprabasal keratins. Human papillomavirus (HPV) infection has been excluded.
The lesion is well circumscribed and cup shaped and only rarely polypoid. The epidermis shows hyperkeratosis, focal parakeratosis, acanthosis and papillomatosis or a flat base. ( Fig. 3.35 ). The upper prickle cell and granular cell layers show features of epidermolytic hyperkeratosis, i.e., marked vacuolation of the keratinocytes with eosinophilic keratin inclusions and irregular keratohyaline granules ( Fig. 3.36 ). More variably, eosinophilic perinuclear keratin condensations resembling the Hailey-Hailey pattern of acantholysis are described. Necrotic keratinocytes with absent nuclei can be seen in the horny layer. A perivascular lymphocytic infiltrate is often present in the papillary dermis.
Epidermolytic acanthoma displays diminished expression of keratins 1 and 10 and increased expression of the hyperproliferative keratins 6 and 16 while other differentiation markers, such as involucrin and loricrin, appear normal.
Identical histologic changes are seen in keratinopathic forms of ichthyosis, linear epidermolytic epidermal nevus, epidermolytic palmoplantar keratoderma, and incidental epidermolytic hyperkeratosis (see Table 3.3 ). Clinical information is usually necessary to avoid diagnostic confusion.
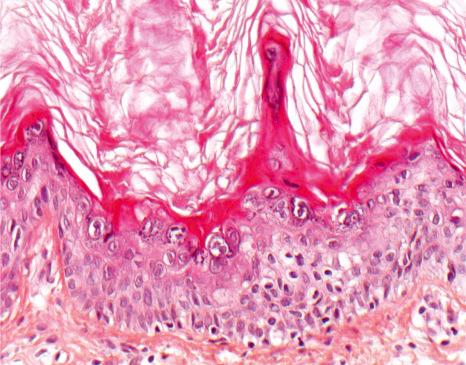
Incidental epidermolytic hyperkeratosis represents a focal non-specific finding of epidermolytic hyperkeratosis in the epidermis overlying or adjacent to an unrelated lesion. It is very common and has been described, for example, in seborrheic keratoses, melanocytic nevi, actinic keratosis, squamous cell carcinoma, and melanoma. In such incidental cases, the changes are limited to the epidermis overlying just one or two dermal papillae in contrast to the much more extensive involvement in other epidermolytic diseases ( Fig. 3.37 ).
Incidental epidermolytic hyperkeratosis can be associated with epidermal and pilar cysts, scars and fibrous histiocytoma, It may also be seen in normal, particularly sun-damaged skin. Incidental histopathologic reaction patterns, such as epidermolytic hyperkeratosis, acantholytic dyskeratosis, and Hailey-Hailey-like acantholysis are presumably related to field canceration and can be regarded as a potentially premalignant change surrounding tumors.
Beyond that, inflammatory conditions, e.g., lichen planus, nummular dermatitis, Grover disease, pityriasis versicolor, and others, may also show the incidental phenomenon of epidermolytic hyperkeratosis.
Ichthyosis Curth-Macklin was originally termed ichthyosis hystrix, a descriptive name for cornification disorders with spiny and dark hyperkeratosis. Ichthyosis Curth-Macklin is characterized by generalized verrucous plaques involving the entire trunk, the flexural surfaces of the extremities, and the palms and soles. This autosomal dominant disorder sometimes resembles EI, but there is no clinical or histologic evidence of blistering.
In ichthyosis Curth-Macklin the mutation affects the variable tail domain (V2) of keratin 1 which results in a failure in keratin intermediate filament bundling and retraction of the cytoskeleton from the nucleus. This is the first in vivo evidence for the crucial role of a keratin tail domain in supramolecular keratin intermediate filament organization and barrier formation.
Histologically, the epidermis is acanthotic and orthohyperkeratotic. The suprabasal keratinocytes are vacuolated and a few of them appear binucleated. In contrast to epidermolytic hyperkeratosis, eosinophilic intracytoplasmic inclusions are not seen ( Fig. 3.38 ). Ichthyosis Curth-Macklin shares many cytologic features with CRIE but lacks parakeratosis (see Table 3.3 ).
The significant ultrastructural observation in ichthyosis Curth-Macklin is the presence of perinuclear concentric shells of tonofilaments. In contrast to keratin mutations of the rod domain in epidermolytic hyperkeratosis, aggregations and clumping of keratin filaments are absent.
Congenital reticular ichthyosiform erythroderma (CRIE) is an autosomal dominant variant of keratinopathic ichthyosis. Patients are born with erythroderma, and initially may be misdiagnosed as having autosomal recessive congenital ichthyosis, i.e., CIE. Then, during early childhood their integument develops an increasing number of small pale white spots ( Fig. 3.39 ). Because of this clinical appearance the genodermatosis has also been termed ichthyosis ‘en confettis’ or ichthyosis variegata. Some patients develop hypertrichosis and palmoplantar hyperkeratosis. There may be a higher risk for keratoacanthoma or squamous cell carcinoma. Although this type of ichthyosis belongs to the group of nonsyndromic ichthyoses, single cases have been described in association with hypogonadism, growth retardation, osteomalacia, or hepatomegaly.
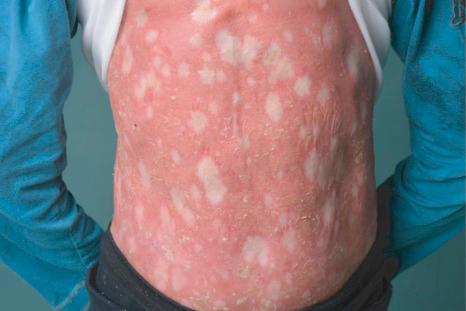
The genetic defect in CRIE is based on specific dominant mutations in KRT10 or in KRT1 . The characteristic formation of white spots has a later onset in KRT1 mutations. These mutations cause disruption of the keratin filament network and mislocalization of keratin to the nucleus. The spontaneous development of multiple patches of normal skin is caused by recombination events in the mutated keratin gene. This intriguing revertant phenotype can be considered a kind of natural gene therapy.
Histologically, the epidermis is pale staining and shows psoriasiform hyperplasia. The horny layer is thickened and parakeratotic. The parakeratotic corneocytes have enlarged nuclei. The keratinocytes of the upper layers show prominent perinuclear vacuolation and contain only a few keratohyalin granules. Their cell borders are well defined and intracytoplasmic eosinophilic granules are absent. Some of the vacuolated keratinocytes are binucleated ( Fig. 3.40 ). While keratin 2e is missing, the other epidermal keratins are regularly expressed. The superficial dermal vessels are dilated, and there is a sparse perivascular inflammatory cell infiltrate with scattered melanophages. Biopsies from the confetti-like spots within the erythrodermic skin reveal a normal differentiating epidermis, both histologically and immunohistochemically.
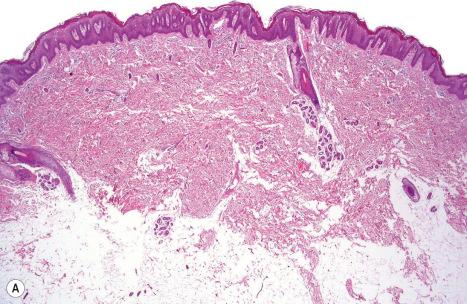
At the ultrastructural level the arrangement of the keratin skeleton is highly disturbed. Immuno-electron microscopy reveals complete absence of keratin filaments in the perinuclear cytoplasm. The number of transitional cells is increased and nick end labeling (TUNEL) for DNA fragmentation shows strong labeling of the parakeratotic corneocytes consistent with an apoptotic mode of cell death. Uptake and processing of melanosomes is irregular. By immunofluorescence dotlike labelling for keratin can be demonstrated in numerous nuclei in the suprabasal epidermis.
The absence of keratin clumping clearly distinguishes CRIE from keratinization disorders characterized by epidermolytic hyperkeratosis. Ichthyosis Curth-Macklin shares the intraepidermal formation of binucleated and vacuolated keratinocytes but lacks parakeratosis and forms perinuclear shells of tonofibrils (see Table 3.3 ).
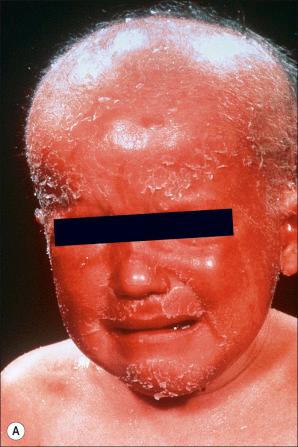
Comèl-Netherton syndrome (Netherton syndrome, ichthyosis linearis circumflexa) is a rare autosomal recessively inherited genodermatosis. It is characterized by the triad of CIE, hair shaft anomalies, and a severe atopic diathesis with high IgE blood levels and eosinophilia. It is believed to affect approximately 1 : 200 000 of the population. Generally, the CIE gradually evolves into a milder ichthyosis linearis circumflexa which is characterized by an erythematous, scaly rash predominantly affecting the trunk and limbs. It is composed of polycyclic, migratory, annular and serpiginous lesions with characteristic two parallel lines of scale at the periphery, the so-called double-edged scale ( Figs 3.41 and 3.42 ). In infancy, erythema and scaling may be widespread, but later the face is often predominantly affected (particularly marked around the mouth and eyes), along with the perineum, and as such the eruption can be mistaken for acrodermatitis enteropathica ( Fig. 3.43 ). Later the scalp, face, and eyebrows may show a yellowish scaling. Comèl-Netherton syndrome is often misdiagnosed as seborrheic dermatitis, atopic dermatitis, and psoriasis vulgaris.
Hairs on the scalp, eyebrows, and eyelashes may be present, sparse or absent at birth. Trichorrhexis invaginata (‘bamboo hair’) does not develop until childhood. These hair anomalies are caused by transient and repeated defects of keratinization, with resultant hair shaft intussusception, and present clinically as coarse and lusterless hair, which is short, brittle, and fragile (see Fig. 3.43 ). Pili torti and trichorrhexis may also occur ( Fig. 3.44 ).
Patients with Netherton syndrome may suffer from life-threatening neonatal dehydration with hypernatremia, failure to thrive, aminoaciduria, and recurrent skin infections often caused by Staphylococcus aureus . At an older age there is a higher risk for HPV-associated papillomatous skin lesions of the groin and perineal regions, spinous cell carcinoma, and giant condyloma of Buschke-Lowenstein. Growth retardation is due to growth hormone deficiency and immune defects. Food allergies against nuts and fish are common and lead to urticaria or angioedema. An impaired epidermal barrier is a potential risk for increased and even toxic absorption of topical medications.
Comèl-Netherton syndrome is caused by mutations in the SPINK5 gene (serine protease inhibitor Kazal-type 5) encoding the serine protease inhibitor LEKTI (lympho-epithelial Kazal-type related inhibitor). Analysis of fetal DNA from amniotic fluid in families at risk for this life-threatening disease allow for an early, rapid, and reliable method of prenatal diagnosis. LEKTI is expressed in the epidermis, thymus, and oral and vaginal mucosa and controls trypsin- and chymotrypsin-like enzymes. The failure of serine protease inhibition accounts for over-desquamation of corneocytes and degradation of desmosomal proteins (e.g., corneodesmosin and desmoglein 1). In addition, the PAR-2 (protease-activated receptor-2) related proinflammatory response is upregulated. In summary, the lack of LEKTI accounts for a unique combination of ichthyotic and inflammatory skin phenotype, which is associated with an extremely impaired epidermal barrier.
For histologic diagnosis, the biopsy should be taken from the scaly margin. A sample from this area will reveal the diagnostic psoriasiform features while biopsies from the center of the lesion show misleading features of atopic dermatitis.
Netherton syndrome shows a psoriasiform dermatitis with parakeratosis and intracorneal neutrophils (sometimes with formation of Munro microabscesses) often indistinguishable from psoriasis vulgaris ( Fig. 3.45 ). New findings described in a series of patients, include compact parakeratosis with large nuclei, subcorneal or intracorneal splitting, presence of clear cells in the upper epidermis or stratum corneum, dyskeratosis, elongated rete ridges without suprapapillary thinning and a superficial dermal inflammatory cell infiltrate rich in neutrophils and/or eosinophils. Small, dark, round or oval granules can be identified within the stratum granulosum. They are diastase-resistant, and periodic acid-Schiff (PAS) and Sudan black positive and are thought to represent an influx of serum exudates resulting from the accompanying dermal inflammation. Similar ‘inclusions’ have been described in psoriasis and atopic eczema and as such they are not specific.
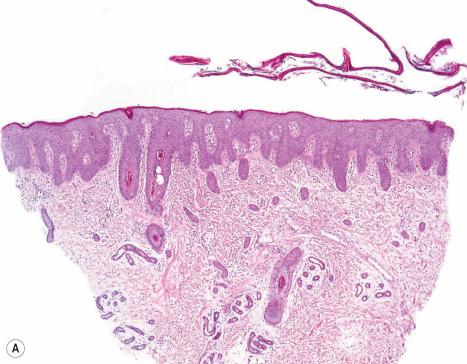
Electron microscopy reveals reduced numbers of LBs in keratinocytes and the presence of lysosomal inclusion bodies with intercellular amorphous deposits in the horny layer.
Immunohistochemistry can demonstrate the absence of LEKTI antigen and is highly specific for the Netherton syndrome both in neonatal erythroderma and scaly rashes of adults. A definite diagnosis can be made by negative staining for LEKTI in the inner root sheath of hair follicles since Netherton syndrome and other diseases may lack the stratum granulosum where LEKTI is normally expressed. However, residual LEKTI expression in the outer epidermis is still demonstrable immunohistochemically in mild forms of Netherton syndrome.
Distinction from psoriasis vulgaris may be histologically extremely difficult (if not impossible) in the absence of clinical information. Other genodermatoses, dermatophytosis, and inflammatory skin diseases with a psoriasiform-like pattern must be differentiated (see Table 3.3 ). Netherton syndrome shares the distinct histologic feature of corneal splitting with other exfoliative disorders of cornification, such as peeling skin syndrome (PSS) and severe dermatitis, multiple allergies, metabolic wasting syndrome (SAM). Atopic dermatitis is another important differential diagnosis.
Peeling skin syndromes (PSSs) are characterized by spontaneous peeling of the stratum corneum without bleeding or pain. Different genetic and clinical entities have to be distinguished.
In peeling skin syndrome type A (familial continual skin peeling, keratolysis exfoliativa congenitale) a generalized lifelong and continued shedding or peeling of the entire skin starts between 3–6 years of age. In contrast with the inflammatory type B, PSS type A lacks signs of inflammation or other symptoms or involvement of mucous membranes and nails. A missense mutation in CHST8 has been described that encodes a Golgi transmembrane N-acetylgalactosamine-4-O-sulfotransferase (GalNAc-4-ST1) that may be important for epidermal homeostasis resulting in increased and continuous desquamation of the stratum corneum.
Histology shows a plane of separation either within the lower part of an otherwise normal horny layer or above the granular cell layer. Ultrastructural analysis reveals an intracellular splitting within the corneocytes.
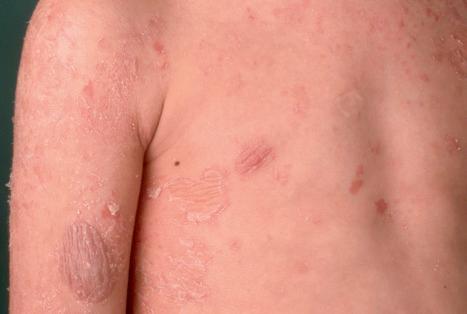
Peeling skin syndrome type B (inflammatory peeling skin disease) presents at birth with ichthyosiform erythroderma and is characterized by lifelong patchy peeling of the entire skin with severe pruritus. Isolated erythematous lesions show flaccid peeling, leaving burning superficially denuded red patches with a peripheral collarette ( Fig. 3.46 ). Easy plucking of the hair and loose nail plates (onychomadesis) are further symptoms. Trigger factors are mechanical stress, low humidity or temperature changes. Pruritus and allergic symptoms of elevated IgE, food allergies, and asthma are reminiscent of Netherton syndrome. Autosomal recessive loss-of-function mutations of CDSN encoding corneodesmosin have been identified as the molecular cause of the disease.
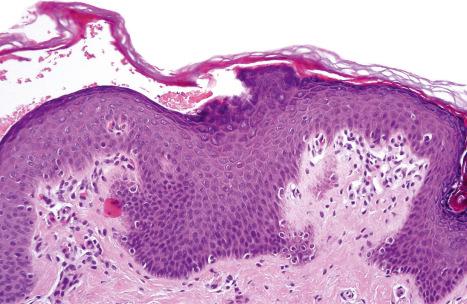
Histologically, the epidermis displays psoriasiform hyperplasia with an absent or reduced granular cell layer and marked parakeratosis. Careful examination reveals detachment of the horny layer from the granular cell layer ( Fig. 3.47 ). Ultrastructurally, a loss of corneodesmosomes can be demonstrated, which is associated with intercellular splitting of the corneocytes from the stratum granulosum. Immunostaining for corneodesmosin and LEKTI may help to distinguish between Netherton syndrome and PSS type B.
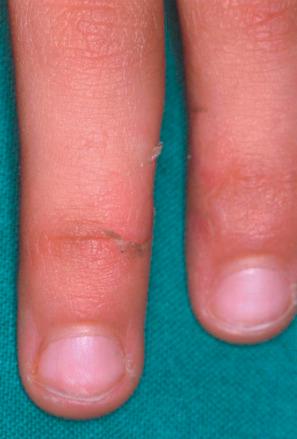
Acral peeling skin syndrome is characterized by superficial painless peeling of the backs of the hand and feet ( Fig. 3.48 ). In children blistering occurs on palms and soles. Only recently it was reclassified as a form of epidermolysis bullosa. Missense mutations in the gene of transglutaminase-5 ( TGM5) represent the molecular cause of this autosomal recessive disease—in contrast to its major differential diagnosis epidermolysis bullosa simplex (EBS), which has an autosomal dominant inheritance. Other differential diagnoses of acral PSS include epidermolysis simplex superficialis and keratolysis exfoliativa. Histologically, the horny layer is detached from the stratum granulosum.
Another type of PSS is related to the CSTA gene, mutations of which cause a deficiency of cystatin A. Initially, this variant was reported as ‘exfoliative ichthyosis’, a nonsyndromic congenital ichthyosis with dry and scaly skin associated with diffuse PPK sensitive to sweat and water exposure. The disease may display clinical features of SEI.
Keratosis linearis-ichthyosis congenita-sclerosing keratoderma (KLICK) is an autosomal recessive genodermatosis presenting with a moderate, non-blistering ichthyosis from birth. Pathognomonic findings are keratotic punctuate plugs and papules arranged in parallel lines and circumscribed follicular keratosis around the wrists, in the folds of arms, axillae, and knees. In addition, a diffuse palmoplantar keratoderma develops including the dorsal side of hands and feet ( Fig. 3.49 ). Other features include a sclerosing flexion deformity of the fingers and constriction bands. There are no other associated features, but there is a report of an associated squamous cell carcinoma at a younger age. Differential diagnosis includes loricrin keratoderma that also features palmoplantar keratoderma and a mild congenital ichthyosis but is less sclerotic and has an autosomal-dominant pattern on inheritance.
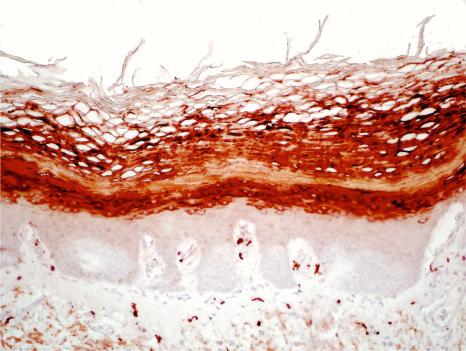
In KLICK a mutation of a proteasome maturation protein ( POMP ) has been identified. This protein serves as a chaperone for proteolytic enzymes degrading unneeded or damaged proteins. This proteasome insufficiency disturbs terminal epidermal differentiation and particularly interferes with processing of profilaggrin.
Histologically, the epidermis is acanthotic and orthohyperkeratotic with focal parakeratosis and shows prominent hypergranulosis with irregular keratohyaline granules. This is reflected by immunohistochemistry for filaggrin which reveals extensive immunoreactivity in the upper epidermis that characteristically extends into the horny layer ( Fig. 3.50 ). On non-volar skin follicular plugging is present. The papillary dermis shows papillomatosis and an inconsistent, mild perivascular lymphocytic infiltrate. Ultrastructure confirms the histologic finding of hypergranulosis and shows abnormally big keratohyaline granules.
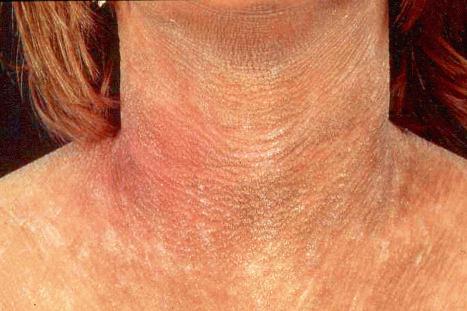
This acronym was defined by Samuelov et al. in 2013 and refers to the cardinal features of severe dermatitis, multiple allergies, and metabolic wasting. SAM is another exfoliative disorder of cornification and thus resembles Netherton syndrome or PSS type B. SAM is caused by loss-of-function mutations in DSG1 . Accordingly, apart from a psoriasiform dermatitis, subcorneal separation and acantholysis within the stratum spinosum and granulosum can be found. At the ultrastructural level half-split desmosomes are evident.
This autosomal recessive inherited disorder combines the features of ichthyosis, spastic bi- or quadriplegia and mental retardation. It is rare, with an incidence of 0.4 per 100 000 of the population albeit it has a higher prevalence of 1 : 10 000 in Northern Sweden.
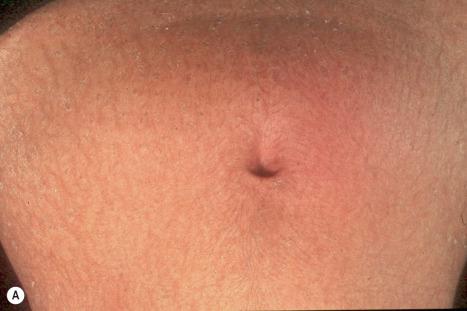
The ichthyosis, which develops in the first year of life with diffuse scaling, affects the entire body with the exception of the central face and is typically intensely pruritic ( Fig. 3.51 ). Later, the skin has a brownish-yellow color and shows a cobblestone-like lichenification. Hyperkeratosis around the umbilicus is said to be characteristic. Erythroderma is not a feature and the hair, nails, and sweat glands are unaffected. The diagnosis should be especially considered in preterm babies with congenital ichthyosis.
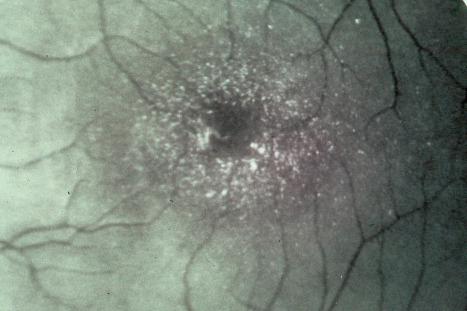
The spasticity, which presents in early childhood, predominantly affects the legs and is often associated with contractures. The majority of patients are wheelchair bound. Kyphoscoliosis may also be present. Mental retardation is typically present, but is not invariable. Epilepsy occurs in up to 40% of patients.
Visual acuity is often impaired and photophobia is a frequent complaint. Macular degeneration associated with crystal deposition is characteristic ( Fig. 3.52 ).
Sjögren-Larsson syndrome results from deficiency of microsomal fatty aldehyde dehydrogenase ( FALDH ). The gene has been mapped to 17p11.2 and multiple mutations, including missense mutations, deletions, and insertions, have been identified. In the pathophysiology of the ichthyosis a disturbed hepoxilin pathway is involved, while an abnormal lipid composition of myelin accounts for the neurological defects. The abnormal level of free fatty alcohols in cultured fibroblasts, direct testing of FALDH activity, or the presence of LTB4 metabolites in urine can provide biochemical screening and/or confirmation of the clinical diagnosis, prior to molecular mutation analysis of the FALDH gene.
Histologically, there is papillomatosis, acanthosis, and basket-weave hyperkeratosis with scattered mild parakeratosis and occasional follicular hyperkeratosis ( Fig. 3.53 ). Epidermal hyperproliferation has been demonstrated. The granular cell layer may be slightly thickened. A moderate lymphocytic infiltrate is sometimes present around the superficial dermal vasculature.
Ultrastructurally, there are lamellar inclusions in the prickle and granular cell layers. Lipid inclusions are not a feature.
Multiple sulfatase deficiency is a severe neuropediatric disorder inherited in an autosomal recessive mode. Pathophysiologically, mutations in the SUMF1 gene block the post-translational modification of sulfatases and result in accumulation of glycosaminoglycans (GAGs) and sulfated lipids. There are severe neonatal, and severe or mild infantile or juvenile forms characterized by progressive loss of mental capacity and motor abilities. Patients usually die before puberty. The ichthyosis is typically mild compared to the severity of the neurological changes. Therefore, ichthyosis in a child with unexplained neurological symptoms should always prompt measurement of steroid sulfatase levels.
Refsum syndrome (hereditary motor and sensory neuropathy type 4, heredopathica atactica polyneuritiformis, phytanic acid deficiency) is a rare type of an autosomal recessive syndromic ichthyosis. The skin changes appear in late childhood and are similar to those seen in ichthyosis vulgaris, including hyperlinear palms. Due to lipid storage, melanocytic nevi may show a yellow hue. Associated symptoms include loss of vision from retinitis pigmentosa, in which night blindness is often the first problem. Other complications are anosmia, cardiac arrhythmias, and a whole spectrum of neurological problems, including bilateral sensorineural deafness, cerebellar ataxia, and peripheral polyneuropathies.
Refsum syndrome is generally caused by a mutation in the PHYH gene encoding peroxisomal phytanoyl-CoA hydroxylase. Mutations in the peroxisomal receptor gene PEX7 account for adult forms. Peroxisomes are involved in the metabolism of bile acid and cholesterol biosynthesis. Elevated levels of phytanic acid in plasma and tissue are diagnostic. Therapy includes a low-phytol diet and apharesis.
Routine histology of a skin biopsy does not differ from ichthyosis vulgaris. When a biopsy is fixed in alcohol and a Sudan stain performed, lipid droplets are found in the keratinocytes, in particular in biopsies from melanocytic nevi. The same inclusions can be shown by ultrastructural examination.
Dorfman-Chanarin syndrome (neutral lipid storage disease with ichthyosis) is a triglyceride storage disease with impaired long-chain fatty acid oxidation resulting in cataracts, hepatosplenomegaly, neurosensorial deafness, myopathy or developmental delay. Affected neonates present as collodion babies or are erythrodermic with scaling. Later the ichthyosis resembles CIE with ectropion, flexural and neck lichenification, and palmoplantar hyperkeratosis. Pruritus and hypohidrosis are common.
The disease is due to mutations in the ABHD5 gene product that is required by triglyceride lipase as a cofactor in the muscle, liver, and brain, but also in white blood cells. The genetic defect leads to acylceramide deficiency resulting in ichthyosis and possibly to associated hepato-neurologic symptoms. Histology shows orthohyperkeratosis with a possibly thinned stratum granulosum. Characteristically, intracellular lipid vacuoles can be demonstrated in the keratinocytes as well as in a variety of other cells, including circulating neutrophils (Jordans anomaly). A skin biopsy fixed in alcohol is useful. Lipid vacuoles may also be found in the obligate carrier parents. Refsum syndrome patients also have epidermal lipid vacuoles, but in Dorfman-Chanarin syndrome patients, the phytanic acid levels are normal.
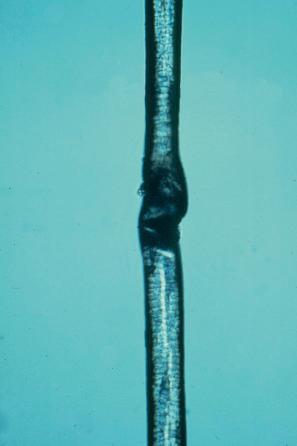
Trichothiodystrophy (syn. Tay syndrome, IBIDS syndrome, PIBIDS syndrome, Amish brittle hair syndrome) represents a heterogeneous group of autosomal recessive neurocutaneous disorders, some of them photosensitive, that share sulfur-deficient brittle hairs. Trichoschisis and brittle hairs are due to an abnormally low hair-shaft sulfur content with a decrease in cysteine. On polarizing microscopy, an alternating light and dark banding (‘tiger-tail pattern’) appears pathognomonic ( Fig. 3.54 ). Trichothiodystrophy is associated with congenital ichthyosis: the acronym IBIDS (‘Tay syndrome’) refers to the clinical findings of ichthyosis (e.g., collodion membrane), brittle hair, intellectual impairment, decreased fertility, and short stature. Other features are microcephaly, dysplasia of nails, failure to thrive, ‘progeria’-like symptoms, cataracts, and photosensitivity (≈ P IBIDS).

In the photosensitive group, DNA-repair anomalies involving various subunits of the transcription factor TFIIH have been identified, while the non-photosensitive group, without a DNA-repair defect, exhibits mutations in the C7ORF11 gene coding for TTDN1 protein. Despite the DNA-repair defect and in contrast to xeroderma pigmentosum, an increased risk of malignancy is not regarded as a feature of photosensitive trichothiodystrophy.
Histology of the ichthyotic skin shows acanthosis with orthohyperkeratosis and a reduced stratum granulosum (see Table 3.3 ).
Neu-Laxova syndrome is an autosomal recessive inherited congenital ichthyosis caused by mutations leading to phosphoglycerate dehydrogenase deficiency. It is characterized by marked intrauterine growth retardation, polyhydramnion, and congenital ichthyosis with a thin, tightly adherent, translucent skin and typical facial dysmorphology that resembles restrictive dermopathy, another neurocutaneous genodermatosis. Associated syndromic features are microcephaly, central nervous system anomalies, short neck, limb deformities, and hypoplastic lungs. Histology of the skin reveals focal parakeratosis.
In ichthyosis prematurity syndrome there is polyhydramnion and the premature neonates may suffer from transient asphyxia. The infants have a thick cheesy membrane which desquamates followed by skin improvement within some weeks. This syndrome is caused by mutations in the fatty acid transport protein 4 gene ( SLC27A4 ). The skin shows compact orthohyperkeratosis and acanthosis.
At ultrastructural level, characteristic masses of lipid membranes in lentiform paranuclear swellings of granular and horn cells can be demonstrated which has led to the designation ichthyosis congenita type 4.
The combination of neurologic manifestations and ichthyosis can be found in at least 16 distinct genetic disorders (see Table 3.2 ). Gaucher disease type II (infantile cerebral Gaucher) is another classic neuro-ichthyosis presenting at birth with collodion membranes. The diagnosis of this fetal metabolic disease can be made by measurement of glucocerebrosidase activity in peripheral blood leukocytes or in extracts of cultured skin fibroblasts. Other examples are cerebral dysgenesis-neuropathy-ichthyosis-palmoplantar keratoderma (CEDNIK) syndrome , arthrogryposis-renal dysfunction-cholestasis (ARC) syndrome , and mental retardation-enteropathy-deafness-neuropathy-ichthyosis-keratoderma (MEDNIK) syndrome. ELOVL4 deficiency presents with erythrokeratoderma and sensorineural deafness, thus resembling keratitis-ichthyosis-deafness (KID) syndrome. Patients with Stormorken syndrome belong to the group of channelopathies affecting calcium homeostasis and is characterized with moderate thrombocytopenia, thrombocytopathia, muscle fatigue, asplenia, miosis, migraine, and dyslexia, altogether with ichthyosis.
Conradi-Hünermann-Happle syndrome is an X-linked dominant congenital ichthyosis with associated chondrodysplasia punctata. It is lethal in the majority of male embryos. Chondrodysplasia punctata is defined as a stippled calcification of the epiphyses. There are several forms but only the type 2 variant presents with severe ichthyosiform erythroderma. Later the erythema clears and persistent whorled scaling following the lines of Blaschko is noted ( Fig. 3.55 ).
Associated symptoms are scarring alopecia, follicular atrophoderma, localized hypo-and/or hyperpigmentation, sectorial cataracts, and skeletal dysplasia, which leads to asymmetric shortening of the long bones or severe kyphoscoliosis. Due to the individual differences in X-inactivation, expression of the disease is rather variable, even within families.
Biochemical analyzes using gas chromatography-mass spectrometry show elevated plasma levels of 8-dehydrocholesterol and 8(9)-cholesterol, resulting from a block of a key enzyme in sterol metabolism, namely the 8–7 sterol isomerase . This enzyme is encoded by the emopamil-binding protein gene, which shows heterozygous mutations in Conradi-Hünermann-Happle syndrome. The developmental abnormalities are presumably caused by interference of cholesterol precursors with sonic hedgehog signaling. Abnormal cholesterol metabolism in the epidermis may account for the ichthyotic phenotype.
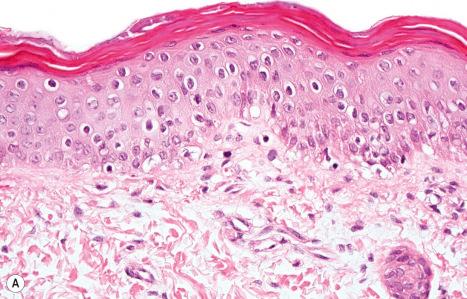
The histologic features resemble those of ichthyosis vulgaris. There is hyperplasia of the epidermis, orthohyperkeratosis, a reduced stratum granulosum, and dilated hair follicle infundibula with keratin plugs. As a pathognomonic finding in newborns, von-Kossa staining demonstrates calcium deposits in the corneocytes of the stratum corneum and hair follicles. This feature allows discrimination from other ichthyoses that share the feature of a reduced stratum granulosum ( Fig. 3.56 ) (see Table 3.3 ). At a later age the calcification is difficult to detect histologically but electron microscopy may reveal cytoplasmic vacuoles and electron-dense calcium crystals in the granular cell layer. In addition, LBs lack their normal lamellar structure.
Follicular ichthyosis (ichthyosis follicularis) is an umbrella term or histologic pattern that is present in many conditions and is defined by follicular orthohyperkeratosis with or without hypergranulosis in the infundibulum. For differential diagnosis see Table 3.3 .
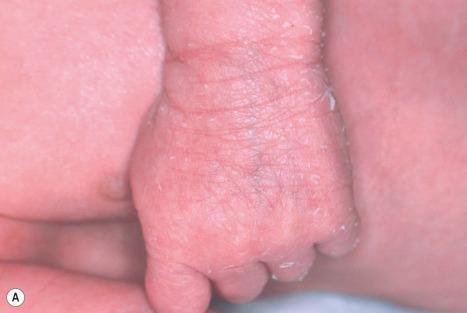
Ichthyosis follicularis with alopecia (atrichia) and photophobia (IFAP syndrome) is an exceedingly rare X-linked, recessively inherited disease in males. Children can be born as collodion babies and characteristically present with generalized filiform follicular keratosis over the entire body including the scalp that often improves during the first year of life ( Fig. 3.57 ).
Other features are ichthyosiform dry skin, generalized total nonscarring alopecia (with absence of eyelashes and eyebrows), and severe photophobia. Ocular findings may include corneal deformity and opacity with surface vascularization. Angular cheilitis, keratotic psoriasiform plaques on the extensor surfaces of the extremities, and nail dystrophy with periungual infection may also be present. Additional findings including hypohidrosis, recurrent respiratory infections, skeletal abnormalities, growth retardation, cryptorchidism or progressive deteriorating neurologic symptoms such as generalized seizures, mental retardation, and cerebellar symptoms. Female carriers are affected by circumscribed hairless, anhidrotic or ichthyotic areas along the lines of Blaschko.
IFAP is caused by missense mutations in the X-linked gene MBTPS2 encoding membrane-bound transcription factor protease, site 2. Different mutations on other sites in the MBTPS2 gene are responsible for keratosis follicularis spinulosa decalvans, Olmsted syndrome, and BRESEK/BRESHECK syndrome.
Membrane bound transcription factor protease site 2 is a zinc metalloprotease essential for cholesterol homeostasis as well as for endoplasmic reticulum (ER) stress response. In cultured cells of IFAP patients, residual enzyme activity is only about a third of wild type activity.
The follicular lesions are characterized by projecting hyperkeratotic plugs showing focal parakeratosis and associated hypergranulosis. Hair follicles are atrophic and lack hair shafts and sebaceous glands ( Fig. 3.58 ). Sweat glands are normal, but hyperkeratosis of the acrosyringia may occlude the openings of sweat ducts ( Fig. 3.59 ).
The psoriasiform plaques show hyperkeratosis with parakeratosis, acanthosis, spongiosis, and a bandlike upper dermal lymphohistiocytic infiltrate.
Other forms of atrichia and follicular keratosis should be considered (see Table 3.3 ). Hereditary mucoepithelial dysplasia (HMD) may also present with photophobia and keratosis pilaris. However, it has an autosomal dominant trait and shows no mutations in MBTPS2 .
This congenital ichthyosis has two allelic variants, one characterized by a whole-body hypotrichosis without atrophoderma (autosomal recessive ichthyosis with hypotrichosis [ARIH]) and one with follicular atrophoderma, i.e., follicular pitting on the dorsal aspects of the hands (congenital ichthyosis, follicular atrophoderma, hypotrichosis, and hypohidrosis [IFAH]). Both phenotypes are associated with autosomal recessive mutations in the ST14 gene, which encodes a matriptase that plays an important role in the epidermal protease network. Clinical heterogeneity may be caused by different types of ST14 mutations. The hair phenotype can be explained by the fact that matriptase is expressed in the cortex cells and shaft of the anagen hair.
Neonatal ichthyosis-sclerosing cholangitis (NISCH) syndrome is characterized by neonatal ichthyosis typically associated with jaundice due to sclerosing cholangitis of variable severity. Other features include hypotrichosis of the scalp with frontal scarring alopecia, and leukocyte vacuoles. Autosomal recessive mutations in CLDN1 encoding claudin-1 have been detected. Claudin-1 is a major component of the tight junctions in the epidermis, cholangiocytes, and hepatocytes. Dysfunctions of claudin 1 account for permeability between epithelial cells resulting in hypercholanemia or epidermal barrier defect.
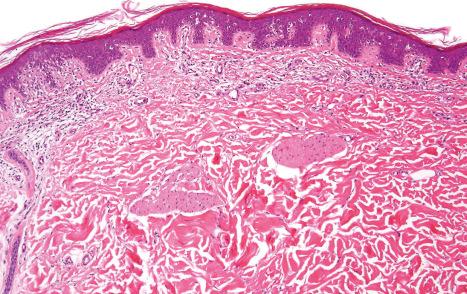
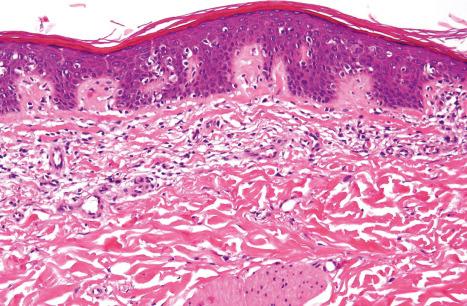
Acquired ichthyosis-like or ichthyosiform conditions refer to patients who develop diffuse ichthyosis-like scaling during their life ( Table 3.4 ). The adult-onset renders the term ‘acquired ichthyosis’ inappropriate. It is an important paraneoplastic manifestation of a number of malignancies: Hodgkin lymphoma is most often encountered, but non-Hodgkin lymphoma, including mycosis fungoides and a range of carcinomas, are also associations. Ichthyosiform skin changes may also accompany malnutrition, human immunodeficiency virus (HIV) and other infectious diseases, sarcoidosis, connective tissue diseases, celiac disease and other gastrointestinal diseases, renal insufficiency, hypothyroidism, and graft-versus-host disease (GVHD). Ichthyosiform skin changes following kava consumption or the administration of lipid-lowering agents and other various drugs have been documented. The features of acquired ichthyosis-like skin conditions most often resemble those of ichthyosis vulgaris both clinically and histologically ( Figs 3.60–3.63 ).
| Etiology | Diseases |
|---|---|
| Dry skin | None |
| Paraneoplastic | Hodgkin and non-Hodgkin lymphoma Multiple myeloma, myelodysplasia |
| Lymphomatoid papulosis Kaposi sarcoma |
|
| Various carcinomas | |
| Infections | Leprosy |
| Tuberculosis | |
| HIV/AIDS | |
| Malnutrition | Pellagra |
| Vitamin A deficiency | |
| Drugs (examples) | Lipid-lowering agents (statins), retinoids |
| Nicotinic acid, clofazimine, allopurinol, cimetidine, | |
| lithium, hydroxyurea | |
| Gastrointestinal diseases | Crohn disease |
| Celiac disease | |
| Gastrectomy | |
| Endocrinopathies | Hyperparathyroidism |
| Hypothyroidism Diabetes |
|
| Miscellaneous | Renal insufficiency |
| Sarcoidosis | |
| Graft-versus-host disease | |
| Dermatomyositis and systemic lupus erythematosus | |
| Down syndrome |
Clinical differential diagnosis includes xerosis cutis, which lacks thick scales, develops at a later age, and can be easily treated by emollients.
Also known as pityriasis circinata, this acquired disorder of keratinization was originally described in the Japanese. It is also not uncommon in South Africans (Bantu) and West Indian blacks, and has been reported in a subpopulation of Italians in Sardinia.
Patients present with persistent, very sharply defined, circular or oval areas of hyper- or hypopigmentation associated with a fine scale ( Fig. 3.64 ). Lesions, which are usually multiple and frequently numerous, are characteristically noninflammatory and asymptomatic. Often, they are confluent. They measure 0.5–28 cm in diameter and are particularly located on the trunk and limbs. The sex incidence is equal. Lesions are sometimes associated with gradual remission during the summer months and relapse in winter. The maximum incidence is in the third to fifth decades. There is often a family history of ichthyosis vulgaris. Familial cases may occasionally be seen.

Pityriasis rotunda sometimes, but not always, appears to be a cutaneous marker of severe internal disease, including tuberculosis, cancer (particularly hepatoma), myeloma, leukemia, cirrhosis, ovarian and uterine disease, malnutrition, diabetes, and favism. Pityriasis rotunda is best be regarded as an acquired circumscribed variant of ichthyosis.
The histologic features are subtle and consist of compact orthohyperkeratosis with a diminished or absent granular cell layer and loss of the epidermal rete ridge pattern. Immunohistochemistry reveals reduced expression of filaggrin and loricrin. Increased pigmentation of the basal keratinocytes may be evident. A mild perivascular chronic inflammatory cell infiltrate is sometimes present in the superficial dermis. A superficial fungal infection, mainly pityriasis versicolor, should always be excluded.
This fairly common condition, which has an autosomal dominant mode of inheritance, is probably a follicular variant of ichthyosis and, indeed, frequently accompanies ichthyosis vulgaris. The age at presentation is most often in the first two decades of life with a peak during adolescence. There is an apparent increased incidence in females, in particular those suffering from hyperandrogenism and obesity. The lesions present as pruritic small follicular keratoses, sometimes containing small, distorted hairs. They are most often found on the lateral aspects of the arms and thighs, although the face, trunk, and buttocks may also be affected ( Fig. 3.65 ). Seasonal variation, with lesions being much more severe in winter, is often documented. There is an increased incidence of atopy.
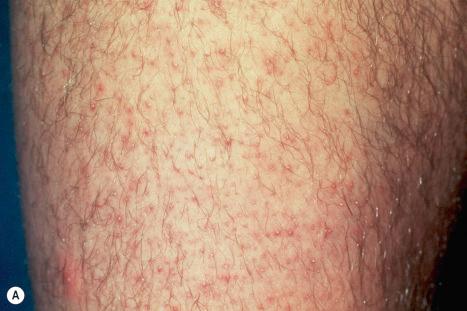
Although keratosis pilaris most often presents as an isolated phenomenon, occasionally it may develop in association with systemic disease, including Hodgkin lymphoma, vitamins B 12 and C deficiency, hypothyroidism, Cushing disease, and treatment with adrenocorticotropic hormone. A keratosis pilaris-like eruption has been described in BRAF kinase inhibitor therapy.
Keratosis pilaris is characterized by follicular dilatation and keratin plugs, which may contain one or several distorted hair shafts ( Fig. 3.66 ). A mild, lymphocytic cell infiltrate surrounds superficial dermal blood vessels and sometimes also involves the hair follicles.
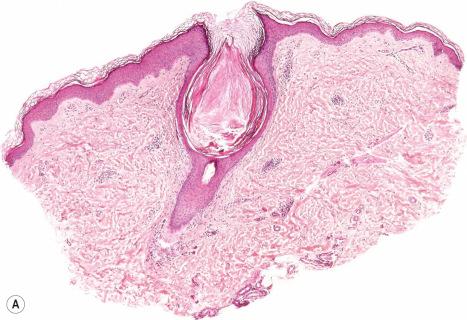
Keratosis pilaris atrophicans combines the features of follicular hyperkeratosis and atrophic scarring. According to different modes of inheritance, clinical presentation, and variable associations three conditions have been described, namely ulerythema ophryogenes, atrophoderma vermiculata, and keratosis follicularis spinulosa decalvans.
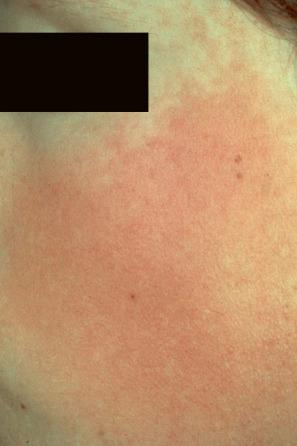
Ulerythema ophryogenes (keratosis pilaris atrophicans faciei, KPAF) presents at birth or in early infancy with follicular papules and surrounding erythema followed by atrophic scarring affecting the lateral aspect of the eyebrows ( Fig. 3.67 ). The cheeks, forehead, temples, and neck may also be involved ( Fig. 3.68 ). Later on, there may be complete loss of eyebrows. Keratosis pilaris affecting the extensor aspects of the arms and thighs is also sometimes present. The condition is believed to be inherited as an autosomal dominant disorder.
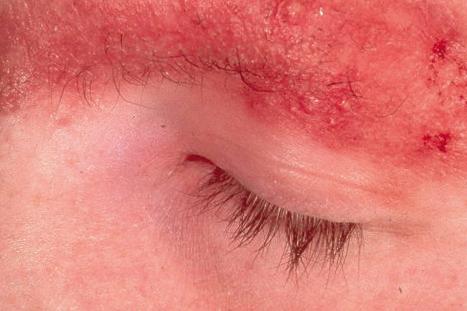
It may be associated with a number of other inherited disorders, including Noonan syndrome, wooly hair, cardiofaciocutaneous syndrome, Cornelia de Lange syndrome, Rubinstein-Taybi syndrome, and partial monosomy 18. The association with Noonan syndrome is of particular importance since such patients suffer from potentially life-threatening congenital pulmonary stenosis. Ulerythema ophryogenes is also associated with atopy.
Atrophoderma vermiculata (ulerythema acneiforme, atrophoderma vermiculatum, atrophoderma reticulata, acne vermoulante, folliculitis ulerythema reticulata, folliculitis ulerythematosa, honeycomb atrophy) is an exceedingly rare form of atrophic keratosis pilaris with an autosomal dominant inheritance. Patients present with follicular keratoses and pitted depressions separated by normal skin (moth-eaten appearance) affecting the cheeks, ears, and forehead (honeycomb atrophy). The disorder presents in patients after 5 years of age. Unilateral nevoid variants following Blaschko lines have also been documented.
Keratosis follicularis spinulosa decalvans is characterized by diffuse atrophic keratosis pilaris associated with scarring alopecia affecting the scalp. Other conditions sometimes present include atopy, palmoplantar hyperkeratosis, photophobia, and punctate keratitis. In some patients it is inherited as an X-linked recessive disorder that is caused by mutations in the MBTPS2 gene encoding membrane-bound transcription factor protease, site 2, which has been mapped to Xp21.13-p22.2. X-linked dominant and autosomal dominant variants have also been described. A pustular variant on the scalp that starts at puberty has been described as ‘folliculitis spinulosa decalvans’.
The pathogenesis of keratosis pilaris atrophicans is unknown. The MBTPS2 , as affected in keratosis follicularis spinulosa decalvans and IFAP, may interfere with sterol control and endoplasmatic reticulum stress response.
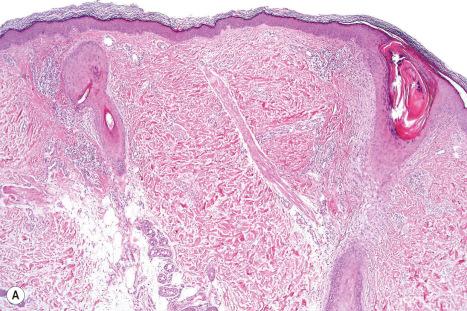
All variants of keratosis pilaris atrophicans are characterized by follicular hyperkeratosis with ostial dilatation, atrophy of the sebaceous gland, and a scanty perifollicular and/or perivascular lymphohistiocytic infiltrate. Comedones and milia may be found. There is variable perifollicular fibrosis that extends into the reticular dermis ( Fig. 3.69 ).
Lichen spinulosus is a rare dermatosis of unknown etiology which particularly affects the extensor surfaces of the extremities, back, chest, buttocks, face, and neck. Occasionally, lesions are generalized. The condition presents in the second and third decades of life as round to oval, 2–6-cm flesh-colored and sometimes pruritic, symmetric plaques composed of multiple 1–3-mm thorny, grouped follicular papules which protrude above the surface of the skin. The condition is of no clinical significance. Lichen spinulosus has been described in association with atopy, Crohn disease, HIV infection, and as an adverse drug reaction.
Lichen spinulosus is characterized by keratotic plugging of dilated follicular infundibula and a superficial perivascular and perifollicular lymphohistiocytic infiltrate. Sebaceous glands may be atrophic or absent. Perforating folliculitis-like features can be superimposed.
There is considerable histologic overlap with keratosis pilaris and the follicular lesions of pityriasis rubra pilaris. The distinction is best made clinically.
Phrynoderma (toad skin) most often develops as a consequence of vitamin A deficiency. Other proposed etiological factors include deficiencies of the vitamin B complex, riboflavin, vitamin C, vitamin E, and essential fatty acids. In Western countries most cases develop as a result of malabsorption. Patients present with xerosis, hyperpigmentation and multiple 2–6-mm, red-brown, dome-shaped papules with a central folliculocentric crater filled with laminated keratinous debris. The elbows and knees are predominantly affected, but lesions may extend to involve the thighs, upper arms, and buttocks.
The papules consist of a cystically dilated follicular infundibulum filled with keratinous debris.
‘Erythrokeratoderma’ or ‘erythrokeratodermia’ refers to a group of genodermatoses characterized by localized erythematous lesions, hyperkeratotic plaques, and, infrequently, mild palmoplantar keratosis. Many of these diseases represent connexin mutations ( Table 3.5 ). Connexin genes code for transmembrane proteins that form gap junctions and are involved in epidermal differentiation. Other connexin mutations, such as the KID/hystrix-like ichthyosis-deafness (HID) syndrome and Vohwinkel syndrome are associated with sensorineural hearing loss. In others, the genetic defect has yet to be identified. According to a consensus conference erythrokeratodermas are now classified as ichthyoses.
| Disease | Inheritance | Locus | Gene | Protein |
|---|---|---|---|---|
| Erythrokeratoderma variabilis (and progressive symmetrical erythrokeratoderma) | AD or AR AD |
1q35.1 1q35.1 |
GJB3 GJB4 |
Connexin 31 Connexin 30.3 |
| Erythrokeratoderma variabilis with erythema gyratum repens-like lesions | AD | 1q35.1 | GJB4 | Connexin 30.3 |
| Keratitis-ichthyosis-deafness syndrome/hystrix-like-ichthyosis deafness syndrome (KID/HID syndrome) | AD | 13q11-12 | GJB2 | Connexin 26 |
| Oculodentodigital dysplasia | AD | 6q22-24 | GJA1 | Connexin 43 |
| Vohwinkel keratoderma | AD | 13q11-12 | GJB2 | Connexin 26 |
| Hidrotic ectodermal dysplasia of Clouston | AD | 13q11-12 | GJB6 | Connexin 30 |
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here