Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Hyperpigmented lesions, presenting at birth and during the first few weeks of life, are quite common. Pigmented lesions can range from small and isolated to large and multiple. They can exist independently, or in association with other signs and symptoms, and may lead to the diagnosis of a congenital or genetic skin disease. In some cases, hyperpigmented ‘birthmarks’ may not be evident at birth but appear weeks to months later. The relatively light color of infant skin can make some pigmentary disorders difficult to appreciate and thus observation over time may be helpful. Also, over time, melanocytes may produce more pigment and differences between normal and hyperpigmented or hypopigmented skin may become more evident. This chapter serves as an aid for clinicians in diagnosing pigmented lesions in the neonatal and infantile period with an approach based on coloration and localization of lesions with a review of diagnostic criteria, associated conditions, and suggested clinical evaluation when applicable ( Table 24.1 ).
| Description of lesions | Location or morphology | Possible diagnoses |
|---|---|---|
| Blue-gray/blue-black patches | Torso Face Shoulder/neck Torso, in association with port-wine stain |
Mongolian spot Nevus of Ota Nevus of Ito Phakomatosis pigmentovascularis |
| Labial macules | Perioral More widespread facial Face/trunk |
Peutz–Jeghers syndrome Carney syndrome LEOPARD syndrome Carney syndrome |
| Face | Centrofacial lentiginosis | |
| Brown sharply defined patches or plaques | Congenital melanocytic nevus Café-au-lait macule |
|
| Small brown macules | Perioral/mucosal Widespread, non-mucosal |
Peutz–Jeghers syndrome LEOPARD syndrome Generalized lentiginosis Inherited patterned lentiginosis |
| Central face/widespread | Carney syndrome | |
| Involves mucosa | ||
| Axilliary/groin/neck only | Neurofibromatosis | |
| Central face only, nonmucosal | Centrofacial lentiginosis | |
| Clustered in a defined body area or segment. Background skin color normal | Segmental lentiginosis Mosaicism |
|
| Clustered in a defined body area or segment. Background skin color darker | Speckled lentiginous nevus | |
| Single | Congenital melanocytic nevus | |
| Linear hyperpigmentation in Blaschko pattern | Flat | Linear and whorled nevoid hypermelanosis (early) Epidermal nevus Incontinentia pigmenti (stage 3) Goltz syndrome Conradi–Hünermann syndrome |
| Raised | Mosaicism Human chimerism Epidermal nevus Incontinentia pigmenti (stage 1, 2) |
Café-au-lait macules (CALM) are well-circumscribed, oval to round, hyperpigmented hairless macules or patches that may be noted at birth, during infancy or throughout childhood. CALM may have the color of ‘coffee with milk’ in lighter skinned individuals but in darker skinned persons the color may be more medium to dark brown. The size of CALM ranges from a few millimeters to over 20 cm and can occur anywhere on the body ( Fig. 24.1 ). Morphologically, the borders of CALM have been described as smooth (resembling the ‘coast of California’) to jagged contours (resembling the ‘coast of Maine’), correlating with specific disorders. However, in general, this feature varies widely and it not a reliable diagnostic sign.
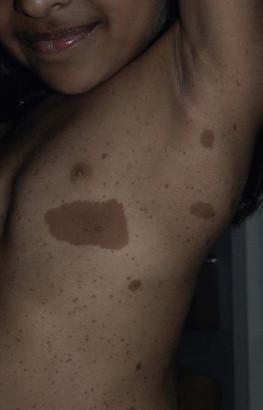
The prevalence of CALM varies in the general population with 2.7% of 4641 newborns having one or more macules in a US study. There are great differences in prevalence amongst various racial groups, with an increased number of CALM seen in darker skin types. A higher prevalence (24.2–36%) of CALM has been reported in older children and young adults as compared with infants. The presence of up to three CALM has been reported in 10–28% of the general population. Multiple CALM, especially six or more, should prompt the clinician to consider underlying disorders ( Table 24.2 ).
| Strong association | Weaker association b | ||
|---|---|---|---|
| Major features | Major features | ||
| Familial inherited CALM a | Multiple (≥2) generations with multiple (≥10) CALM and other NF-1 stigmata (−) | Tuberous sclerosis | Hypopigmented macules, facial angiofibromas, CNS tubers, retinal hamartomas, renal cysts, angiomyolipoma, cutaneous collagenomas, seizures, mental retardation, cardiac rhabdomyoma, periungual fibromas, subependymal giant cell astrocytomas |
| Neurofibromatosis type I | Skin-fold freckling, neurofibromas, Lisch nodules, neurocognitive deficits, optic glioma, skeletal dysplasia, macrocephaly | Bloom syndrome | Short stature, photosensitivity, susceptibility to malignancy, chronic lung disease, immunodeficiency, cryptorchidism |
| Neurofibromatosis type II | Acoustic neuromas, meningiomas, cataracts, schwannomas, neurofibromas | Ataxia-telangiectasia syndrome | Progressive cerebellar ataxia, lymphoreticular malignancy, cutaneous/ocular telangiectasia, immunodeficiency, hypogonadism |
| Legius syndrome | Macrocephaly, skin-fold freckling, developmental disorders | Silver–Russell syndrome | Congenital short stature and low birthweight, craniofacial and body asymmetry, limb anomalies, microcephaly |
| Watson syndrome | Short stature, pulmonary stenosis, low intelligence, skin-fold freckling | Bannayan–Riley– Ruvalcaba syndrome | Congenital macrosomia, megalencephaly, lipomas, intestinal polyps, vascular anomalies |
| Ring chromosome syndrome | Microcephaly, mental retardation, short stature, skeletal anomalies | Noonan syndrome | Facial dysmorphism, pulmonic valve stenosis, webbed neck, pectus excavatum, mental retardation, short stature, cryptorchidism, hematologic malignancies |
| Hereditary spinal neurofibromatosis | Skin-fold freckling, spinal root neurofibromas | Faconi anemia | Bone marrow failure, limb anomalies, mental retardation, microcephaly, predisposition to malignancy |
| McCune–Albright syndrome | Polyostotic fibrous dysplasia, endocrinopathies | Turner syndrome | Short stature, lymphedema, congenital heart disease, valgus deformity |
| LEOPARD syndrome (multiple lentigines syndrome) | Multiple lentigines, hypertelorism, pulmonic valve stenosis, EKG abnormalities, sensorineural deafness, growth retardation, genitourinary abnormalities | ||
| Multiple endocrine neoplasia syndrome type I | Parathyroid adenoma, pituitary adenoma, pancreatic islet adenoma, lipomas, gingival papules, facial angiofibromas, collagenomas | ||
a Arnsmeier, Riccardi and Paller proposed that absence of two consecutive generations of neurofibromas and other non-pigmentary changes of NF-1 allows the diagnosis of familial CALM.
b Syndromes listed have been described with CALM but these are not considered part of the diagnostic criteria, and may not be seen in all cases.
Although the diagnosis is generally made through clinical examination, histologic examination of CALM shows increased melanin content in keratinocytes and melanocytes without evidence of melanocyte proliferation. On electron microscopy, giant melanosomes may be seen. While establishing the diagnosis of CALM, these microscopic features are not useful in determining specific systemic associations (such as neurofibromatosis).
The differential diagnosis of CALM includes a congenital melanocytic nevus, speckled lentiginous nevus, lentigo, Becker's nevus and forms of pigmentary mosaicism such as nevoid hypermelanosis. Often the distinction will become evident over time. Individual lesions of urticaria pigmentosa or a solitary mastocytoma may also be mistaken for CALM and can be distinguished by the Darier's sign (the development of urticaria following firm stroking of a mastocytoma) (see Chapter 28 ).
Treatment of isolated CALM is generally not necessary. Laser therapy, typically with ‘Q-switched’ lasers (Q-switched ruby, Q-switched alexandrite, and Q-switched Nd : YAG), has been attempted for cases considered extensive or disfiguring, but results are inconsistent and repigmentation may occur. CALM may also paradoxically darken with laser treatment and result in postinflammatory hyperpigmentation (usually transient).
Segmental pigmentation disorder (SPD or mosaic hyperpigmentation, pigmentary mosaicism) refers to a hyperpigmented or hypopigmented patch often with a sharp midline demarcation and a less distinct lateral border, commonly located on the trunk. Lesions are often noted in infancy and are described in a segmental block-like pattern resembling a ‘checkerboard’ or following the lines of Blaschko. Those lesions that less commonly involve non-truncal sites (such as the extremity or neck) usually extend onto the torso, and solitary facial involvement has been described. The majority of patients with SPD have no extracutaneous abnormalities. SPD is thought to be due to somatic mutations resulting in cutaneous mosaicism. Extensive cases can be variably associated with chromosomal anomalies. The natural history of SPD has not been reported, but persistence of the hyperpigmentation is expected. A rare biopsy done on a patient with hyperpigmented SPD showed increase melanin in the basal layer, similar to CALM.
CALM are usually the first presenting sign in NF-1, and multiple CALM can be seen in 90% of patients with NF-1 on the trunk, buttocks and legs. In fair-skinned individuals, CALM may be initially overlooked on cutaneous examination. The number of CALM increases over time during the first decade of life and lesions tend to grow in proportion to the child and may darken with sun exposure. Axillary and/or inguinal freckling is usually absent at birth but present by age 3–5 years, although it may be noted earlier. This common feature tends to be the cutaneous sign, along with multiple CALM, that establishes the diagnosis and is felt to be pathognomonic for NF-1. Multiple CALM can be seen in other variants of NF, including neurofibromatosis type 2, segmental NF-1 and hereditary spinal neurofibromatosis. A more extensive discussion of NF can be found in Chapter 29 .
Legius syndrome (neurofibromatosis type 1-like syndrome) is a recently identified autosomal dominant disorder caused by a mutation in the SPRED1 gene. The SPRED1 gene is responsible for making spred-1 protein which inhibits the Ras/MAPK pathway. Affected individuals typically have CALM and axillary freckling. However, neurofibromas, typical osseous lesions, Lisch nodules of the iris and central nervous system tumors are characteristically absent. Some patients with Legius syndrome have been described with macrocephaly, a Noonan-like facial appearance, learning disorders, developmental delay and/or attention deficit/hyperactivity disorder in childhood. Several families with Legius syndrome have been described with lipomas as well. Many individuals may meet the NIH diagnostic criteria for NF-1 but gene testing will be negative for mutations in the neurofibromin gene. It is uncertain if there is an increased risk of malignancies. However, some authors have proposed less stringent monitoring in Legius patients as compared to NF-1 patients. Screening for developmental delay and behavioral and learning problems is recommended as well as examination and counseling by a clinical geneticist familiar with Legius syndrome.
Multiple familial CALM have been described in several families without the other stigmata of NF-1, including the absence of NF-1 gene mutation. SPRED1 had not been tested in these families. This diagnosis should only be made in an older child without the other stigmata of NF-1 with a clear family history of multiple CALM without other signs of NF-1.
The cardinal features of McCune–Albright syndrome (MAS) include polyostotic fibrous dysplasia, endocrinopathies and precocious puberty as well as CALM. MAS is a rare and sporadic disorder caused by a postzygotic mutation in the gene, GNAS1 . The GNAS1 gene encodes for the alpha subunit of the guanine nucleotide-binding protein, causing loss of GTPase activity and increased stimulation of the adenylate cyclase system, resulting in proliferation and autonomous hyperfunction of hormonally responsive cells. This autosomal dominant mutation is only compatible with life when it occurs in individuals as a postzygotic somatic mutation with resulting mosaicism, which in turn results in marked phenotypic variability.
The CALM seen in MAS are often large, and segmental with favored distribution over the torso and buttocks. They may also follow the lines of Blaschko ( Fig. 24.2 ). The border of the CALM in MAS is often described as ‘jagged’ and irregular. These cutaneous findings may be present at birth or within the first few years of life and have been noted in approximately 53–98% of patients with MAS. Melanotic macules of the oral mucosa have also been reported.
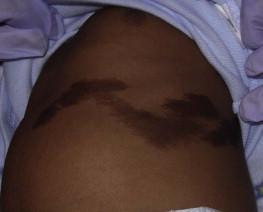
Extracutaneous findings in MAS include polyostotic fibrous dysplasia, where bone is replaced by fibrous tissue resulting in asymmetry, bony growths and pathologic fractures; this finding is rarely seen at birth and tends to manifest over time, usually within the first decade. In addition, endocrine abnormalities that may occur include precocious puberty, hyperthyroidism, Cushing syndrome, hyperparathyroidism, hyperprolactinemia, and hypersomatotropism. Neonatal cholestasis, acholic stools and jaundice have been described as presenting symptoms in neonates with MAS.
Due to the variable clinical expression and segmental nature, diagnosis of MAS may be difficult in infancy. Cutaneous findings of MAS must be differentiated from NF-1. The CALM found in NF-1 tend to be smaller, lighter in color, and more scattered in distribution. Histologic studies would not be helpful in differentiating these disorders. Radiologic studies for MAS may not reveal bony abnormalities in the neonatal period and CALM often precede the development of bony changes by 3 months to 9 years. When MAS is suspected, close observation for endocrine abnormalities is warranted. In those with known or suspected MAS, a referral to orthopedics and endocrinology is recommended. Prognosis for MAS is generally favorable and the development of malignancy is rare. Extensive osseous dysplasia early in life, however, portends a poorer prognosis.
The presence of dermal melanocytes in the skin can give rise to several clinically appearing blue-gray, green or black lesions on the skin including congenital dermal melanocytosis (Mongolian spots), Nevus of Ota, Nevus of Ito and blue nevi. The blue color is due to the Tyndall effect: the scatter of light passing through a turbid medium, such as the dermis, as it strikes melanin particles.
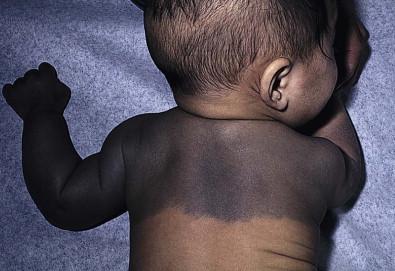
Mongolian spots are common birthmarks present in over 80% of black and Asian infants compared with only up to 10% of Caucasians infants. They present at birth, or early infancy, as blue-gray, blue-green, or blue-black macules most often noted over the sacrum and gluteal regions ( Fig. 24.3 ) and less often on extensor extremities and the posterior shoulder. Size can range from a few millimeters to over 10 cm and single or multiple lesions may be present. The borders of Mongolian spots are ill-defined and tend to fade gradually into the surrounding skin color. The coloration will often stabilize in infancy and fade during the first decade of life. Only a small number of lesions persist (3–4%) after adolescence and it has been suggested that these are likely not distinct entities but analogous to Nevus of Ota or Ito. Clinically, these persistent lesions are often blue-black homogenous or speckled patches present in a segmental distribution.
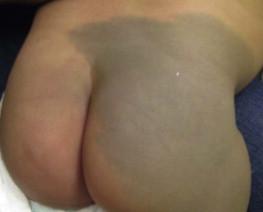
In the Mongolian spot, elongated, slender and slightly wavy dendritic cells containing melanin granules are found in the mid and deep dermis. These dendritic melanocytes are present in low numbers and scattered between collagen bundles, both of which are orientated parallel to the skin surface. There is an absence of melanophages. The reason for regression of Mongolian spots and failure of regression of other dermal melanocytic lesions such as nevus of Ito or Ota with age is currently unknown.
Mongolian spots are common, and associated abnormalities are rare. Dermal melanocytosis may be associated with certain lysosomal storage diseases, including gangliosidosis type 1 (GM1), Niemann–Pick disease, Hunter syndrome, α-mannosidosis syndrome, and Hurler syndrome. Dermal melanocytosis is generally extensive in distribution often involving the ventral and dorsal aspects of the trunk in addition to the sacrum and extremities. Mongolian spots may also co-exist within CALM in NF-1. Dermal melanocytosis is part of the criteria for a subgroup of neurocristopathies labeled ‘phakomatosis pigmentovascularis’ (PPV), in which vascular malformations of the skin, pigmented nevi and Mongolian spots occur together in the same patient (see below). Cleft lip malformation has been associated with adjacent dermal melanocytosis although the significance of the association is unknown.
Typical Mongolian spots will fade over time and treatment is usually not needed. For those lesions that persist, laser therapy has been reported to cause lightening with variable success.
Mongolian spots are clinically diagnosed by their common location, congenital or neonatal onset, and morphology, and a biopsy is most often not needed. Other blue lesions include Nevus of Ito, Nevus of Ota, and congenital blue nevus. Location and persistence over time will help in differentiating these entities. A biopsy can be helpful in verifying the dermal location of melanocytes if the diagnosis is in question.
Bluish hyperpigmentation of the skin along the ophthalmic and maxillary branches of the trigeminal nerve is known as Nevus of Ota ( Fig. 24.4 ). These patches can be brown, blue-gray or blue-black, gray or purple and have a speckled or mottled appearance with poorly-demarcated borders. Nodular areas similar to blue nevi can be present. Nevus of Ota often affects one side of the face and bilateral involvement is described in only 5–10% of patients. Distribution of Nevus of Ota most commonly involves the ophthalmic and maxillary branches with involvement of all three branches of the trigeminal nerve being the least common distribution. This condition is more commonly seen in Asians and has a female preponderance. They may be present at birth or appear within the first year of life, or later between the ages of 11–20 years; mid-childhood onset is rare. Spontaneous resolution does not occur as seen in Mongolian spots. Pathology of cutaneous lesions shows a greater concentration of elongated, dendritic dermal melanocytes scattered within the collagen bundles in the upper dermis (compared with Mongolian spots).
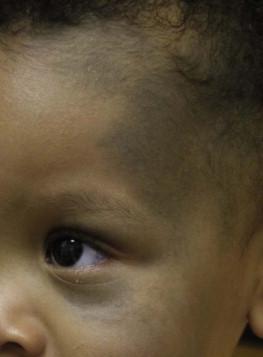
In addition to cutaneous involvement, Nevus of Ota involving the ocular sclera can be seen in two-thirds of patients. Pigmentation of the sclera, conjunctiva, cornea, iris and choroid, and less commonly, the optic nerve, retrobulbar fat, orbital periosteum and extraocular muscles can occur. Oral mucosal pigmentation and pigmentation of the tympanic membrane can occur as well.
Most Nevi of Ota pose a potential cosmetic concern but few may be associated with complications. Open-angle glaucoma occurs in approximately 10% of patients due to proliferation of melanocytes in the anterior chamber angle. The most concerning complication is the development of melanoma within pigmented areas. Malignant degeneration has been reported and may include the eye (choroid, iris, or orbit), skin, and brain. Sturge–Weber syndrome, Klippel–Trenaunay syndrome, neurofibromatosis, inflammatory vascular disease, cerebral arteriovenous malformation, multiple hemangiomas, and spinocerebellar degeneration have been described occurring in association with Nevus of Ota. Rare occurrences of ipsilateral hearing loss with Nevus of Ota have been reported. Nevus of Ota occurring with nevus flammeus is seen in variants of phakomatosis pigmentovascularis. Leptomeningeal melanosis and Nevus of Ota can occur concurrently as well.
Nevus of Ota tends to increase in size and color intensity over time. An ophthalmologic examination is recommended for those with involvement of periorbital skin, and yearly examinations for those with ocular pigmentation. Dermatologic follow-up is recommended periodically and removal of stable lesions for melanoma prophylaxis is unnecessary since the risk is quite low. Opaque make-up may be an option as a means of providing cosmetic improvement. Laser surgery with Q-switched devices (ruby, Nd : YAG or alexandrite) has been shown to reduce the coloration safely and remains the treatment of choice.
The differential diagnosis of Nevus of Ota in a neonate includes a CALM, speckled lentiginous nevus, congenital blue nevus and ochronosis.
The Nevus of Ito is an analogous lesion to Nevus of Ota with a distribution over the posterior supraclavicular and lateral cutaneous branchial nerves. These blue-gray poorly-demarcated patches may present over the shoulder, supraclavicular neck, upper arm, scapular and deltoid areas ( Fig. 24.5 ). Bilateral nevus of Ito has been reported in a patient with a speckled lentiginous nevus, and bilateral Nevus of Ota with a unilateral Nevus of Ito has been noted. Cutaneous melanoma and malignant cellular blue nevus have been rarely reported in Nevus of Ito and extracutaneous findings are not seen.
Blue nevi are dermal melanocytomas that represent benign proliferations of pigment producing dendritic dermal melanocytes ( Fig. 24.6 ). Dermoscopy of common blue nevi reveals uniform blue coloration. Blue nevi are rarely seen at birth, but congenital forms have been described. Most are acquired, with onset in childhood or adolescence, with predilection for the scalp and face, dorsal aspects of the distal extremities and sacral area. Three types of blue nevi have been described: (1) cellular blue nevus, (2) common blue nevus, and (3) the combined nevus.
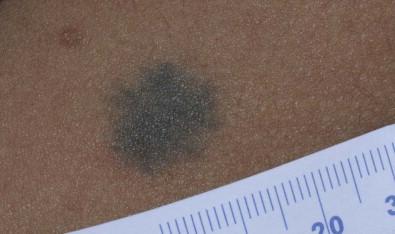
Cellular blue nevi seen at birth are rare and have a predilection for the scalp, sacrococcygeal area and buttocks. They present with blue-gray or blue-black nodules or plaques with a smooth or irregular surface texture. Although most congenital cellular blue nevi are small in size (1–3 cm), larger and ‘giant’ lesions can be seen, most commonly on the scalp. Melanoma has been described particularly in larger lesions on the scalp. Multiple satellite blue nevi in association with a large congenital cellular blue nevus have also been reported. Other related variants of congenital blue nevi include the pauci-melanotic cellular blue nevi, epithelioid blue nevi (also known as pigmented epithelioid melanocytoma), congenital plaque-type blue nevi and neurocristic hamartomas. Multiple blue nevi may be seen in Carney complex (see below) or without associated abnormalities. A rare, congenital, plaque-like type of blue nevus, showing a circumscribed area with numerous macules and papules has been described. Blue nevi may arise within speckled lentiginous nevi as well as within other dermal melanocytoses such as Nevus of Ota.
Blue-black nodules, papules and plaques seen at birth or infancy are suggestive of blue nevi. A biopsy may be needed to confirm the diagnosis and differentiate lesions from melanoma. Common blue nevi will show pigmented dendritic melanocytes singly or in small aggregations in the reticular dermis, surrounded by thickened collagen. Histopathology of cellular blue nevi, in contrast, shows cellular islands of large spindle-shaped or epithelioid cells with little or no melanin, in addition to pigmented dermal dendritic melanocytes. Lesions may penetrate deep into the subcutaneous fat.
Infants with blue nevi that are not easy to monitor due to location and color, as well as those undergoing rapid growth or symptoms (ulceration, bleeding), should be considered for prophylactic surgical excision. Clinically stable, small common blue nevi without unusual or atypical clinical or dermoscopic features may be monitored. Although malignant blue nevi are rare, new atypical features or symptoms should be evaluated histologically via biopsy or conservative excision.
Mosaicism is the presence of two or more genetically distinct cell lines in a single individual, which may arise from a variety of genetic defects from separate single mutations, groups of mutations or chromosomal differences. Cutaneous mosaicism is associated with diverse pigmentary patterns along the lines of Blaschko or in segmental or block-like patterns, such as the pigmentary anomalies found with hypomelanosis of Ito and McCune–Albright syndrome. Many hyperpigmented conditions present in segmental or Blaschko-linear patterns and may be isolated skin findings or coincide with systemic manifestations ( Box 24.1 ).
Early (macular) epidermal nevus
Incontinentia pigmenti
Linear and whorled nevoid hypermelanosis
Human chimerism
Chromosomal mosaicism
Naegeli–Franceschetti–Jadassohn syndrome
X-linked cutaneous amyloidosis (female carriers)
Focal dermal hyperplasia
Conradi–Hünermann disease, X-linked variant
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here