Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Human growth is an astonishing process. Its beginnings are intertwined with the enormously intricate mechanisms that transform a single cell into a complex embryo. Once formed, the human fetus and then child continue to grow over the course of approximately 15 years, reaching a body mass more than 10,000,000 times greater than that of the zygote from which the child originated. The early growth rate is enormous. The fetus grows in length at more than 100 cm per year, but, by birth, the linear growth rate has slowed to 50 cm per year and by midchildhood to 5 cm per year. This decline in the growth rate is briefly interrupted by the pubertal growth spurt but then resumes, causing body growth to gradually grind to a halt as the child approaches adulthood. During the period of body enlargement, growth of the different organs, such as the kidney, heart, lung, liver, and bones, occurs synchronously, orchestrated to maintain body proportions.
In the majority of children, the largely mysterious mechanisms governing body growth proceed like clockwork, yielding an adult height most often between 1.5 and 2 m. However, in some children, body growth is either greater than or less than the normal range, often prompting the concerned family to seek medical evaluation.
The healthcare provider is then called upon to answer two questions. First, why is the child’s growth outside the normal range and second what, if anything, should be done about it. The first question, “why,” is important for several reasons. Many children are short or tall because they have inherited from their parents a large set of normal polymorphisms that place the child in one tail of the normal height distribution or the other. Such polygenic short or tall stature is generally benign. However, some children are short or tall because of a single gene abnormality that may have important health implications, such as a propensity to cardiac malformations or to malignancies. Other children are short because of a subtle systemic acquired disease, such as inflammatory bowel disease (IBD), autoimmune thyroiditis, or a central nervous system (CNS) tumor causing pituitary dysfunction. Thus the healthcare provider who evaluates children’s growth faces the challenge of sorting out the few children with important underlying pathology from the many children with a benign condition.
The second question, “what to do about it,” can also be challenging. If the diagnostic evaluation uncovers an underlying, treatable disorder, such as celiac disease, the course is clear. But often, no simple treatable cause is found. Then, the provider and the family must try to assess whether the short or tall stature is sufficiently abnormal and sufficiently distressing to the child, or likely to be distressing in the future, so that the possible benefits of medical therapy outweigh the effort, costs, discomforts, and potential risks of intervening.
Addressing these two central questions requires a deep understanding of normal childhood growth and the many disorders that affect it, the determination to look diligently for underlying causes, as well as the willingness and ability to listen compassionately to the concerns of the child and family. In this chapter, we have tried to present the healthcare provider with a broad overview of the knowledge necessary for this endeavor.
In children, many tissues and organs grow simultaneously. However, most children who present to the pediatric endocrinologist for evaluation of growth do so because the child’s height is abnormal. The height is primarily determined by the length of the long bones in the lower extremities and the height of the vertebral bodies. These bone lengths in turn are determined by the action of the skeletal growth plates, thin layers of cartilage located near the ends of the long bones and vertebrae.
Within the growth plates, the chondrocytes are arranged in columns parallel to the long axis of the bone ( Fig. 11.1A ). In the upper portion (closer to the ends of the bone) of the columns, the chondrocytes proliferate ( Fig. 11.1B ). In the lower portion of the columns, the chondrocytes stop dividing and instead physically enlarge ( Fig. 11.1B ). These two processes, cell proliferation and cell hypertrophy, lead to chondrogenesis, the production of cartilage. In isolation, this chondrogenesis would cause progressive thickening of the cartilaginous growth plate. However, simultaneously, at the bottom of the growth plate, the newly formed cartilage is remodeled into bone. The net result is that new bone tissue is progressively created at the bottom of the growth plates, the overall bone grows longer, and the child grows taller.
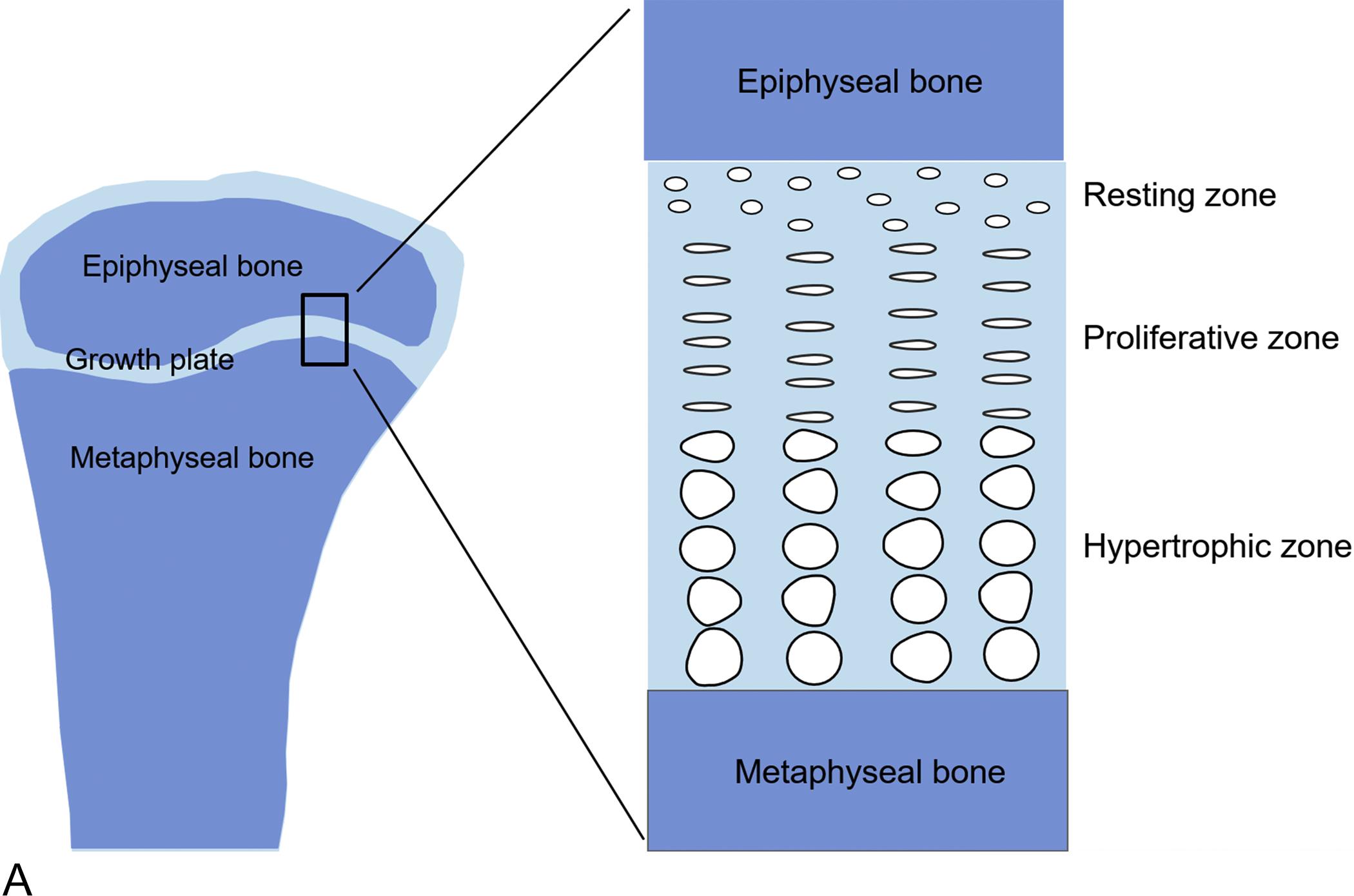
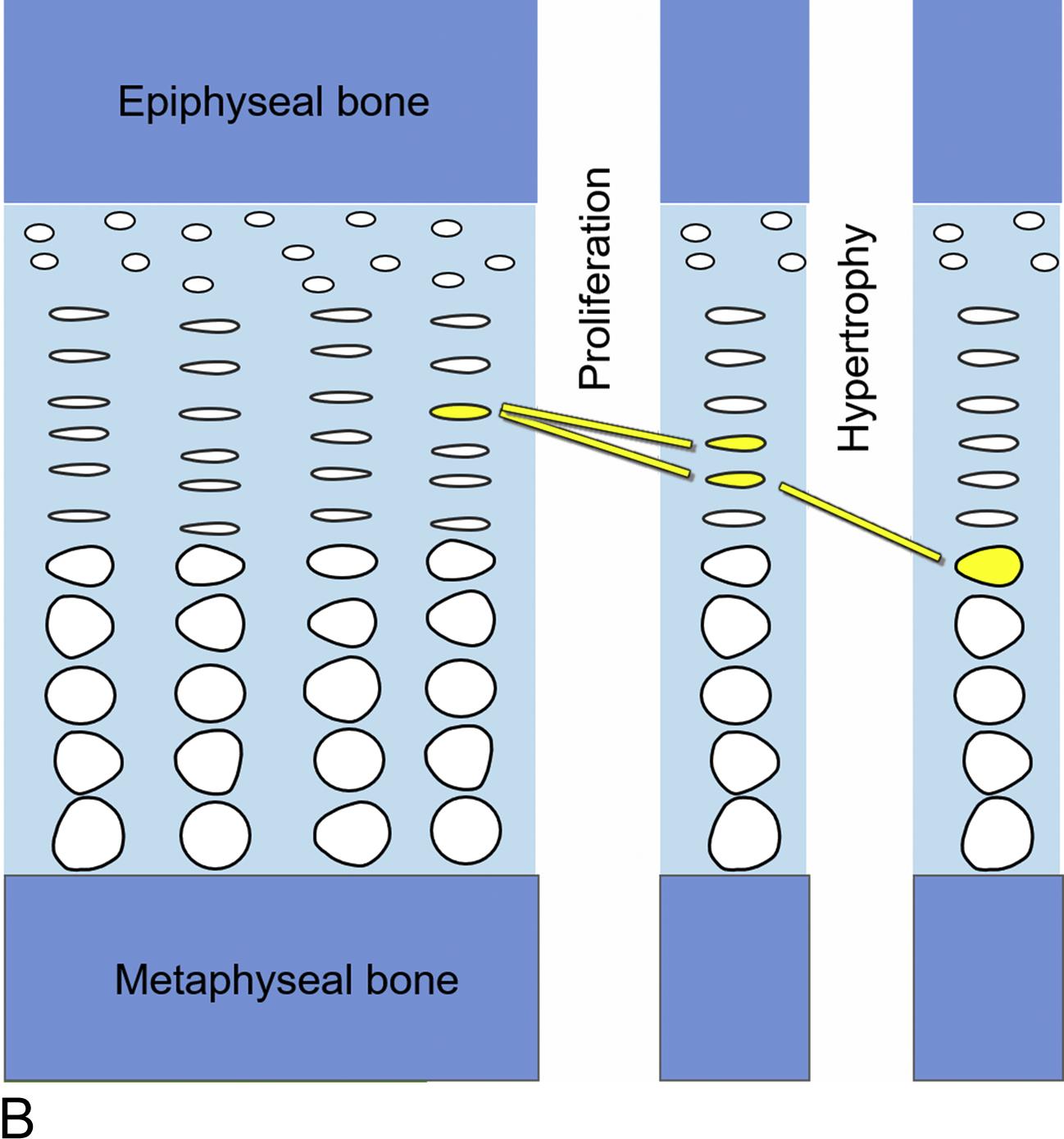
Thus linear growth (height gain) in children results from growth plate chondrogenesis. Therefore short stature is caused by decreased growth plate chondrogenesis and tall stature is caused by increased growth plate chondrogenesis.
Because linear growth results from growth plate chondrogenesis, the regulation of linear growth results from the regulation of growth plate chondrocytes. Factors that stimulate growth plate chondrocyte proliferation and hypertrophy stimulate linear growth, whereas factors that inhibit growth plate chondrocyte proliferation and hypertrophy inhibit linear growth. This regulation of growth plate chondrocyte function occurs at multiple different levels; endocrine signals, inflammatory cytokines, nutritional intake, paracrine/autocrine signals, extracellular matrix effects, and intracellular systems can all modulate chondrocyte proliferation and hypertrophy and therefore affect the rate of linear growth. Consequently, abnormalities at any of these levels can cause short or tall stature.
The pituitary gland lies in the sella turcica , the hypophyseal fossa of the sphenoid bone, which is located in the center of the cranial base. The concept of the pituitary as a “master gland” controlling the endocrine activities of the body has become outdated and has been replaced by an appreciation of the importance of the brain, particularly of the hypothalamus, in regulating hormonal production and secretion. Nevertheless, the pituitary gland remains central to our understanding of the regulation of growth, metabolism and homeostasis, response to stress, lactation, and reproduction.
Embryologically, the pituitary gland is formed from two distinct sources, namely Rathke’s pouch, a diverticulum of the primitive oral cavity (stomodeal ectoderm), which gives rise to the adenohypophysis, and the neural ectoderm of the floor of the forebrain, which gives rise to the neurohypophysis, the posterior lobe. The adenohypophysis normally constitutes 80% of the weight of the pituitary and consists of the pars distalis (also known as the pars anterior or anterior lobe ), the pars intermedia (also known as the intermediate lobe ), and the pars tuberalis (also known as the pars infundibularis or pars proximalis ).
Much of our knowledge of normal hypothalamopituitary development is derived from animal, particularly rodent, models. In the mouse, a thickening of the ectoderm in the midline of the anterior neural ridge, forming the hypophyseal placode, heralds the onset of pituitary development at 7.5 dpc (days postcoitum). The formation of a rudimentary Rathke’s pouch follows at 9 dpc, with formation of a definitive pouch by 12 dpc and subsequently, the anterior pituitary consisting of five different cell types secreting six different hormones ( Fig. 11.2 ).
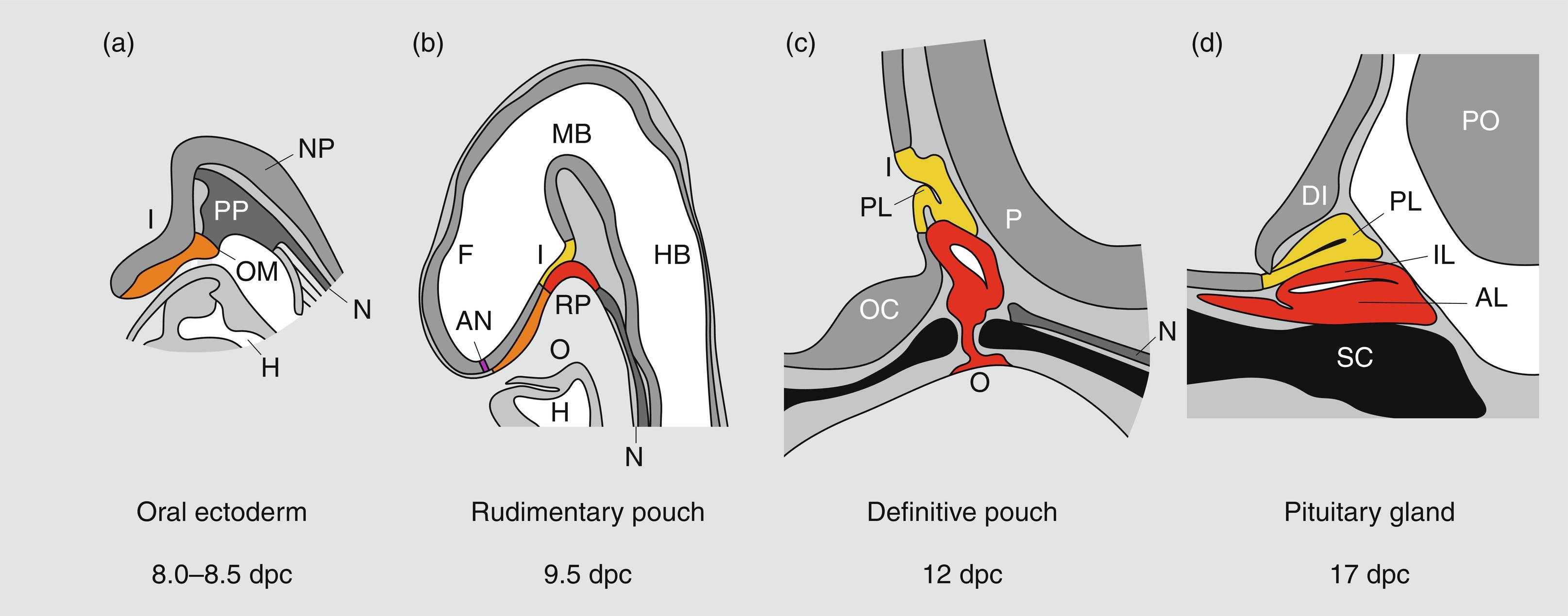
In humans, the pars distalis is the largest portion of the adenohypophysis and houses the great majority of hormone-producing cells. In contrast with the mouse, the pars intermedia is rudimentary and consists of several cystic cavities lined by a single layer of cuboidal epithelium as it largely disappears during embryogenesis. The pars distalis and intermedia are separated by a cleft, a vestigial structure of Rathke’s pouch from which it develops. This structure may often develop as a cyst (Rathke’s cleft cyst). The pars tuberalis represents an upward extension of the pars distalis onto the pituitary stalk and may contain a limited number of gonadotropin-producing cells. The posterior pituitary (neurohypophysis) consists of the infundibular stem or hypophyseal stalk, the median eminence of the tuber cinereum, and the infundibular process (posterior lobe, neural lobe). The posterior pituitary contains the terminal axonal projections of magnocellular neurons from the paraventricular and supraoptic nuclei of the hypothalamus; these produce oxytocin, which is required during lactation and parturition, and vasopressin, which is required for osmotic regulation. It has no known function in the regulation of growth and will not be discussed further in this chapter.
Rathke’s pouch, the origin of the adenohypophysis, can be identified in the 3-mm embryo during the third week of pregnancy in humans. Rathke’s pouch then begins to develop, resulting in a complete pouch disconnected from the oral ectoderm by the end of the sixth gestational week. Growth hormone (GH)-producing cells can be identified by 9 weeks of gestation. It is at about this time that the vascular connections between the anterior lobe of the pituitary and the hypothalamus develop, although it has been demonstrated that hormonal production by the pituitary can occur in the absence of connections with the hypothalamus. Somatotropes are thus frequently demonstrable in the pituitary of an anencephalic newborn. Nevertheless, it appears likely that the initiation of development of the anterior pituitary is dependent on responsiveness of the oral ectoderm to inducing factors from the ventral diencephalon. Infrequently, the craniopharyngeal canal (marking the embryonic migration of Rathke’s pouch) remains patent and may contain small nests of adenohypophyseal cells—giving rise to a pharyngeal hypophysis that may be capable of hormone synthesis.
Maintained apposition and interaction between the oral ectoderm and neuroectoderm is critical for normal anterior pituitary development. Experimental manipulation of embryos from several species, as well as Rathke’s pouch explant experiments in rodents, have shown that signals from the diencephalon are essential not only for the induction and maintenance of Rathke’s pouch, but also for the regionalization within the pouch that allows the emergence of the different endocrine cell types. During gestation, proliferating progenitor cells are enriched around the pouch lumen, and they appear to delaminate as they exit the cell cycle and differentiate. During late mouse gestation and the postnatal period, anterior lobe progenitors reenter the cell cycle and expand the populations of specialized, hormone-producing cells. At birth, all cell types are present, and their localization appears stratified, based on cell type. Current models of cell specification in the anterior lobe suggest that opposing gradients of fibroblast growth factor (FGF) and bone morphogenic protein (BMP) signaling pattern the progenitor cells within Rathke’s pouch before they move on to the anterior lobe where they differentiate. Several studies have revealed that normal pituitary development is dependent upon a complex cascade of transcription factors and signaling molecules that are expressed in a spatiotemporal manner.
Signaling molecules implicated in pituitary development are either intrinsic, emanating from the oral ectoderm, such as sonic hedgehog (Shh), or extrinsic from the neuroectoderm, such as Nkx2.1, FGFs (e.g., FGF 8), and BMPs (e.g., BMP4) ( Fig. 11.3 ). These molecules may activate or repress transcription factors, such as Hesx1, Lhx3, and Lhx4. They may also act as morphogens creating the appropriate environment for cell differentiation, thus playing a critical role in cell fate. Such signaling molecules include members of the Shh family, FGFs, transforming growth factors-β (TGFs)/BMPs, Wingless/Wnts, and molecules in the Notch pathway to mention a few.
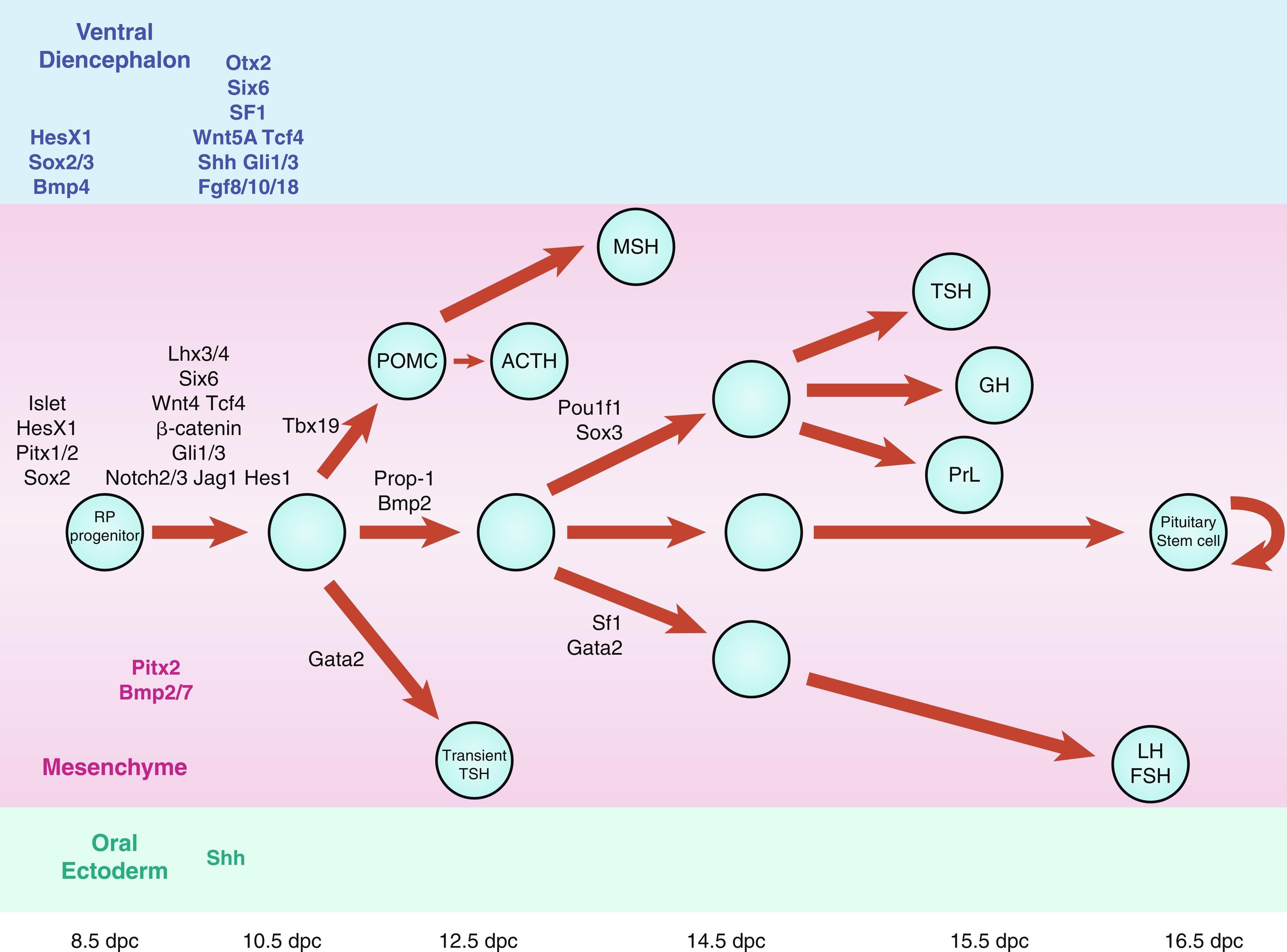
Recently, Davis and colleagues have challenged the current dogma of pituitary cell specification, showing evidence that, in mice, the pattern of cell specification that results in the rostral location of gonadotropes, the caudal location for somatotropes and a more intermediate location for corticotropes and thyrotropes, does not appear to be the result of an ordered cell cycle exit, as previously suggested. All anterior lobe cell types appear to begin the differentiation process concurrently (E11.5-E14.5), rather than in a temporally discrete manner.
To date, not many pituitary phenotypes have been reported in association with mutations in these signaling molecules. Importantly, the Wnt signaling pathway has recently been implicated in pituitary tumorigenesis. For example, there is clear evidence that the Wnt/β catenin pathway is involved in the pathogenesis of craniopharyngioma, a rare tumor in the hypothalamopituitary region.
Multiple pituitary-specific transcription factors are involved in the determination of pituitary cell lineages and cell-specific expression of anterior pituitary hormones (see Fig. 11.3 ). Several homeodomain transcription factors have been shown to be involved in human anterior pituitary development and differentiation. Defects in several of these have now been associated with various combinations of pituitary hormone deficiencies ( Table 11.1 ). Because additional gene defects have been implicated in abnormal murine hypothalamopituitary development, it seems likely that the number of known human genetic defects will expand.
| Gene | Pituitary Deficiencies | MRI Phenotype | Inheritance | Other Phenotypic Features |
|---|---|---|---|---|
| POU1F1 | GH, TSH, prolactin | Small or normally sized anterior pituitary | AR and AD | |
| PROP1 | GH, TSH, LH, FSH, prolactin, evolving ACTH deficiencies | Small, normal or enlarged anterior pituitary – may evolve over time | AR | |
| HESX1 | Isolated GH deficiency through to panhypopituitarism with TSH, LH, FSH, ACTH, and prolactin deficiencies | Optic nerve hypoplasia, absence of the septum pellucidium, ectopic posterior pituitary, anterior pituitary hypoplasia | AR and AD | Developmental delay, visual abnormalities, SOD |
| LHX3 | GH, TSH, LH, FSH, prolactin with late ACTH deficiencies | Small, normal, or enlarged anterior pituitary | AR | Short neck with limited rotation |
| LHX4 | GH, TSH, ACTH, and gonadotrophin deficiencies | Small anterior pituitary, ectopic posterior pituitary, cerebellar abnormalities, corpus callosum hypoplasia | AD (variable penetrance) | |
| SOX3 | GH, TSH, LH, FSH, ACTH deficiencies. Most commonly isolated GH deficiency | Anterior pituitary and infundibular hypoplasia, ectopic posterior pituitary, corpus callosum abnormalities including cysts; persistent craniopharyngeal canal | X-linked recessive | Learning difficulties |
| SOX2 | LH, FSH variable GH deficiency | Anterior pituitary hypoplasia, optic nerve hypoplasia, SOD, hypothalamic hamartoma | AD (usually de novo) | Microphthalmia, anophthalmia, micropenis, sensorineural deafness, gastrointestinal tract defects with esophageal atresia |
| GLI2 | GH, TSH, and ACTH with variable gonadotrophin deficiencies | Anterior pituitary hypoplasia | AD | Holoprosencephaly, cleft lip and palate, anophthalmia, postaxial polydactyly, imperforate anus, laryngeal cleft, renal agenesis |
| GLI3 | GH, TSH, LH, FSH, ACTH | Anterior pituitary hypoplasia | AD | Pallister-Hall syndrome Postaxial polydactyly, hamartoma |
| OTX2 | GH, TSH, LH, FSH, and ACTH deficiencies | Normal or small AP, pituitary stalk agenesis, ectopic posterior pituitary, Chiari I malformation | AD (usually de novo) | Microcephaly, bilateral anophthalmia, developmental delay, cleft palate |
| FGFR1 | GH, TSH, LH, FSH, and ACTH deficiencies | Normal or small anterior pituitary, corpus callosum agenesis | AD | ASD and VSD, brachydactyly, brachycephaly, preauricular skin tags, ocular abnormalities, seizures |
| FGF8 | GH, TSH, ACTH, gonadotrophin, and ADH deficiencies | Absent corpus callosum, optic nerve hypoplasia, holoprosencephaly | AD or AR | |
| PROKR2 | GH, TSH, ACTH and gonadotrophin deficiencies | Hypoplastic corpus callosum, normal or small anterior pituitary | AD | |
| ARNT2 | GH, TSH, ACTH, LH, FSH, ADH deficiencies | Absent PP, thin stalk, thin corpus callosum, delayed myelination | AR | Hip dysplasia, hydronephrosis, vesicoureteric reflux, neuropathic bladder, microcephaly, prominent forehead, deep-set eyes, retrognathia |
| TCFL7 | GH | Absent posterior pituitary, anterior pituitary hypoplasia, optic nerve hypoplasia, partial agenesis of corpus callosum, thin anterior commissure | AD | |
| IGSF1 RNPC3 KCNQ1 |
GH (transient/partial), TSH, prolactin GH deficiency GH, ACTH, TSH, and gonadotrophin deficiencies |
Normal in the majority of cases. Frontoparietal hygroma, hypoplasia of the corpus callosum, and small stalk lesion reported Small anterior pituitary with normal stalk and eutopic posterior pituitary Normal/small AP |
X-linked recessive AR AR |
Macroorchidism, delay in puberty Maternally inherited gingival fibromatosis |
In the human adult, the pituitary has a mean weight of 600 mg, with a range of 400 to 900 mg. Pituitary weight is slightly greater in women than in men, and typically increases during puberty and pregnancy. In the newborn, pituitary weight averages about 100 mg. The pituitary normally resides in the sella turcica immediately above and partially surrounded by the sphenoid bone. The volume of the sella turcica provides a good measure of pituitary size, which may be reduced in the child with pituitary hypoplasia. It is important, however, to recognize that considerable variation in pituitary size occurs normally. The pituitary is covered superiorly by the diaphragma sellae, and the optic chiasm is directly above the diaphragma. The anatomic proximity between the optic chiasm and the pituitary is important because hypoplasia of the optic chiasm may occur together with hypothalamic/pituitary dysfunction, as in the condition of septooptic dysplasia (SOD), and because pituitary tumors may in turn impact on the optic chiasm, leading to visual impairment. The patient with congenital blindness or nystagmus should be initially evaluated and then subsequently monitored carefully for hypopituitarism, as this can evolve. In addition, suprasellar growth of a pituitary tumor may initially manifest with visual complaints or evidence of progressive impairment of peripheral vision, particularly bitemporal hemianopia.
The existence of a portal circulatory system within the pituitary is critical to normal pituitary function. The blood supply of the pituitary derives from the superior and inferior hypophyseal arteries, branches of the internal carotid. The anterior and posterior branches of the superior hypophyseal artery may terminate within the infundibulum and the proximal portion of the pituitary stalk. Hypothalamic peptides, produced in neurons that terminate in the infundibulum, enter the primary plexus of the hypophyseal portal circulation and are transported by means of the hypophyseal portal veins to the capillaries of the anterior pituitary. This portal system thus provides a means of communication between the neurons of the hypothalamus and the hormone-producing cells of the anterior pituitary. The blood supply of the neurohypophysis is separate, deriving from the inferior hypophyseal artery. Regulation of the posterior lobe of the pituitary does not involve the hypophyseal portal circulation but, rather, is mediated through direct neural connections.
The definitive Rathke’s pouch comprises proliferative progenitors that will gradually relocate ventrally, away from the lumen as they differentiate. A proliferative zone containing progenitors is maintained in the embryo in the perilumenal area and was recently found to persist in the adult. The exact nature of progenitor cells in the pituitary gland, however, remains unknown. Members of the Sox family of transcription factors are likely involved in the earliest steps of pituitary stem cell proliferation and the earliest transitions to differentiation. The transcription factor Prophet of Pit1 (PROP1) and the NOTCH signaling pathway may then regulate the transition to differentiation. It has been proposed that the niche of progenitors may be the marginal zone around the lumen of Rathke's pouch, between the anterior and intermediate lobes of mouse pituitary, because cells in this region are able to give birth to all five pituitary hormone cell lineages (see Fig. 11.3 ).
Stem cells have been shown to play a role in tumorigenesis in some tissues, and their role in pituitary hyperplasia, pituitary adenomas, and tumors is an important area for future investigation. The ability to cultivate and grow pituitary stem cells in a predifferentiation state might also be helpful for the long-term treatment of pituitary deficiencies. Indeed, a seminal study resulted in the efficient self-formation of a three-dimensional anterior pituitary in an aggregate culture of mouse embryonic stem (ES) cells. Various endocrine cells were generated, and these cells were able to respond to trophic hormones. When these ES-derived cell aggregates were implanted under the kidney capsule in hypophysectomized mice, corticosterone was produced. These studies may therefore reflect the first step toward stem cell treatment.
Terminally differentiated secreting cells are not distributed randomly in a patchwork-like fashion throughout the pituitary gland. Instead, these cells appear to organize themselves in same-cell–type networks. The connectivity between the cells of this network is important to deliver coordinated secretory pulses of hormones to their target tissues and to facilitate the coordinated physiological response to stimuli.
Human GH is produced from somatotrope cells within the anterior pituitary as a single-chain, nonglycosylated, 191-amino-acid, 22-kDa protein that comprises a core of four helices in a parallel/antiparallel orientation with two disulfide bonds between cysteines 53–165 and 182–189. The 217-amino-acid GH precursor is cleaved to remove the signal peptide before secretion.
GH is homologous with several other proteins produced by the pituitary or placenta, including prolactin, chorionic somatomammotropin (CS, placental lactogen), and a 22-kDa GH variant hGH-V, secreted only by the placenta. The latter differs from pituitary GH by 13 amino acids. The genes for these proteins have probably descended from a common ancestral gene, even though the genes are now located on different chromosomes.
GH1 is located on the long arm of chromosome 17 (17q22-24) within a cluster of five homologous genes encompassing a distance of about 65 kb ( CSHP [CS pseudogene], CSH-1 [CS gene], GH-2 and CSH-2 ). Expression of GH1 is regulated by the highly polymorphic proximal promoter and a locus control region (LCR) 15–32 kb upstream of the gene that confers the pituitary-specific and high-level expression of GH. Normally, the vast majority of GH (75%) produced by the pituitary is of the mature 22-kDa form. Alternative splicing results in deletion of amino acids 32 through 46, yielding a 20-kDa form that normally accounts for less than 10% of pituitary GH. The remainder of pituitary GH includes desamidated and N-acetylated forms, as well as various GH oligomers. A 17.5-kDa variant that results from complete skipping of exon 3 and lacks amino acids 32–71 is much less abundant (1%–5%).
GH can be identified in fetal serum by the end of the first trimester. The pulsatile pattern characteristic of GH secretion largely reflects the interplay of multiple regulators, including two hypothalamic regulatory peptides: GH-releasing hormone (GHRH) and somatostatin (somatotropin release–inhibiting factor [SRIF]). GHRHR encodes a G-protein–coupled receptor comprising seven transmembrane domains. The expression of GHRHR is upregulated by pituitary-specific transcription factor 1 ( POU1F1 ) and is required for the proliferation of somatotrophs.
Regulation of GH production by GHRH is largely transcriptionally mediated and is dependent on stimulation of adenylate cyclase and increases in intracellular cyclic adenosine monophosphate (cAMP) concentrations. Solid tumors secreting GHRH are a rare cause of GH excess. GHRH is used diagnostically, especially for the identification of adult GH deficiency, when it is frequently used in combination with arginine. However, it is not conventionally used for therapeutic purposes in patients with GH deficiency.
The actions of the 14-amino-acid peptide somatostatin appear to regulate the timing and amplitude of pulsatile GH secretion, rather than GH synthesis. The binding of somatostatin to its specific receptor results in an inhibition of adenylate cyclase activity and a reduction in intracellular calcium concentrations. The pulsatile secretion of GH observed in vivo is believed to result from a simultaneous reduction in hypothalamic somatostatin release and increase in GHRH activity. Conversely, a trough of GH secretion occurs when somatostatin release is increased in the presence of diminished GHRH activity. The net effects of these two hypothalamic hormones is modulation of GH secretion, as well as the timing and amplitude of peaks, resulting in pulsatile GH secretion. Multiple neurotransmitters and neuropeptides are involved in regulating the release of these hypothalamic factors, including serotonin, histamine, norepinephrine, dopamine, acetylcholine, gamma-aminobutyric acid (GABA), thyroid-releasing hormone, vasoactive intestinal peptide, gastrin, neurotensin, substance P, calcitonin, neuropeptide Y, vasopressin, corticotrophin-releasing hormone, and galanin. These factors are implicated in the alterations of GH secretion observed in a wide variety of physiological states (such as stress, sleep, hemorrhage, fasting, hypoglycemia, and exercise) and form the basis for a number of GH-stimulatory tests used in the evaluation of GH secretory capacity/reserve. GH secretion is also impacted by a variety of nonpeptide hormones, including androgens, estrogens, thyroxine, and glucocorticoids. The precise mechanisms by which these hormones regulate GH secretion are complex, potentially involving actions at the hypothalamic and pituitary levels. Practically speaking, hypothyroidism and glucocorticoid excess may each blunt spontaneous and provocative GH secretion (and therefore should be corrected before GH testing). Sex steroids, at the onset of puberty or administered pharmacologically, appear to be responsible for the rise in GH secretion characteristic of puberty. Synthetic hexapeptides capable of stimulating GH secretion have been developed and termed GH-releasing peptides (GHRPs). These peptides, later recognized as analogues of the gastric hormone ghrelin, are capable of directly stimulating GH release and enhancing the GH response to GHRH. These agents have the potential advantage of oral administration, and, in the patient with an intact pituitary, may be capable of greatly enhancing GH secretion. When these agents were administered chronically to elderly patients and to some GH-deficient children, the amplitudes of GH pulses were significantly increased. Ghrelin-mimetic ligands were used to characterize a common receptor termed the GH secretagogue receptor (GHS-R) for the GH-releasing substances. The GHS-R is a G-protein–coupled receptor, which is distinct from the GHRH receptor. The receptor is strongly expressed in the hypothalamus, but specific binding sites for GHRPs have also been identified in other regions of the CNS and peripheral endocrine and nonendocrine tissues in both humans and other organisms. Ghrelin is a 28-amino-acid peptide that has been identified as the endogenous ligand for GHS-R. It is expressed predominantly in the stomach, but smaller amounts are also produced within the bowel, pancreas, kidney, the immune system, placenta, pituitary, testis, ovary, and hypothalamus. Ghrelin is a unique gene product that requires octanoylation for normal function. Intravenous, intracerebroventricular, and intraperitoneal administration of ghrelin in animal models stimulates food intake and obesity and raises plasma GH concentrations, and, to a lesser extent, prolactin (PRL) and adrenocorticotropic hormone (ACTH) concentrations. In addition, it influences endocrine pancreatic function and glucose metabolism, gonadal function, and behavior. It also controls gastric motility and acid secretion and has cardiovascular and antiproliferative effects. Both ghrelin and GHRPs release GH synergistically with GHRH but the efficacy of these compounds as growth-promoting agents is poor. Variants in the ghrelin receptor have been identified as a possible cause of idiopathic short stature (ISS) and GH deficiency. However, note that mouse models with targeted deletion of the receptor ( ghsr -/- ) have a near normal phenotype. A second peptide encoded by the same gene as ghrelin has been identified and termed obestatin . This gene appears to regulate weight but not GH secretion.
In addition to the complex regulatory processes described previously, the synthesis and secretion of GH are also regulated by feedback by the insulin-like growth factor (IGF) polypeptides. IGF receptors have been identified in the pituitary. Inhibition of GH secretion by IGF-1 has been demonstrated in multiple systems. In addition, inhibition of spontaneous GH secretion has been demonstrated in humans treated with subcutaneous injections of recombinant IGF-1. GH also regulates its own secretion by acting directly on hypothalamic GH receptors (GHRs), via a short loop negative feedback.
During puberty, sex steroids increase GH pulse amplitude contributing to the very high serum concentrations of IGF-1 characteristic of puberty. GH secretion begins to decline by late adolescence and continues to fall throughout adult life. Indeed, puberty may be considered with some justification a period of “acromegaly,” whereas aging (with its characteristic decrease in GH secretion) has been termed the somatopause.
In addition to aging, a wide variety of physiological conditions affect GH secretion. These include stage of sleep, nutritional status, acute fasting, exercise, stress, and sex steroids. Ho and associates have reported that neither age nor sex influenced the integrated serum concentrations of GH when the effects of estradiol were removed from analysis. The effects of testosterone on serum IGF-1 concentrations may be at least in part independent of GH because individuals with mutations of the GHR still experience a rise in serum IGF-1 during puberty.
The pulsatile nature of GH secretion is readily demonstrable by frequent serum sampling, especially when coupled with sensitive assays for GH. Such assays demonstrate that, under normal conditions, serum GH concentrations are less than 0.2 ng/mL between bursts of GH secretion. It is consequently impractical to assess GH secretion by a single, random serum sample. Maximal GH secretion occurs during the night, especially at the onset of the first slow-wave sleep (stages III and IV). Rapid-eye-movement (REM) sleep is, on the other hand, associated with low GH secretion.
Normal young men generally experience, on average, 12 GH secretory bursts per 24 hours. Obesity is characterized by decreased GH secretion, reflected by a decreased number of GH secretory bursts. Fasting increases the number and amplitude of GH secretory bursts, presumably reflecting decreased somatostatin secretion. The impact of the pulsatile secretory nature of GH secretion on its biological actions remains uncertain.
The GHR comprises an extracellular, hormone-binding domain, followed by a single membrane-spanning domain, and a cytoplasmic domain. Two genomic GHR isoforms that exist only in humans have arisen from ancestral homologous recombination. They differ in the retention or deletion of exon 3. Exon 3 of the GHR has been shown to be deleted in a number of normal individuals. This delta-3 GHR polymorphism has been shown by some, but not all, investigators to affect responsiveness to GH and to be associated with birth size and postnatal growth. There are also short isoforms of the GHR because of alternative splicing, resulting in a truncated receptor with loss of a large portion of the cytoplasmic domain.
The GHR is a member of the class 1 hematopoietic cytokine family and is highly homologous with the prolactin receptor and shares sequence homology with many of the receptors for interleukins (ILs), as well as receptors for erythropoietin (EPO), leptin, granulocyte-macrophage colony-stimulating factor, and interferon.
Examination of the crystal structure of the GH-GHR complex revealed that the complex consists of one molecule of GH bound to two GHR molecules, indicating a GH-induced receptor dimerization—which is necessary for GH action. Interestingly, a genetically engineered fusion complex of GH and the GHR has been shown to have a significantly improved efficacy and a dramatically longer half-life compared with GH alone when tested in rodent models.
The GHR, like its family group member EPO-R, is preformed as a dimer and is transported in a nonligand bound state to the cell surface. GH then binds in a sequential manner to the GHR dimer where the first GHR binds to the stronger site 1 of the GH molecule followed by the second GHR binding to the weaker site 2. Binding of GH results in a conformational change whereby rotation of the GHRs results in repositioning of the intracellular domains and of Box11-1–associated Janus Kinase 2 (JAK2), a major GHR-associated tyrosine kinase. As a result, JAK2 is autophosphorylated and activated, which in turn leads to cross-phosphorylation of distal tyrosine residues of GHR. This enables SH2 ( Src homology 2) domain molecules to dock to these sites. The GHR itself appears to have no intrinsic kinase activity. It is likely that colocalization of two JAK2 molecules by the dimerized GHR results in transphosphorylation of one JAK2 by the other, leading to JAK2 activation. Signal transducer and activator of transcription (Stat)5a and 5b molecules contain SH2 domains and bind to these phosphorylated tyrosine sites, and then they in turn become phosphorylated. Phosphorylated Stat5 molecules (homo- and hetero-) dimerize and translocate to the nucleus where they bind deoxyribonucleic acid (DNA), as dimers or as tetramers, and activate target genes. Importantly, the dimerization of the GHR by GH is exploited by the use of Pegvisomant, a genetically modified form of GH, which binds to one receptor only and therefore cannot dimerize the receptor. It therefore acts as a GH antagonist and can be used to treat pituitary gigantism and acromegaly.
Both Stat5a and Stat5b can be activated by GH, and they have both overlapping and distinct functions. Gene inactivation mouse models have shown that Stat5b is of greater importance for stimulation of growth than Stat5a.
Activation of JAK-STAT signaling occurs rapidly, within minutes after GH stimulation, but is transient because of the tight control of the termination of signaling. This negative regulation of signaling occurs at several levels: GHR internalization, suppressors of cytokine signaling (SOCS), protein tyrosine phosphatases (PTPs), and protein inhibitors of activated Stats (PIAS).
In humans, the circulating GH-binding protein (GHBP) appears to derive from proteolytic cleavage of the extracellular domain of the GHR. GHBP has an identical affinity for GH as GHR. It binds to GH with high specificity and affinity, but with relatively low capacity. It increases GH half-life in the circulation and may serve a function in the transportation of GH to target tissues and subsequent binding to the receptor. GHBP is, like GHR, present in many tissues, but GHBP in the circulation is mostly derived from the liver. Although GHR and GHBP are regulated by and are very sensitive to GH, and GHR and GHBP often change in parallel, measurement of plasma GHBP has not been shown to reflect GHR and GH responsiveness. Measurement of serum GHBP concentrations may, however, aid in identifying patients with GH insensitivity (GHI) caused by genetic abnormalities of the GHR. Patients with GHI from nonreceptor abnormalities, abnormalities of the intracellular portion of the GHR, or inability of the receptor to dimerize may have normal serum concentrations of GHBP. Recently, a complex of the GH and the GHBP molecule has been shown to be more effective than GH alone—indicating a physiological and possible therapeutic role for GHBP.
According to the somatomedin hypothesis, the anabolic actions of GH are mediated through the IGF polypeptides. Although this hypothesis is at least in part true, it appears that GH is capable of stimulating a variety of effects that are independent of IGF activity. Indeed, the effects of GH and IGF are on occasion contradictory, as for example, the “diabetogenic” actions of GH and the glucose-lowering activity of IGFs. Green and colleagues attempted to resolve some of these differences in a “dual-effector” model in which GH stimulates precursor cells, such as prechondrocytes, to differentiate. When differentiated cells or neighboring cells then secrete IGFs, these peptides act as mitogens and stimulate clonal expansion. This hypothesis is based on the ability of IGF peptides to work not only as classic endocrine factors that are transported through the blood but as paracrine or autocrine growth factors. GH also stimulates a variety of metabolic effects, some of which appear to occur independently of IGF production, such as lipolysis, amino acid transport in diaphragm and heart, and production of specific hepatic proteins. Thus there are multiple sites of GH action, and it is not entirely clear which of these actions are mediated through the IGF system and which might represent IGF-1–independent effects of GH.
The IGFs (or somatomedins) constitute a family of peptides that are at least in part GH-dependent and that are believed to mediate many of the anabolic and mitogenic actions of GH. Although they were originally identified in 1957 by their ability to stimulate [35S] sulfate incorporation into rat cartilage, it has been established over the ensuing 45 years that they are involved in diverse metabolic activities.
IGF-1, previously termed somatomedin-C , is a basic polypeptide of 70 amino acids, whereas IGF-2 is a slightly acidic polypeptide of 67 amino acids. The two peptides are structurally related, sharing 45 of 73 possible amino acid positions. They have approximately 50% amino acid homology to insulin. Like insulin, both IGFs have A and B chains connected by disulfide bonds and an intervening connecting (C-peptide) region. This structural homology explains the ability of both IGFs to bind to the insulin receptor and for insulin to bind to the type 1 IGF receptor. On the other hand, structural differences explain the failure of insulin to bind to the IGF-binding proteins.
In human fetal serum, IGF-1 concentrations are relatively low and are positively correlated with gestational age. A correlation between fetal cord serum IGF-1 concentrations with birth weight has been reported by some groups, although others have reported no correlation. IGF-1 concentrations in human newborn serum are generally 30% to 50% of adult concentrations. There is a slow, gradual rise in serum concentrations during childhood, with attainment of adult concentrations at the onset of sexual maturation. During puberty, IGF-1 concentrations increase to 2 to 3 times the concentrations seen in adults. Thus concentrations during adolescence correlate better with Tanner stage (bone age) than with chronological age. Girls with gonadal dysgenesis show no adolescent increase in serum IGF-1, clearly establishing the association of the pubertal rise in IGF-1 with the production of sex steroids. Estrogen stimulates GH secretion, which increases hepatic IGF-1 production. It is of note, however, that patients with GHI because of GHR mutations show a pubertal rise in serum IGF-1, which may be mediated by direct effects of androgen on IGF-1.
After adolescence, or at least after 20 to 30 years of age, serum IGF-1 concentrations demonstrate a gradual and progressive age-associated decline. It has been suggested that this decline may be responsible for the negative nitrogen balance, decrease in body musculature, and osteoporosis characteristic of aging. Although this provocative hypothesis remains unproven at this time, it has generated considerable interest in the potential use of GH and/or IGF-1 therapy in normal aging.
Human newborn concentrations of IGF-2 are generally 50% of adult concentrations. By 1 year of age, however, adult concentrations are attained with little, if any, subsequent decline even out to the seventh or eighth decade of life. This is in contrast to rodents, whose IGF-2 expression declines early following birth. Lack of rodent models has impeded our understanding of the physiological role of IGF-2 persistence in humans.
IGFs bind (although generally with low affinity) to insulin receptors, thus providing an explanation for their insulin-like activity. In addition, IGFs bind to at least two classes of IGF receptors.
The type 1 IGF receptor is closely related to the insulin receptor. Both are heterotetramers composed of two identical membrane-spanning alpha subunits and two identical intracellular beta subunits. The alpha subunits contain the binding sites for IGF-1 and are linked by disulfide bonds. The beta subunits contain a transmembrane domain, an adenosine triphosphate (ATP)-binding site, and a tyrosine kinase domain, which constitutes the presumed signal transduction mechanism for the receptor.
Although the type 1 IGF receptor has been commonly referred to as the IGF-1 receptor , the receptor is capable of binding both IGF-1 and IGF-2 with high affinity—and both IGF polypeptides appear capable of activating tyrosine kinase by binding to this receptor. Affinity of the type 1 receptor for insulin is generally 100-fold less, thereby providing one of the mechanisms for the mitogenic effects of insulin. IGF-1 acts primarily through IGF1R but can bind with lower affinity to the highly homologous insulin receptor (IR), and to IGF1R/IR heterodimers. Vice versa, insulin is able to signal through the IGF1R. Signs of such alternative signaling can become apparent in pathological IGF-1 or insulin signaling.
The type 1 IGF receptor mediates IGF actions on multiple cell types, and these actions are diverse and tissue specific. In general, it is believed that all of the effects of IGF receptor activation are mediated by tyrosine kinase activation and phosphorylation of substrates, which activate specific cellular pathways, leading to various biological actions. Among these effects is induction of cell growth through activation of the cell cycle machinery, maintenance of cell survival (prevention of apoptosis) mediated by effects on the Bcl family members, and induction of cellular differentiation, which occurs by as yet incompletely characterized mechanisms.
The substrates phosphorylated by the IGF receptor include members of the insulin receptor substrate family (particularly IRS-1 and IRS-2); knockout of these genes results in poor growth in mice (as well as insulin resistance). In addition, several other signaling molecules respond to IGF receptor activation. Blockade of the type I receptor has been proposed as a cancer therapy.
Mutations that decrease IGF signaling lead to an extension of life expectancy in nematodes, flies, and mice. It is unclear, however, what relevance the IGF-1/IR has for human longevity. Recent data have suggested that loss-of-function variants in IGF1R are overrepresented among female centenarians, suggesting a role of this pathway in modulation of human life span.
The type 2 IGF receptor, however, bears no structural homology with either the insulin or the type 1 IGF receptor. The type 2 receptor does not contain an intrinsic tyrosine kinase domain or any other recognizable signal transduction mechanism and has been found to be identical to the cation-independent mannose-6-phosphate (CIM6P) receptor, a protein involved in the intracellular lysosomal targeting of a variety of acid hydrolases and other mannosylated proteins. Unlike the type 1 IGF receptor, which binds both IGF polypeptides with high affinity and insulin with 100-fold lower affinity, the type 2 receptor only binds IGF-2 with high affinity. IGF-1 binds with substantially lower affinity, and insulin not at all. Most studies have indicated that the classic mitogenic and metabolic actions of IGF-1 and IGF-2 are mediated through the type 1 IGF receptor, with its tyrosine kinase signal transduction mechanism.
Although insulin and the IGFs share significant structural homology, and despite the structural-functional similarity of the insulin and type 1 IGF receptors, the IGFs differ from insulin in that the IGFs circulate in plasma complexed to a family of binding proteins. These carrier proteins extend the serum half-life of the IGF peptides, transport the IGFs to target cells, and modulate the interaction of the IGFs with their surface membrane receptors. Six distinct IGFBPs have been identified. Additional lower affinity IGF binding proteins (named IGFBP related proteins , IGFBPrPs ) were found by in silico searches for homology to the known IGFBPs; many of these molecules were previously known in other contexts, serving roles in normal or neoplastic growth.
Under most conditions, the IGFBPs appear to inhibit IGF action—presumably by competing with IGF receptors for IGF polypeptides. This concept is supported by the observation that IGF analogues with decreased affinity for IGFBPs generally appear to have increased biological potency. In addition, IGFBP-3 appears to inhibit cell growth even in the absence of added IGF—suggesting a direct inhibitory role of the binding protein. Under specific conditions, however, several of the IGFBPs apparently are capable of enhancing IGF action—perhaps by facilitating IGF delivery to target receptors.
Evidence indicates that IGFBPs are essentially bioactive molecules that, in addition to binding IGFs, have a variety of IGF-1–independent functions. These include growth inhibition in some cell types, growth stimulation in other tissues, direct induction of apoptosis, and modulation of the effects of other non-IGF growth factors. These effects of IGFBPs are mediated by binding to their own receptors. Because IGFBP-3 is regulated by GH, it is intriguing that in vivo, IGFBP-3 enhances IGF-1 action when given to hypophysectomized rats (rather than inhibiting it). The mechanisms involved in this effect have not been elucidated but may explain the limited effects of IGF-1 therapy on the growth of Laron patients.
The relative amounts of each of the IGFBPs vary among biological fluids. IGFBP-1 is the major IGFBP in human amniotic fluid. IGFBP-2 is prominent in cerebrospinal fluid and seminal plasma. IGFBP-3 is the major IGFBP in normal human serum and demonstrates clear GH dependence. Among the IGFBPs, IGFBP-3 and IGFBP-5 are unique in that they normally circulate in adult serum as part of a ternary complex consisting of IGFBP-3 or IGFBP-5, an IGF polypeptide, and an acid-labile subunit (ALS).
Analysis of IGFBPs is further complicated by the presence of IGFBP proteases, capable of various levels of IGFBP degradation. Initially reported in the serum of pregnant women, proteases for IGFBP-3, -4, and -5 have already been demonstrated in a variety of biological fluids. Proteolysis of IGFBPs complicates their assay by both Western ligand blotting and radioimmunoassay methodologies and must be taken into consideration when concentrations of the various IGFBPs in biological fluids are reported. The physiological significance of limited proteolysis of IGFBPs remains to be determined, although evidence suggests that protease activity results in decreased affinity of the IGFBP for IGF peptides. Recently, recessive mutations in the metalloproteinase pregnancy-associated plasma protein A2 ( PAPP-A2 ) were associated with short stature. The mutations lead to increased binding of IGF-1 within the ternary complex and reduced free IGF-1.
The critical role of the IGF system in fetal and postnatal growth was demonstrated in a series of elegant gene knockout studies in mice. Unlike GH and GHR knockouts, which are near normal size at birth, Igf1 null mice have a birth weight 60% of normal. Postnatal growth is also abnormal. A similar prenatal and postnatal growth phenotype has been observed in a reported human case of an Igf1 gene deletion. Igf2 null mice also show diminished pre- and postnatal growth. The Igf2 gene is imprinted in humans, such that only the paternal gene is expressed. Therefore loss-of-function mutations on the paternal allele cause growth impairment whereas mutations on the maternal allele do not. When the gene for the type 1 IGF receptor is knocked out (Igf1r null mice), the mice are severely growth retarded. In humans, dominant and recessive IGF1R mutations are associated with intrauterine growth retardation (IUGR), postnatal growth failure, and learning difficulties (see section on Disorders of Childhood Growth for details). The relationship between GH and IGF-1 in controlling postnatal growth was analyzed in mouse mutants lacking GHR, IGF-1, or both. This demonstrated that GH and IGF-1 promote postnatal growth by independent and common functions.
Thyroid hormone is required for normal linear growth. Newborns with hypothyroidism are of normal birth length, suggesting that fetal growth is not greatly affected by fetal thyroid hormone production. In contrast, severe, long-standing postnatal hypothyroidism can impair linear growth and delay skeletal maturation. Mutations in either thyroid hormone receptor-alpha or -beta cause some impairment in linear growth, suggesting that both of these receptors contribute to linear growth regulation. Thyroid hormone has both a direct effect on growth plate chondrocytes, particularly supporting chondrocyte hypertrophy, and an indirect effect, mediated by a stimulatory effect on growth hormone secretion.
Glucocorticoids, when present in concentrations that exceed physiological levels, impair linear growth. This linear growth impairment arises in part from a direct effect of glucocorticoids on growth plate chondrocyte proliferation. Glucocorticoids may also indirectly affect growth plate chondrogenesis by altering GH secretion. However, not all studies show an effect, and any effects may be caused by the glucocorticoid-induced obesity. Circulating concentrations of IGF-1, a marker and mediator of GH action, do not appear to be reduced by glucocorticoid excess, which would be consistent with the idea that the decreased GH concentrations are primarily caused by obesity. The inability of GH treatment to fully compensate for the linear growth impairment caused by glucocorticoid excess further suggests that the growth impairment is primarily caused by a direct action of glucocorticoids on growth plate chondrocytes.
In adolescents, both male and female, estrogens stimulate linear growth, contributing to the pubertal growth spurt. In boys, the estrogen that stimulates the adolescent growth spurt is produced by aromatization of testicular and adrenal androgens. The growth acceleration is mediated in part by the stimulatory effect of estrogens on GH secretion. In addition, estrogen appears to have direct actions on growth plate chondrocytes. Estrogen stimulates growth plate senescence, the developmental program in the growth plate that is responsible for the progressive slowing of linear growth with age. Consequently, estrogen exposure leads to more rapid “aging” of the growth plate, earlier growth cessation, and earlier epiphyseal fusion. This effect of estrogen accounts for the early epiphyseal fusion in children with precocious puberty and the late epiphyseal fusion in individuals with hypogonadism, aromatase deficiency, and estrogen resistance.
Thus the effect of estrogen on linear growth is a “two-edged sword” in that it has two opposing effects on adult height. Estrogen stimulates the linear growth rate, which would be expected to increase adult height, but also accelerates growth plate senescence, which would be expected in decreased adult height. In general, the latter effect predominates. As a result, untreated precocious puberty can cause initial childhood tall stature but eventual adult short stature, whereas untreated hypogonadism or delayed puberty can cause adolescent short stature but eventual adult tall stature. These concepts also suggest that gonadotropin-releasing hormone (GnRH) analogues and aromatase inhibitors would increase adult height (see section on Treatment of Short Stature, later in this chapter).
Androgens also can accelerate linear growth and thus contribute to the pubertal growth spurt. Androgens, secreted by the gonads or adrenal glands can be aromatized to estrogens in various peripheral tissues, including adipose tissue and thereby affect linear growth. Aromatase is also expressed in growth plate cartilage ; thus local conversion into estrogen may occur. In addition to androgen’s role as a precursor for estrogen, androgen per se appears also to stimulate linear growth. For example, dihydrotestosterone, which is not aromatizable, appears to accelerate linear growth in adolescent boys. Animal and in vitro studies suggest that these effects are mediated, at least in part, through a direct effect on the growth plate.
Insulin positively regulates human fetal growth. Consequently, fetal growth retardation occurs in individuals with decreased fetal insulin production because of permanent neonatal diabetes mellitus and those with decreased insulin sensitivity because of severe biallelic insulin receptor defects. Conversely, increased fetal growth occurs in pregnancies complicated by gestational diabetes. This macrosomia, which primarily involves weight rather than length, is thought to be caused by the high glucose concentrations stimulating high fetal insulin concentrations.
Inadequate nutritional intake causes functional GH insensitivity, with decreased circulating IGF-1 concentrations and increased GH concentrations. Proposed mechanisms include decreased insulin concentrations, causing downregulation of GHR expression and increased FGF-21 concentrations, causing a decrease in hepatic GHR and phosphorylated STAT5 (which is involved in GH signal transduction). The increased FGF-21 concentrations in malnutrition may impair GH sensitivity not only in the liver but also in the growth plate. In addition, undernutrition can decrease thyroid hormone concentrations, as part of the nonthyroidal illness syndrome, increase cortisol concentrations, and decrease sex steroids (mediated by decreased leptin and hence gonadotropins), all of which may contribute to the decrease in linear growth.
Conversely, overnutrition can stimulate linear growth. Obese children tend to be tall with an increased bone age. Because of the accelerated skeletal maturation and, in girls, the earlier puberty, there is little effect on adult height. Proposed mechanisms by which obesity affects linear growth include increased levels of estrogens because of peripheral aromatization and increased free IGF-1, leptin, prolactin, and adrenal androgens.
Impaired linear growth often occurs in children with systemic inflammatory disorders, including juvenile idiopathic arthritis (JIA), IBD, and cystic fibrosis. The underlying mechanisms are likely complex and involve multiple mediators, including malnutrition and glucocorticoid excess. In addition, there is evidence that elevated levels of proinflammatory cytokines contribute to the poor growth. Various proinflammatory cytokines, including tumor necrosis factor-α (TNF-α), IL-1β, and IL-6, act directly on growth plate chondrocytes to suppress bone growth. Proinflammatory cytokines may also act indirectly on the growth plate by decreasing circulating IGF-1, sex steroid, and thyroid hormone concentrations. The effects of these cytokines may be mediated in part by upregulation of an intracellular protein called SOCS, which decreases GHR signaling.
Many different autocrine/paracrine factors are secreted by growth plate chondrocytes and act locally on other chondrocytes. These factors serve to orchestrate the complex processes of proliferation and differentiation in the growth plate. Consequently, genetic defects in these local growth factors, their receptors, or other genes involved in growth factor signaling can impair growth plate chondrogenesis and therefore linear growth of the child. Depending on the specific paracrine system involved and the severity of the genetic abnormality, the bones might be both short and malformed, presenting clinically as a chondrodysplasia, or short but not malformed, presenting clinically as isolated short stature. Following are some examples of autocrine/paracrine growth factors that regulate growth plate chondrocytes and are clinically important.
C-type natriuretic peptide (CNP) was named based on its structural similarity to atrial and brain natriuretic peptides. However, CNP serves different functions. It acts locally in the growth plate to stimulate chondrocyte proliferation and hypertrophy. Consequently, loss-of-function mutations in NPR2, the receptor for CNP, cause short stature whereas gain-of-function mutations cause tall stature (see section on Disorders of Childhood Growth for details). CNP analogues are in clinical trials to augment bone growth in achondroplasia (ACH).
FGFs, acting through FGF receptor-3 (FGFR3), negatively regulate growth plate chondrogenesis. As a result, gain-of-function mutations in FGFR3 impair linear growth. Depending on the severity of the mutation, the clinical presentation can include several skeletal dysplasias with severe short stature. Recent evidence suggests that milder gain-of-function mutations can present as isolated short stature without an evident skeletal dysplasia. Conversely, loss-of-function mutations cause tall stature.
IGF-1 and -2 act through the receptor IGF1R to stimulate growth of many tissues. In the growth plate, they act on chondrocytes to stimulate proliferation and hypertrophy. IGF-1, produced primarily in the liver, acts in an endocrine fashion on the growth plate (see section Regulation of Linear Growth-Endocrine Regulation of Linear Growth), but IGF-1 is also thought to be produced locally in the growth plate, by cells in the perichondrium and/or chondrocytes, and act locally on chondrocytes as a paracrine factor. In rodents, both endocrine and paracrine effects are important, but the relative importance in humans is unclear. IGF-2 acts primarily as a paracrine factor and is expressed at high levels in fetal tissues. The IGF2 gene is imprinted and consequently is expressed only by the paternal allele. In humans, mutations in IGF1, IGF2, and IGFR1 can all impair both fetal and postnatal growth (see section on Disorders of Childhood Growth for details) indicating that both IGF-1 and IGF-2 are important for growth before and after birth.
Parathyroid hormone-related protein (PTHrP) is expressed in the chondrocytes near the ends of the long bones in the embryo and in the resting zone of the growth plate postnatally. It diffuses down through the growth plate. PTHrP acts through PTH1R (the same receptor used by PTH) and, in part, through Gs-alpha, to prevent proliferative zone chondrocytes from undergoing hypertrophic differentiation until they reach a sufficient distance from the source of PTHrP. Indian hedgehog (IHH) is produced by chondrocytes that have just begun hypertrophic differentiation. IHH stimulates production of PTHrP, forming a negative feedback loop. Mutations in genes that encode components of this system impair bone growth. For example, loss-of-function mutations in PTH1R cause Blomstrand lethal chondrodysplasia, and gain-of-function mutations in PTH1R cause Jansen metaphyseal chondrodysplasia. Similarly, loss-of-function mutations in IHH cause brachydactyly type A1 or short stature with nonspecific skeletal abnormalities, and loss-of-function mutations in Gs-alpha cause Albright hereditary osteodystrophy. Mutations in genes affecting subsequent steps in the PTHrP signal transduction pathway cause acrodysostosis.
Growth plate chondrocytes secrete a cartilage matrix that serves a structural function, allowing the growth plate cartilage to bear the considerable mechanical forces required of the skeletal system. The cartilage matrix also provides the milieu through which endocrine and paracrine factors must travel to reach growth plate chondrocytes. This matrix is a complex mixture of molecules, including multiple types of collagen, for example, collagen types II and X, and proteoglycans, such as aggrecan. Mutations in many of the encoding genes, for example, COL2A1 , COL10A1 , and ACAN (aggrecan), cause chondrodysplasias. Heterozygous mutations in ACAN can also present as short stature with advanced bone age (see section on Disorders of Childhood Growth for details). Mutations in the collagen 1 genes, COL1A1 and COL1A2 , cause osteogenesis imperfecta. Affected patients have bone fragility, reflecting the important role of collagen type I in bone matrix, but also can have short stature.
Proliferation and hypertrophic differentiation of growth plate chondrocytes is also under complex control by intracellular factors. For example, the short stature homeobox-containing gene ( SHOX) encodes a transcription factor that is required for normal growth plate function and consequently mutations in SHOX cause short stature. Another example of an intracellular system that regulates growth plate chondrocyte function is the RAS-mitogen-activated protein kinase (MAPK) pathway. It is a signal transduction system that regulates cell proliferation and differentiation in many tissues. Ligand binding to tyrosine kinase receptors causes signal transduction involving phosphorylation of a series of proteins. Mutations that augment RAS-MAPK signaling lead to Noonan syndrome (NS), which includes short stature.
Linear growth is extremely rapid in the human fetus and progressively slows during infancy and childhood. This deceleration is briefly interrupted by the pubertal growth spurt but then the deceleration resumes, causing growth to eventually cease. The decline in linear growth is caused by a developmental program intrinsic to the growth plates termed growth plate senescence . This developmental program includes a functional decline in chondrocyte proliferation and also a gradual involution of the growth plate with a decrease in the number of chondrocytes and a decrease in the height of the growth plate. Growth plate senescence advances until chondrocyte proliferation essentially ceases. At that point, the growth plate is no longer causing bone elongation, and the now-inert cartilage is remodeled into bone, an event termed epiphyseal fusion . Thus epiphyseal fusion is not the cause, but rather the effect, of growth cessation. The bone age (usually assessed by a radiograph of the nondominant hand and wrist) serves as a radiological marker for growth plate senescence. As a consequence, the bone age provides an estimate of how far senescence has proceeded in a child and therefore how much of the child’s growth potential has been expended and how much remains. For this reason, the bone age is useful for prediction of adult height.
Growth-inhibiting conditions, such as malnutrition, hypothyroidism, and GH deficiency, generally slow not only the growth rate, but also the rate of growth plate senescence. This delay in growth plate senescence is reflected by the delay in the bone age. From an evolutionary standpoint, this delay in growth plate senescence likely serves an important function in the malnourished child, allowing growth to slow to conserve nutrients, while conserving much of the growth potential for better times.
In contrast, estrogen accelerates growth plate senescence, explaining the advanced bone age and loss of adult height in children with precocious puberty and explaining the delayed bone age and increase in adult height of adolescents with hypogonadism, aromatase deficiency, and estrogen resistance. For example, in a man with biallelic mutations in estrogen receptor-alpha, linear growth continued at a slow rate well into adulthood, resulting in tall stature, and epiphyseal fusion occurred at 35 years of age. This effect of estrogen on growth plate senescence also explains the potential therapeutic use of GnRH analogues and aromatase inhibitors to increase adult height.
Catch-up growth occurs in children following a period of growth inhibition. Linear growth can be inhibited by many different conditions, including malnutrition, hypothyroidism, growth hormone deficiency, and glucocorticoid excess. If the condition persists long enough, the child’s height standard deviation score (SDS) for age will decline (decreasing percentile on height curve). Often the growth-inhibiting condition will eventually resolve, for example, if food becomes more available, thyroid hormone or growth hormone replacement is initiated, or pharmacological glucocorticoids can be stopped. In this situation, the child’s linear growth rate usually does not just increase to equal the normal growth rate for age. Instead, the child’s growth rate exceeds normal, causing the height SDS to improve. This catch-up growth is often not complete in that the child’s height SDS does not return all the way back to the value before the growth inhibition, and the adult height may still be somewhat compromised.
For some time, it was thought that catch-up growth results from a mechanism within the CNS that assesses the child’s actual body size, compares this actual body size to age-appropriate set point, and then adjusts the growth rate accordingly. After a period of growth inhibition, this hypothesized mechanism would sense the height deficit and increase the growth rate to cause catch-up growth. However, more recent evidence suggests that the primary mechanism responsible for catch-up growth resides not in the CNS but rather in the growth plates and involves delayed growth plate senescence. Growth-inhibiting conditions slow growth plate senescence. When the growth-inhibiting condition resolves, the growth plates are less senescent than normal and therefore grow more rapidly than normal and for a longer period of time, causing catch-up growth. In some conditions, the rate of human catch-up growth fits this hypothesis, but in others conditions the high initial rate of catch-up growth suggests the possibility of other contributing mechanisms.
Childhood growth patterns and adult height are the result of interactions between genetic and environmental factors. Since Francis Galton’s famous study in 1885, it has been known that a child’s height is strongly correlated with the height of his or her parents. Later large-scale studies focusing on height variability between twins showed that environmental factors have greater importance during early childhood, whereas genetic factors have a more decisive influence on the height of adolescents and adults. Recently, the specific genes that control normal height variation have begun to be elucidated by genome-wide association studies (GWAS), which search for statistical associations between adult height and single nucleotide polymorphisms. These studies have identified common allelic variants (usually with a minor allele frequency > 5%) that individually exert only a small phenotypic effect, but together determine much of the variability of height. This approach was used to identify variants in more than 700 loci that have a significant influence on height. Each of these variants is responsible for approximately 1 mm in height variability per allele and in combination, explain approximately 25% of the heritability in adult height. In addition, 83 lower-frequency coding variants (minor allele frequency ≤ 1%) have been identified as being responsible for up to 2 cm of height per allele.
Accurate measurement is the critical cornerstone of all growth evaluations. Although it is often dismissed as simple (after all, how many parents track their kids’ growth on a wall in their homes?), two factors make it in fact challenging. First, because growth is defined as the change in length or height over a certain period of time, the two measurements at each end of that time interval must be performed according to identical technique; any deviations by one or the other measurer can introduce artifactual microaccelerations or microdecelerations into the calculated growth that become amplified errors when the velocity is annualized for analysis ( Box 11.1 ). Second, intrinsic to identical technique is correct positioning each and every time, which can be challenging with an infant or child who is fidgety, anxious, bored, developmentally unable to understand or execute the instructions, or physically challenged in achieving and maintaining the correct position (e.g., because of hypotonia, hypertonia, contractures, balance issues, leg length asymmetry, malformations, injury, or illness). Further compounding the difficulty in maintaining identical technique each and every time are both personnel issues (the same child is commonly measured by different staff members at serial visits because of the time that has lapsed or different locations of care) and equipment issues (which must be calibrated regularly and used appropriately). A final source of error can occur at the recording stage, when the measurement is entered into the medical record, such as transposing digits, other typographical error, or inadvertently recording the measurement in inches or centimeters when it had been measured in the other unit.
Standing height should be measured with a wall-mounted stadiometer. The child should be standing erect against the wall or backboard with shoes off, any interfering hair accessories removed, and the head in the Frankfurt plane (the lower margins of the orbits and the upper margins of the ear canals all lying in the same horizontal plane), that is, looking straight ahead. The head plate, which is lowered on top of the head for the measurement reading, must be firmly fixed such that it is perpendicular to the wall or backboard. Flip-up horizontal bars (floppy arms) on weighing scales, frequently used to measure height, are subject to great errors because of the child’s slumping posture and considerable variation in the angle of the horizontal bar.
Recumbent length is similarly measured for infants and toddlers using a recumbent length board, composed of a firm platform, a fixed head plate, and a moveable footplate for reading the measurement. Two people are required for accurate length measurement: one to hold the head in the Frankfurt plane, looking straight up, and the second to keep the knees straight and bring the measuring board to the feet, which must also be secured perpendicular at the ankles. Many practitioners, measuring infants unassisted, lie the patient on the paper covering the examining table, mark off with a pen on the paper the positions of the head and feet, remove the patient, and then use a measuring tape to quantify the distance between the two pen markings. This common technique is highly inaccurate because of incorrect positioning of the infant, movement and crumpling of the paper, and failure to get perpendicular markings by the pen.
Despite well-established, generally accepted protocols for measuring children, studies have shown that it is frequently done improperly. Observation of 55 US primary care practices (44 pediatric, 11 family practice) found measurements were considered accurate (defined as ≤ 0.5 cm) only 30% of the time, and differed from measurements of the same children by the study investigators by an average of 1.3 cm. Because the average error exceeded the difference between normal (5 cm/year) and subnormal (4 cm/year) growth velocities, measurement error likely contributes to both inappropriate investigation and/or subspecialist referrals of normally growing children and missed detection of true growth problems in others. However, this is readily corrected through training. Randomization of these same 55 practices to educational intervention and control groups found that measurement accuracy rates rose persistently in the intervention group at 3 months (55% vs. 37% in the control group) and 6 months (70% vs. 34% in the control group) later. Although many busy primary care practices measure patients only once, triplicate measurements are taken (with the average recorded) as a means of improving accuracy when precision is key (e.g., for children undergoing growth evaluations and treatment in endocrine clinics or participating in research studies).
With the proper equipment, experienced personnel, and a cooperative child, the standing height percentile can be assessed with considerable accuracy. In contrast, even with good measurement technique, the error in short-term growth velocity assessment is problematic. The relative error (absolute error divided by the measurement) is far greater for growth velocity than for height primarily because the denominator for the growth velocity (the small difference between two successive measurements) is so much smaller than the denominator for the height. For example, for a 5-year-old child with an observed height at the third percentile, the 95% confidence interval spans the second to fourth percentile but the confidence interval for a 12-month height velocity spans the eighth to 52nd percentiles. As a result, even with 1-year growth velocity, children can easily be misclassified as having a normal versus abnormal growth velocity. Trying to measure growth velocity over periods less than a full year results in a relative measurement error that is even greater. Growth velocity, measured over a year, was found to have little diagnostic validity to identify children with isolated idiopathic GH deficiency or Turner syndrome. Height velocity had to be measured over a 3-year period to achieve acceptable validity. Thus overreliance on calculated growth velocity may be misleading. However, it is important for the clinician to identify children with poor height and/or weight gain, which can indicate an important underlying disorder. As an alternative to calculated growth velocities, declines in height and/or weight percentiles on a standard growth chart can provide valuable clues to a diminished growth rate.
Because normal growth follows a predictable pattern, growth charts are constructed depicting this pattern as sequential centile curves showing the distribution of selected body measurements (usually length or height, weight, and weight-for-length or body mass index [BMI]) in children of a given country. Growth is fastest during the fetal period and gradually slows during infancy, with an approximate length increase of 25 cm over the first year of life and 10 to 15 cm over the second year. During infancy, children commonly cross length centiles (physiological rechanneling) as they transition from prenatal to postnatal growth determinants; maternal and pregnancy health and nutrition have a major impact on the former, whereas the latter normally aligns with the child’s genetic height potential. After the second birthday, the linear growth rate continues to decline, but more gradually, and shifting centile channels is abnormal. With the onset of puberty, statural growth accelerates, resulting in the pubertal growth spurt, after which the decline in linear growth rate resumes, causing height gain to gradually grind to a halt. However, attainment of adult body composition continues in the transition period that follows.
The population selected for reference is important when judgments are made about the height and growth of an individual. Multiple growth charts have been constructed for US children in past decades, differing in the racial and socioeconomic composition of the reference populations upon which they were based. Growth charts for children of other countries are likewise available, reflecting the different height distributions of the local populations. The most recent and currently recommended US growth charts, published in 2000 by the Centers for Disease Control and Prevention (CDC; available at https://www.cdc.gov/growthcharts/cdc_charts.htm ), are based on several cross-sectional data sources and introduced US pediatric BMI charts. Because the CDC and other national charts are used for clinical judgment despite being reference charts (i.e., observationally describing how children in the populations grow), the World Health Organization (WHO) set out to create growth standard charts (i.e., depicting how healthy children should grow). The 2006 WHO growth charts (available at: https://www.cdc.gov/growthcharts/who_charts.htm ) are based on longitudinal measurements from birth to age 2 years of 882 children from six different countries, representing racial diversity yet growing under “ideal” conditions, and cross-sectional data on other children after age 2 years. Thus the CDC recommends use of the 2006 WHO growth charts under age 2 years and the 2000 CDC growth charts after age 2 years in assessing growth of US children. One important distinction is that the WHO charts are based on breastfed infants versus the mostly formula-fed infants of the CDC charts, resulting in differently shaped weight curves that can alter the perceived rates of failure to thrive depending on the feeding modality of the infants assessed clinically.
Two special categories of growth charts are also helpful. Velocity charts plot annual increments rather than height (distance) and are tempo adjusted, allowing for the variation in timing and intensity of the adolescent growth spurt. These charts may be helpful to assess the growth patterns of children with slower or more rapid tempo of maturation (“early and late bloomers”), whose variations are effectively lost in the cross-sectional height charts that average growth across peers in different developmental periods. One limitation to such longitudinal growth charts is that their data were derived from relatively small, nonrepresentative population samples recorded a long time ago. A second limitation is that the large relative error in growth velocity measurements, discussed earlier, creates both uncertainty in the individual child’s data and also in the reference data upon which the growth velocity charts were based. Thus based on studies of measurement error, Voss and colleagues concluded, “Although velocity charts are attractive in concept, they seem to be no more discriminating than height charts in practice, and may be clinically deceptive unless interpreted with great care.” Alternatively, growth charts that show height, rather than growth velocity, but are based on longitudinal data, may be helpful for children with slow or rapid tempo of maturation.
The second special category of growth charts includes specific charts for children with particular genetic syndromes. These charts are used in essentially the same way as the charts for normal children. Deviation of growth from the appropriate disease-related growth curve suggests the possibility of a second underlying problem, such as acquired hypothyroidism in a patient with Down or Turner syndrome.
The skeleton does not grow proportionally. The upper-to-lower body segment ratio starts at 1.7 at birth, indicating a trunk considerably longer than the lower extremities. However, after birth, the lower extremities grow more rapidly than the trunk, and the ratio drops to approximately 1.0 by 10 years of age. Early cessation of linear growth, such as that which occurs in precocious puberty, results in persistently childlike proportions (short limbs compared with the trunk), whereas prolonged growth, such as occurs in hypogonadism, creates a long-limbed habitus. Limb to trunk proportions can be assessed by three different techniques, all incorporating standing height measurement for comparison. For the upper-to-lower segment ratio, the lower body segment is measured from the upper border of the pubic symphysis to the floor while the patient is standing straight. This lower segment length is subtracted from the standing height to determine the upper segment length and then divided by the lower segment length to determine the ratio, which can be compared with reference data. The second technique measures the arm span (between the tips of the middle fingers with the arms fully extended perpendicular to the torso, again with the child standing straight against the wall) for comparison with the standing height. Normally arm span is less than standing height before age 8 years, equal to height at ages 8 to 12 years, and greater than height above age 12 years. The third technique measures the sitting height, which is divided by the standing height to yield the sitting height index. The sitting height index is sensitive for detecting disproportionate growth of the trunk (vertebrae) versus the lower extremities and can be used to screen for specific causes of disproportionate short stature, such as SHOX deficiency and hypochondroplasia. Excellent reference data are available from NHANES III ( Table 11.2 ).
| Age Group (years) | Mean Age (years) | Percentiles | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| N | M | SD | 5th | 10th | 15th | 25th | 50th | 75th | 85th | 90th | 95th | ||
| Males | |||||||||||||
| 2.0–2.9 | 2.46 | 619 | 56.9 | 2.01 | 53.7 | 54.4 | 54.9 | 55.6 | 56.9 | 58.3 | 59.0 | 59.6 | 60.3 |
| 3.0–3.9 | 3.45 | 508 | 55.5 | 1.8 | 52.6 | 53.2 | 53.6 | 54.3 | 55.6 | 56.8 | 57.5 | 58.0 | 58.7 |
| 4.0–4.9 | 4.47 | 553 | 54.4 | 1.8 | 51.6 | 52.2 | 52.6 | 53.2 | 54.4 | 55.6 | 56.3 | 56.7 | 57.4 |
| 5.0–5.9 | 5.43 | 496 | 53.5 | 1.7 | 50.8 | 51.4 | 51.8 | 52.4 | 53.5 | 54.6 | 55.3 | 55.7 | 56.3 |
| 6.0–6.9 | 6.45 | 262 | 52.7 | 1.6 | 50.2 | 50.7 | 51.1 | 51.7 | 52.7 | 53.8 | 54.4 | 54.8 | 55.5 |
| 7.0–7.9 | 7.47 | 273 | 52.1 | 0.0 | 49.7 | 50.2 | 50.6 | 51.1 | 52.2 | 53.2 | 53.8 | 54.2 | 54.8 |
| 8.0–8.9 | 8.46 | 267 | 51.7 | 0.1 | 49.3 | 49.8 | 50.2 | 50.7 | 51.7 | 52.8 | 53.3 | 53.7 | 54.3 |
| 9.0–9.9 | 9.50 | 281 | 51.4 | 0.1 | 49.0 | 49.5 | 49.9 | 50.4 | 51.4 | 52.4 | 53.0 | 53.4 | 53.9 |
| 10.0–10.9 | 10.45 | 288 | 51.2 | 1.5 | 48.8 | 49.3 | 49.7 | 50.2 | 51.2 | 52.2 | 52.8 | 53.1 | 53.7 |
| 11.0–11.9 | 11.44 | 275 | 51.0 | 1.4 | 48.7 | 49.2 | 49.6 | 50.1 | 51.1 | 52.1 | 52.6 | 53.0 | 53.6 |
| 12.0–12.9 | 12.47 | 204 | 51.0 | 1.4 | 48.7 | 49.2 | 49.5 | 50.0 | 51.0 | 52.0 | 52.6 | 52.9 | 53.5 |
| 13.0–13.9 | 13.47 | 192 | 51.0 | 1.4 | 48.7 | 49.2 | 49.5 | 50.0 | 51.0 | 52.0 | 52.6 | 52.9 | 53.5 |
| 14.0–14.9 | 14.49 | 186 | 51.1 | 1.5 | 48.7 | 49.2 | 49.6 | 50.1 | 51.1 | 52.1 | 52.6 | 53.0 | 53.6 |
| 15.0–15.9 | 15.45 | 182 | 51.2 | 1.5 | 48.8 | 49.3 | 49.6 | 50.2 | 51.1 | 52.2 | 52.7 | 53.1 | 53.7 |
| 16.0–16.9 | 16.45 | 196 | 51.3 | 1.5 | 48.9 | 49.4 | 49.7 | 50.3 | 51.3 | 52.3 | 53.2 | 53.8 | |
| 17.0–17.9 | 17.45 | 191 | 51.4 | 1.5 | 49.0 | 49.5 | 49.8 | 50.4 | 51.4 | 52.4 | 53.0 | 53.4 | 53.9 |
| 18.0–18.9 | 18.45 | 172 | 51.6 | 1.5 | 49.1 | 49.6 | 50.0 | 50.5 | 51.5 | 52.6 | 53,1 | 53.5 | 54.1 |
| 19.0–19.9 | 19.43 | 160 | 51.7 | 1.5 | 49.2 | 49.7 | 50.1 | 50.6 | 51.7 | 52.7 | 53.3 | 53.7 | 54.3 |
| 20.0–29.9 | 24.96 | 1620 | 52.3 | 1.5 | 49.8 | 50.3 | 50.7 | 51.2 | 52.2 | 53.3 | 53.8 | 54.2 | 54.8 |
| 30.0–39.9 | 34.72 | 1452 | 52.4 | 1.6 | 49.9 | 50.4 | 50.8 | 51.3 | 52.4 | 53.4 | 54.0 | 54.4 | 55.0 |
| 40.0–49.9 | 44.35 | 1195 | 52.2 | 1.5 | 49.9 | 50.4 | 50.8 | 51.3 | 52.3 | 53.4 | 53.9 | 54.3 | 54.9 |
| 50.0–59.9 | 54.89 | 835 | 52.1 | 1.5 | 49.8 | 50.3 | 50.7 | 51.2 | 52.2 | 53.3 | 53.8 | 54.2 | 54.8 |
| 60.0–69.9 | 64.83 | 1129 | 51.9 | 1.5 | 49.7 | 50.2 | 50.6 | 51.1 | 52.1 | 53.1 | 53.6 | 54.0 | 54.5 |
| 70.0–79.9 | 74.16 | 798 | 51.7 | 1.4 | 49.5 | 50.0 | 50.4 | 50.9 | 51.8 | 52.8 | 53.3 | 53.7 | 54.2 |
| 80.0–90.9 | 84.09 | 566 | 51.5 | 1.4 | 49.3 | 49.8 | 50.1 | 50.6 | 51.5 | 52.5 | 53.0 | 53.3 | 53.9 |
| Females | |||||||||||||
| 2.0–2.9 | 2.45 | 584 | 56.9 | 1.9 | 52.9 | 53.5 | 54.0 | 54.7 | 55.9 | 57.2 | 57.9 | 58.4 | 59.1 |
| 3.0–3.9 | 3.46 | 586 | 55.3 | 1.8 | 52.1 | 52.7 | 53.2 | 53.8 | 55.0 | 56.2 | 56.9 | 57.4 | 58.1 |
| 4.0–4.9 | 4.43 | 528 | 54.1 | 1.6 | 51.5 | 52.1 | 52.5 | 53.1 | 54.3 | 55.4 | 56.1 | 56.5 | 57.2 |
| 5.0–5.9 | 5.46 | 543 | 53.2 | 1.6 | 50.9 | 51.5 | 51.9 | 52.5 | 53.6 | 54.7 | 55.4 | 55.8 | 56.4 |
| 6.0–6.9 | 6.47 | 273 | 52.5 | 1.6 | 50.4 | 51.0 | 51.4 | 52.0 | 53.1 | 54.2 | 54.8 | 55.2 | 55.8 |
| 7.0–7.9 | 7.44 | 268 | 52.1 | 1.4 | 50.1 | 50.7 | 51.0 | 51.6 | 52.7 | 53.7 | 54.3 | 54.7 | 55.3 |
| 8.0–8.9 | 8.47 | 247 | 51.8 | 1.5 | 49.8 | 50.4 | 50.7 | 51.3 | 52.3 | 53.4 | 54.0 | 54.3 | 54.9 |
| 9.0–9.9 | 9.43 | 273 | 51.7 | 1.5 | 49.6 | 50.1 | 50.5 | 51.0 | 52.1 | 53.1 | 53.7 | 54.1 | 54.7 |
| 10.0–10.9 | 10.43 | 260 | 51.6 | 1.5 | 49.5 | 50.0 | 50.3 | 50.9 | 51.9 | 52.9 | 53.5 | 53.9 | 54.4 |
| 11.0–11.9 | 11.46 | 288 | 51.6 | 1.4 | 49.4 | 49.9 | 50.2 | 50.8 | 51.8 | 52.8 | 53.3 | 53.7 | 54.3 |
| 12.0–12.9 | 12.46 | 221 | 51.7 | 1.5 | 49.3 | 49.9 | 50.2 | 50.7 | 51.7 | 52.7 | 53.3 | 53.6 | 54.2 |
| 13.0–13.9 | 13.45 | 227 | 51.8 | 1.5 | 49.3 | 49.8 | 50.2 | 50.7 | 51.7 | 52.7 | 53.2 | 53.6 | 54.2 |
| 14.0–14.9 | 14.47 | 216 | 52.0 | 1.5 | 49.4 | 49.9 | 50.2 | 50.7 | 51.7 | 52.7 | 53.3 | 53.6 | 54.2 |
| 15.0–15.9 | 15.47 | 189 | 52.1 | 1.5 | 49.4 | 50.0 | 50.3 | 50.8 | 51.8 | 52.8 | 53.3 | 53.7 | 54.2 |
| 16.0–16.9 | 16.46 | 224 | 52.2 | 1.5 | 49.5 | 50.0 | 50.4 | 50.9 | 51.9 | 52.9 | 53.4 | 53.8 | 54.3 |
| 17.0–17.9 | 17.45 | 213 | 52.4 | 1.4 | 49.6 | 50.1 | 50.5 | 51.0 | 52.0 | 53.0 | 53.5 | 53.9 | 54.4 |
| 18.0–18.9 | 18.43 | 186 | 52.5 | 1.4 | 49.8 | 50.3 | 50.6 | 51.1 | 52.1 | 53.1 | 53.6 | 54.0 | 54.6 |
| 19.0–19.9 | 19.48 | 191 | 52.6 | 1.3 | 49.9 | 50.4 | 50.8 | 51.3 | 52.3 | 53.2 | 53.8 | 54.2 | 54.7 |
| 20.0–29.9 | 24.91 | 1842 | 52.9 | 1.5 | 50.2 | 50.8 | 51.1 | 51.7 | 52.7 | 53.8 | 54.3 | 54.7 | 55.3 |
| 30.0–39.9 | 34.85 | 1837 | 52.8 | 1.5 | 50.5 | 51.0 | 51.4 | 51.9 | 52.9 | 53.9 | 54.5 | 54.9 | 55.5 |
| 40.0–49.9 | 44.28 | 1328 | 52.8 | 1.5 | 50.5 | 51.1 | 51.4 | 51.9 | 52.9 | 53.9 | 54.5 | 54.9 | 55.4 |
| 50.0–59.9 | 54.83 | 979 | 52.8 | 1.5 | 50.4 | 50.9 | 51.3 | 51.8 | 52.8 | 53.8 | 54.3 | 54.7 | 55.2 |
| 60.0–69.9 | 64.82 | 1110 | 52.3 | 1.5 | 50.1 | 50.6 | 50.9 | 51.4 | 52.4 | 53.4 | 54.0 | 54.3 | 54.9 |
| 70.0–79.9 | 74.46 | 893 | 52.0 | 1.5 | 49.5 | 50.0 | 50.4 | 50.9 | 51.9 | 52.9 | 53.5 | 53.9 | 54.4 |
| 80.0–90.9 | 84.45 | 599 | 51.1 | 1.5 | 48.8 | 49.3 | 49.7 | 50.2 | 51.2 | 52.2 | 52.8 | 53.2 | 53.8 |
Disproportions can occur not only between the limbs and trunk but also in specific limb segments, categorizing patients into specific diagnostic groups. Rhizomelia refers to disproportionate shortening of the proximal limbs (humerus and femur), mezomelia to disproportionately shortened middle portion of the limbs (radius/ulna and tibia/fibula), and acromelia to disproportionately shortened distal limbs (hands and feet). Thus every child who presents with a growth problem should be evaluated for disproportionality because this information helps to narrow the differential diagnosis, including the possibility of skeletal dysplasias.
Normal variation in rates of maturation among children and adolescents impacts growth prognosis, as children with more time to grow can achieve taller adult heights. An anteroposterior (AP) radiograph of the nondominant hand and wrist is used to determine the child’s bone age as a quantitative determination of somatic maturation. The bone age assesses the degree to which the skeleton has ossified. In the human embryo, the skeleton first forms out of cartilage, and then later the cartilage is remodeled into bone. This process continues throughout childhood. In the long bones, this ossification begins as a primary ossification center in the middle of the bone shaft and then spreads outward. Later, secondary ossification centers form in the epiphyses and also spread outward. However, the primary and secondary ossification centers remain separated during childhood by a thin layer of cartilage, termed the growth plate , which is responsible for bone elongation and therefore height gain. In adolescence, as growth comes to a halt, the growth plate itself is finally ossified, an event termed epiphyseal fusion , leaving cartilage only at the articular surface. Because bone is more radiopaque than cartilage or soft tissues, a radiograph of the left hand and wrist reveals how far along the ossification has proceeded in an individual child. The image is then compared with published standards (Greulich and Pyle or Tanner-Whitehouse [TW] ). There are separate sets of standards for boys and girls. If, for example, a patient’s bone age image is most similar to the standard for a 9-year-old child, then the patient is said to have a bone age of 9 years. Recently, an automated method to determine bone age has been developed.
Just like other auxological measures, bone ages have normal ranges, based on age and gender. The normal ranges define the normal variance between bone age and chronological age of a child. Discordance among skeletal centers, with maturity levels differing between bone groups, can be a normal variant. Because bone age assignment is such a subjective measure, it is prudent for pediatric endocrinologists to make their own readings rather than simply rely on radiologists’ reports. This will eliminate interreader variability between repeated studies, thereby improving the validity of longitudinally following bone age progression in a child.
The bone age mirrors the tempo of physical development of the child, particularly reflecting the progression of growth plate senescence, and indicates the remaining growth potential. Knee films can be used instead at very young ages when hand/wrist films are not yet informative because of developmentally appropriate lack of ossification. The bone age is also more closely associated than the chronological age with the timing of central puberty in some situations, for example, boys with congenital adrenal hyperplasia, familial male-limited precocious puberty, and idiopathic short stature. However, surprisingly, the bone age does not predict pubertal onset better than does chronological age in normal children.
Even before obtaining a bone age, a clinician can surmise a child’s rate of maturation by examining their teeth; the degree of dental maturation tends to correlate with skeletal maturation (i.e., delayed dentition can imply delayed bone age).
Skeletal maturation, like skeletal growth, is highly regulated by endocrine, nutritional, and genetic factors. In general, factors that positively regulate linear growth, such as thyroid hormone, GH, estrogen, and nutritional intake, also positively regulate skeletal maturation, whereas factors that negatively regulate growth, such as glucocorticoids in high concentration, also negatively regulate skeletal maturation.
Adult height predictions are used to try to determine which children are growing toward a normal adult height. The “normal” range for adult height can be defined by the outer centiles demarcated on the country’s growth chart, recognizing that the centiles selected for this purpose (e.g., 5 th and 95 th or 3 rd and 97 th ) are arbitrarily chosen.
Because height is a highly hereditable trait, a more specific target can be established from the heights of the child’s biological parents. It is important to measure the parents’ heights rather than rely on self-reports, which are often inaccurate. The two parents’ heights can be combined using the formulae for gender-adjusted midparental height ( Box 11.2 ), which adjust for the 13-cm (5-inch) difference between the mean adult male and female heights, and provide the midparental height ± 2 SD (1 SD would be about 5 cm, or 2 inches) as the “normal” adult height range for their offspring. However, the adjusted midparental height assumes that the child’s height is inherited from the parents in a polygenic fashion, with multiple inherited polymorphisms contributing to the child’s height. It does not apply to monogenic conditions. If, for example, one of the two parents is short because of a heterozygous mutation in SHOX and the other parent is of normal height, the offspring will tend to be either short, like the affected parent, or of normal stature, like the unaffected parent, not halfway in between. Even for polygenic inheritance, empirical data indicate that children of short parents tend not to be as short as their parents and tall children tend not to be as tall as their parents. For example, when the midparental height is –2 SD, the children’s heights tend to be closer to –1 SD.
The simplest approach to adult height prediction is to assume that the child’s height centile will remain fairly constant until adulthood. However, this method is not accurate and may fail if the child has an advanced or delayed bone age, a condition that affects growth, or an early or late timing of puberty.
There are three popular methods for predicting adult height that take skeletal maturation (i.e., bone age) into account, and all are gender specific. The most commonly used of the three methods, the Bayley-Pinneau prediction tables from 1952, uses longitudinal data derived from healthy US children living in Cleveland to estimate the fraction of growth remaining at each bone age (by the Greulich-Pyle method ). Adult height is predicted by combining the child’s bone age with the height measured at the time of the radiographic study. The prediction tables and the prediction error are provided in the Greulich and Pyle Atlas. The Greulich and Pyle bone age (read either manually or with an automated system) can also be used in a more sophisticated mathematical prediction model (currently available online), which appears to have a slightly greater accuracy than the Greulich and Pyle tables in children with idiopathic short stature. The TW method, derived from British children, uses TW standards for bone age assessment, in which each bone in the hand and wrist is assessed individually. In addition to the bone age and measured height, this method factors in chronological age, parental heights, and in girls, the occurrence of menarche. The third method for predicting adult height is the Roche-Wainer-Thissen (RWT) method. This method takes into account the weight or nutritional status of the child and uses recumbent length instead of standing height (calculated as 1.25 cm greater than standing height). The five predictor variables in the RWT method are recumbent length, weight, bone age, chronological age, and parental heights. An additional method of height prediction has more recently been developed that uses more sophisticated mathematical methods and may show better accuracy than previous methods. It is currently available online.
Unfortunately, even using bone age, height predictions are not highly accurate. The prediction error is considerable but does decrease with age. For example, using the Bailey-Pinneau tables for girls, the prediction can err for a 9-year-old child by approximately 7.4 cm or 3 inches (2 SD of measurement error), and by approximately 3 cm or 1.2 inches for a 13-year-old child. Furthermore, these prediction methods and the associated error estimates are based on data from healthy children. There may be systematic errors in the setting of illness or in idiopathic short stature. For example, these methods overpredict adult height in untreated precocious puberty, Turner syndrome, and primordial short stature. Sources of inaccuracy in adult height prediction include the inaccuracy of the bone age estimation itself, the inability to predict pubertal onset and tempo, and the difficulty taking into account effects of disease. A small difference in bone age determination can lead to a great difference in adult height prediction, especially during the pubertal growth spurt. A computer simulation, comparison study of three methods for adult height prediction—Bayley-Pinneau, RWT, and Khamis-Roche (calculates adult height without bone age from the child’s height, weight, and midparental height, using gender and age-specific coefficients) —found poor overall agreement among the three methods (κ = 0.21 for boys and negative for girls) in predicting adult height under the 1.2 nd percentile (the threshold for the US Food and Drug Administration [FDA]-approved indication of growth hormone treatment for idiopathic short stature). It is important for both clinicians and patient-families to understand the considerable limitations of height prediction.
Much of the normal variability of height between individuals is caused by a polygenic influence. Multiple genetic variants, both common and rare, each having a small or moderate effect on linear growth, combine to determine the individual’s height. However, short or tall stature, especially when severe or associated with other abnormalities, can also be primarily monogenic, that is, caused by a single variant that has a strong effect on linear growth. These monogenic causes may be identified because they follow classic Mendelian inheritance patterns, usually autosomal dominant or autosomal recessive and less often X-linked. Clinically, the inheritance pattern is investigated by eliciting a careful family history of the heights of the extended family members, which can be visualized with a written pedigree. For a more detailed discussion, see section on Diagnostic Approach to the Patient with Short Stature later in this chapter. When the pedigree suggests a polygenic inheritance pattern, the clinician can calculate the adjusted midparental height, which provides an estimate of the likely adult height of the child, based on the heights of the two parents. This topic is discussed in more detail in a previous section of this chapter: Clinical Evaluation of Growth—Prediction of Adult Height
Many disorders affect childhood growth, particularly decreasing growth to cause short stature. Many of the major causes are discussed later. However, the list is not exhaustive. An extensive, organized list can also be found within the International Classification of Pediatric Endocrine Diagnoses (ICPED, http://www.icped.org/ ).
Short stature results from decreased growth plate chondrogenesis. The decreased chondrogenesis can be caused by factors intrinsic to the growth plate (primary growth impairment) or can be secondary to factors elsewhere in the body that secondarily affect growth plate chondrocytes (secondary growth impairment) ( Fig. 11.4 ).
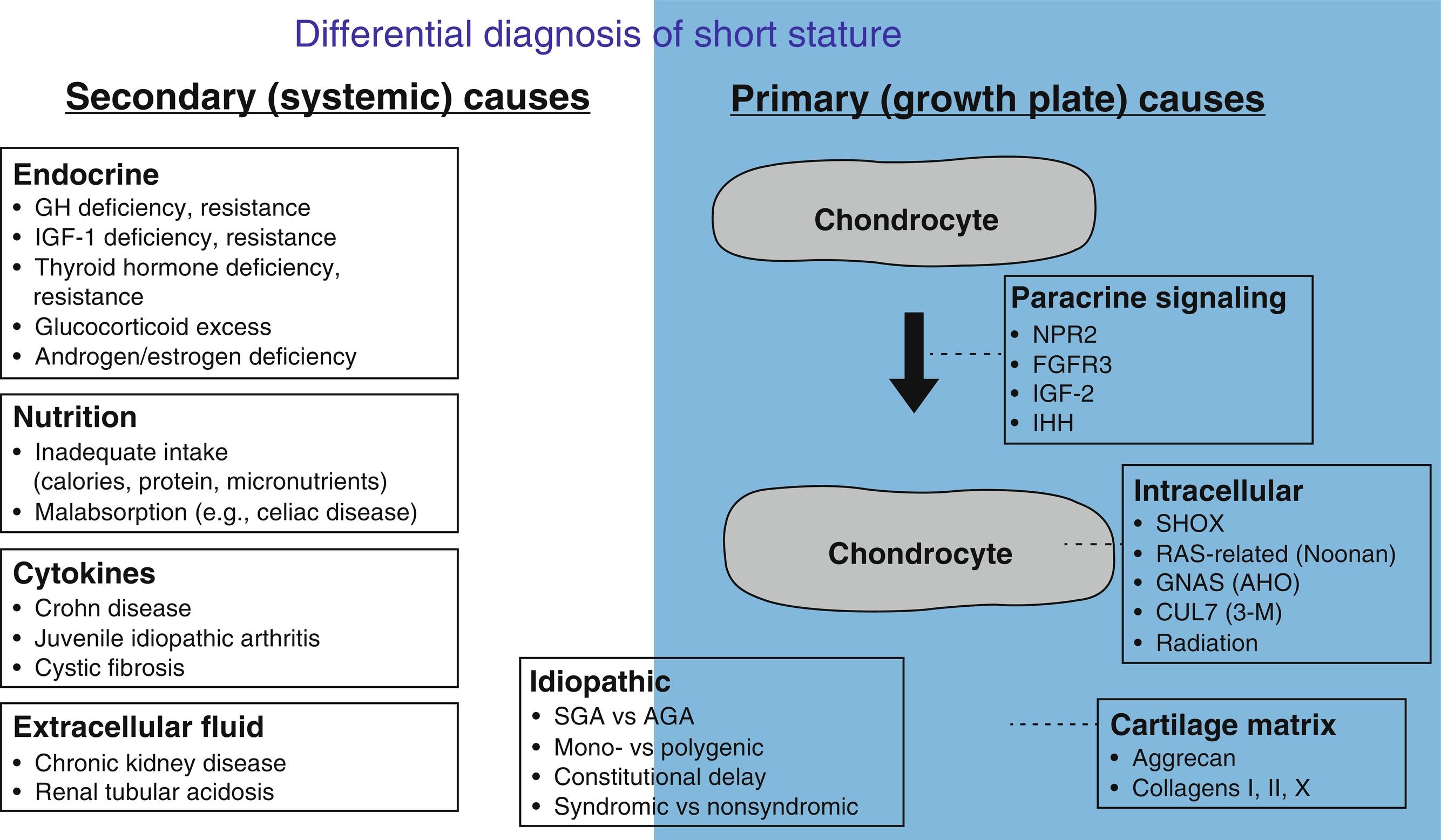
Primary growth plate impairment can involve abnormalities in paracrine signaling, cartilage matrix, or intracellular factors. In some conditions, the defect in growth plate chondrogenesis causes bones to be not only short but also malformed, in which case the condition is called a skeletal dysplasia or, more precisely, a chondrodysplasia , for example, ACH. Often, genetic defects that affect the growth plate also affect the development of other tissues, leading to a syndrome that includes not only short stature but also other congenital anomalies and organ dysfunction, for example, NS or pseudohypoparathyroidism 1A.
Secondary growth impairment can involve nutritional deficiencies, endocrine abnormalities, inflammatory disorders, and extracellular fluid abnormalities. Often these disorders are caused by a defect in some organ system other than the skeleton, such as the kidneys, pituitary, thyroid, or immune system. These disorders occurring elsewhere in the body then lead to an abnormal concentration of some molecules that are needed for normal growth plate chondrocyte function. These molecules can include steroid or polypeptide hormones, proinflammatory cytokines, or small molecules, such as phosphate or hydrogen ion.
Both primary and secondary growth impairment can be monogenic, oligogenic, polygenic, or nongenetic. Some conditions affect fetal growth, and thus the child is born small for gestational age. Other conditions affect only postnatal growth so that the child is born at a size appropriate for gestation age, and the short stature develops subsequently. Yet other conditions affect both prenatal and postnatal growth.
Adequate nutrition is required to support optimal statural growth, and there is often a lag between weight deceleration/acceleration and height deceleration/acceleration. Malnutrition can result from insufficient dietary intake, malabsorption or some other gastrointestinal process, or a combination of the two.
Malnutrition is the most common cause of poor growth globally, with insufficient dietary intake (total calorie and/or protein-calorie malnutrition) often related to food insecurity and poverty. Malnutrition is also common in higher socioeconomic communities, when energy demands (e.g., increased because of illness, sports or activities) are not met by intake (e.g., because of unstructured meals, picky eating, fear of obesity). Decreased nutrient intake can result from oropharyngeal malformations (e.g., Pierre Robin sequence, cleft lip/palate), abnormal oral-motor function (e.g., pervasive developmental delay), as well as loss of appetite because of certain medications (e.g., stimulants for treatment of attention deficit hyperactivity disorder or chemotherapeutic agents). Eating disorders (e.g., anorexia nervosa) involve disturbed eating behaviors related to an altered body image or food avoidance because of phobias or sensory aspects of food. Malabsorption can also cause malnutrition, and disorders such as celiac disease, IBD, or cystic fibrosis can present as isolated growth failure. Bone age and puberty are often delayed.
In addition to malnutrition, deficiency of certain micronutrients can impair statural growth. The frequent occurrence of concurrent deficiencies complicates the research and literature on any isolated micronutrient. Although iron deficiency is often found in children with nutritional growth stunting and should be corrected, multiple metaanalyses and systematic reviews of randomized controlled intervention trials of iron supplementation showed that iron therapy had no significant effect on growth in iron-replete children. In contrast, human growth retardation (and male hypogonadism) from zinc deficiency was first reported by Prasad and colleagues in 1963, and several, but not all, systematic reviews show height and/or weight increase from zinc supplementation in children. Zinc ion is required for activity of over 300 enzymes (zinc metalloenzymes), including DNA polymerase, ribonucleic acid (RNA) polymerase and thymidine kinase, which are important for nucleic acid and protein synthesis and cell division. Bone contains large amounts of zinc where it plays an important role in skeletal development, and zinc deficiency reduces IGF-1 production, cellular IGF-1 responsiveness, and growth response to GH treatment of children who are GH deficient.
Careful assessment of weight and height progression will identify children who are not gaining weight and growing appropriately. Taking a detailed dietary history is essential to establishing this diagnosis, such as a 3-day diet diary for analysis of any macro- or micronutrient deficits. Treatment is, of course, nutritional repletion, which can involve patient-family education, behavior modification, dietary supplements and in severe cases, tube feedings, as well as treatment of any underlying gastrointestinal issues. However, it is important to have in mind, that several conditions that cause short stature are associated with low BMI (or have low BMI as a component of their phenotype), such as Silver-Russell syndrome (SRS), NS, and Bloom syndrome.
Disorders of GH/IGF axis comprise a large and heterogeneous group of conditions with distinct phenotypes ( Table 11.3 ). Clinical and hormonal evaluations are usually the cornerstones of diagnoses of these disorders.
| Hormone Measurement | |||||||
|---|---|---|---|---|---|---|---|
| Gene | Inheritance | Growth Pattern | GH | IGF-1 | IGFBP-3 | Additional Features | |
| GH deficiency | multiple | AR/AD | varying severity/postnatal | ↓ to ↓↓↓ | ↓ to ↓↓↓ | ↓ to ↓↓↓ | Midline defects, deficiency of other pituitary hormones |
| Bioinactive GH | GH1 | AR/AD | varying severity/postnatal | ↑↑ | ↓↓ | ↓↓ | Growth and IGF-1 improve after exogenous GH |
| GH insensitivity (GHI) | GHR | AR > AD | severe/postnatal | Nl to ↑↑ | ↓ to ↓↓↓ | ↓ to ↓↓↓ | Growth and IGF-1 do not improve after exogenous GH. Low GHBP in 70% of cases |
| GHI associated with immune dysfunction | STAT5b | AR > AD | severe/postnatal | ↑↑ | ↓↓ | ↓↓ | GHI phenotype with immune dysfunction and elevated PRL |
| Ternary complex defects | IGFALS | AR | mild/postnatal | Nl to ↑ | ↓↓ | ↓↓↓ | Laboratory abnormality disproportionate to mild short stature, (IGFBP-3 lower than IGF1 concentrations) |
| PAPPA2 | AR | varying severity | ↑↑ | ↑↑↑ | ↑↑↑ | Mild microcephaly. Low free IGF-1 a | |
| IGF1 defects | IGF1 | AR/AD | severe/pre- and postnatal | ↑↑ | ↓↓↓↓ | Nl or ↑ | Microcephaly, sensorineural deafness, developmental delay, intellectual disability |
| IGF2 defects | IGF2 | ADp | severe/pre- and postnatal | Nl or ↑ | Nl or ↑ | Nl or ↑ | Phenotype resembling Silver Russell syndrome and low IGF-2 concentrations a |
| Bioinactive IGF-1 | IGF1 | AR | severe/pre- and postnatal | ↑↑ | ↑↑↑↑ | Nl | Microcephaly, sensorineural deafness, developmental delay, intellectual disability, insulin resistance a |
| IGF insensitivity | IGF1R | AD > AR | varying severity/pre- and postnatal | Nl to ↑↑ | Nl to ↑↑ | Nl to ↑↑ | Variable clinical findings in the dominant forms. Microcephaly, developmental delay and intellectual disability may be present |
GH deficiency is a rare disorder with a prevalence of approximately 1 in 4000 during childhood. GH deficiency may be isolated (IGHD) or combined with other hormone deficiencies, when it is known as combined or multiple pituitary hormone deficiency (CPHD/MPHD). The condition may be congenital or acquired ( Table 11.4 ). Early diagnosis and treatment is critical to ensure optimal final height outcomes. The diagnosis of GH deficiency is based on a combination of auxology, exclusion of other pathology leading to short stature, biochemical investigation of the GH-IGF-1 axis, and imaging of the hypothalamopituitary area. More recently, a molecular diagnosis may be sought in those with “idiopathic” GH deficiency or MPHD (see Table 11.1 ); however, the majority of patients with IGH deficiency or with CPHD remain to be explained in terms of etiology. Before evaluation of the GH-IGF-1 axis, other diagnoses, such as familial short stature, hypothyroidism, Turner syndrome, celiac disease, chronic illness, such as Crohn disease or renal failure, and skeletal dysplasias should be excluded. Consensus guidelines on the diagnosis of GH deficiency in childhood were published in 2000, ( https://doi.org/10.1210/jcem.85.11.6984 ) and suggest investigation into the following scenarios:
Severe short stature, defined as a height more than 3 SD below the mean.
Height more than 1.5 SD below the midparental height.
Height more than 2 SD below the mean and a height velocity over 1 year more than 1 SD below the mean for chronological age, or a decrease in height SD of more than 0.5 over 1 year in children over the age of 2 years.
In the absence of short stature, a height velocity more than 2 SD below the mean over 1 year or more than –1.5 SD sustained over 2 years, which may occur in GH deficiency presenting in infancy or in organic acquired GH deficiency.
Signs indicative of an intracranial lesion.
Signs of MPHD.
Neonatal symptoms and signs of GH deficiency.
| 1. Congenital |
|
|
|
|
|
|
|
|
|
|
|
|
| 2. Acquired Causes |
|
|
|
|
|
|
|
|
|
|
The presentation in the neonatal period can include hypoglycemia, prolonged conjugated hyperbilirubinemia (usually with associated ACTH deficiency), and micropenis. Birth size is typically within the normal range, although there may be a reduction of approximately 10%, which occurs typically late in pregnancy. Growth velocity is reduced during the first year of life in children with severe GH deficiency with growth failure manifesting by 6 months of age, but the phenotype evolves after 1 year of age in those with milder GH deficiency. The earliest manifestations are a reduction in height velocity followed by a reduction in height SDS adjusted for mean parental height SDS. The child’s height SDS will ultimately fall below –2 SD, the time taken depending on the severity and duration of GH deficiency. A child with severe GH deficiency often has midface hypoplasia, hypotonia, a high-pitched voice, immature appearance, delayed dentition, thin sparse hair, slow nail growth, and truncal adiposity. GH deficiency is also associated with effects on cognition. In mice with GH deficiency, reduced spatial learning and memory has been reported whereas in untreated children with GH deficiency IQ, verbal comprehension and processing speed are reduced. In children and adults, the neurocognitive defects in GH deficiency are improved by GH therapy. With prompt recombinant human GH (rhGH) treatment, the outlook for final height is excellent. Other hormone deficiencies may require treatment; in particular those with structural abnormalities of the hypothalamopituitary region are more likely to evolve and develop other hormone deficiencies.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here