Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Multiple malformation syndromes may be identifiable by ultrasound following the identification of one abnormality and associated investigations.
There are thousands of malformation syndromes, many too rare for the scope of this book and many not amenable to ultrasound identification. However, identification of any malformation should immediately alert the sonographer to look for further abnormalities. This chapter will be a guide for possible syndrome diagnosis following the initial identification of an abnormality.
A number of abnormalities may be identified which will lead to a specific examination for other features. The individual abnormalities will be discussed in other chapters and in this chapter there will be particular reference to possible malformation syndromes.
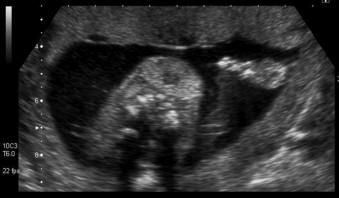
Fetal hydrops is defined as the presence of excess fluid in more than one body cavity, a combination of two or more from skin oedema, pleural effusions, pericardial effusion and ascites ( Figures 8-1 to 8-3 ).
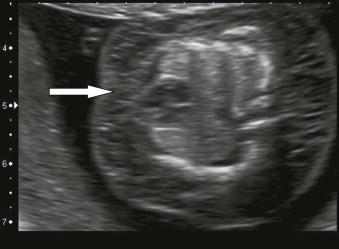
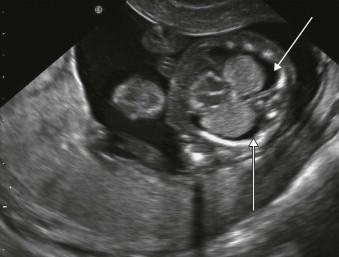
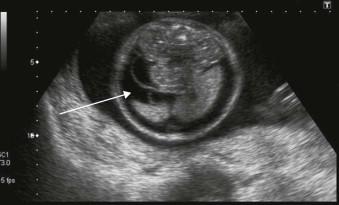
Hydrops requires an imbalance between intravascular and extravascular fluid. The driving force out of the vascular space is the difference between the capillary hydrostatic pressure and the colloid oncotic pressure of interstitial fluid. Colloid oncotic pressure depends on both the concentration of osmotically active molecules and the efficacy with which differences in oncotic pressure can cause water shifts. The fetal capillaries are more permeable, thus colloid oncotic pressure is less of a driver than after birth and fetal tissues are more compliant resulting in an increased loss of intravascular fluids before a rise in interstitial colloid oncotic pressure checks the flux.
| Abnormality Group | % Cases |
|---|---|
| Chromosomal causes | 63 |
| Infection | 7.3 |
| Cardiovascular | 2.1 |
| Fetal akinesia | 8.3 |
| Multiple malformation | 2.1 |
| Other anatomical defect | 3.1 |
| Haematological cause | 1.1 |
| Unknown cause | 10.3 |
| Cause | <24–28 weeks' gestation | >24–28 weeks' gestation |
|---|---|---|
| Chromosomal | 34.5% | 5.4% |
| Cardiovascular | 9.0% | 24.2% |
| Genetic or syndromal | 8.8% | 8.2% |
| Infective | 4.8% | 5.8% |
| Pulmonary | 7.4% | 13.2% |
| Unknown | 15.0% | 22.1% |
| High Output Cardiac Failure | 1° Cardiac Causes | Non-Cardiac Causes |
|---|---|---|
| Fetal anaemia | Supraventricular tachycardia (SVT) | Infection |
| Fetal thyrotoxicosis | Hypertrophic cardiomyopathy | Metabolic syndromes |
| Twin-to-twin transfusion | Dilated cardiomyopathy | Lung malformations |
| Arteriovenous malformation | Structural cardiac abnormalities | Chromosomal |
| Fetal/placental tumours | Fetal bradycardia | Single gene disorders |
| Lymphatic abnormalites | Fetal akinesia | |
| Musculoskeletal |
Lymph flow is increased in the fetus but delayed development of the lymphatics will prevent this. Clearance of lymph fluid is dependent on the outflow pressure in the great veins of the neck. If the negative pressure is not maintained, lymph flow is reduced, with the consequence of development of interstitial fluid and hydrops. Hence a rise in central venous pressure (CVP) is a final common pathway for many causes of fetal hydrops ( Figure 8-4 ).
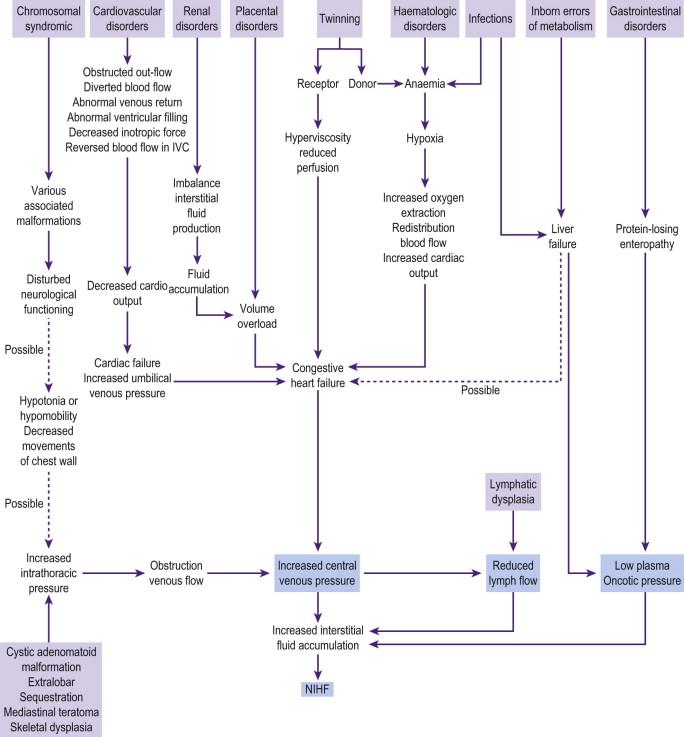
Fetal anaemia raises the CVP secondary to high cardiac output, and anoxia due to anaemia leads to increased capillary permeability.
Accumulation of osmotically active lactic acid may underlie some metabolic causes of hydrops ( Figure 8-5 ).
The presence of fetal hydrops in the first trimester is a sign of impending demise and the survival rate is practically 0%. Major chromosomal abnormalities including 45X will account for the majority of cases. Identification of major chromosomal abnormalities by first-trimester screening with raised nuchal translucency and abnormal serum biochemistry has reduced the underlying chromosomal burden of hydrops, as many of these high-risk fetuses will have aborted prior to the development of hydrops.
Raised nuchal translucency may proceed to fetal hydrops in later pregnancy. There may initially appear to be an improvement followed by hydrops either identified at the 20-week scan or later. Persistence of the nuchal fold without the development of fetal hydrops would raise concerns that the baby has Noonan or a Noonan-related syndrome.
Fetal hydrops was historically divided into immune and non-immune hydrops, as the commonest underlying cause was fetal anaemia secondary to Rhesus incompatability. Since the introduction of anti-D this complication has become much rarer, and although other blood group antigens can cause red blood cell incompatability, it is now an uncommon cause of fetal anaemia.
Fetal anaemia remains a significant cause of hydrops but the underlying causes of the anaemia are different ( Table 8-4 ).
| Blood Group Incompatibility (Haemolytic) | Rhesus, ABO, Kell |
| Aplastic anaemia | Diamond-Blackfan syndrome |
| Inherited anaemia | α Thalassaemia |
| Red cell enzyme disorders | Pyruvate kinase deficiency, glucose 6 phosphate isomerase deficiency |
| Red cell membrane defects | Platelet abnormality, platelet antibodies |
| Fetal haemorrhage | Haemophilia |
| Feto-maternal haemorrhage | |
| Twin–twin transfusion | |
| Infection (aplastic) | Parvovirus, CMV |
Specific features on ultrasound may give an indication as to the underlying cause of the fetal hydrops. There are other signs that are less specific but may help with directing further investigations, these include:
Increased peak systolic velocity (PSV) in the middle cerebral artery Doppler (MCAD) → anaemia ( Figure 8-6 ).
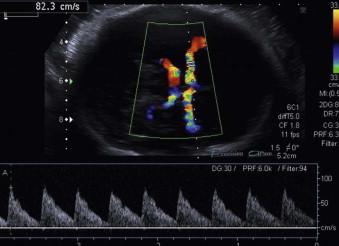
Pericardial effusion and ascites → anaemia.
Hepatosplenomegaly → metabolic syndrome, infection.
A fetal bleed should be suspected if a heterogeneous mass lesion is identified on ultrasound: an intracranial haemorrhage is the most common manifestation of a fetal bleed ( Figure 8-7 ).
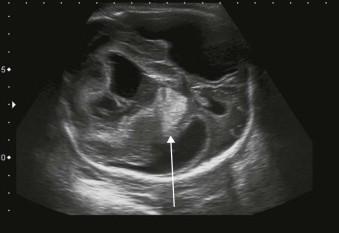
Fetal thrombocytopenia secondary to alloimmune thrombocytopenia is a recognized cause of fetal intracranial haemorrhage. This is due primarily to antibodies against the platelet antigen HPA1a. It occurs in 80% of cases in the first pregnancy and may present before 20 weeks. Intravenous immunoglobulin (IVIG) may help to reduce its severity and the risk of intraventricular bleed. The risk of recurrence is approximately 80% (see Chapter 19 , Haematological Disorders).
Thrombocytopenia can also be caused by haemophagocytic lymphohistiocytosis, a rare autosomal recessive disorder.
Other causes of fetal haemorrhage include fetal haemophilia. Factor VIII and factor IX deficiency are the commonest haemophilia syndromes but they do not cause fetal bleeds; however factor V and factor X deficiencies can present antenatally. Clotting studies antenatally remain unsatisfactory as the blood volume required for such analysis is not possible at present from a live fetus.
Mutations in Col4A1 and Col4A2 have recently been identified as genetic causes of porencephalic cysts. These mutations may cause fetal anaemia as a consequence of haemorrhage, although the more usual presentation is as ultrasound-detected intra-cranial abnormalities in the third trimester.
Aplastic anaemia secondary to Diamond-Blackfan syndrome is a rare disorder which is inherited as an autosomal dominant disorder in 10–25% of cases. It is secondary to mutations in a number of ribosomal proteins. Many affected patients are steroid-dependent, but can maintain a haemoglobin count of around 7 mg/100 mL. No prenatal treatment with steroids has been attempted. A number of cases will arise as de novo mutations, so the absence of a family history will not exclude this diagnosis. Fetal infection with cytomegalovirus (CMV) and parvovirus may cause aplastic anaemia. Identification of parvovirus infection is particularly important as fetal blood transfusion may be required for survival of the fetus prior to spontaneous recovery. Fetal hydrops secondary to CMV is multifactorial.
Haemolytic anaemia is caused by a defect in a number of red cell enzymes but these rarely cause a severe enough anaemia antenatally to cause hydrops. Mutations in all four of the α-thalassaemia genes will cause fetal hydrops from the lack of production of the α chains in haemoglobin.This is incompatible with life and will always cause fetal demise. In this situation the mother may become very unwell due to mirror syndrome.
Fetal leukaemia may cause hydrops and in Down syndrome there may be a transient myeloproliferative disorder, however the anaemia may be so severe as to cause still birth ( Figure 8-8 ).
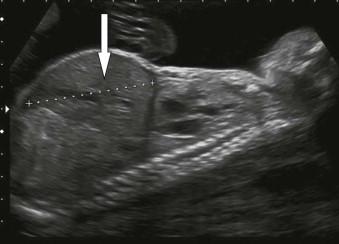
Fetal thyrotoxicosis may cause fetal hydrops though this is rare. Fetal thyrotoxicosis is normally secondary to maternal Graves' disease and is autoimmune in aetiology. Ultrasound findings may include fetal goitre, tachycardia and growth restriction. Fetal hypothyroidism may occur due to over-treatment of maternal thyrotoxicosis or very rarely primary hypothyroidism in the baby. A fetal goitre can be identified on ultrasound and fetal thryroid function can be assessed on cordocentesis ( Figure 8-9 ).
See Chapter 9 , Assessment of Twin Gestation.
AV malformations such as a Vein of Galen malformation have a very poor prognosis, not only because they cause high-output cardiac failure secondary to shunting of blood, but they may also cause bleeding and hydrocephalus secondary to a mass effect. Vein of Galen malformations have been associated with capillary malformation-arteriovenous malformation disorder which is an autosomal dominant disorder secondary to RASA1 mutations. This disorder can be highly variable within a family. Examination of both parents for the presence of haemangiomas may suggest a diagnosis of a capillary haemangioma syndrome.
Chorioangiomas of the placenta can cause fetal hydrops which may be severe and it may be necessary to occlude the feeding vessels of the tumour by laser ablation if hydrops presents pre-viability. The aetiology of tumour-induced hydrops is multifactorial: from the high-output cardiac failure caused by anaemia and/or arteriovenous shunting in sacrococcygeal tumours to the vascular compression hydrops of pharyngeal teratomas.
Fetal bradycardia can be caused by structural cardiac lesions, particularly left isomerism ( Figure 8-10 ).
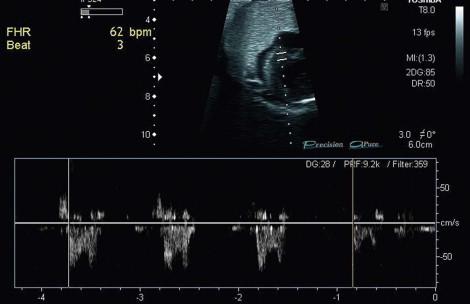
More commonly however fetal bradycardia is related to complete heart block resulting from the destruction of the cardiac conduction system by maternal antibodies. Anti-Ro and anti-La antibodies have been particularly implicated and a maternal history of systemic lupus erythematosus (SLE) should be sought.
Channelopathies can rarely cause fetal hydrops. Autosomal recessive long QT syndrome (Jervell–Langer–Nielson syndrome) causes severe prolongation of the QT interval and sensorineual deafness. Long QT syndrome is normally an autosomal dominant disorder; 12 causative genes have been identified to date and there are others still to be discovered. Long QT 1-3 account for about 60% of all cases of long QT syndrome. Late-onset fetal bradycardias have been noted, but rarely cause fetal hydrops. The fetal bradycardia may be first noted in labour, however, and be confused with fetal distress. Catecholaminergic polymorphic ventricular tachycardia (CPVT) may also present antenatally with fetal bradycardia, and although documented in a case report of fetal hydrops, it is uncertain if this was the primary cause. Postnatal investigation needs to be undertaken in all cases as complete heart block may lead to sudden death. CPVT is a highly dangerous condition with 30% chance of sudden death by the age of 40 years. The resting ECG and 24 hour tape are normal and it requires an exercise ECG to reveal rhythm disturbances, which is not possible until approximately the 4th year of life.
Investigation of a fetal bradycardia should include:
A fetal echocardiogram looking for evidence of congenital heart disease including left isomerism.
Maternal blood for anti-Rho antibodies and anti-nuclear antibody (ANA).
Parental ECGs should be performed at some stage looking for evidence of long QT syndrome as well as asking for a history of fainting and sudden death within the family.
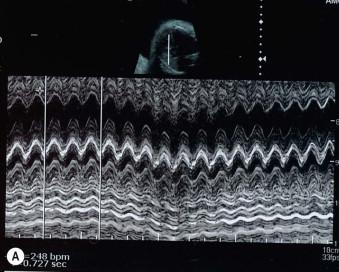
These are supraventricular in origin in the majority of cases with re-entry pathways the major cause of sustained fetal tachycardia ( Figure 8-11A and B ). Sustained fetal tachycardias are much more likely to cause fetal hydrops than intermittent tachycardias, but hydrops has been described in the latter condition. An intermittent tachycardia may well be missed and require recurrent examinations prior to being identified. It should be considered in any baby with unexplained hydrops in the third trimester. Treatment is usually started with digoxin and or flecanide. Treatment of the baby needs to continue for the first few weeks of life until there has been adequate maturation of the conduction system.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here