Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Seizures occur during the neonatal period in approximately 2 to 4 per 1000 live births. Seizures are a neurologic emergency: they are often caused by acute brain injury or treatable underlying conditions and rapid, appropriate treatment can improve treatment success. , Seizure management includes rapid and thorough diagnostic evaluation, as seizures are frequently the presenting symptom for electrolyte abnormalities, infection, hypoxic-ischemic injury, and intracranial hemorrhage. Seizures in this age group have unique features requiring an age-appropriate approach to classification and diagnosis using an electroencephalogram (EEG). An appreciation for the multifactorial effects of antiseizure medications (ASMs) and seizure burden on the rapidly developing brain is essential.
This chapter will focus on seizure diagnosis and classification in the neonatal population, diagnostic, treatment, and prognostic considerations in confirmed neonatal seizures, and the ongoing controversies associated with each of these topics.
EEG is an essential tool for seizure diagnosis in neonates. Neonates often exhibit paroxysmal movements, which can be difficult for even skilled providers to distinguish from seizures clinically. When experienced neonatal healthcare providers evaluated standardized videos of seizures and seizure mimics, they identified seizures correctly only 50% of the time. Furthermore, prospective cohort studies have demonstrated that up to 80% of neonatal seizure burden may be electrographic only. , Significant underdiagnosis occurs if seizures are diagnosed using only clinical observation. Electrographic seizures can persist when clinical manifestations resolve, even when clinical neonatal seizures are appropriately identified and treated. This phenomenon has been termed electroclinical uncoupling and is estimated to occur in up to 25% of neonates. Recent evidence suggests that early identification and treatment of electrographic seizures may increase the odds of successful seizure treatment. ,
In 2011, the American Clinical Neurophysiology Society developed guidelines for monitoring and diagnosing neonates at risk for seizures. These guidelines recommend long-term conventional video EEG monitoring for multiple neonatal populations, including neonates with unexplained paroxysmal events, encephalopathy, central nervous system infection or trauma, suspected perinatal stroke or hemorrhage, suspected inborn errors of metabolism or genetic epilepsies, and children who require cardiopulmonary bypass.
Historically, a seizure in neonates has been defined as sudden, repetitive, rhythmic, evolving EEG activity with a minimum of 2 μV peak-to-peak voltage that lasts a minimum of 10 seconds. More recently, the International League Against Epilepsy (ILAE) expanded the definition to include shorter events if the duration is sufficient to demonstrate clear onset, evolution of the frequency and morphology, and resolution of the abnormal discharge. Discharges without clear evolution of the rhythmic activity are considered to be brief rhythmic discharges (BRDs). BRDs are thought to confer an increased seizure risk. The traditional definition of status epilepticus in older children and adults, which includes a failure to return to baseline cognitive function, is difficult to apply to neonates. Neonatal status epilepticus has instead been operationally defined as seizures lasting greater than 50% of any 1-hour epoch of EEG recording.
The American Clinical Neurophysiology Society guidelines recommend conventional video EEG (cEEG) as the gold standard for seizure diagnosis in neonates. Amplitude-integrated EEG (aEEG) is a simplified brain-monitoring tool that has been widely adopted in many units for ease of bedside monitoring.
In neonates, the International 10–20 system for EEG is simplified to approximately 20 scalp electrodes accompanied by an electrocardiogram lead and respiratory belt. Interpretation requires specialized education to learn preterm and term neonates’ normal patterns in wakefulness and sleep and the appearance of important pathologies, including seizures. EEG technologists and bedside nursing staff are essential to help identify the plethora of potential artifacts commonly observed on cEEG recordings.
aEEG is a widely available bedside tool that is easy to apply and interpret at the bedside. It uses a limited montage (usually 1–3 channels to cover the frontal and central or parietal regions). The maximum and minimum amplitude recorded during a specified time epoch are plotted on a compressed time scale and semilogarithmic y -axis for amplitude to display a trend tracing for each electrode pair. A seizure is suspected when a sudden and sustained increase in amplitude is noted on both upper and lower margins of the aEEG tracing. A concurrent review of the raw EEG signal is helpful to confirm an evolving, monomorphic waveform consistent with seizure. Limitations of aEEG include low sensitivity to detect low amplitude, brief, or very focal seizures occurring in an area not directly adjacent to the electrodes. In 125 neonates greater than 34 weeks postmenstrual age who were evaluated with concurrent cEEG and aEEG, neonatologists were able to use aEEG to identify between 12% and 38% of the individual seizures seen on cEEG, and overall aEEG use identified between 22% and 57% of neonates with seizures.
Common EEG and aEEG artifacts include patting, sucking, intravascular line drips, ventilation artifact, and extracorporeal membrane oxygenation artifact. , Artifacts may be more difficult to distinguish from seizures on aEEG due to limited channels, lack of video, and absent technologist input to resolve electrical artifacts. Prospective cohort studies have found dramatically differing rates of events concerning for seizures depending on the type of neurophysiology monitoring used, particularly in the preterm neonatal population. In two similar prospective cohorts of preterm neonates, 48% of children had seizures when aEEG was used for diagnosis compared with 5% of children when gold standard cEEG was used for diagnosis. Such divergent results suggest that, while aEEG may serve as an excellent screening tool (especially in resource-limited settings), gold-standard cEEG is necessary for optimal seizure management. ,
For most monitoring systems, aEEG can be displayed at the bedside (using the leads placed for the full montage recording) while the cEEG is recorded and made available for remote access by the neurophysiologist. This approach has the advantage of providing both a bedside tool for immediate use by the neonatology team and the gold-standard recording for definitive seizure identification. Furthermore, using the same machine and recording for both the aEEG and traditional EEG display can facilitate communication between teams as annotations regarding clinical condition or medication administration by the bedside team can be communicated easily with the neurophysiologist and vice versa.
Quantitative EEG (qEEG) has been increasingly used to predict and aid in identifying neonatal seizures. qEEG analysis, including relative and absolute spectral analysis, has been used to evaluate neonatal EEG seizure risk in neonates with encephalopathy. Ongoing work is focused on translating qualitative use of EEG background to predict seizure risk , into quantified and more easily applied metrics.
Seizure detection algorithms developed in adults have low accuracy for detecting seizures in neonates. Recent efforts to develop neonatal-specific seizure detection algorithms have demonstrated decreased time to treatment and seizure burden when partnered with physician review. Initial neonatal qEEG development has focused on term and near-term neonates, but efforts are underway to develop qEEG techniques specific for preterm neonates.
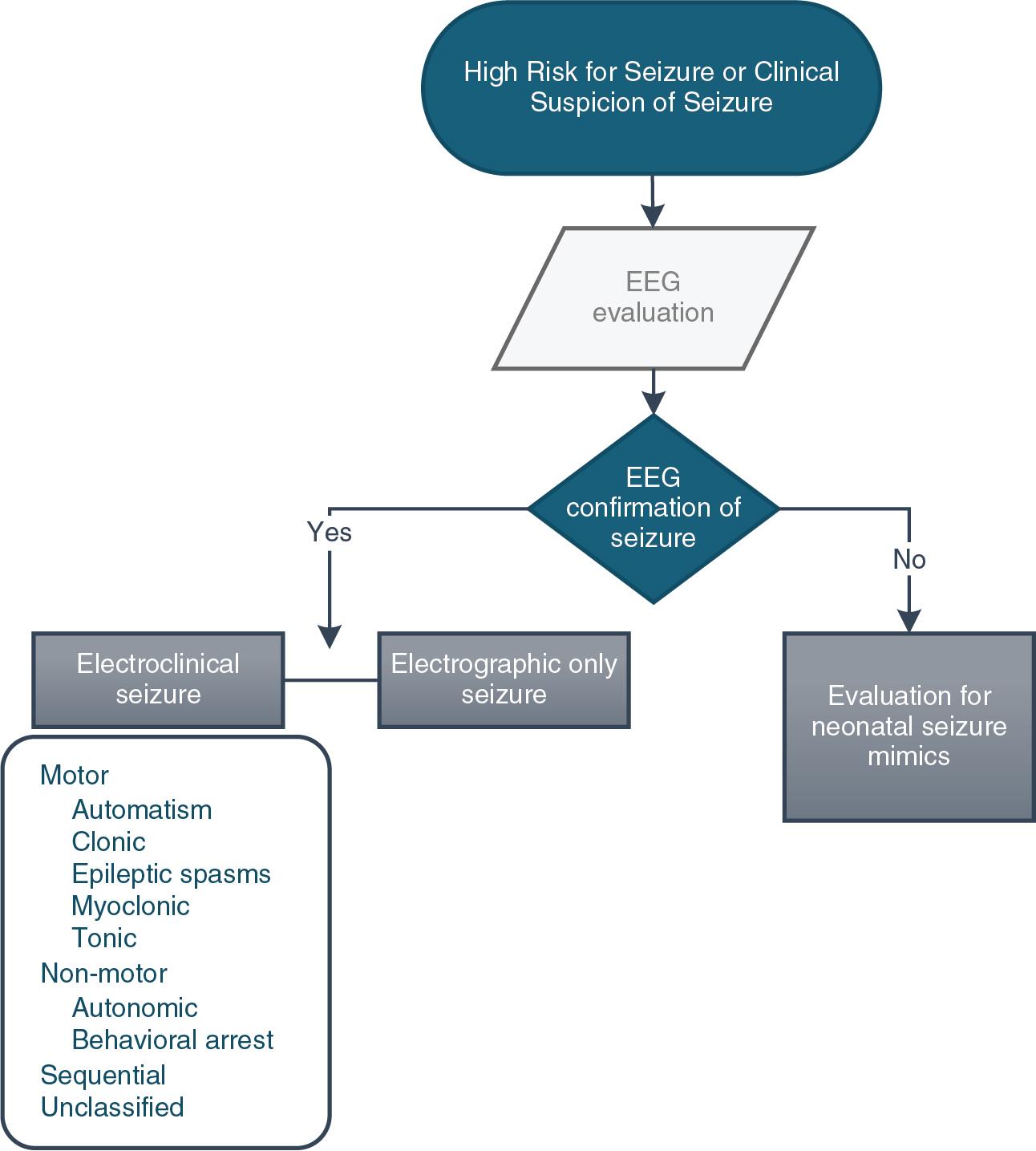
There have been multiple historical classification systems for neonatal seizures, which typically do not fit neatly into the seizure classifications used for older children or adults. In 2021, the ILAE published an updated framework to classify neonatal seizures ( Fig. 9.1 ). The updated classification system emphasizes the need to incorporate EEG evaluation and explicitly recognizes electrographic-only seizures due to their high frequency and importance in neonates. , , Generalized seizures are not included in the classification as seizure onset is always focal in neonates. Electroclinical seizures are categorized as motor seizures, nonmotor seizures (autonomic changes or behavioral arrest), sequential seizures, or unclassified ( Table 9.1 ).
| Seizure Type | Characteristics | Nonepileptic Mimics |
|---|---|---|
| Electrographic only |
|
|
| Motor: automatisms |
|
|
| Motor: clonic |
|
|
| Motor: epileptic spasms |
|
|
| Motor: myoclonic |
|
|
| Motor: tonic |
|
|
| Nonmotor: autonomic |
|
|
| Nonmotor: behavioral arrest |
|
|
| Sequential |
|
|
Neonatal seizures with automatisms usually consist of oral-lingual-buccal movements and are frequently accompanied by alternations of consciousness. They may be associated with fluctuations in blood pressure, heart rate, and oxygen saturation but are often clinically subtle unless accompanied by other seizure types.
Clonic seizures consist of rhythmic movements, which are nonsuppressible and unaltered by repositioning. Focal clonic seizures are the most consistently correctly identified type of neonatal seizure and are often associated with injury localized to a specific site, such as a perinatal stroke or other cerebrovascular event. Multifocal clonic seizures are more common in neonates with multifocal or generalized brain injury such as hypoxic-ischemic encephalopathy. Clonic seizures in a neonate may be mistaken for nonepileptic tremor, jitteriness, or clonus.
Myoclonic movements are extremely rapid (<100 msec) jerks of one or multiple limbs. Myoclonus can represent seizure or originate at more distal regions of the central nervous system, including the brainstem and spinal cord. Myoclonic seizures are most commonly seen in neonatal-onset genetic epilepsies and inborn errors of metabolism. ,
Epileptic spasms in neonates are rare. Their clinical presentation is similar to older-onset infantile spasms (which typically present around 6 months of age), with sudden flexion or extension of the proximal and truncal muscles. Spasms are longer than myoclonic movements but briefer than tonic seizures and usually occur in clusters. Subtle forms can consist of clusters of abnormal eye movements or head nodding. This rare seizure type is classically associated with inborn errors of metabolism.
Tonic seizures are marked by sustained flexion or extension lasting seconds to minutes. Tonic seizures can include neck or head version, forced eye deviation, or trunk and limb involvement. Neonatal tonic seizures are highly variable, including focal, unilateral, or bilateral asymmetric, and are typically associated with early infantile-onset epileptic encephalopathies. , , ,
Autonomic seizures are paroxysmal alterations in cardiovascular, respiratory, vasomotor, or pupillary function with EEG epileptic correlate. Isolated autonomic abnormalities are rarely caused by seizure; less than 5% of studies obtained for isolated apnea have an ictal correlate. Autonomic seizures are usually associated with intraventricular hemorrhage, temporal lobe or occipital lesions, and occasionally with early-onset epileptic encephalopathies. , , ,
Ictal behavioral arrest is rare in isolation. More commonly, a behavioral arrest is part of a sequential seizure. ,
Neonatal seizures are sequential when the ictal pattern progresses through a series of phases associated with EEG changes. Within the individual ictal event, the lateralization and composition of ictal features may vary. This type of seizure is associated with genetic epilepsies, particularly KCNQ2 encephalopathy. ,
Neonates have a wide array of paroxysmal movements, which can be easily mistaken for seizures. The most common mimics include tremulousness, nonepileptic myoclonus, dystonic movements, and more rarely hyperekplexia.
Tremor and jitteriness can occur in both ill and healthy neonates. The movements are characterized by rhythmic oscillation of varying amplitudes and stereotypy. Tremor may be asymmetric and either spontaneous or induced by stimulation or movement. Tremor and jitteriness may be differentiated from seizure by suppression with flexion, restraint, or repositioning. Tremor and jitteriness can occur in neonates with hypothermia, or secondary to metabolic derangements, particularly hypoglycemia and hypocalcemia. Tremor and jitteriness are also seen in neonates exposed to some maternal medications classes, including selective serotonin reuptake inhibitors (SSRI), cocaine, and marijuana.
Nonepileptic myoclonus, like myoclonic seizures, is characterized by rapid (<100 msec) jerks of one or more areas of the body. In the preterm neonate, benzodiazepines may cause medication-induced myoclonus thought to be due to developmental differences in the γ-aminobutyric acid (GABA) receptors, which abate as neonates mature. , Nonepileptic myoclonus has been described in the setting of intracranial infection, intracranial hemorrhage, periventricular leukomalacia, and genetic and metabolic disorders. , , ,
In an otherwise healthy neonate with a normal neurologic exam and jerking movements occurring only during sleep, benign neonatal sleep myoclonus should be considered. Benign neonatal sleep myoclonus is arrhythmic, may increase with attempts to physically suppress the movements, occurs at all sleep stages, and stops when the child is wakened. Benign neonatal sleep myoclonus will typically resolve by 3 months of age, although a few infants may have movements up to a year of age.
Dystonia is a sustained involuntary contraction of opposing muscle groups, often resulting in a twisted or abnormal posture. In neonates, dystonia or opisthotonic posturing is most commonly seen in encephalopathic neonates who have sustained an injury to the basal ganglia. Less common causes include inborn error of metabolism (e.g., monoamine neurotransmitter disorders, maple syrup urine disease , ). Nonepileptic bilateral tonic extension not having a correlate on EEG may represent “brainstem release” in the setting of extensive cortical dysfunction or posterior fossa pathology.
Hyperekplexia is a rare but important cause of hyperkinetic paroxysmal events in neonates that is characterized by nonextinguishable exaggerated startle reflex accompanied by hyperreflexia and hypertonia. Hyperekplexia is most commonly associated with mutations of the glycine receptor, but can also be seen in molybdenum cofactor deficiency. , Untreated hyperekplexia is associated with death secondary to apneic spells, which can be mitigated using scheduled benzodiazepines. ,
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here