Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
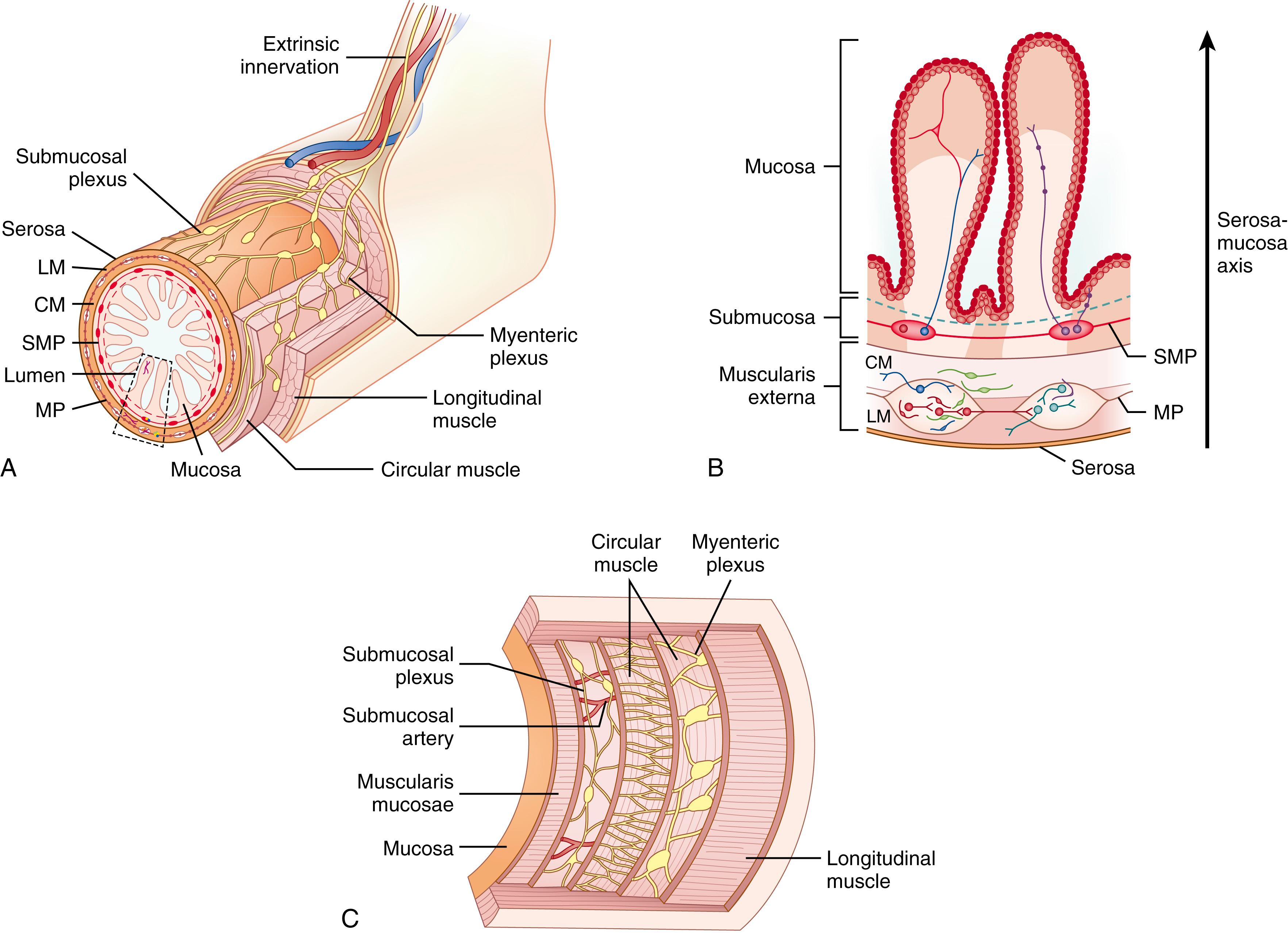
A critical modulator of gastrointestinal (GI) motility and secretion is the enteric nervous system (ENS). The ENS is a highly unique integrated neuronal circuitry, which, in contrast to other components of the peripheral nervous system, can and often does mediate reflex activity independent of the central nervous system (CNS). The ENS consists of two major plexuses ( Fig. 84.1 ): the myenteric plexus (Auerbach plexus), located between the external circular and longitudinal muscle layers, and the submucosal plexus (Meissner plexus), which itself is divided even further in larger mammals.
Although the ENS often acts independently, it also undergoes continuous, bidirectional communication with the CNS as well as other cell types, including immune, epithelial, and endocrine cells. All of these interactions play important roles in influencing the various physiologic roles within the intestine.
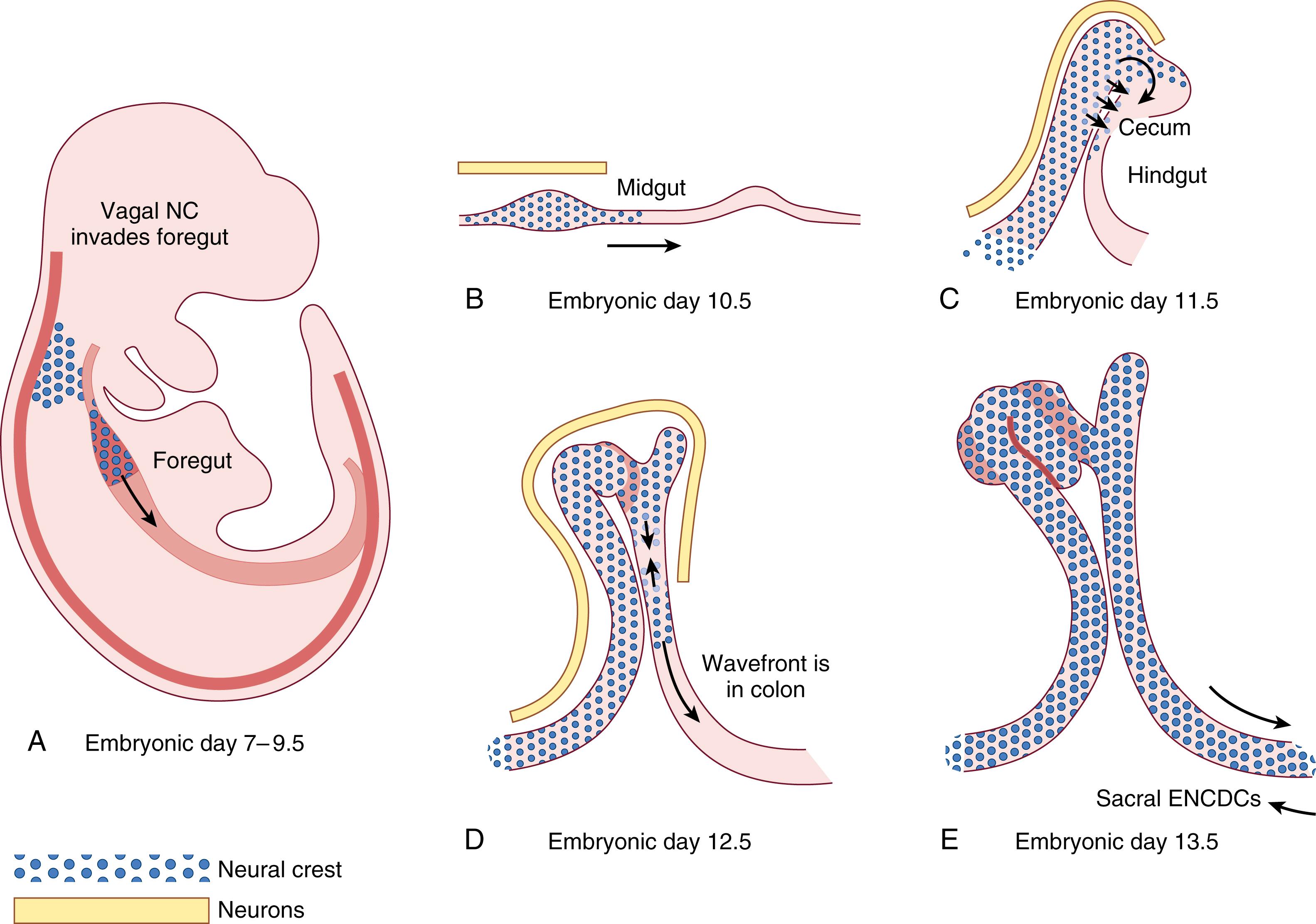
Enteric neurons and glial cells are derived from the migration of neural crest cells. Crest-derived cells from the vagal crest have been shown to colonize the entire bowel while those originating from the sacral crest contribute additional cells to the gut caudal to the umbilicus. , However, the vagal neural crest is the major source of ENS precursors. Premigratory neural crest cells from either site are multipotent; these pre-enteric neural crest–derived cells (pre-ENCDCs) enter the foregut and begin to migrate through the bowel just prior to week 4 in utero. As the ENCDCs migrate, they proliferate and differentiate into neurons, glia, and ganglia to form a network throughout the bowel ( Fig. 84.2 ). By week 7, this migration is complete. Phenotypic expression of the neural crest cells is ultimately dictated by the enteric microenvironments they confront at their site of terminal differentiation. After the initial gut colonization, ENCDCs undergo an inward radial migration, which results in the formation of the myenteric and submucosal plexuses. Although beyond the scope of this chapter, it is important to note that each of these stages is highly regulated by an extensive matrix of receptors, ligands, morphogens, and signaling proteins, extensively reviewed elsewhere. , However, correct progression through all of these phases is critical for the functions discussed below.
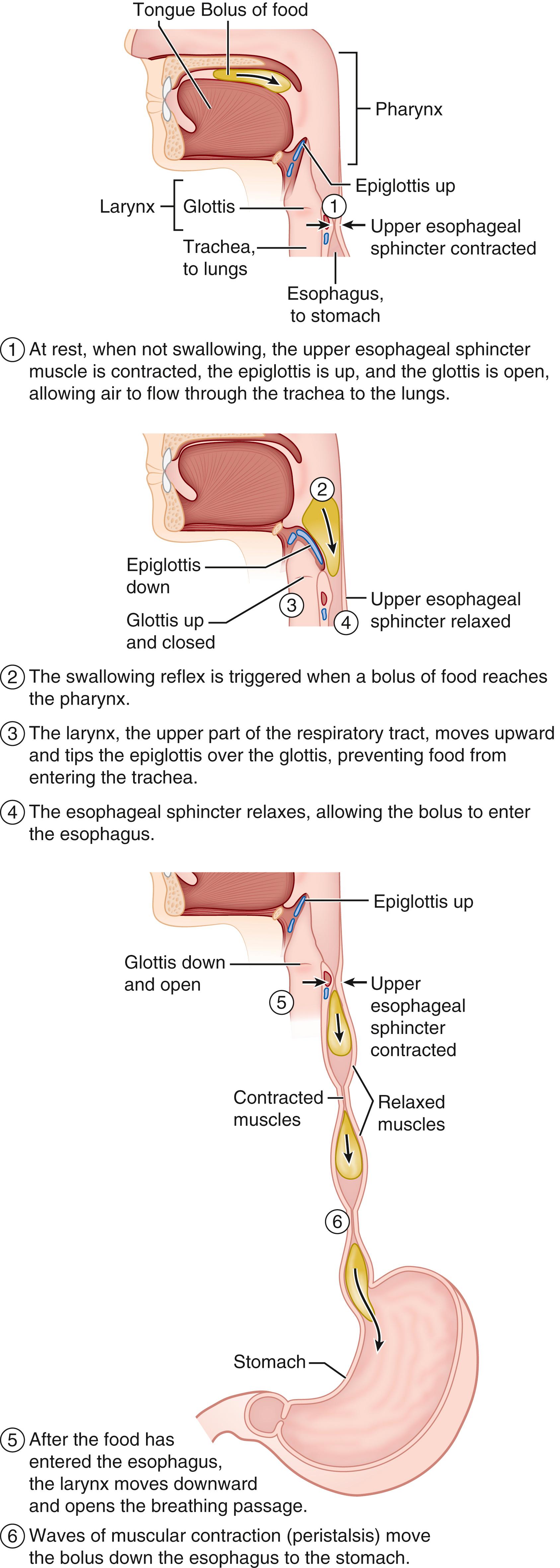
Ingestion of milk by the newborn infant depends on the ability to suck, in order to express milk from the breast or bottle into the oral cavity; to swallow, to propel milk from the oral cavity into the pharynx and then from the pharynx into the esophagus; and to coordinate sucking and swallowing with breathing. This process can be divided into the four phases of deglutition. The first phase is the oral preparatory phase, during which milk is drawn into the mouth by negative suction, mixed with saliva forming a liquid bolus. The second phase is the oral phase, in which the tongue projects posteriorly while contacting the palate to propel the bolus into the pharynx and through the relaxed open upper esophageal sphincter (UES). The third phase is the pharyngeal phase, during which the pharyngeal constrictors contract sequentially, clearing the residue of the bolus through the UES and into the esophagus. During the final phase, the esophageal phase, esophageal peristalsis propels the bolus down the length of the esophagus through the relaxed lower esophageal sphincter (LES) into the stomach ( Fig. 84.3 ).
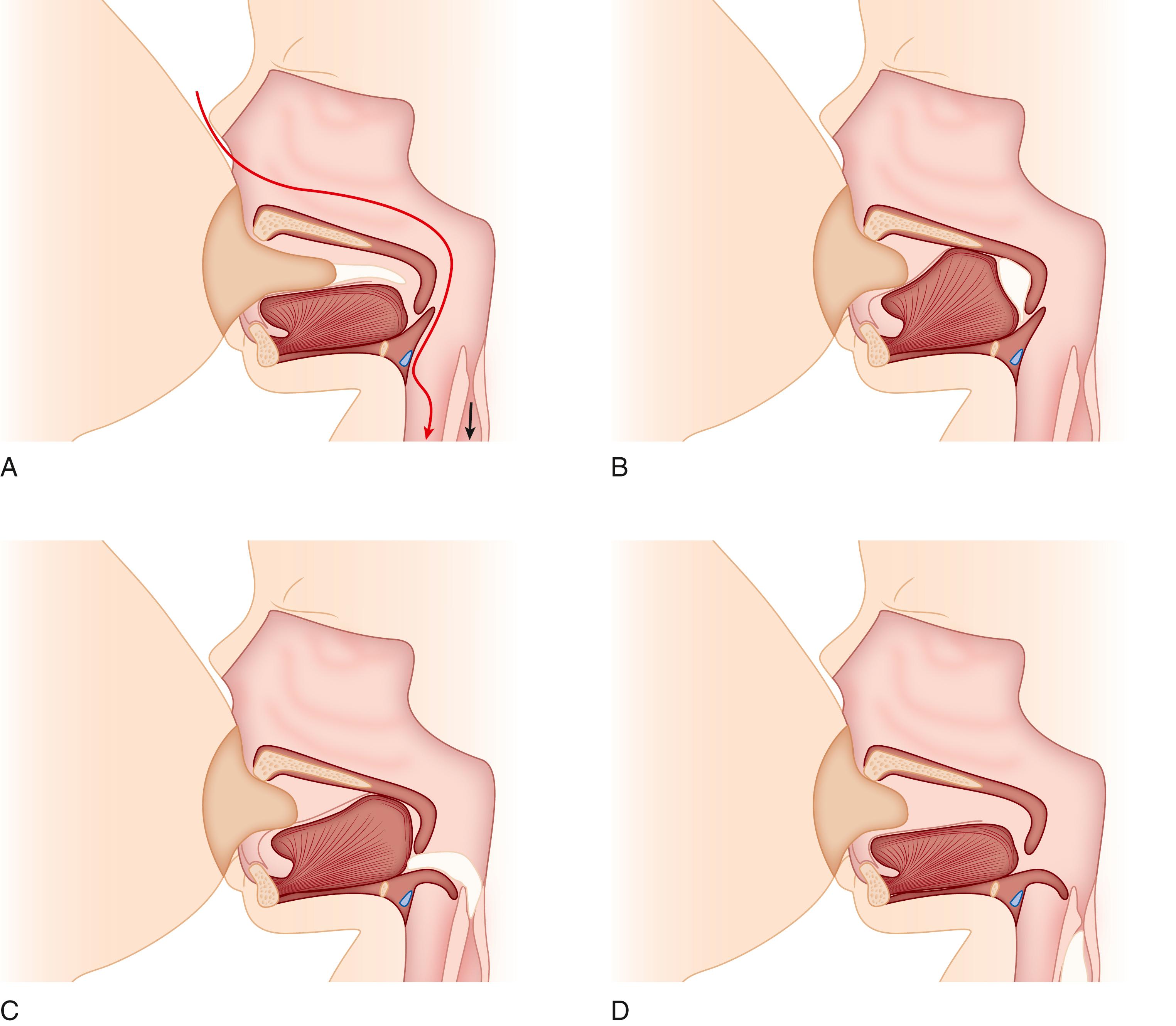
The orientations of the neonatal tongue, gums, palate, larynx, epiglottis, and pharynx are specifically adapted to facilitate the entry of a liquid bolus into the esophagus. Sucking and swallowing is dependent on both the anatomic integrity of the feeding apparatus and the maturation of complex oropharyngeal neuromuscular control mechanisms. The primary mechanism of induction of bolus propagation progresses from swallow induced at a gestational age of approximately 26 weeks to esophageal distension at approximately 32 weeks. Most neonates are capable of a coordinated suck and swallow by 34 weeks. ,
Development of sucking begins in utero and continues well after birth. Mouthing and lingual movements begin as early as 15 weeks gestation and progress to coordinated sucks as early as 28 weeks. Lingual movements begin with simple forward thrusting at approximately 21 weeks gestation and progress to cupping, which is a midline depression of the tongue, at approximately 28 weeks. Anterior protrusion coordinated with posterior retraction of the tongue is consistently observed by 28 weeks gestation and can be triggered by oral-facial stimulation in utero. The development of lingual movements coincides with myelination of the brain stem. In premature neonates, sucking patterns continue to mature through 36 weeks gestation.
The oral cavity in the newborn infant is adapted to facilitate passage of a liquid bolus initiated by sucking ( Fig. 84.4 ).The buccal fat pads and palate stabilize the lateral and superior walls of the cavity. The lips seal over the nipple, and the tongue presses against the soft palate to form a closed chamber in the oral cavity. The large tongue pulls inferiorly like a piston, creating a negative suction in the oral cavity, and the gums synchronously oppose this action to produce a positive expression pressure. This sucking action is the same during bottle- and breast-feeding, with the exception of the tongue, which is projected farther forward during breast-feeding.
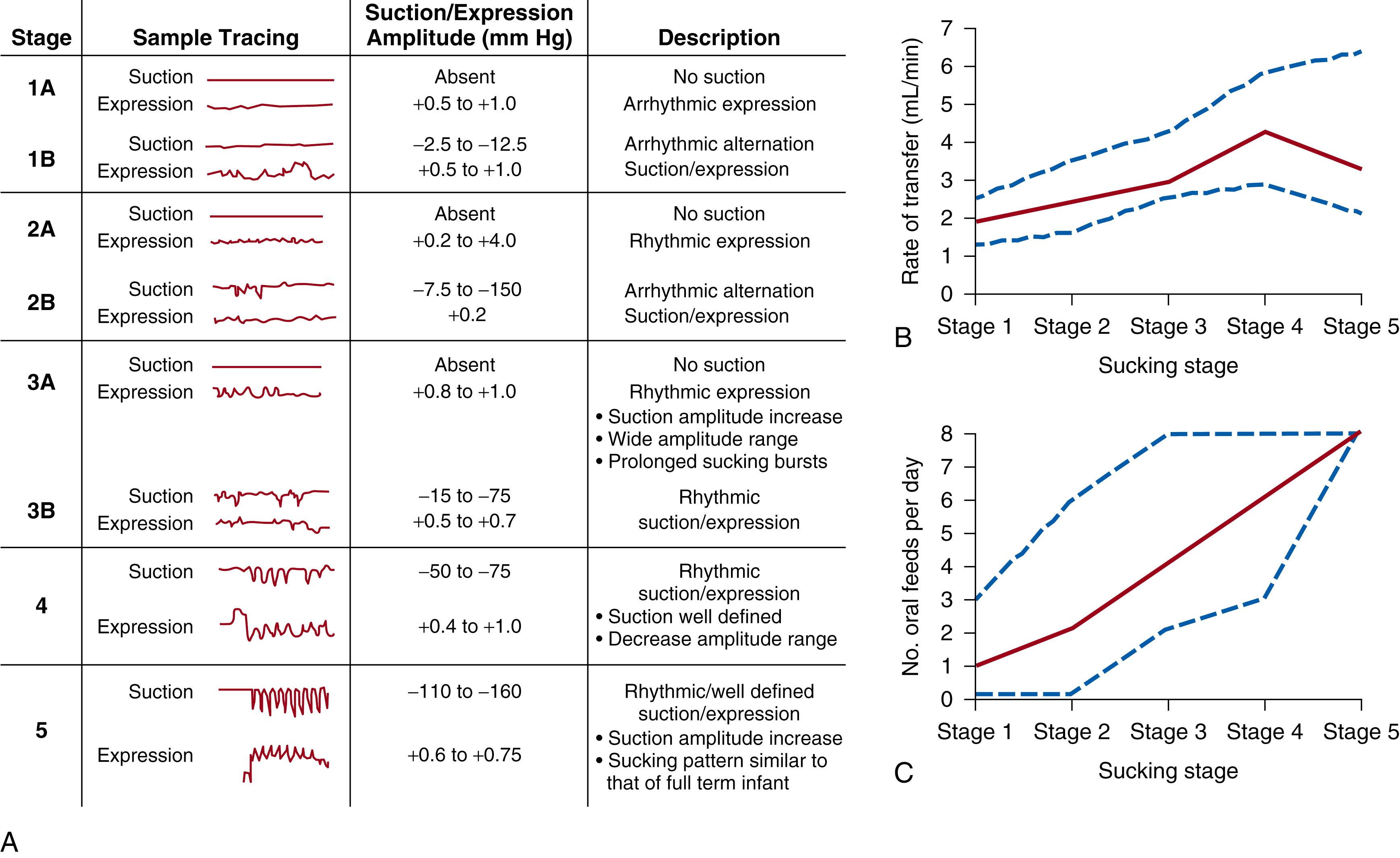
The development of oral motor skills starts with immature patterns of arrhythmic expression without suction (stage 1) and continues to a fully developed rhythmic expression-suction pattern (stage 5) ( Fig. 84.5 ). Both preterm and term infants exhibit maturation of sucking patterns postnatally, with preterm infants displaying the ability to attain full oral feeding at earlier post-gestational ages compared with those born at term. , , As the infant matures, the oral cavity enlarges, allowing manipulation of pureed spoon feedings. At 3 to 4 months of age, tongue movements develop that move a food bolus from the front of the tongue back towards the pharynx. At 6 months, infants will occlude their lips to remove pureed foods from a spoon and will move the food towards the midline with their tongue for chewing. As alveolar ridges and teeth develop during the next year, the infant develops increased tongue mobility that allows lateralization of solid foods for mature mastication.
In the fetus and the neonate, the propagation of a liquid bolus through the pharynx and into the esophagus, known as swallowing, proceeds directly from the act of sucking. The distinction between bolus formation and swallowing matures as the mouth, the pharynx, the esophagus, and the neural networks that control the coordination of these structures develop. Coordination of swallowing involves a complex interplay of neuromuscular fibers, cortical processing, and brain stem reflexes.
As the milk bolus is sucked into the oral cavity, afferent sensory input to the brain stem triggers contraction of longitudinal and transverse intrinsic lingual muscles, distal elevation of the tongue, and a medial peristaltic wave that travels posteriorly along the tongue. As the oral contents enter the pharynx, the soft palate and laryngeal musculature elevate, closing the nasopharynx and epiglottis while forming a conduit for passage of the milk bolus through the pharyngo-esophageal junction. Incoordination of this clearance from the oral cavity in premature infants may lead to residual overflow into the oropharynx, which may result in aspiration when the airway is not protected.
The pattern of muscular contractions that coordinate the oropharyngeal and esophageal phases of swallowing is centrally integrated by two regionally distinct groups of medullary interneurons, the nucleus tractus solitarius and the nucleus ambiguus, which together form the “swallowing center.” The nucleus tractus solitarius acts as a pattern generator, whereas the nucleus ambiguus regulates the transmission of the signal, together coordinating the sequential activation of motor neurons responsible for pharyngeal and esophageal contraction and UES and LES relaxation. , , Coordination becomes more efficient with maturity as sequential steps in the swallowing pathway occur in a more orderly manner.
Respiration in the neonate depends on the normal development of the anatomic relationships of the pharynx, larynx, and esophagus, their respective protective reflexes, and the coordination of breathing patterns with swallowing. In utero, the pharynx is a conduit for fluid that facilitates the ingestion of up to 750 mL of amniotic fluid each day. Swallowing contributes to amniotic fluid homeostasis, and the swallowed fluid appears to be trophic for mucosal development of the GI tract. , However, after birth, the pharynx performs a dual function, regulating the passage of food into the esophagus and air into the larynx.
Mature term infants possess an airway protection mechanism that is triggered before pharyngeal swallowing. In contrast, recently born term infants and preterm infants rarely synchronize breathing and swallowing. Shortly after birth, term infants exhibit periods of rapid swallowing that are associated with unstable breathing, depressed respiratory rate, and reduced minute volume. , Premature infants alternate bursts of rapid swallowing with periods of rapid breathing. , When fluid enters the neonatal larynx, vagal afferents trigger a reflex of rapid swallowing, apnea, laryngeal constriction, hypertension, and bradycardia. , Although protective against aspiration, this reflex can threaten the infant and may exacerbate uncoordinated breathing and swallowing patterns. The coordination of breathing and swallowing depends on interneuronal pathways between the medullary swallow and respiratory centers; these pathways are poorly developed in premature infants.
The esophagus is a hollow muscular tube, closed proximally and distally by muscular sphincters. The UES surrounds the upper part of the esophagus, and the LES surrounds the junction between the esophagus and the stomach. The main function of the esophagus is to propel swallowed food or fluid into the stomach, where digestion and absorption take place. This occurs through a series of peristaltic contractions of circular muscle in the esophageal body, in concert with appropriately timed relaxation of the UES and LES.
The UES and the proximal third of the esophageal body are composed of striated muscle. More distally, there is a transition zone where striated and smooth muscle intermix. The distal half to two thirds of the esophageal body and the LES are composed of smooth muscle.
The intrinsic innervation of the esophagus is made up of networks of nerves and ganglia that comprise the myenteric and submucosal plexuses (see Fig. 84.1 ). The myenteric plexus, located between the longitudinal and the circular layers of the tunica muscularis, regulates contraction of the outer muscle layers. The submucosal plexus, located within the submucosa, regulates secretion and the peristaltic contractions of the muscularis mucosae. Extrinsic innervation of the esophagus involves neurons located in the brain stem and spinal cord. Sympathetic and parasympathetic neurons originating in the nucleus ambiguus and vagus nerve supply the upper striated muscle and the UES. Neurons from the dorsal motor nucleus and cervical and thoracic sympathetic trunks supply smooth muscle and the LES.
The UES serves the dual role of accommodating boluses from the pharynx into the esophagus and maintaining a physical barrier to refluxed contents from the stomach entering the larynx. UES function is controlled by a variety of reflexes that involve afferent inputs to the motor neurons innervating the UES. These reflexes elicit either contraction or relaxation of the tonic activity of the UES.
UES pressure is generated predominantly by tonic contraction of the cricopharyngeus muscle. During the esophageal phase of swallowing, a propagated bolus exerts pressure to open the UES while the cricopharyngeus relaxes and the hyoid and larynx elevate. , Feeding difficulties may arise in premature infants who lack sufficient pharyngeal intrabolus pressure or lack coordination between oropharyngeal swallowing and UES relaxation. , Reflex pathways of the vagus nerve and brain stem mature with age, and this maturation coincides with improved coordination of the timing of this sequence. , Esophageal distension, such as by refluxed gastric contents, causes UES relaxation in premature infants, which may lead to retrograde passage into the oropharynx. However, premature infants exhibit higher UES resting pressures during states of arousal or behavioral strain, which may serve as a protective mechanism.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here