Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
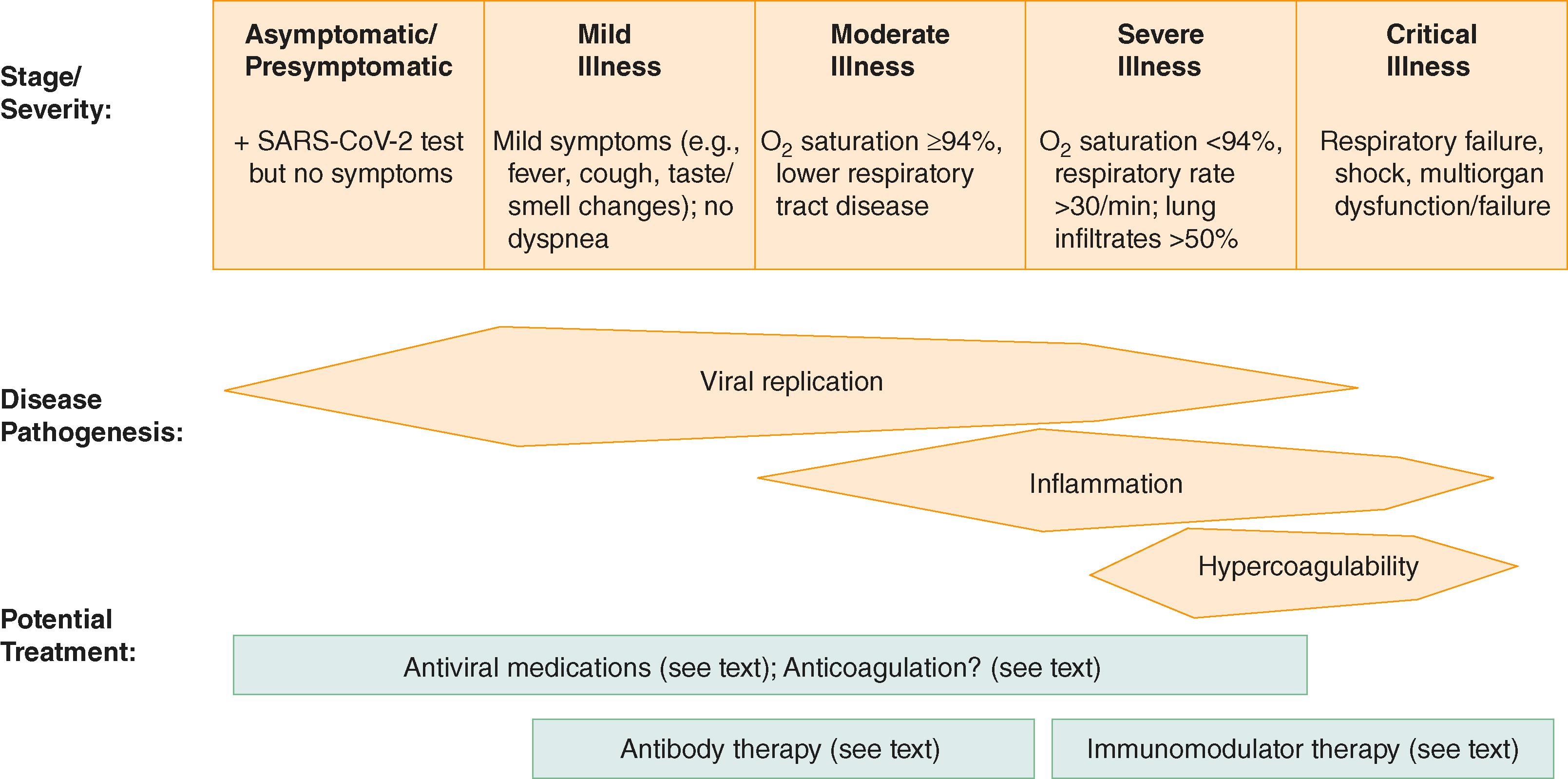
The approach to treating coronavirus-2019 disease (COVID-19) must first consider the severity of the patient’s disease, which can range from mild to moderate respiratory or systemic symptoms, to severe disease (i.e., dyspnea, tachypnea, and hypoxia), to critical illness (i.e., respiratory failure, shock, and/or multiorgan failure; see Fig. 336-1 ). The second consideration is the phase of illness. The third consideration is the type of intervention: antiviral therapy, immunomodulatory medications, combination therapy, and therapies directed at preventing or treating complications, such as thrombosis.
The pathogenesis that drives the different phases of the illness also informs treatment decisions ( Fig. 337-1 ). Viral replication is most active early in disease, whereas excess inflammation occurs later in the course of disease. Regardless of the phase of illness, it is safe to continue angiotensin-converting enzyme 2 (ACE2) inhibitors, angiotensin-receptor blockers, and therapeutic nonsteroidal anti-inflammatory drugs.
Most patients with mild to moderate COVID-19 will recover with supportive care, such as hydration, antipyretics, and analgesics. However, close monitoring for clinical worsening is mandatory, particularly for patients who are at increased risk for progression, such as patients who are older, who have certain chronic conditions (e.g., cardiovascular disease, chronic lung disease, obesity, immunocompromising conditions), or who are taking immunosuppressive medications. Worsening shortness of breath, which typically occurs about 1 week after the onset of symptoms, or other severe or progressive symptoms should prompt in-person evaluation. Patients with COVID-19 may deteriorate rapidly, and hospitalization is indicated for monitoring and treatment if patients develop progressive dyspnea, weakness, lethargy, or confusion or if they become tachypneic or hypoxic or have a markedly abnormal chest radiograph.
Passive immunity can be induced by monoclonal antibodies that bind to the SARS-CoV-2 spike protein, block viral entry into cells, and thereby reduce replication. Monoclonal antibodies can result in more rapid viral clearance and reduce rates of hospitalization or death by approximately 70 to 85% when given within a few days of the onset of symptoms to outpatients who have mild to moderate COVID-19 and are at high risk for progression (see Fig. 337-1 ), provided that they are infected with susceptible viral variants. However, antibodies that were successful against earlier SARS CoV-2 variants have less activity against newer variants; as a result of changing variants, anti–SARS-CoV-2 monoclonal antibodies are currently not authorized for treatment or prophylaxis in the United States. The decision to use antibodies and the doses among them should be guided by data on current variants and their susceptibility.
Convalescent plasma, which is derived from patients who previously have been infected, has shown mixed results. In one study, it reduced the risk of hospitalization in unvaccinated outpatients treated with 8 days of symptoms, but other studies showed no benefit. Currently it is authorized for use in immunocompromised individuals, but it should only be considered if the plasma is likely be active against the circulating variant.
Antiviral medications are useful in high-risk patients who are early in their disease course ( Fig. 337-2 ). However, none of the available drugs are currently authorized for lower risk patients.
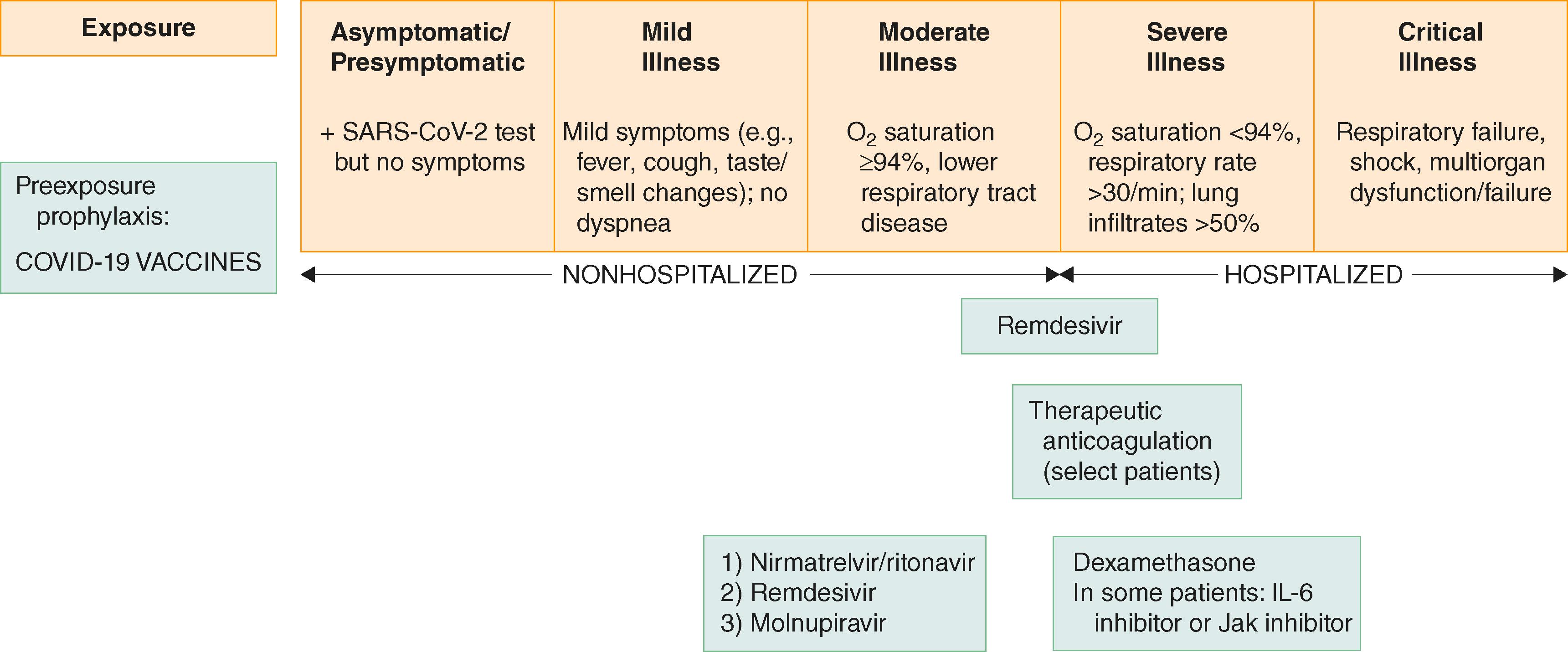
Nirmatrelvir is an oral protease inhibitor that is coadministered with ritonavir to boost its blood level by inhibiting its metabolism by the cytochrome P-450 system. In unvaccinated nonhospitalized patients who have COVID-19 with a high risk of clinical progression and who are treated within 5 days of the onset of symptoms, nirmatrelvir/ritonavir can reduce the risk of hospitalization or death by about 90%.
Remdesivir, which is a nucleotide pro-drug, binds to the SARS-CoV-2 RNA polymerase, acts as a chain terminator, and thereby inhibits RNA synthesis and viral replication. In high-risk nonhospitalized patients treated within 7 days of the onset of symptoms, remdesivir (200 mg on day 1 and 100 mg on days 2 and 3) for 3 days can reduce hospitalization by 87%, even though it does not reduce nasopharyngeal SARS-CoV-2 levels.
Molnupiravir, which is an oral inhibitor of SARS-CoV-2 replication, induces mutations that compromise the virus’s viability. In nonhospitalized adults who have had 5 or fewer days of mild or moderate COVID-19 symptoms plus at least one risk factor for developing severe COVID-19, molnupiravir (800 mg twice daily for 5 days) reduced the risk of hospitalization or death by about 30%. However, this benefit may not extend to persons who have been vaccinated.
Currently nirmatrelvir/ritonavir is typically the first choice in most high-risk patients. In a patient who cannot receive nirmatrelvir/ritonavir because of drug-drug interactions, remdesivir is usually the second choice. When remdesivir is not feasible or appropriate, molnupiravir should be considered.
The role of inhaled budesonide (800 µg twice daily for 14 days) in high-risk outpatients is uncertain, and inhaled fluticasone is of no benefit. Ivermectin has not been shown to be of benefit. Neither anti-platelet therapy with aspirin nor anticoagulant therapy with apixaban is recommended in outpatients who do not have other indications for their use.
Hospitalized patients with COVID-19 benefit from selected medications and from careful attention to oxygenation, intensive care, and organ support. Clinicians should follow the latest guidelines (see https://www.covid19treatmentguidelines.nih.gov/about-the-guidelines/whats-new/ and https://www.idsociety.org/practice-guideline/covid-19-guideline-treatment-and-management/ ).
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here