Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The author would like to acknowledge Frandics Chan, MD, PhD, for his contributions to prior editions of this chapter.
Coronary artery disease in children can be acquired, but the majority of anomalies are congenital. Among acquired coronary abnormalities, Kawasaki disease (KD) is the most common. Other causes include sequelae from trauma, vasculitides, radiation injury after oncologic treatment, and rare cases of familial hyperlipidemia and idiopathic infantile arterial calcification. Congenital coronary anomalies can be characterized as abnormalities in origin, abnormalities associated with the coronary vessel, and abnormalities associated with congenital heart disease. Boxes 77.1 and 77.2 outline the common acquired and congenital coronary anomalies seen in children.
Kawasaki disease
Behçet disease
Polyarteritis nodosa
Trauma (pseudoaneurysm)
Kawasaki disease
Heart transplant-associated coronary artery vasculopathy
Radiation
Behçet disease
Polyarteritis nodosa
Trauma (dissection)
Homozygous familial hypercholesterolemia
Idiopathic infantile arterial calcification
Anomalous aortic origin of the left coronary artery (AAOLCA)
Anomalous aortic origin of the right coronary artery (AAORCA)
Anomalous coronary artery origin from the pulmonary artery (ALCAPA)
Coronary fistulae
Congenital coronary stenosis
Congenital coronary aneurysms
Myocardial bridging
Tetralogy of Fallot
Transposition of the great arteries
Truncus arteriosus
Pulmonary atresia with intact ventricular septum
Transthoracic echocardiography (TTE) remains the first-line imaging tool for evaluating children with suspected coronary anomalies. In experienced hands, the coronary origins and proximal coronary courses are reliably imaged. When further anatomic detail of luminal dimensions, ostial morphology, or evaluation of more distal portions of the coronary anatomy is necessary, other imaging modalities are necessary. Catheter angiography is the reference standard for the determination of coronary luminal abnormalities, such as coronary stenosis. Additional techniques that further characterize the arterial wall and myocardial perfusion can be performed during catheterization, such as intravascular ultrasound (IVUS) and fractional flow reserve measurement. However given the invasive nature of catheter angiography, electrocardiographic (ECG)-gated coronary computed tomography angiography (CCTA) and cardiac magnetic resonance angiography (CMRA) are the main diagnostic modalities utilized. Both techniques yield three-dimensional images of the heart, with the coronary vessels shown in the context of adjacent cardiac structures. Visualization is facilitated by various postprocessing techniques, such as curved multiplanar reformatting and endoluminal views via virtual angioscopy. CCTA has excellent diagnostic accuracy in detecting coronary stenosis, and several studies show that it is now comparable to catheter angiography. These capabilities have made CCTA and CMRA the preferred method for the diagnosis and characterization of coronary anomalies.
CCTA provides excellent visualization of coronary anatomy, including the distal coronary branches. Images synchronized to the cardiac cycle can be acquired with prospective or retrospective ECG synchronization. Prospective ECG-triggering yields a snapshot of the heart at a predetermined portion of the cardiac cycle, whereas with retrospective ECG-gating, data is acquired throughout the cardiac cycle and can therefore be reconstructed at multiple time-points in the cardiac cycle and can be used to produce cine images of the heart. However, because data is acquired throughout the cardiac cycle, images obtained with retrospective ECG-gating generally incur a greater radiation dose compared with prospective ECG-triggering. With 64-slice scanners, the typical scan time in either mode is less than 10 seconds. Newer generation dual source and volumetric computed tomography (CT) scanners provide full anatomic coverage of a pediatric thorax in less than a second or in a single heartbeat, freezing respiratory and cardiac motion, and obviating the need for sedation in many cases. As CCTA uses ionizing radiation to produce images, measures should be taken to minimize the dose of ionizing radiation delivered while maintaining image quality. Strategies for CCTA dose optimization are listed in Box 77.3 and have been detailed in Chapter 66 .
Limit scan range to anatomy of clinical interest.
Center the patient within the gantry.
Lower tube potential and tube current to account for patient size. Consider automated algorithms.
Technique should be adjusted to the lowest practical radiation dose that will yield diagnostic image quality based on the clinical indication.
Prospective ECG-triggering should be used when possible.
For coronary imaging, the narrowest temporal acquisition window possible should be used.
Iterative reconstruction should be used on all scans.
Heart rate lowering medications should be considered.
The lack of ionizing radiation makes CMRA an attractive alternative to CCTA. CMRA can also provide information regarding ventricular size, systolic function, and tissue characterization (such as perfusion imaging, myocardial delayed enhancement, and T1 mapping). CMRA for assessment of the heart and great arteries is often performed using a three-dimensional, cardiac-gated, navigated echo, T2-prepared, steady-state, free-precession sequence (see Chapter 67 ). Coronary vessel visualization with this sequence is based on the intrinsic long-T2 value of blood, so no gadolinium contrast administration is required.
One of the main drawbacks of CMRA is that it lags in spatial resolution compared with CCTA, which can provide isotropic submillimeter resolution. Additionally, despite the lack of ionizing radiation, CMRA often requires anesthesia in patients younger than 8 years of age, and the risks of anesthesia versus radiation exposure must be weighed.
Embryologically, the spatial configuration of the aortic sinuses adjacent to the pulmonary trunk act as a stimulus for coronary bud development. In a structurally normal heart with normal coronary artery anatomy, the left main coronary artery (LMCA) and right coronary artery (RCA) arise from the left and right sinuses of Valsalva, respectively. The LMCA then divides into the left anterior descending (LAD) and circumflex (Cx) coronary arteries. Many anatomic variations of the coronary arteries exist, with normal variants typically having no clinical significance ( Fig. 77.1 ).
In cases where there is associated congenital heart disease, nomenclature of the coronary arteries should refer to the coronary dependent territory as opposed to the site of coronary origin. For example, in congenitally corrected transposition of the great arteries (L-transposition of the great arteries) where the morphologic left ventricle lies to the right, the coronary artery in the right-sided atrioventricular groove is named the left circumflex artery (see Chapter 75 ). When derangement of the ventricles is so severe that the ventricular morphology cannot be clearly identified, then the coronary arteries should be described relative to physical landmarks—for example, left-sided atrioventricular branch instead of left Cx, right-sided atrioventricular branch instead of RCA, anterior interventricular septal branch instead of LAD, and posterior interventricular septal branch instead of posterior descending artery (PDA).
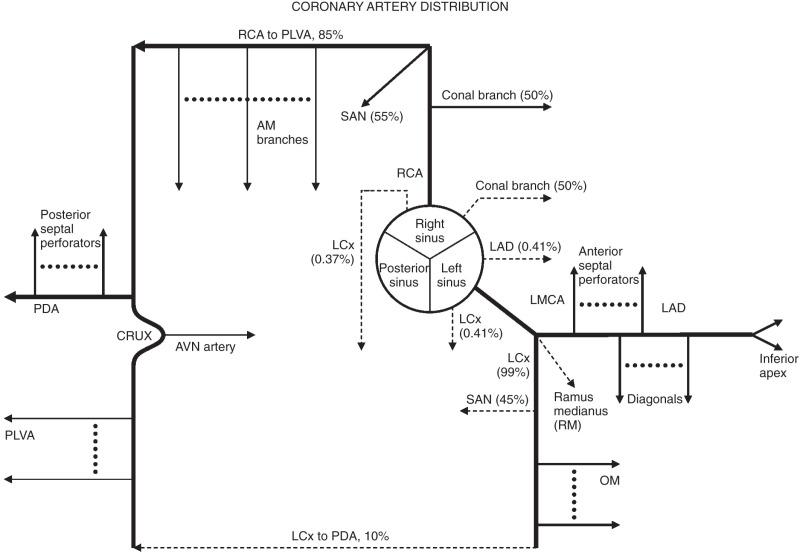
After its takeoff from the right sinus of Valsalva, the RCA courses anteriorly into the right atrioventricular groove and gives rise to several branches. The right conal artery arises as the first branch of the right coronary artery 50% to 60% of the time, arising directly from the right sinus of Valsalva in 40% to 50% of cases. The sinoatrial nodal artery arises from the proximal RCA in the majority of cases and from the proximal left Cx in the other ( Fig. 77.2A ). The midportion of the RCA gives rise to the acute marginal branches, which supply the RV free wall ( Fig. 77.2B ). The distal portion of the RCA wraps around the inferior surface of the heart. An atrioventricular node branch often arises from the RCA near the crux of the heart ( Fig. 77.2D ). In 70% to 85% of the population, the RCA gives rise to a PDA that courses within the posterior interventricular sulcus to supply the basilar inferior wall of the left ventricle ( Fig. 77.2C ). This pattern is called a right-dominant coronary system . There can be codominance, where the RCA supplies the PDA and the Cx supplies additional posterior left ventricular branches. The least common pattern is left-dominance, where the PDA arises from the Cx.
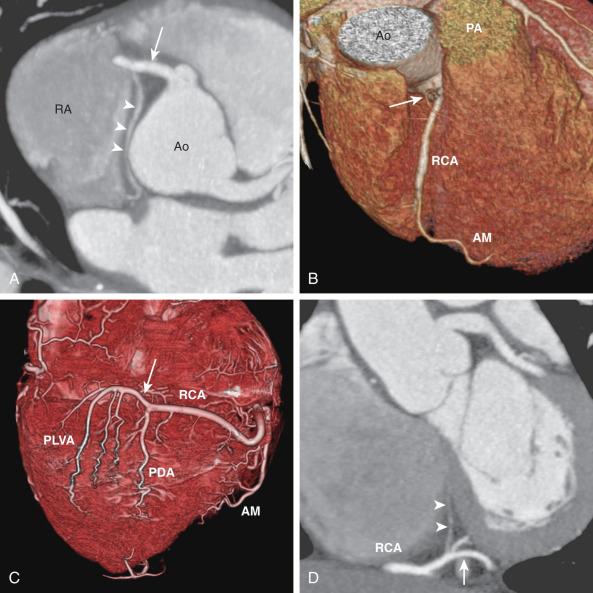
The LMCA originates from the left sinus of Valsalva and courses to the left for a short distance before it bifurcates into the LAD and Cx coronary arteries. Occasionally, the LMCA trifurcates into an LAD coronary artery, a Cx coronary artery, and a ramus intermedius branch ( Fig. 77.3A ). This ramus intermedius branch supplies the anterior left ventricular wall. The LAD artery gives rise to two sets of vessels: an epicardial set called the diagonal branches and an intramuscular set called the anterior septal perforator branches . The diagonal branches are responsible for perfusing the anterior left ventricular wall, whereas the septal perforator branches are responsible for the anterior septum. The distal LAD artery typically wraps around the cardiac apex and terminates at the inferior wall of the apex ( Fig. 77.3B ). In a minority of cases, the Cx artery gives off a sinoatrial nodal branch ( Fig. 77.3C ) before it enters the left atrioventricular groove. The Cx artery then gives rise to a number of obtuse marginal branches, which supply the lateral left ventricular wall (see Fig. 77.3B ).
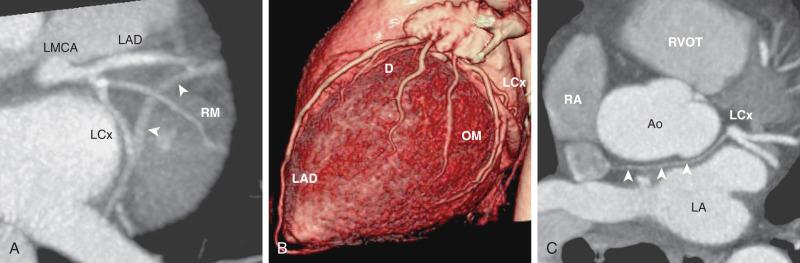
The true prevalence of congenital coronary artery anomalies is unknown. According to adult catheter angiographic data, 0.6% to 1.5% of patients were found to have coronary artery anomalies. The largest study of this type reviewed more than 126,000 coronary studies and revealed a prevalence of 1.3%, with the majority (80%) felt to have no clinical significance. The two most common anomalies are separate origins of the LAD and Cx arteries arising from the left sinus of Valsalva (0.41%) ( e-Fig. 77.4A ) and a retroaortic Cx artery arising from the right sinus or the RCA (0.37%) ( e-Fig. 77.4B ). Both configurations are clinically benign. Other examples of benign anomalies are an absent Cx artery with a superdominant RCA that reaches the anterior atrioventricular groove and a high takeoff of either coronary (>1 cm above the sinotubular junction).
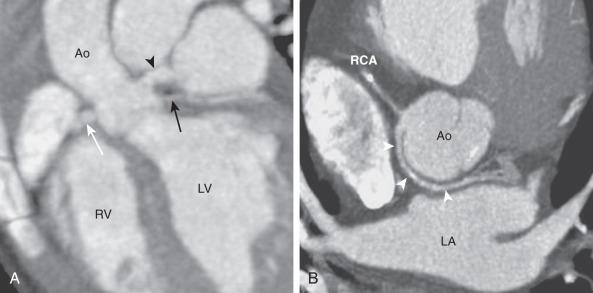
An anomalous aortic origin of the LMCA (AAOLCA) may originate from the right sinus of Valsalva or may arise as a single coronary artery with the RCA. Before bifurcating into the LAD and Cx arteries, the LMCA must travel toward the left by one of four routes: anterior to the right ventricular outflow tract (RVOT), within the interventricular septum beneath the RVOT (intraseptal), between the aorta and the pulmonary artery (interarterial), or posterior to the aorta. Similarly, an anomalous aortic origin of the RCA (AAORCA) may originate from the left sinus of Valsalva or as a single coronary with the LMCA. It typically has an interarterial course toward the right atrioventricular groove, although other routes are possible. While the anterior and posterior courses are considered clinically benign, the interarterial course ( Fig. 77.5 ) is associated with sudden cardiac death. This association was originally described in several autopsy series, with AAOLCA found more frequently than AAORCA. Several studies and case reports have subsequently established this association, with congenital coronary anomalies reported as the second-most common cause for sudden cardiac death in adolescent athletes.
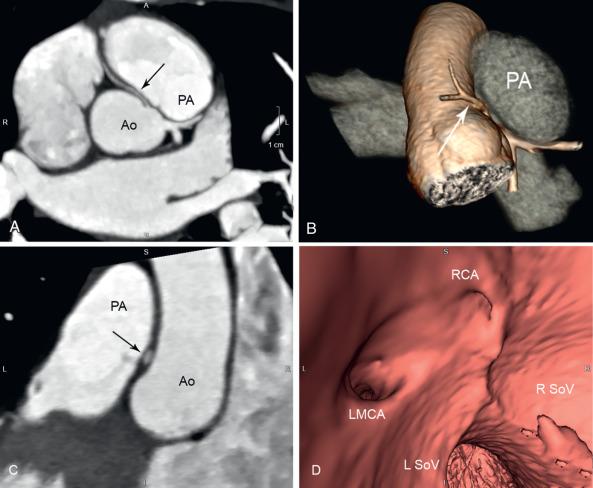
The true prevalence of anomalous aortic origin of the coronary arteries is unknown. The majority of patients are asymptomatic and a major adverse cardiac event is often the initial presentation. Based on prior angiographic studies, the prevalence of AAOLCA with an interarterial course is estimated at 0.02% to 0.05% of the population, and the prevalence of AAORCA is estimated at 0.1%. Given that AAORCA appears to be three to five times more common than AAOLCA in the general population, the prior autopsy studies suggest that AAOLCA is a higher risk lesion.
The pathophysiology of sudden cardiac death has not been conclusively determined. Most patients with sudden cardiac death had no significant clinical findings or known premonitory symptoms. In about 30% of the cases, syncope or chest pain occurred within 2 years of death, but resting ECG and stress ECG have been reported as normal. The prevailing thought is that the risk of sudden cardiac death is increased with significant physical exertion, such as strenuous athletic activity. Presumed risk factors for a lethal arrhythmia are transient ischemia caused by inadequate coronary flow through a narrowed “intramural” lumen compressed by the aorta and pulmonary artery, a slitlike opening caused by an acute takeoff (<45 degrees) of the anomalous coronary artery, and the presence of an ostial ridge or flap that also occludes the ostium. An intramural lumen refers to a portion of the coronary artery where the tunica media is shared with the aortic wall and there is no interposed adventitial layer.
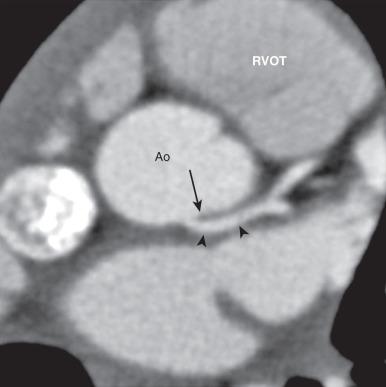
In addition to interarterial coronary arteries, sudden death has rarely been reported with AAOLCA arising from the noncoronary sinus without an interarterial course. In this configuration, the alignment of the LMCA origin and the anterior interventricular groove is such that the LMCA may have a more acute takeoff angle from the noncoronary sinus ( e-Fig. 77.6 ). This acute takeoff angle creates a narrowing at the ostium that is thought to be a factor leading to ischemia and sudden death.
In neonates and young children, echocardiography can delineate the coronary origins to suggest the anomalous coronary types previously discussed. In adolescents and young adults, the coronary arteries may not be adequately assessed with echocardiography. Confirmation of the diagnosis and detailed characterization of the high-risk features previously mentioned is better performed by CMRA and CCTA. In particular, the presence of an intramural course should be evaluated by cross-sectional imaging, as this is poorly characterized by echocardiography. Current data suggests the risk of sudden cardiac death is low before 10 years of age, and so many centers defer surgical intervention until after this age. Further cross-sectional imaging can likely be deferred in most routine cases until around 10 years of age. This has the benefit of avoiding sedation or anesthesia in the majority of cases. Depending on the expertise and equipment available to individual centers, CMRA can be considered the first test of choice to avoid radiation exposure. If CMRA is not feasible due to technical or diagnostic reasons, then CCTA presents an excellent choice. Diagnostic catheter angiography does not have much of a role for this indication.
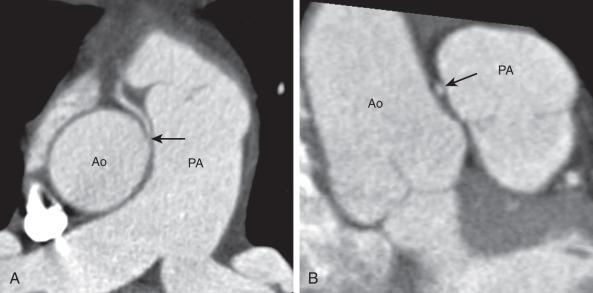
Morphologic features considered clinically relevant are: ostial size/morphology (including the presence of an acute angle takeoff from the aorta, a slitlike origin, and the presence of an ostial ridge), interarterial narrowing suggestive of an intramural course, length of the intramural portion, location in relation to the sinotubular junction and aortic commissures, and coronary dominance. Determination of the intramural course can be difficult with CCTA and CMRA. Correlation with surgical pathology suggests that CCTA findings of a slitlike origin, an acute angle at vessel takeoff, and an elliptical cross-section with parallel course along the aortic root are related to an intramural coronary segment ( Fig. 77.7 ). Endoluminal views and volume rendered reconstructions are helpful for ostial characterization and surgical planning.
Although there is a strong association with sudden cardiac death, the absolute risk among individuals, particularly in the absence of participation in competitive sports, is unknown. There is marked practice variation among congenital heart surgery centers due to this uncertainty. Recent data from a multicenter registry by the Congenital Heart Surgeons Society involving 23 pediatric centers reported 52% of AAORCA and 67% of AAOLCA had undergone surgery at a median age of 12.6 years. The type of surgical repair is dependent on the coronary anatomy. In those with an intramural course, an unroofing procedure is the technique of choice. This involves incising the common wall between the aorta and intramural coronary. Aortic valve commissural resuspension may be necessary in cases where the intramural coronary courses behind the commissure. Direct reimplantation or creation of a neo-ostium and ostioplasty is performed when there are separate ostia and no significant intramural course. A pulmonary artery translocation can be performed in the case of a single coronary with evidence of coronary compression and/or ischemia. A bypass graft can be performed but has a very limited role due to concerns with competitive flow from the native coronary.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here