Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Our understanding of why some children are born with imperfections while the majority are born with normal anatomy has made a tremendous improvement through the last half century. These often disfiguring anomalies are commonly seen by both general practitioners and specialists but the spectrum of congenital vascular malformations (CVMs) is so wide and complicated that it often requires a wide range of specialists dedicated to their study and treatment as a challenging problem.
The term “congenital vascular malformation” (CVM) is used to describe malformed vessels resulting from arrested development during various stages of embryogenesis. , It is the outcome of a congenital defect in the vascular system and is therefore present at birth, although it is not always clearly identifiable immediately. CVMs may involve one or all three circulatory systems – arterial, venous, and lymphatic – either as a predominant form (e.g., venous malformation) or as a mixed condition (e.g., hemolymphatic malformation). The lesions originating from the “early” stage of embryogenesis keep the unique condition of continuing to grow regardless of type.
CVMs may be confused with a more common vascular anomaly, the infantile or neonatal hemangioma. , Although both CVMs and hemangiomas are vascular anomalies, the conditions are fundamentally different not only in their anatomic, histologic, and pathophysiologic findings but also in their clinical course ( Fig. 170.1 ). , A true hemangioma is a vascular tumor that originates from endothelial cells. It usually appears in the early neonatal period as a rapidly growing tumor. It has a distinctive course characterized by a proliferation phase of early, rapid growth followed by an involutional phase of slow regression. Hemangiomas are therefore, “self-limited” to grow in general; involution is usually complete before age 12 years. In contrast, CVMs are “self-perpetuating” continuing to grow when stimulated and generally distinctive at birth as inborn errors, and they grow steadily in parallel with the child’s systemic growth. CVMs never disappear or regress.
In the early 20th century, many physicians, including Maurice Klippel and Paul Trénaunay (1900), reported a congenital anomaly involving skin, soft tissue, bone, and blood vessels. , Owing to limited anatomic and pathophysiologic knowledge and a lack of appropriate diagnostic studies, this condition was not properly understood and was simply named after the describing physicians, such as Klippel–Trénaunay syndrome (KTS) , and Parkes Weber syndrome (PWS). , These eponymous conditions included various clinical findings (e.g., soft tissue swelling, long-bone growth discrepancy), but the primary vascular lesion and secondary nonvascular lesion were not appropriately identified. This resulted in significant confusion about the true nature of the problem, as well as a lack of essential information about the cause, anatomy, and pathophysiology of these complex vascular anomalies. , Modern diagnostic and therapeutic modalities have provided valuable information on the cause and anatomo-pathophysiology of CVMs. ,
The Hamburg Classification was established to accommodate the contemporary understanding of CVMs based on anatomy and pathophysiology based on modern diagnostic criteria. , However, it is still useful for understanding the combined forms of vascular malformations in particular. For example, the vascular components of KTS are now called hemo-lymphatic malformations (HLMs) in the Hamburg classification (a combination of venous, lymphatic, and capillary malformations), whereas the components of PWS consist of venous, lymphatic, capillary, and arteriovenous malformations ( Fig. 170.2 ).
The term angiodysplasia has been replaced with the term congenital vascular malformation to minimize confusion. , Finally, the term capillary or cavernous hemangioma has erroneously been used to describe a venous malformation and should be removed. , Finally, the term hemangioma is correctly used only to identify a genuine hemangioma (i.e., a vascular tumor), not a vascular malformation.
The Hamburg classification ( Box 170.1 ) appropriately classifies CVMs according to the underlying anatomic, histologic, pathophysiologic, and hemodynamic status derived from different embryonic stages. , , It is based on a consensus of CVM experts who met at the Seventh International Workshop on Vascular Malformations in Hamburg, Germany, in 1988. This classification has shown its excellent clinical applicability and has been well accepted as a modern classification system. It originally divided CVMs into five types based on the predominant vascular component: arterial malformation (AM), , venous malformation (VM), , arteriovenous malformation (AVM), , lymphatic malformation (LM), , and combined vascular malformation represented by the HLM. , The initial consensus did not include the capillary malformation (CM) because it was believed to lack the clinical significance of other CVMs. However, the addition of CMs was proposed and upheld at subsequent consensus meetings. , The Hamburg classification with further modification to accommodate embryological subclassification has been accepted worldwide as a practical guideline for the CVM management, however it is far from perfect, and further improvements are necessary before it completely replaces the old terminology and classifications.
∗ Modified from the original classification based on the consensus reached at the international workshop in Hamburg, Germany, 1988.
Arterial defects
Venous defects
Arteriovenous shunting defects
Lymphatic defects
Combined vascular defects – hemolymphatic defects
Capillary defects
† Represents arrested development at different stages of embryonic life, with extratruncular being an earlier stage and truncular a later stage. Both forms may coexist.
Infiltrating, diffuse
Limited, localized
Stenosis or obstruction
Hypoplasia, aplasia, hyperplasia
Membrane; congenital spur
Dilatation
Localized (aneurysm)
Diffuse (ectasia)
Each vascular malformation is subclassified into extratruncular and truncular forms, based on its embryologic stage of development (see Box 170.1 ). , The clinical behavior of every vascular malformation is dependent on its embryologic characteristics, closely linked to the stage of embryogenesis when arrested development occurred. The varied embryologic characteristics result in a wide range of clinical presentations, unpredictable clinical courses, and erratic responses to treatment, along with potentially high rates of recurrence. The embryologic subclassification of CVMs into truncular and extratruncular allows clinicians to predict the clinical course, response to treat ment, and likelihood of recurrence. , Further information on their distinctive characteristics is included in subsequent sections.
Another classification system, introduced by Mulliken et al., is based on the hemodynamic status of the CVM and provides an excellent guideline for clinical management. In the ISSVA (International Society for the Study of Vascular Anomalies)/Mulliken classification, all CVMs are divided into two groups, depending on blood flow: fast-flow lesions and slow-flow lesions. This helps in understanding the complex nature of the hemodynamic status of a CVM. It also accommodated old eponym/syndrome-based classification. Currently, many specialists have adopted parts of this classification system and combined it with the Hamburg classification to enhance the efficacy of overall clinical implication.
Due to confusing nomenclatures and definitions in times past, epidemiologic data often misstate the true incidence and prevalence of CVMs. In a comprehensive study of the incidence and geographic distribution of congenital vascular anomalies, Kennedy published data based on 238 studies reporting more than 20 million births. The overall incidence of CVM was 1.08%, ranging from 0.83% in data obtained from hospital records, birth certificates, and retrospective questionnaires to 4.5% in data from intensive examinations of children. This study highlighted the variability in reporting methods due to differences in terminology and inconsistent diagnostic criteria.
In a large study sponsored by the World Health Organization, Stevenson and colleagues surveyed 426,932 live and stillborn births and reported an overall frequency of major and minor malformations of 12.7% in single births and 4.6% in multiple births.
Tasnadi et al. reported overall incidence of the CVM in 1.2% among 3573 3-year-old children they studied. Among them, they found VM and/or AVM in 0.45%, CM (port-wine skin lesion) in 0.42%, LM/primary lymphedema in 0.14%, and mixed CVM showing phlebectasia, nevus and limb-length discrepancy in 0.34%.
CVMs have a male-to-female ratio of 1:1 and VMs are generally known as the most common type. Among VMs, the extratruncular type is the most frequent lesion, and presents in either diffuse or localized forms. Eifert et al. reported on the prevalence of deep venous anomalies/truncular VMs among 257 VMs; phlebectasia was the most frequent (36%), followed by aplasia or hypoplasia of the deep venous trunks (8%) and venous aneurysms (8%). However, more than 70% of the CVMs are mixed, and these complex forms can include arterial, capillary, venous, or lymphatic elements as well. When both extratruncular LM/lymphangiomas and truncular LMs/primary lymphedema are combined, the overall incidence is equal or higher to those of the VM.
During the first half of the 20th century it was believed that all congenital malformations were inherited, and the terms congenital and inherited were used interchangeably. A remarkable step forward was the finding that chromosomal abnormalities can cause certain types and combinations of congenital malformations
A number of genetic and chromosomal abnormalities have been implicated in the development of CVMs. These have been associated with: (1) exposure to damaging chemical compounds during the first trimester of pregnancy; (2) infections such as rubella, cytomegalic inclusion disease, herpesvirus, and toxoplasmosis; (3) thalidomide and other drugs such as aminopterin, cyclophosphamide, quinine, anticonvulsant drugs, cortisone, and corticotropin; and (4) alcohol, tobacco, and cocaine abuse. Maternal diseases and exposures including goiter, diabetes mellitus, thyroid disease, tuberculosis, hypoxia, and carbon monoxide and lead poisoning have also been identified as potential causes of CVMs. , ,
Although most CVMs are sporadic, autosomal dominant inheritance has also been confirmed to cause various CVMs based on genetic study of families to identify mutated genes. These mutated genes that have an important role in angiogenesis encode tyrosine kinase receptors and intracellular signaling molecules in some patients. The endothelial-specific angiopoietin receptor TIE2/TEK, located on 9p21 was identified to cause familial mucocutaneous CVMs. Further studies have shown that somatic mutations in angiopoietin receptor gene TEK resulted in single or multiple VMs and led to loss of TIE2 receptor function and upregulated expression of other vascular endothelial growth factors such as βTGF and βFGF, which exacerbated the severity of the lesion.
Vascular endothelial growth factor (VEGF) has also been found to induce penetration of capillary vessels into the avascular epidermis. A defective response of endothelial cells to VEGF as the consequence of abnormal signaling of VEGF receptors results in CVMs if the differentiation is abnormal and there is an arrest in the development of normal vascular tissue. The persistence of the embryonic vascular system also causes additional abnormal development.
More recently a high susceptibility locus within the HLA locus on chromosome 6p21.32 has been identified with a de velopment of truncular VM. Due to deletion or duplication, the number of copy number variations is significantly associated with an increased chance of developing truncular VMs. The region contains 211 known genes. In addition, the increased expression of matrix metalloproteinase-9 in intramuscular VM lesions suggests that VMs have the ability for angiogenesis to provoke invasive growth while expanding slowly due to the increase in hydrostatic pressure. Progesterone receptors in VMs are also suspected to cause lesion progression when hormonal levels change.
Hereditary hemorrhagic telangiectasia (HHT) is now confirmed as an autosomal dominant condition caused by a “loss-of-function mutation” in the genes encoding activin receptor-like kinase-1 (ACVRL1) and endoglin (ENG), specialized transforming growth factor-beta (TGF-β) superfamily receptors. Although these are CVMs, these lesions develop progressively in the brain, lungs, liver and intestine, and are generally diagnosed at a later age.
CM–AVM syndrome (combined capillary malformation and AV malformation) is another autosomal dominant condition caused by RASA1 mutations with the AVM affecting the brain, spine, face and extremities. These familial/syndrome-based AVMs associated with RASA1 mutations behave differently from the sporadic type and remain relatively stable with minimal progression, although the symptomatology varies with anatomic location.
The AVM associated with Parkes Weber syndrome (PWS) is also associated with the RASA1 mutation, but these AVM lesions generally have a slow progression as a part of PWS in comparison to the AVM belonging to sporadic type. Together with other vascular malformation components, VM and LM, it often results in marked tissue overgrowth of affected muscle, bone and subcutaneous fat. As a part of syndromes of tissue overgrowth (e.g., Cowden and Banyan–Riley–Ruvalcaba syndromes), PTEN mutations are also responsible for the focal tissue overgrowth in hamartomas that frequently contain AVMs. AVMs caused by PTEN mutations are the most aggressive type of AVM, recurring rapidly following therapy with the tendency to develop new AVM lesions at different sites.
When the embryo begins its exponential growth, an equivalent growth of the vascular system is necessary for the proper development of the fetus. If this growth does not occur there is a high risk of CVM. The long, complex process of angiogenesis increases the risk of developmental defects in comparison to other tissues from the same mesodermal origin, such as bone, muscle, and connective tissue.
Embryonic blood vessels originate from the blood island of Pander, which represents the masses of vasoformative cells that undergo a complicated process of evolution. Undifferentiated capillary plexus becomes the initial primitive vasculature; subsequently, some capillaries undergo regression and coalescence with others to form a reticular structure. This reticular structure is efficient to meet the embryo’s needs during the early, rapid-growth stage of embryogenesis. This extratruncular stage occurs before the final truncular stage, during which formation of the actual artery, vein, and lymphatic trunks occurs to complete the differentiation. The characteristics of the embryonic cells change drastically between the extratruncular and truncular stages. , , The clinical behavior of the vascular defect depends on the stage at which arrested development occurs. Any defect occurring before the truncular stage maintains the embryonic characteristics of mesenchymal cells origin.
Extratruncular CVMs represent arrested development during early embryonic life, while the vascular system is in the reticular stage. , Therefore, these embryonic remnants of mesodermal origin retain the characteristics of mesenchymal cells (angioblasts). They have the potential to grow and proliferate when stimulated internally (e.g., by menarche, pregnancy, hormonal release) or externally (e.g., by trauma, surgery). Therefore, all extratruncular lesions, regardless of type, carry a significant risk of recurrence after inadequate treatment that provokes a dormant or silent lesion to grow rapidly. , By nature, an extratruncular lesion behaves like an infiltrating process, invading the surrounding structures. Because bones, muscles, soft tissues, and blood vessels all originate from the same mesoderm, there is no plane of partition between them. Therefore, such a lesion, whether diffuse or circumscribed, produces a pressure effect on the surrounding tissues, in addition to the hemodynamic impact on the affected vascular system.
Truncular CVMs arise when arrested development occurs later, during the vascular trunk formation stage of embryonic development. , Truncular lesions have lost the embryonic characteristics of the mesenchymal cells, together with the potential to grow and proliferate. These lesions do not carry the same risk of recurrence as extratruncular lesions, but they are generally associated with more serious hemodynamic consequences than their extratruncular counterparts. Based on their pathoanatomic condition, truncular lesions are further divided into obstructive and dilated lesions. , All truncular lesions appear as formed vessels with various degrees of developmental defects, ranging from incomplete or immature lesions (aplasia or hypoplasia) to overdeveloped lesions (hyperplasia). A truncular VM may present as a persistent fetal remnant (truncal) vessel that does not involute normally (e.g., sciatic vein, marginal or embryonic vein) or as a defective vessel trunk (e.g., vein web, venous aneurysm/ectasia) to result in various extents of stenosis to obstruction as well as dilatation. , ,
In terms of pathophysiologic significance, CVMs can be divided into two types: those with a mechanical impact on surrounding structures, producing a compression effect, and those with a hemodynamic impact on the affected circulation. The relative significance of these variants depends on the type of CVM as well as its embryologic subtype.
In general, all extratruncular lesions have various degrees of mechanical and hemodynamic impact. The initial mechanical impact is generally of little significance, because compression is the result of steady growth of the infiltrating lesion. Rapid expansion of the lesion increases the mechanical impact by compressing surrounding tissues. A simple lesion can result in serious damage if it is located near vital structures (e.g., those involving breathing, sight, hearing, eating) and can even become life-threatening (e.g., airway obstruction). In general, the hemodynamic impact is usually far more serious than the mechanical impact. High flow CVMs (e.g., AVMs) have greater hemodynamic impact than low-flow lesions (e.g., VMs) because the pathoanatomic structure of arteriovenous shunting does not have the resistance of the normal capillary system.
All truncular lesions, regardless of type, have a mainly hemodynamic impact. This hemodynamic impact is variable, depending on the type of CVM. For example, the impact of a truncular LM that manifests clinically as primary lymphedema remains limited to the lymphatic transportation system, seldom involving the accompanying venous system.
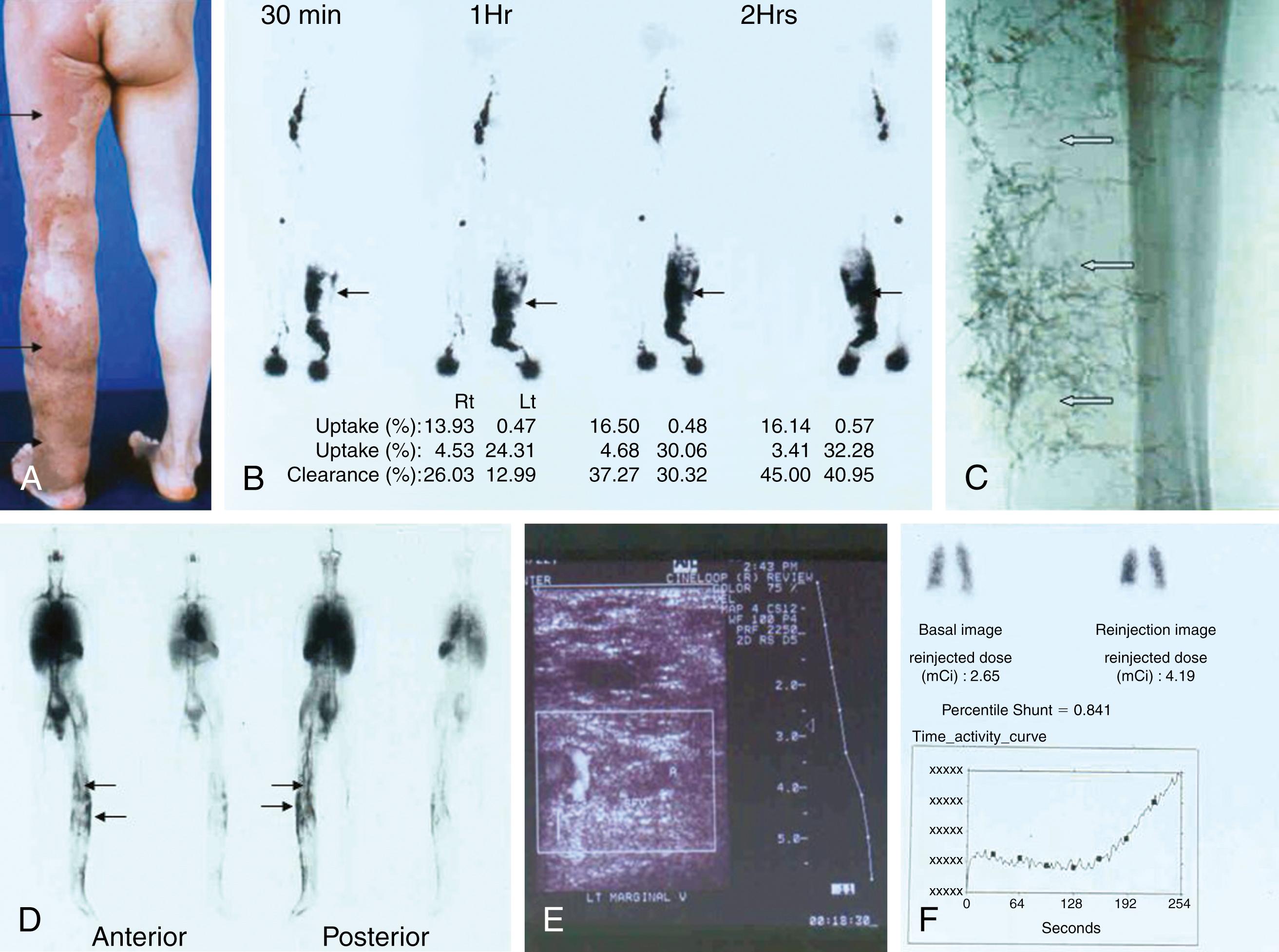
In contrast, a truncular VM affecting the lower extremity (e.g., femoral vein aplasia or hypoplasia) often presents with more serious hemodynamic impacts, resulting in chronic venous insufficiency. When venous insufficiency is combined with lymphatic insufficiency caused by coexisting truncular LM/primary lymphedema, its pathophysiologic impact (e.g., phlebolymphedema) is substantially increased.
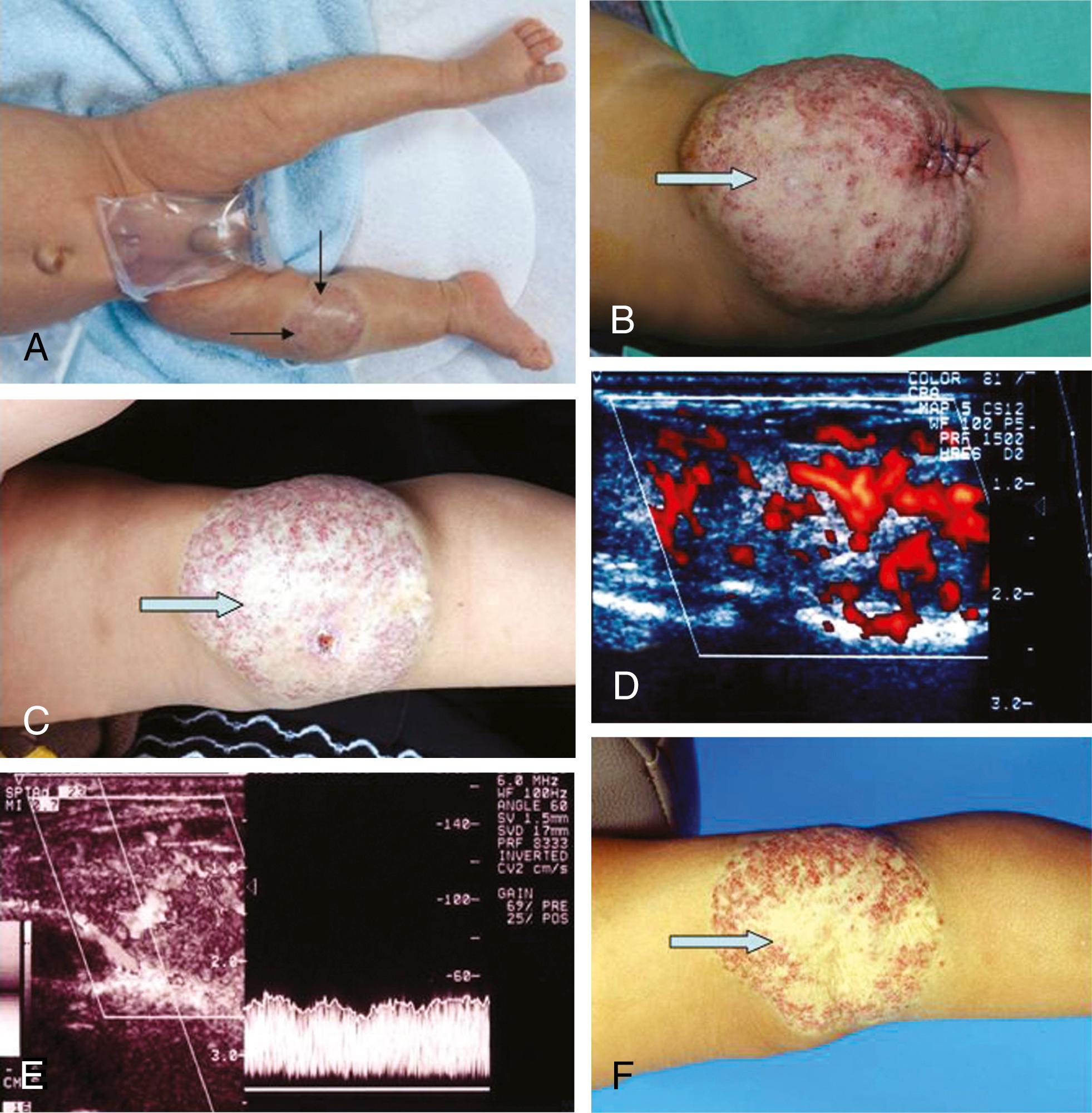
Among truncular VMs, , , a venous aneurysm is a unique condition, defined as a segmental venous dilatation 1.5 to 2 times the normal vein size. This is potentially a source of venous thrombosis and pulmonary embolism, especially when the deep system is involved. The most common symptoms are a painful mass and localized swelling. In some cases, a massive pulmonary embolism is the first symptom. In children, venous dilatation is most common in the internal jugular vein and is most noticeable during crying or other physical effort. The incidence of venous aneurysms is similar in men and women. Gillespie et al. reported on 39 venous aneurysms in 30 patients, the majority located in the deep venous system of the lower extremities, with five in the internal jugular vein and four in the superficial venous system. Diagnosis was made by phlebography, color-flow duplex scanning, and/or MRI. The size of the aneurysms ranged from 1.7 to 6 cm. Three patients had deep venous thrombosis, and three had pulmonary embolism.
Truncular AVMs, which are true fistulous “direct” connections between the arterial and venous system with no nidus interposed, have the most powerful impact on the vascular system although they are extremely rare. The shunting of arterial blood to the venous system may result in a substantial degree of arterial steal from the distal arterial vessels, as well as overloading the proximal and distal venous system, especially when the lesion is located in the extremities. , , , This may result in chronic venous hypertension distal to the lesion and high output cardiac failure due to the progressive increase in central venous overloading.
The pathophysiologic impact of a CVM is not limited to the primary lesion. It may have a significant secondary impact on related tissues or organs. For example, an intraosseous CVM has an important effect on the musculoskeletal system by stimulating the epiphyseal plate and inducing abnormal long-bone growth, resulting in leg-length discrepancy. Compensatory scoliosis with or without pelvic tilt can have clinical consequences on several physiologic functions (e.g., abnormal gait, cardiopulmonary impingement). Other systems, such as the gastrointestinal, cardiopulmonary, and genitourinary systems, may also experience secondary pathophysiologic effects because of the location of the CVM (e.g., chylo-reflux). ,
The presentation of extratruncular CVMs depends on the vascular system involved, the location of the lesion, and whether the lesion is localized. They may affect the arterial, venous, lymphatic, or capillary system either as an independent lesion (CM, VM, LM, AM, AVM), or they may affect two or more of these vessel types (HLM). Each of these vascular systems may also be the site of truncular malformations, such as arterial aneurysms and ectasias, venous aneurysms and phlebectasias, and congenital/primary lymphedema when the defective development should occur in the late stage of embryogenesis. Hence, the variations in presentation are countless. An extratruncular CVM lesion may appear to be localized to one small area on initial assessment but, like the tip of an iceberg, it turns out to be an extensive malformation of the deep tissues revealed only by further diagnostic assessments. A complete clinical examination is usually sufficient to establish a diagnosis. However, diagnosis may be difficult in small children, and it may take some time for the lesion to become fully identifiable. , , ,
A CM appears as a port-wine stain on the skin with a distinctive color; it is different from the common vascular birthmark known as nevus flammeus neonatorum. Birthmarks are pinkish discolorations affecting the nuchal region (“stork bite”) or the face along the eyelids, forehead, or lips (“angel’s kiss”). In contrast, CMs can present anywhere on the body and can range from small localized lesions to extensive lesions covering the skin in multiple areas. CMs are often accompanied by other abnormal physical findings beneath the skin, heralding other CVMs. For example, a CM in the face is a warning sign for an intracranial CVM known as Sturge–Weber syndrome, with ipsilateral ocular and leptomeningeal vascular malformations.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here