Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The OMT (Oberg, Manske, Tomkin) classification divides congenital tumorous conditions of the upper extremity into four broad categories based on the tissue involved: vascular, neurological, connective tissue, and skeletal.
While the great majority of congenital lesions are benign, many can cause local destruction, systemic dysregulation, and life-long morbidity.
The clinician should undertake a careful and methodical approach to evaluating, diagnosing, and treating a child with a tumorous condition. Many of the lesions presented herein can mimic others. A multidisciplinary approach is advocated and usually results in the most optimal outcomes.
Vascular anomalies of the upper limb are divided into tumors (including hemangiomas) and malformations. Hemangiomas proliferate and then spontaneously involute, whereas malformations tend to expand slowly over time.
Nerve tumors in children commonly present as peripheral nerve sheath tumors or adipose lesions of the nerve and are usually associated with other major comorbid conditions such as neurofibromatosis.
Connective tissue tumors are rare and difficult to diagnose because they mimic each other and different rare conditions. Index of suspicion should be high as seemingly aggressive lesions are usually benign, yet seemingly benign lesions can be sinister.
The most common skeletal tumorous dysplasias are the osteochondromatoses and enchondromatoses. These conditions display significant morbidity and carry a high risk of malignant transformation.
In carefully selected patients, surgical resection of the aforementioned conditions can be predictably performed with thoughtful preoperative planning and adherence to basic principles. Technical refinements and pearls are outlined.
Significant variation and overlap coexist simultaneously in the etiology, morphology, and molecular basis of many of these conditions. New discoveries in the biochemical pathways responsible for many lesions have revolutionized care today, and hold promise for future medical therapy.
![]() Access video content for this chapter online at Elsevier eBooks+
Access video content for this chapter online at Elsevier eBooks+
Congenital and pediatric tumorous conditions represent a broad saet of mostly rare and unusual, but somewhat inter-related, conditions. Although pathologically distinct, many of these conditions share similarities and overlap in their clinical manifestations and molecular underpinnings. A common theme is their persistent entrenchment in confusing and inaccurate terminology and outdated classification systems. Because diagnosis and (hence) treatment hinges on accurate and precise nomenclature, much recent work by the scientific community has focused on clarifying, reclassifying, and more precisely defining these conditions – especially as their molecular mechanisms are elucidated.
Another common theme shared by these conditions is that many mimic each other and other rare lesions. The clinician must have a critical eye and remain suspicious when the clinical picture is unclear. Nearly half of patients referred to a specialized vascular anomalies center will have an incorrect initial diagnosis. For most of the conditions outlined herein, the ideal environment for evaluation and decision-making is a multidisciplinary clinic with experienced clinicians, including radiologists, dermatologists, surgeons, hematologists/oncologists, and others.
The last common theme seen throughout the ensuing discussions is generalized or localized overgrowth and the dysregulation of molecular signaling pathways that control it. A few of the major growth-modulating molecular pathways have been precisely delineated in the last decade, and newer medical treatments that target these pathways have been developed and are being fervently studied. A few of the conditions discussed share similar dysregulated pathways, and thus, one targeted treatment may be applicable to multiple conditions. This arena of molecular research has exploded and holds significant promise for the future of many of these conditions.
This chapter will review congenital and pediatric tumorous conditions that specifically apply to the upper extremity. The first few sections focus on the general approach and treatment of a child with a growth, outlining common considerations and principles. Subsequently, each anatomically separate category of vascular, neurological, connective tissue, and skeletal will be discussed with a discussion of individual conditions within these categories. The common themes discussed above will be echoed throughout and serve to unify these seemingly disparate entities.
The temporal distinction between pediatric (occurring before skeletal maturity) and congenital (present at birth) may be blurred at times. Although a few of these conditions present at birth or in infancy, most patients discussed will either have clinically silent lesions or have the predilection to develop them, not overtly manifesting until later in childhood or adolescence.
A clearly thought-out, step-wise approach to diagnosis and evaluation is necessary. The most important underpinning of safe and efficacious care is establishing the correct diagnosis. Many of these conditions are rare, and the nomenclature and nosology are inconsistent and confusing. Furthermore, many of these conditions mimic each other or different rare diseases; thus making an accurate diagnosis paramount.
The initial time and energy devoted to determining a high-probability diagnosis pays substantial dividends in avoiding complications, unfavorable outcomes, and distraught patients. Because of the high degree of clinical overlap between these conditions, it is not uncommon to overtreat innocuous lesions that seem aggressive or undertreat sinister lesions that seem harmless. A high-probability diagnosis begets high-probability treatment. One way to ensure this is by having multiple experienced clinicians in a multidisciplinary clinic and/or scheduled conference to oversee diagnoses and treatment recommendations. If there are doubts about diagnosis, tissue biopsy should be pursued.
As always, the patient should be assessed with a thorough history and physical examination. In the younger age group, the history is often more reliable than the physical examination. This population does not follow commands predictably, is easily distracted, and cannot articulate symptoms well. It is particularly challenging to decipher subtle sensory or motor neuropathies. The surgeon should rely heavily on parental descriptions, especially if they relate a change in function or child behavior. Having the child play with simple toys in the office can help uncover functional upper extremity deficits. The patient’s pediatrician may have a unique perspective especially if there has been a trend in symptoms. Lastly, an important but oft-neglected option is to serially examine the patient over several clinic visits.
The history should elicit time of onset, growth rate, character of pain, functional symptoms, sensory disturbance, and other relevant findings. The physical examination should focus on the area of concern first, then the area distal. Characteristics of the lesion to interrogate include: location, size, contour, tenderness, adherence to surrounding tissues, borders (clear or indistinct), compressibility, and the presence of distal changes including: sensory and motor disturbance, contractures, skin changes, deviations and malalignments, and other neurologic findings such as Tinel’s sign. In many cases, it is worthwhile to obtain baseline functional data including length and circumference of the affected and unaffected limbs early in life and to follow these parameters as the child grows.
When evaluating a patient with a mass, valuable details of clinical history and examination can often be missed. This issue is particularly problematic in younger patients who may withdraw and not comply with examination. It is helpful to create a formal list of symptoms and physical signs to check, like a surgical checklist. The list should include items related to history (e.g. onset, duration, growth rate), observation (e.g. digital cascade, bulges, color, visible pulsation), and direct examination (e.g. Allen test, active range of motion, temperature, pitting edema). A systematic list-based approach ensures that no important details are missed.
The great majority of vascular anomalies have skin manifestations and can be diagnosed by history and physical examination alone. However, the remainder of tumorous conditions outlined herein will likely require imaging and/or tissue evaluation. The ideal imaging technique will vary depending on the lesion, with considerable variability even within one diagnosis group (e.g. vascular lesions). The initial radiological test for most pediatric soft tissue lesions defaults to ultrasound. Ultrasound is ubiquitous, rapid, and requires no sedation in smaller children. It distinguishes fast-flow and slow-flow vascular anomalies, often being the only imaging needed to differentiate between a vascular tumor and a vascular malformation. Ultrasound can also rule out vascular lesions, thus making it helpful for soft-tissue masses without skin manifestations. In multidisciplinary vascular anomalies clinics, the ultrasound study can be done by a qualified interventional radiologist using a portable machine, thus saving time and cost, and yielding immediate answers.
Plain radiography is usually the best initial imaging test for suspected bony lesions, or soft-tissue lesions that may involve the bone (e.g. venous malformation or infantile myofibromatosis). Plain radiographs are needed in case of joint deviations or malalignment. Many bone tumors tend to have classic radiographic appearances. Ominous features such as bone destruction, lysis, scalloping, periosteal reaction, matrix ossification, and soft-tissue invasion should alert the clinician to the presence of a malignancy. Although primary congenital malignant tumors are extremely rare, longitudinal malignancy risk is high in patients with congenital dysplasias such as neurofibromatosis, enchondromatosis, and osteochondromatosis.
Computed tomography (CT) can delineate characteristics and degree of involvement of bony lesions. It is also helpful for detecting interosseous involvement or skeletal distortion caused by vascular malformations. CT angiography is an important adjunct in understanding difficult vascular malformations that may envelope end-arteries in the upper extremity. Newer rendering techniques can provide impressive 3D images that help the surgeon understand the relationship of the lesion to its neighboring tissues. 3D imaging can also help with pre-surgical planning if bony reconstruction is planned. Lastly, positron emission tomography (PET) scan has become a valuable adjunct in stratifying malignancy risk of peripheral nerve sheath tumors.
Magnetic resonance imaging (MRI) with contrast (gadolinium) is the gold standard for characterization of soft tissue and vascular lesions. Fat-suppressed T1-weighted images with contrast can differentiate between a vascular tumor and a vascular malformation. T2-weighted sequences show vascular channels well and can guide minimally invasive treatment. MRI can distinguish between lesions that mimic venous malformation, such as fibro-adipose vascular anomaly (FAVA) or intramuscular hemangioma (Enzinger type), both of which most commonly occur in the extremities. It is also useful in characterizing neurological and connective tissue tumorous conditions, as will be discussed later.
Angiography is indispensable in the diagnosis, assessment, and treatment of fast-flow vascular malformations. The vascular architecture of the malformation can be precisely determined and arterial suppliers and draining veins identified. Indirect and direct phlebography techniques are also sometimes useful for diagnostic and therapeutic management of the venous malformations of the extremity.
Despite the wide variability of lesions discussed in this chapter, certain principles of treatment remain true. There are lesions that are amenable to surgical treatment, and others that are best approached non-surgically. The surgeon, in consultation with the patient/family and other members of the team, can help inform these decisions. Lesions that respond best to surgery are limited in size and adjacent tissue involvement. Excising these lesions can sometimes be curative; in contrast, debulking large or extensive lesions usually provides temporary improvement due to a high likelihood of recurrence or continued expansion as the child grows.
The surgeon should always consider the long-term effects of an operation, especially if a complete resection is not possible. Re-operating on lesions such as vascular anomalies can have substantially decreasing benefits with exponentially increasing risk after each reoperation. If the lesion came back once despite an extensive operation, it is likely to come back again. Many vascular malformations can never be absolutely eradicated; re-expansion or recurrence is the rule. This is an important consideration in preoperative planning, patient counseling, and future interventions. The fourth dimension (time) is a harsh judge of surgical decision-making. The ideal way to cultivate a sense of future expectations is with experience and meticulous documentation. The surgeon who takes on rare lesions in children is obligated to measure, photograph, and record obsessively, and assess outcomes honestly.
Surgical principles gain acceptance with time and must be periodically reassessed and refined, particularly in rapidly advancing fields. In contrast, techniques change frequently and are modulated by technology. The surgical principles outlined in Table 37.1 evolved mainly with experience in vascular anomalies but apply equally well to other lesions in the extremity. Caveats and pearls of surgical dissection in the hand and digits are outlined in Tables 37.2 and 37.3 .
| Preoperative planning | Delineate anatomy and goals of procedure clearly and correlate with physical examination and imaging Provide clear counseling to parents and children |
| Precise instrumentation | Utilize fine instruments and magnification |
| Hemostasis | Use tourniquet control and keep field as dry as possible to enhance visualization |
| Well-defined borders | Outline the extent of resection and abide by it As much as possible, stay within anatomic boundaries |
| Incision placement | Design high mid-axial incisions in the digit and zigzag in the palm Avoid unnecessary scarring and consider future interventions |
| Thorough dissection | Limit procedure to a well-defined area but dissect/excise thoroughly For debulking, dissect no more than half of a digit or limb at a time |
| Do | Preserve superficial veins Reconstruct arterial defects Use drains liberally Immobilize postoperatively Be prepared to amputate symptomatic, non-functional or painful digits or limbs |
| Do not | Dissect vascular anomalies intraneurally Dissect partially within muscle groups (e.g. VM) Close under tension |
| Follow-up | Be compulsive about follow-up Document extensively and follow patients long term |
|
||
 |
 |
 |
| Diffuse VM of the palm, all vital structures identified and tagged | Motor branch of the ulnar nerve over green background | Dissection/resection complete |
|
|||
 |
 |
 |
 |
| Diffuse VM | Ulnar neurovascular bundle | Radial neurovascular bundle | Closure showing mid-axial approach |
Immobilization is an important tenet of care after surgical treatment of extremity lesions in children. Immobilization allows time for the soft and hard tissues to heal while avoiding motion-induced pain and swelling. While the risks of immobilization can easily outweigh its benefits in adults, the opposite is true in children. Children do not develop the same degree of stiffness and adhesions as adults, whereas they are more likely to disrupt a suture line or a skin graft with excess motion. We recommend immobilization for the great majority of extirpative procedures in the upper extremity, especially if flaps or skin grafts are utilized.
Rehabilitation can begin immediately after immobilization is discontinued. If healing of the soft and hard tissues is complete, there should be very few restrictions. In contrast to adults who start motion early and ramp up slowly, the ideal paradigm in children is to restrict early but allow near full return to activities once healing is complete.
Two to three weeks of immobilization is the minimum after extirpative procedures in the pediatric upper extremity. The authors use short-arm casts in older children and adolescents and long-arm cast in infants and young children. If significant postoperative swelling is expected, a non-circumferential temporary splint is placed first and then exchanged for a cast a few days later.
During the past five decades, there has been a steady, almost exponential increase in our knowledge of vascular anomalies. A biological classification system has evolved and is under constant refinement. Attentive physical examination and readily available imaging will yield an accurate diagnosis in most cases. A multidisciplinary team can offer individualized treatment with some degree of predictability. Improvements in minimally invasive radiological procedures have dramatically transformed treatment of these lesions. With discovery of biological pathways responsible for these lesions, widening application of medical therapies shows promising outcomes. Nevertheless, in many patients, surgical treatment is often the best remedy and should not be viewed as the last resort. Carefully planned and well-orchestrated procedures and rehabilitation can yield remarkable outcomes.
In the late 1970s, Mulliken and Folkman proposed a working hypothesis that vascular anomalies tissue could be distinguished by its cellular characteristics. Surgical specimens were analyzed by selected histological stains, electron microscopy and autoradiography (uptake of tritiated thymidine into DNA). Two types of lesions were present: those that showed endothelial hyperplasia, called hemangiomas, and those that did not, called malformations. A binary schema based on biologic activity was proposed and accepted by the International Society for the Study of Vascular Anomalies (ISSVA) in 1996. This system has been corroborated by both radiological studies and immunohistological staining. Nevertheless, the classification system has been changed many times and continues to evolve. A vascular lesion may be moved from one category to another or may be discarded completely with expansion of clinical experience and basic science investigation. For the past 30 years, this system has been very helpful to the upper limb surgeon in the proper diagnosis and management of these lesions. For the latest revision, the reader is referred to the ISSVA website.
The classification system divides vascular anomalies into two broad categories: vascular tumors and vascular malformations. Vascular tumors (e.g. hemangiomas) exhibit rapid cellular hyperplasia. In contrast, malformations represent a localized defect in vascular morphogenesis. Hemangiomas have a biphasic growth curve with rapid proliferation early in life and subsequent involution. Malformations have a propensity to slowly expand throughout life. Vascular malformations can be broadly classified into slow-flow and fast-flow lesions. Slow-flow lesions include capillary, lymphatic (LM), venous (VM) malformations or mixed lesions. Fast-flow lesions include arteriovenous malformations (AVM) and others.
Vascular anomalies can occur anywhere in the hand and upper extremity and can involve all components of soft tissue, bone, and joint. Table 37.4 ![]() presents typical signs and diagnostic modalities for more common lesions. With a few exceptions, hemangiomas are usually absent at birth or present as a small stain. Patients present with a growing, vascular, high-flow lesion in early infancy. These lesions have a deep red appearance and strong Doppler signals throughout. In contrast, vascular malformations can present any time in life. Although they are likely present at birth, they may be clinically silent for many years. Vascular malformations can be associated with various overgrowth conditions, which particularly affect the limbs. A few rare but pertinent conditions are outlined in Table 37.5 .
presents typical signs and diagnostic modalities for more common lesions. With a few exceptions, hemangiomas are usually absent at birth or present as a small stain. Patients present with a growing, vascular, high-flow lesion in early infancy. These lesions have a deep red appearance and strong Doppler signals throughout. In contrast, vascular malformations can present any time in life. Although they are likely present at birth, they may be clinically silent for many years. Vascular malformations can be associated with various overgrowth conditions, which particularly affect the limbs. A few rare but pertinent conditions are outlined in Table 37.5 .
| Diagnostic workup | Condition | ||
|---|---|---|---|
| Hemangioma | CM | VM | |
| Physical examination | ++++ | ++++ | ++++ |
| Ultrasound | +++ | − | + |
| MRI, MRV, MRA | ++ | − | +++ |
| Radiographs | − | − | ++ phleboliths bone |
| CT | + | − | + |
| Angiography | − | − | − |
| Biopsy | + | − | + |
 |
 |
 |
|
| LM | Combined | AVM | |
| Physical examination | ++++ | ++++ | ++++ |
| Ultrasound | + | + | +++ |
| MRI, MRV, MRA | +++ | +++ | +++ |
| Radiographs | + bone | + bone | + bone |
| CT | + | + | + |
| Angiography | − | − | ++ |
| Biopsy | + | + | + |
 |
 |
 |
|
| Vascular tumors | Vascular malformations |
|---|---|
| Slow-flow vascular malformations | |
|
|
| Fast-flow vascular malformations | |
|
|
Vascular anomalies can present with and/or display wide-ranging local or systemic symptoms and functional sequelae. While some may remain asymptomatic for years, others can cause local problems such as pain, deficits in motion, contractures, neuropathy, chronic wounds, and vascular steal with ulceration and necrosis. Less commonly, large lesions can precipitate systemic consumptive coagulopathy, deep venous thrombosis and embolism, and heart failure.
The etiology of vascular anomalies is not known but is a subject of intense scientific research. More recently, there have been significant breakthroughs uncovering responsible factors in the molecular pathways of a few entities that also mediate overgrowth. The discoveries, in turn, have led to the development of multiple new medications that show promise in treating difficult lesions. For a comprehensive discussion, the reader is referred to other resources.
The full gamut of vascular anomalies and associated overgrowth conditions is beyond the scope of this chapter. The next few sections will outline the more common lesions that can affect the upper extremity and the best approaches for diagnosis and treatment.
Vascular anomalies are complex problems and are best approached by a multidisciplinary team. In our center, a weekly conference and a separate weekly clinic take place where the team meets to discuss individual patients. Representatives from radiology, general (pediatric) surgery, hematology, anesthesiology, plastic surgery, dermatology, and scientists from the laboratory attend. Other specialists such as neurosurgeons, orthopedic surgeons, oral surgeons, ophthalmologists, endocrinologists, and head and neck surgeons are called as needed. A comprehensive approach to patient care is assembled, with immediate availability of ultrasound imaging and expert opinion. This approach has been monumental in the correct diagnosis and treatment of hundreds of patients.
Infantile hemangioma is usually single and involves the upper limb in 15% of cases. The median time of appearance is at age 2 weeks. When dermis is involved, the skin initially appears pink but can evolve to deep red. Deeper lesions may cause a pale, bluish discoloration ( Fig. 37.1 ). This lesion grows very fast during the first 9 months (proliferating phase), begins to shrink by 12 months (involuting phase), and involution is complete by 5 years (involution phase) leaving residual telangiectasias, fibrofatty residuum and redundant, atrophic skin.
Diagnosis can be made by history and physical examination in the great majority of cases. Hand-held Doppler demonstrates fast flow. Ultrasonography shows fast flow, elevated resistance and increased venous drainage. On MRI scans, hemangiomas are isointense on T1, hyperintense on T2, and enhance during the proliferating phase. Rarely is biopsy needed. Immunostaining for GLUT1, a glucose transporter specific to IH, will usually differentiate this lesion.
Most infantile hemangiomas are managed by observation because 90% are small, localized, and do not impair function. Larger lesions that proliferate quickly can sometimes ulcerate and bleed. To protect against this, topical antibiotic cream and a petrolatum gauze barrier may be used. Ulcerated wounds are treated with local wound care and rarely need biologic coverage such as skin grafts. Those that are small but cause functional problems can be injected with steroids or treated topically (see below).
The first-line agent for systemic treatment of infantile hemangioma is oral propranolol. The US Food and Drug Administration approved propranolol for use in proliferating infantile hemangioma in 2014. Propranolol is dosed between 2 ando 3 mg/kg per day unless there are other comorbidities that would require a lower dose. Oral steroids, once the gold standard agent, are now recommended only if there are contraindications or inadequate response to oral propranolol. For superficial lesions with thin dermis, the use of timolol maleate ophthalmic solution has recently been advocated. Reports demonstrate efficacy with minimal adverse effects, but there is concern about safety especially in younger patients due to direct systemic absorption without first-pass filtering by the liver (oral agent). Recommended dosing suggests avoiding more than 1 drop twice daily when treating younger patients.
Surgery in the upper limb during the proliferation phase is indicated only for problematic ulcerations or large lesions interfering with function ( Fig. 37.2 ). During infancy, anesthetic risks are higher and the tumor is fast-flow, increasing risks of blood loss and iatrogenic injury due to poor surgical visibility. After involution, the hemangioma can leave redundant skin with fibrofatty soft tissue. These lesions can be unsightly and, on rare occasion, excess skin redundancy can interfere with function. Small lesions can be excised in one stage, whereas larger ones require a staged approach.
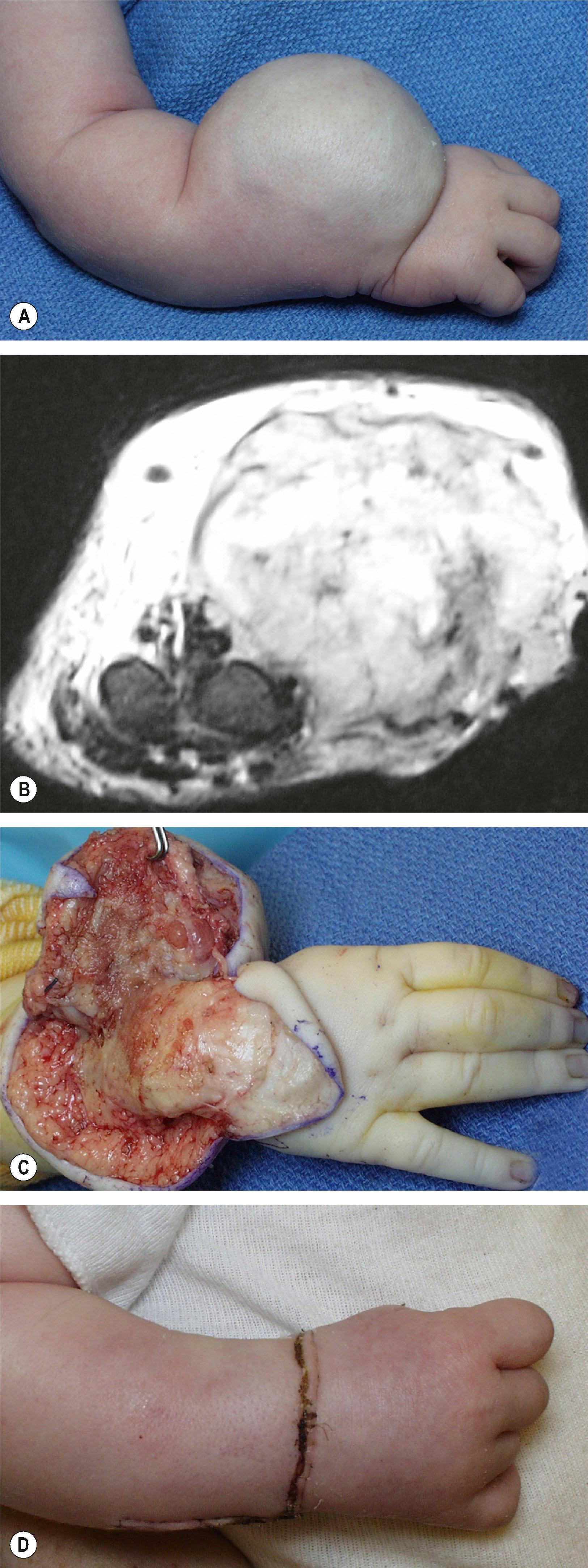
The upper limb surgeon must be familiar with the congenital variants of hemangioma. In contrast to infantile hemangioma, congenital hemangioma (CH) is a rare lesion fully grown at birth that does not exhibit the proliferation and regression so typical of the more common variant. These lesions appear differently with a red-violaceous color, coarse telangiectasias, central pallor, and a peripheral halo. There are two variations of CH: the rapid involuting congenital hemangioma (RICH) and the noninvoluting congenital hemangioma (NICH) ( Fig. 37.3 ). Both lesions are commonly seen in the extremities and have an equal sex distribution. RICH involutes rapidly after birth; 50% of the lesion is gone by 7 months and involution is complete by 14 months of age. Fibrofatty deposits are not present. NICH does not regress and persists as raised, lumpy, plaque-like lesions with a characteristic peripheral halo. A persistent fast flow component remains unchanged.
Diagnosis is made by physical examination, clinical course, and ultrasonography. The history and characteristic appearance of these lesion (halo, central pallor) are the main clues to a diagnosis. CH lesions do not stain for GLUT1.
RICH does not require operative management early in life, as it undergoes accelerated involution. Only large lesions in other parts of the body may need drug therapy. NICH may require systemic propranolol or intralesional corticosteroid injection in problematic regions of the upper limb. Large, lumpy masses within the palm of the hand, interdigital webspaces, or those causing muscle or tendon impairment are best excised under tourniquet control.
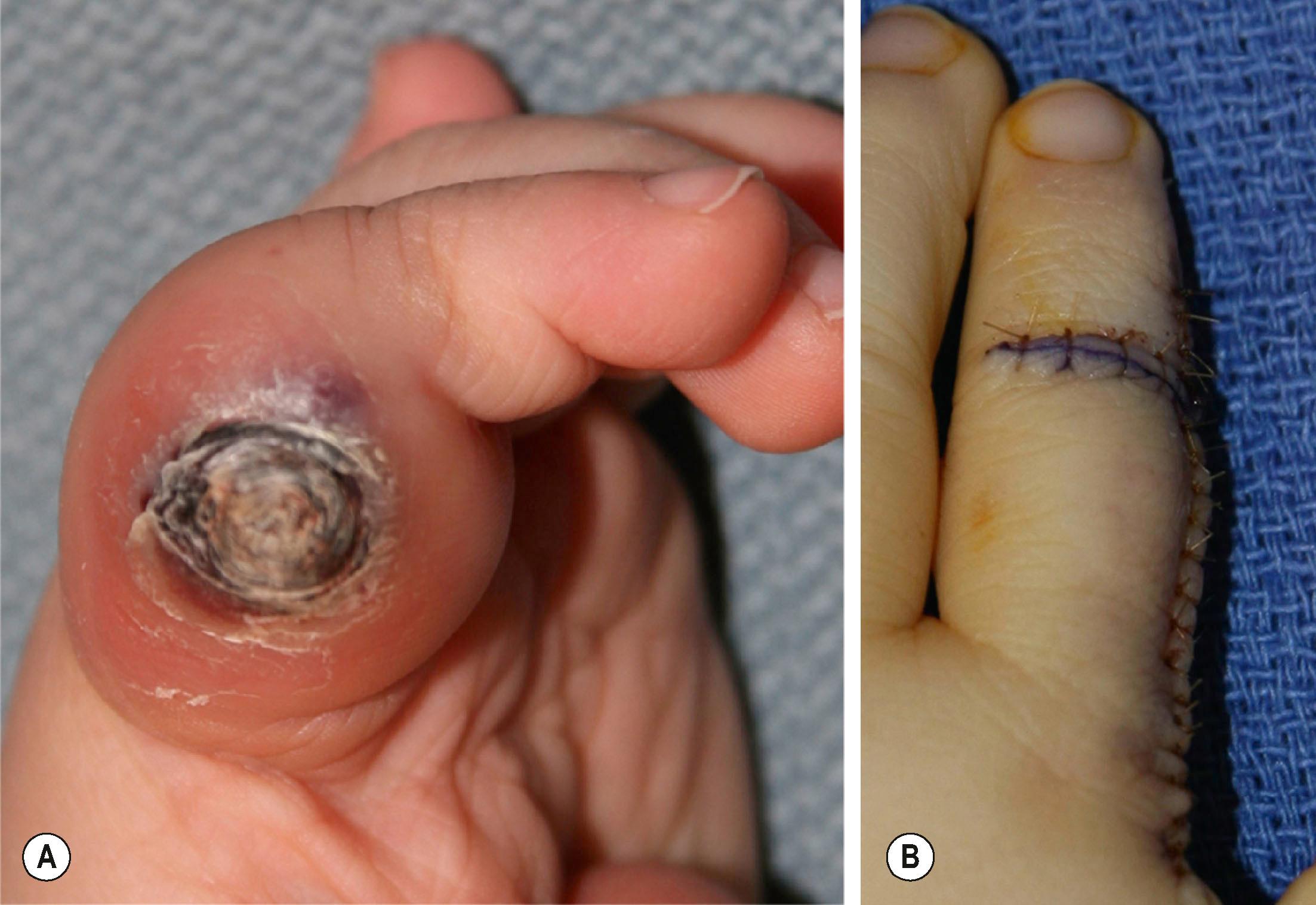
Pyogenic granuloma (PG) is a solitary red papule that grows rapidly, forming a stalk. It has been called a lobular capillary hemangioma. It is small with an average diameter of 6.5 mm. The male to female ratio is 2:1. It commonly involves skin and, in the upper extremity, it is most problematic on the glabrous surfaces of the digits, in nail folds ( Fig. 37.4 ), fingertips, and within webspaces and flexion creases. It appears as a red papule growing on the end of a stalk – though immature lesions can be flat with no stalk. Persistent bleeding and constant ulceration are common. PG is distributed all over the body, including the mucous membranes, cheeks, lips, oral cavity, eyelids, and forehead. Histological examination will differentiate PG from other similar entities.
Patients present with classic symptoms of a red, friable papule with intermittent profuse bleeding. Diagnosis is made by physical examination because the appearance is classic.
Numerous treatments have been described for PG: curettage, shave excision, laser therapy, chemical treatment (silver nitrate), or excision. Complete excision is the preferred treatment. Most lesions are small and can be excised and closed directly. Skin graft coverage may be necessary for large defects.
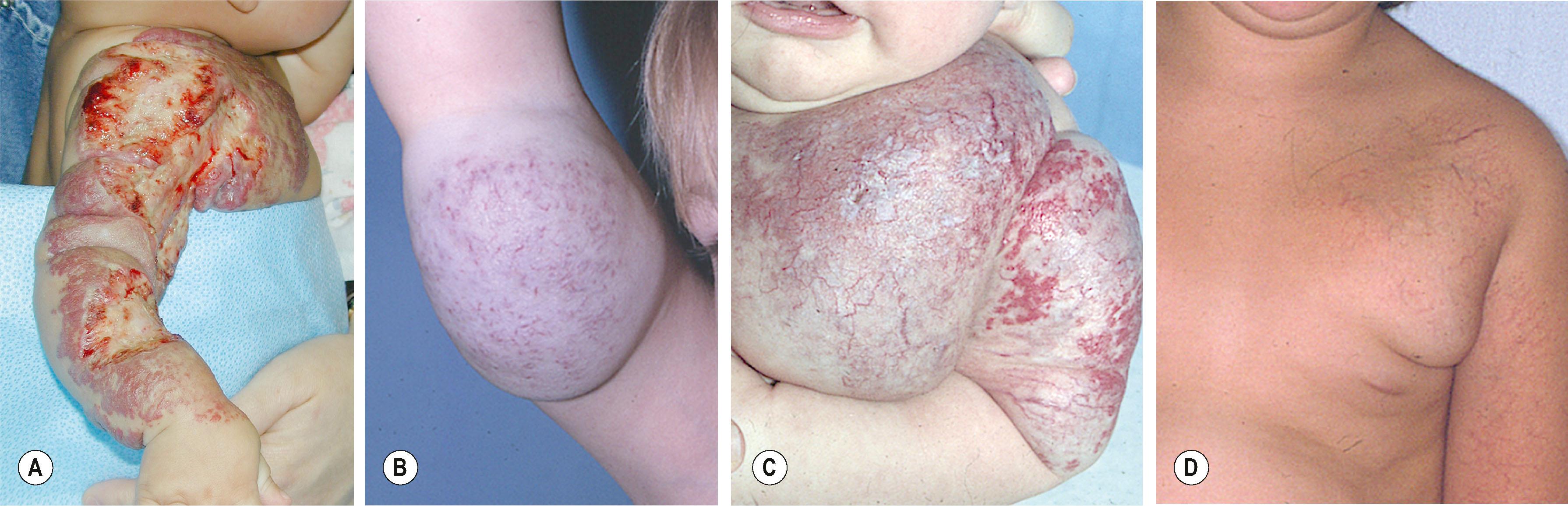
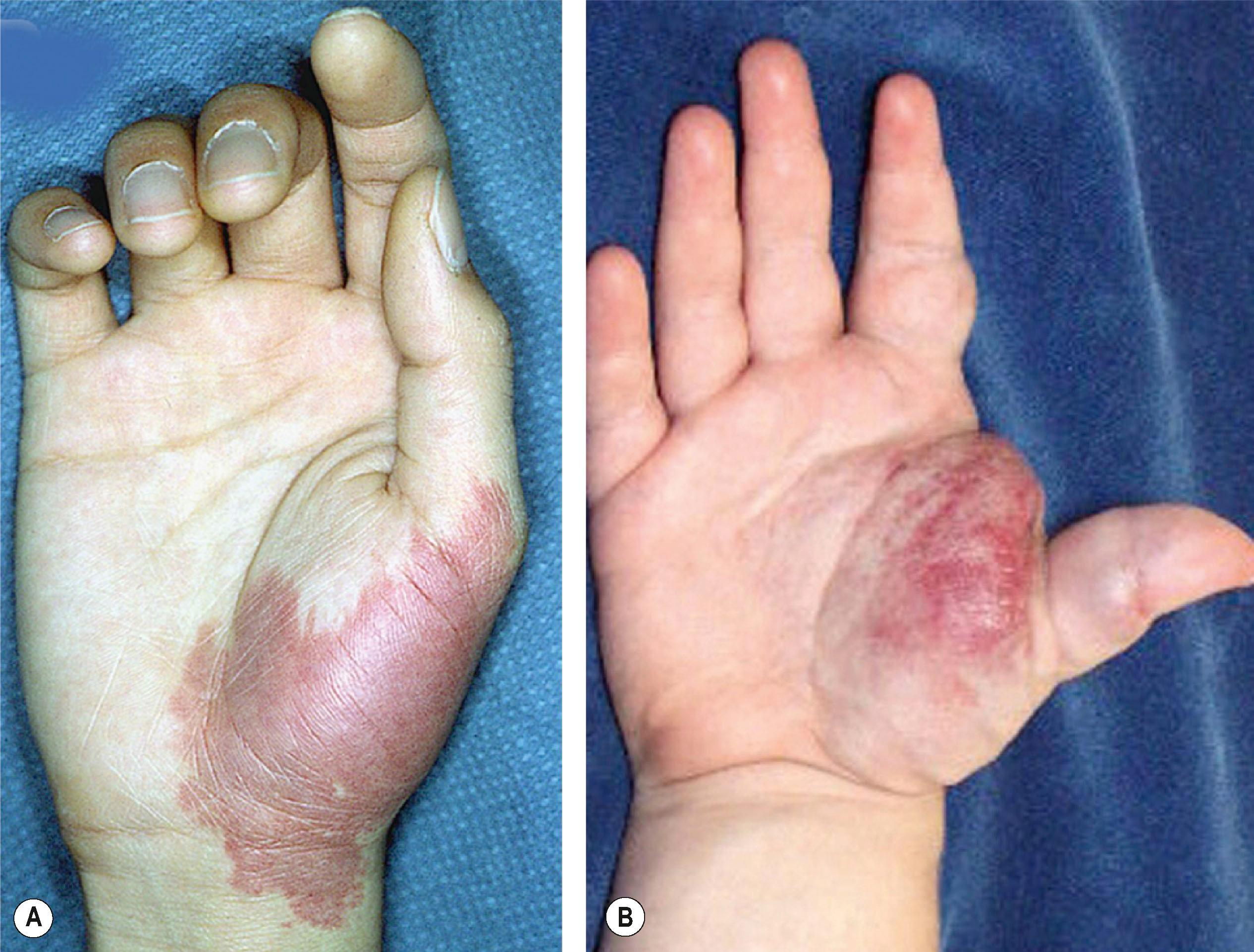
Capillary malformation (CM) represents dilated capillaries in the superficial dermis. CM is most often solitary, but varies in size and may occur in any region of the extremity ( Fig. 37.5 ). With time and aging, pink lesions will often darken and develop some fibrovascular overgrowth. They can be associated with deeper vascular lesions, both slow-flow (Klippel–Trenaunay, CLOVES [congenital lipomatous overgrowth with vascular malformations, epidermal nevi, and skeletal/spinal anomalies]), and fast-flow (Parkes–Weber, CM–AVM, PTEN-associated AVM) (see Table 37.7 ) and with skeletal and soft-tissue hypertrophy. A CM on the face in one or more of the trigeminal nerve distributions is indicative of Sturge–Weber syndrome. These patients are at risk for seizures, retinal detachment, glaucoma, and stroke.
| Parkes–Weber | Ostler–Rendu–Weber | CM–AVM | PTEN–AVM | |
|---|---|---|---|---|
| Eponym/ name | Parkes–Weber; AVM with AVF | Hereditary hemorrhagic telangiectasia (HHT) | CM–AVM | PTEN-hamartoma syndrome (PTHS), Cowden, BRRS |
| Vascular malformation | Diffuse AVM with AVFs lymphedema | CM, visceral AVMs in liver and lungs | CM–small, pink | Multiple AVMs, VM |
| Skin appearance | Pseudo CM on skin lymphedema | Telangiectasia lips, fingers mucosal-nasal epistaxis | Pinkish lesion with halo | CM; possible VM bluish discoloration |
| Hemodynamics | Fast-flow | Fast-flow AVFs | Fast-flow in 30% | Fast-flow AVM in 50% |
| Cardiac compromise | Common | Rare, anemia | Possible | None |
| Arteriovenous fistuli | Multiple, often aneurismal | None | Multifocal | |
| Lateral/deep venous anomalies | None | None | None | Clusters abnormal veins |
| Coagulopathy | None | None | None | Unusual |
| Pattern | Lower → upper extremities | Lips, intraoral | Legs > arms/hand | |
| Genetics | Unknown | HHT1 mutation of endoglin HHT2 mutation ALK1 |
Mutation RASA1 ; positive family history | PTEN mutation |
| Associated anomalies | Rare pelvic AVM; Cobb syndrome | AVM cerebral, spinal, liver and pulmonary | Cerebral AVM 7% | Macrocephaly; penile freckling (males); lipomas |
| Limb hypertrophy | Major, disproportionate osteolysis can occur | None | Absent | Absent |
| Symptoms | Warmth, pain, hypertrophy, sweating | Nasal epistaxis GI bleeding anemia | Warmth, pain, mass | Pain, mass effect; function |
| Venous stasis, skin ulcers | Ischemic changes; pseudo Kaposi changes | Telangiectasia ± bleeding | None | Absent |
| Clinical prognosis | Poor; progressive expansion | Good | Progressive with AVM | Progressive |
 |
 |
 |
 |
Diagnosis is made by physical examination. Lesions with deep tissue involvement must be studied with additional imaging.
No treatment is necessary for extremity CMs. Pulsed dye laser (585 nm) can effectively decrease the intensity of the cutaneous blush. Patients (including infants) are often treated awake and with topical anesthesia. More dramatic outcomes are obtained in the head and neck region and face than in the trunk and extremities. Multiple treatments are often necessary. Outcomes in younger children are better than adults; results show 90% lightening of lesions, 50–90% improvement, and 20% minimal change. Many will re-darken over time.
Lymphatic malformation (LM) results from an error in the embryonic development of the lymphatic system. Since lymphatic and venous systems have a common origin, combined lymphatico-venous malformations (LVM) occur frequently. LM is characterized by the size of the malformed channels: microcystic, macrocystic, or combined. Most lymphatic lesions are noted at birth or within the first two years of life. Large macrocystic lesions may be slightly compressible; whereas, those with smaller channels are rubbery, a distinguishing feature. The overlying skin may appear normal, have a bluish hue, or contain clear or pink vesicles. These vesicles can weep and be a portal for bacterial entry. Dark vesicles are indicative of intralesional bleeding.
Dermal involvement often overlies a large lymphatic lesion and this skin may be thick and exhibit cutaneous puckering, much like the “peau d’orange” skin seen in lymphedema. These cutaneous vesicles may coalesce, drain clear fluid, bleed and lead to chronic wounds, infection, and sepsis.
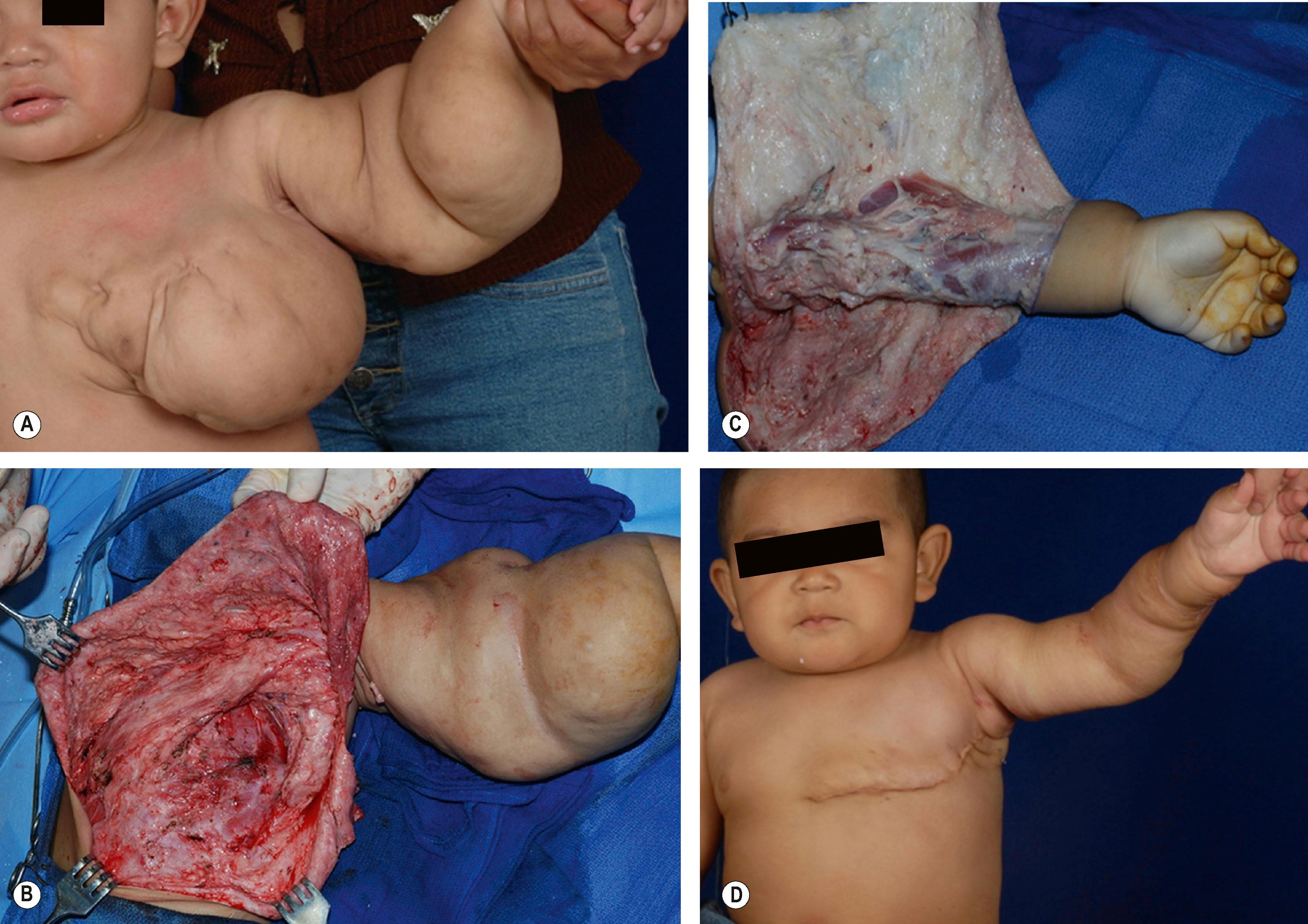
LM is characteristically macrocystic in the head and neck (“cystic hygroma”), mediastinum and axilla ( Fig. 37.6 ) and microcystic in the distal forearm, wrist, and hand. LMs may be accompanied by increased amounts of dysplastic fat adding to the bulk of the malformation. It is unusual for LMs to directly involve muscle tissue as they are primarily found within the subcutaneous tissue planes. The eponym Gorham disease is used when there is skeletal involvement. Extensive LM may be associated with skeletal atrophy.
Like many other vascular anomalies, the great majority of LMs are diagnosed by history and physical examination. Small, superficial lesions do not require further diagnostic workup. Large or deep lesions may require MRI scans to confirm the diagnosis, define the three-dimensional characteristics of the LM, and plan treatment. These scans show characteristic fluid-filled spaces, multiple septae of variable thickness, hyperintensity on T 2 -weighted MRI sequences, and slight rim enhancement following contrast administration. Diffuse enhancement is not present. Direct puncture of macrocystic lesions further delineates the architecture of the lesion prior to sclerotherapy.
Large venous channels are often present and bleeding into a large macrocyst is the rule rather than the exception. Ultrasonography shows multiloculated macrocysts with “rim enhancement” and hypo-echoic microcystic lesions with little flow. Histologic evaluation of LM shows abnormally walled channels filled with eosinophilic, protein rich fluid and collections of lymphocytes. Positive immunostaining is seen with lymphatic markers D2–40 and LYVE-1.
Because LM does not typically involve muscle tissue, it is unlikely to cause contractures. A patient who presents with a diagnosis of lymphatic malformation of the forearm but exhibits digital contractures should be assessed with tissue biopsy to confirm the diagnosis.
LM is a benign condition, and intervention for small asymptomatic lesions is not necessary. More aggressive management is reserved for symptomatic lesions that cause pain, infection, compression neuropathies, significant deformity, or functional impairment. An infected LM within the extremity can be controlled with local wound care, oral antibiotics, and occasionally, intravenous antimicrobial therapy. The rapidly advancing “wild fire” infections seen in these lesions can cause sepsis but will regress just as rapidly with appropriate treatment.
Superficial lesions with weeping vesicles can be treated with local injection of bleomycin or carbon dioxide laser. Depending on the degree of involvement, each of these approaches can be quite effective. A newer modality of treatment that is slowly gaining traction is use of topical sirolimus. Multiple case series have shown effectiveness for various vascular anomalies, including LM. The lesion should be superficial enough to absorb the topical medication. An ongoing multicenter randomized controlled trial promises to provide validated data regarding safety and efficacy for cutaneous microcystic LM.
Sclerotherapy is the first-line management for large, problematic macrocystic LMs. Several sclerosants have been used: doxycycline, sodium tetradecyl sulfate (SDS), ethanol, bleomycin, and OK-432. SDS is the most widely used. Ethanol has the highest complication rate and is particularly toxic to neurovascular structures (see Fig. 37.9 ). OK-432 is very effective but not widely available. Multiple injection sessions are required for all but the smallest lesions. Swelling, compartment syndromes, and skin extravasation are the most common complications encountered in the upper limb. We prefer sclerotherapy for the macrocystic LMs primarily located in the head and neck, axilla, and proximal portions of the upper extremity. Microcystic lesions are best treated surgically because they do not respond well to sclerotherapy.
Surgical resection of LM is reserved for: (1) symptomatic lesions causing bleeding or fluid loss, infection, pain, or interference with function; (2) those that cannot be adequately treated with sclerotherapy (usually microcystic); and (3) small, well-localized lesions which can be resected for cure. For large upper extremity LMs, staged excision is much safer than large radical resections. It is often best to start with the bulkiest chest wall, axillary and arm regions before extending more distal into the forearm and hand (see Fig. 37.6 ). Areas of involved skin are preferentially removed and only one side of a forearm, wrist, hand, or digit are debulked at one operation. Flap compromise, wound dehiscence and infection are early problems and tendon adherence, neuromas and joint stiffness may be encountered later.
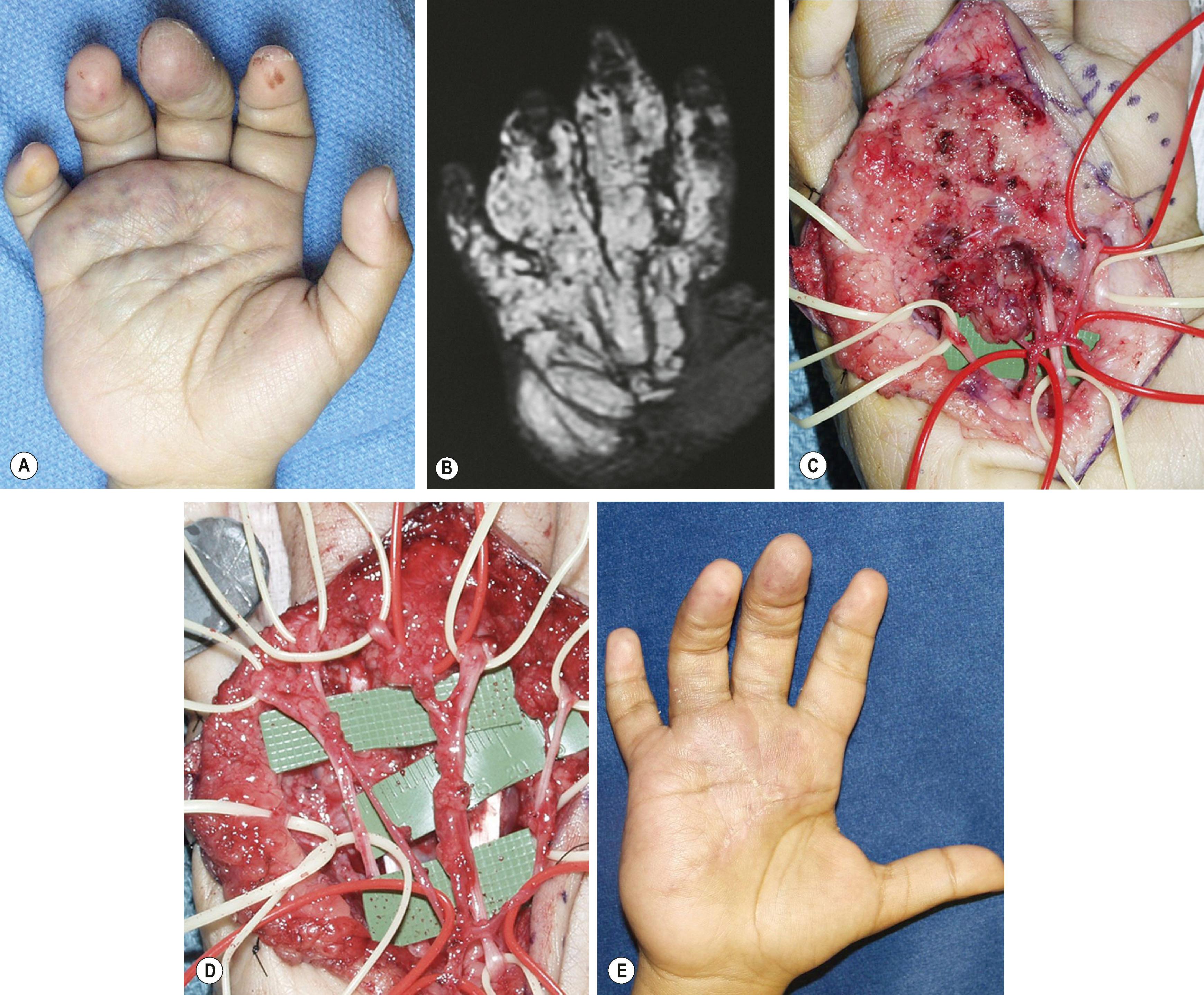
Because of its complex anatomy, the palmar hand is the most difficult region to dissect for an LM. There are no natural planes within the indurated, weeping lymphatic tissue, which may have been scarred by previous inflammation or other treatment. However, smooth, areolar connective tissue planes can usually be found around both nerves and arteries, and should be followed meticulously. Of all surgical principles outlined previously, limited and thorough dissection is most important for LM in the hand (see Table 37.2 ). In contrast to the digits and thumb, postoperative edema is almost impossible to control and may affect joint mobility and tendon gliding. In the digits and distal palm, all LMs are microcystic and can be accompanied by a large amount of aberrant adipose tissue.
Dorsal LM of the hand and digits is common and is the easiest to debulk ( Fig. 37.7 ). Involved dorsal skin should be excised and replaced by thick split-thickness or full-thickness skin grafts, ideally in anatomic subunits. Mid-axial incisions appear better long term. Incisions through the glabrous surfaces of the palm or digit have a predilection for hypertrophic scarring. Liposuction, even with ultrasonic technique, is not a good option because of the firm, rubbery consistency of microcystic LM and potential for injury to underlying structures.
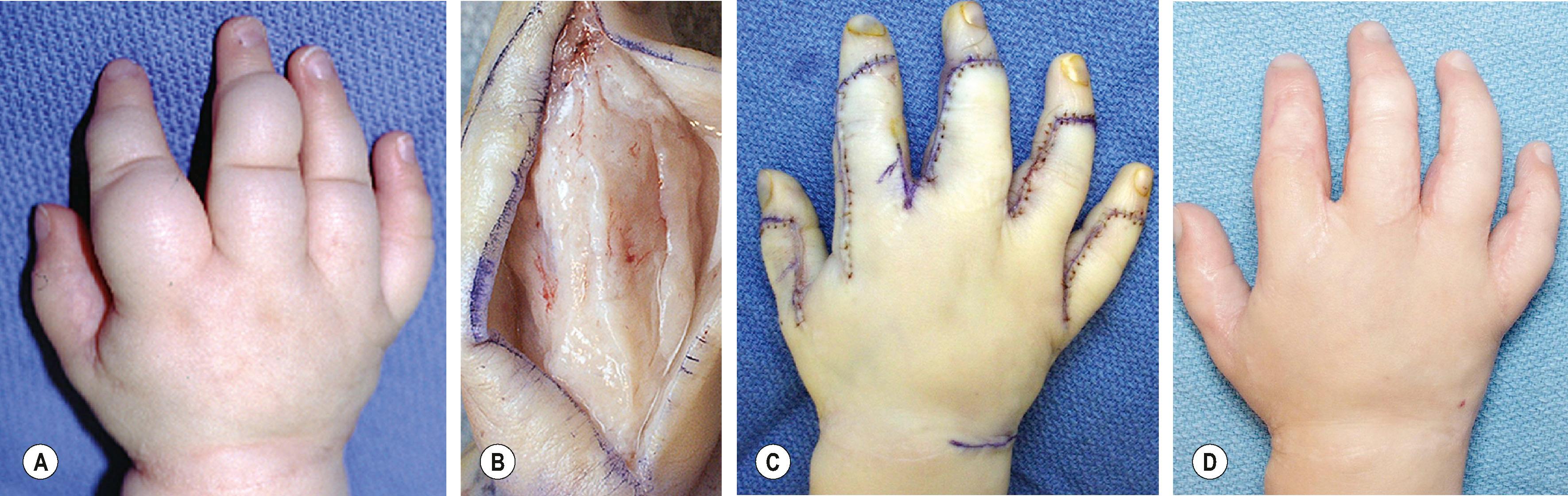
The degree of functional gain and appearance is proportional to the extent of the malformation. Frequency of complications such as persistent drainage, wound dehiscence, and skin loss is less than 8%; secondary neuromas and hypertrophic scarring are much more common (20%). With appropriate postoperative therapy and splinting, joint stiffness can be minimized. Upper extremities with large, heavy, edematous hands will always be assisting extremities and digits with limited range of motion will never be normal due to the scar from previous surgeries and residual LM within the remaining soft tissue. However, extremities with well-localized LM can be expected to function normally following well-planned and executed procedures.
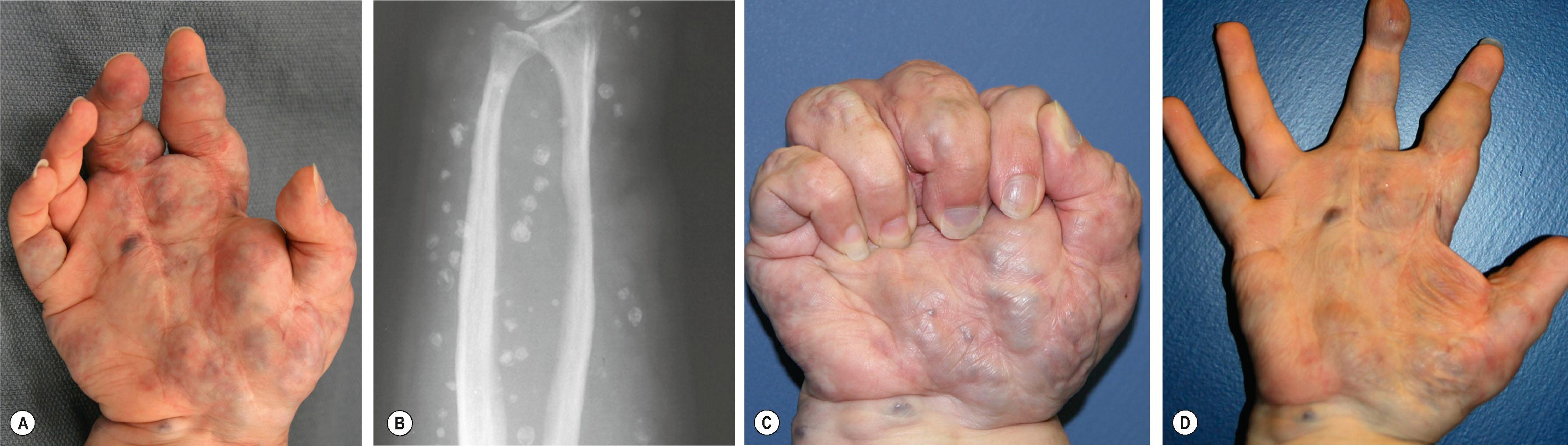
Venous malformation (VM) is the most common vascular malformation that the upper extremity surgeon will encounter. It is comprised of thin walls and abnormal smooth muscle cells, arranged in seemingly disorganized clumps. VMs show gradual dilatation and expansion with time and growth. Although VMs are present at birth, they are not always obvious. Within the first 5–6 years of life, all but the smallest or deepest lesions will become clinically evident. Less than 10% are clinically indolent until adolescence. Approximately, 40% of all VMs involve the extremities and more than half of these involve deeper structures including muscle, bone, and nerve. They grow commensurately with the child and can expand disproportionately during the adolescent growth spurt. VMs are typically sporadic and solitary, and half harbor a somatic mutation in the endothelial receptor TIE2.
VM range from small, localized skin lesions to diffuse malformations involving all tissue planes, including bones and joints. These lesions become engorged when the limb is in a dependent position and decompress when the limb is held above the level of the heart (see Fig. 37.8 ). Most VMs are located in the subcutaneous plane superficial to the muscular fascia, but in contrast to LM, VM may involve muscles directly. Also in contrast to LM, which forms distinct microcystic and macrocystic channels, the configuration of VM is random.
There is hormonal modulation with medium and large-sized venous malformations. These often increase in size during the adolescence growth spurt and menses, and in response to anti-ovulant medication and pregnancy. In women who experience an exacerbation of their symptoms during pregnancy, postpartum reduction in size does not typically occur. Patients must be followed carefully through adolescence and subsequent pregnancies.
The clinical pathophysiology of VMs stems primarily from flow characteristics within the lesions. When a VM is large enough, the flow through its channels is slow and blood can remain stagnant. Stagnant blood clots more easily, causing localized swelling, inflammation, and pain. Over time, these clots can calcify and organize into phleboliths. VMs in smaller children are usually asymptomatic – because they are smaller. As the child grows, and the VM with them, the hallmark symptoms start to become more bothersome. The conservative way to address these VM-related symptoms is with compression and antithrombotic therapy. The goal of both treatments is to avoid clot formation. Compression works externally to keep channels collapsed and minimize stagnant blood. Antithrombotic therapy, usually low-dose aspirin, works systemically by altering coagulation.
The eponymous genuine diffuse phlebectasia of Bockenheimer represents a subset of patients with severe diffuse upper extremity venous malformation. These malformations characteristically involve multiple tissue planes including muscle and bone. Large caliber ectatic veins may be deep or superficial and often place the patient at risk for clot formation and subsequent pulmonary embolism. The stagnation seen in these malformations can cause a systemic consumptive coagulopathy – promoting pathological clotting in the malformation (termed localized intralesional coagulation, LIC) while inhibiting natural hemostasis systemically.
Functional problems with venous malformations are related to the size, weight, and location of the VM. The most common functional problem in the upper extremity is pain followed by paresthesias. These are usually the result of local inflammation around intralesional thrombi which may be adjacent to major nerves. Areas of phlebothrombosis or local hemorrhage become swollen; firm and painful, especially when compressive garments are applied. Most symptoms are aggravated after exercise involving repetitive movements such as lifting or gripping. Motor dysfunction and stiffness usually result from nerve injury or scarring, both due to surgery or sclerotherapy. Ethanol is particularly toxic to the surrounding tissues and carries the highest risk of functional loss.
A variety of lesions, sometimes associated with other conditions, can harbor or mimic a venous malformation. These include verrucous venous malformation, glomuvenous malformation, blue rubber bleb, and fibro-adipose vascular anomaly (FAVA). These lesions and conditions, though rarer than common VM, have a predilection for the extremities and are reviewed in Table 37.6 ![]() . FAVA is reviewed more extensively later in the chapter.
. FAVA is reviewed more extensively later in the chapter.
| Common VM | BRBNS | GVM | VVM | FAVA | Diffuse VM | ||
|---|---|---|---|---|---|---|---|
| Name, eponym | Venous malformation | Blue rubber bleb nevus syndrome | Glomuvenous malformation; glomangioma | Verrucous venous malformation; verrucous hemangioma | Fibro-adipose vascular anomaly | Genuine phlebectasis of Bockenheimer; diffuse VM | |
| Skin appearance | Bluish discoloration, present at birth; wide distribution | Dark blue spots present at birth | Thickened and purple early; present at birth; cobblestone appearance | Dark purple stain early, evolving to roughened superficial surface, and later thick hyperkeratotic vascular lesions with prominent scales | Bluish to purple; usually deep and intramuscular with minimal skin manifestations | Diffuse; large, tortuous venous channels | |
| Genetics | 50% mutation in endothelial TIE2 | Unknown | Mutation in glomulin gene; chromosome 1 p21–22 | Unknown | PIK3CA mutations in some but not all | Unknown | |
| Pattern | Sporadic; common; sexes equal | AD; sexes equal | AD; sexes equal; | Rare, sporadic; sexes equal | Sporadic; sexes equal | Sporadic; sexes equal | |
| Hemodynamics | Slow-flow | Slow-flow | Slow-flow | Slow-flow | Slow-flow | Slow-flow | |
| Limb affected | All parts of body | All parts of body | All parts of body | Extremities suprafascial | Extremities, usually intramuscular, contractures common | Upper extremity | |
| Skeletal involvement | Present, unusual | None | None | None | None | Present, diffuse, pathologic fractures | |
| Visceral involvement | Yes | Yes: particularly bowel anemia common |
None | None | None | None | |
| Histopathology | Deficient smooth muscle in vessel walls; dilated vessels; thrombus formation | Similar to VM, well-delineated, thin-walled dilated vessels | Cuboidal glomus cells | Epidermis with hyperkeratosis overlying dilated capillaries. GLUT-1 positive | Dense fibrous tissue, fat and lymphoplasmacytic aggregates within atrophied skeletal muscle | Similar to common VM | |
 |
 |
 |
 |
 |
 |
 |
|
As with most vascular anomalies, the great majority of VMs are diagnosed by history and physical examination. Dependent position of the anomaly will confirm the diagnosis: VM will enlarge due to reduced venous return in the extremity ( Fig. 37.8 ). Small, superficial, asymptomatic lesions require no further workup. However, larger and/or deeper lesions should be evaluated by MRI to: (1) confirm diagnosis; (2) define the three-dimensional extent of the lesion; and (3) plan long-term treatment. VM is hyperintense on T 2 -weighted MRI sequences. In contrast to LM, VM will enhance with contrast, show phleboliths as signal voids, and may involve muscle. Ultrasound may be used for some localized lesions: characteristic findings are slow-flow anechoic–hypoechoic channels separated by more solid regions of variable echogenicity. Phleboliths are hyperechoic with acoustic shadowing. CT scans can be helpful to delineate bony involvement. Histological diagnosis of VM is rarely necessary, but may be indicated if there is a suspicion of malignancy or if the imaging is not confirmatory. Arteriography is not helpful.
Compression garments are the first line of treatment and are used to reduce blood stagnation within the lesion, thereby reducing the chance of expansion, LIC, phlebolith formation, and pain. Those with pain and large lesions are given daily aspirin (81 mg) to prevent clotting. Large lesions are at risk for consumptive coagulopathy: a vicious cycle of blood stagnation, stimulation of thrombin, conversion of fibrinogen to fibrin, and subsequent fibrinolysis. This type of chronic coagulopathy can paradoxically promote both thrombosis and bleeding. Though seemingly contradictory, the treatment for this type of coagulopathy is anticoagulation. Treatment decreases the consumption, increasing the clotting factors, and improving systemic clotting. Low molecular weight heparin (LMWH) is considered for those with significant LIC or those at risk for disseminated intravascular coagulation (DIC). Patients who are at risk for (or have developed) thrombotic problems, such as a pulmonary embolus, require long-term anticoagulation or a vena caval filter. These patients should be carefully managed by an expert hematologist, especially when undergoing surgical procedures.
Fibrinogen is the main parameter to follow in patients with consumptive coagulopathy due to vascular anomalies. Anticoagulation should be titrated to achieve fibrinogen levels above 200 mg/dL, especially around surgical procedures.
Sclerotherapy is the next treatment option for symptomatic lesions that cause pain, functional impairment, a mass effect, or major contour problems. Sclerotherapy does not discriminate: it injures all the tissues with which it comes in contact. It should be used carefully in the upper extremity, especially around major nerves and end-arteries. Although sclerotherapy reduces the size of the malformation, it generates scar and does not remove the malformation. The VM can re-expand, and multiple sessions are invariably needed.
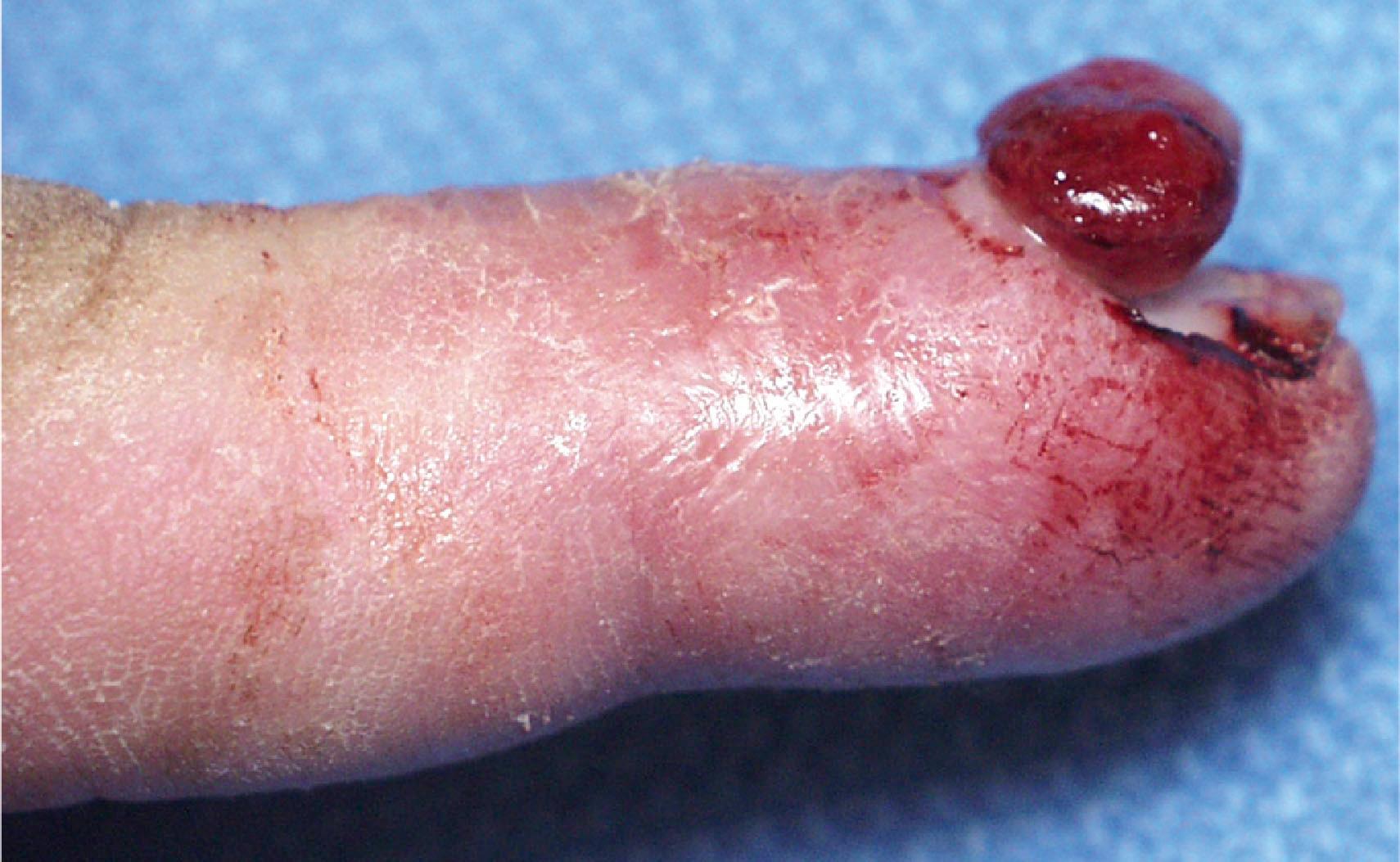
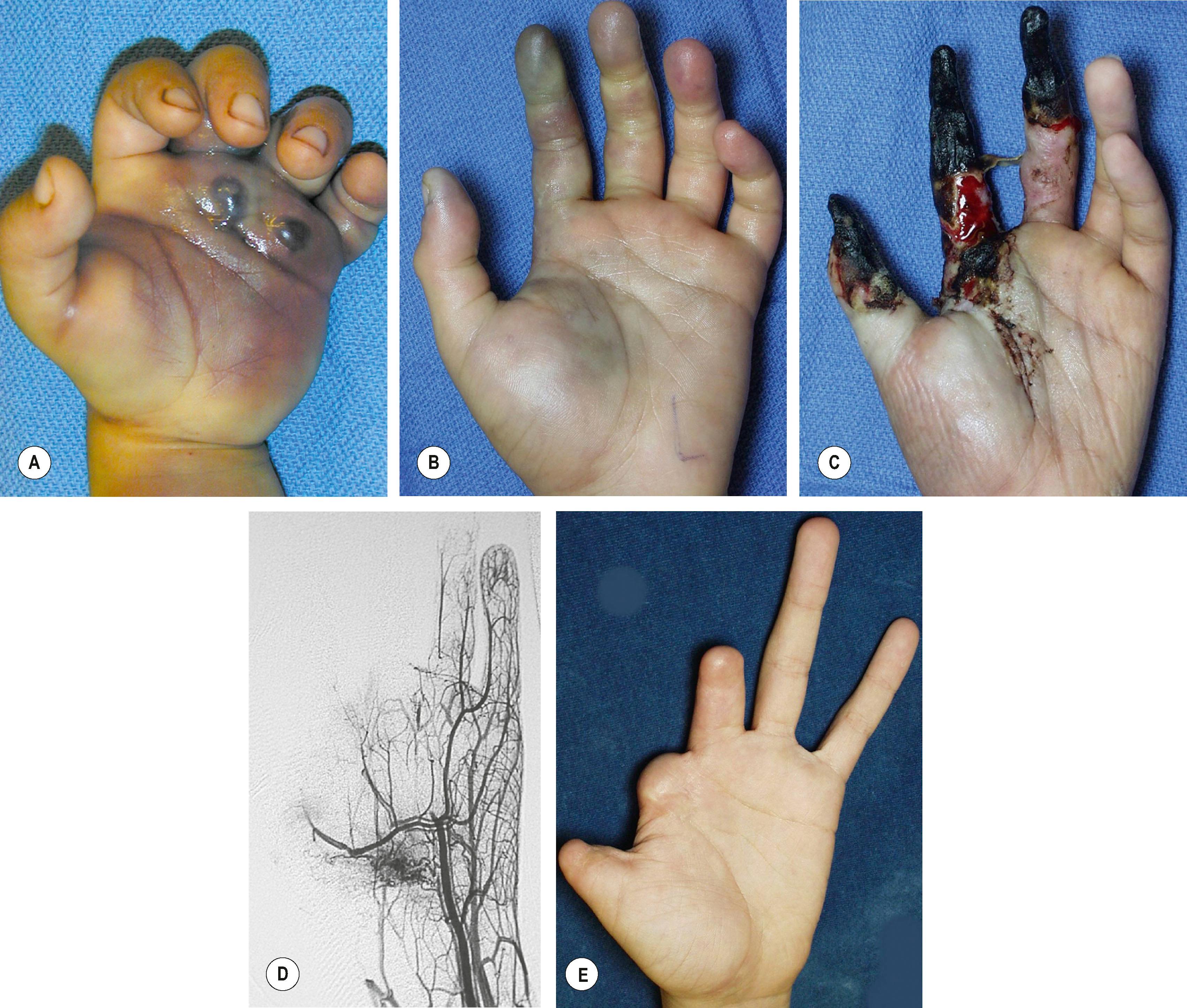
Direct puncture sclerotherapy is used for VM. Most patients are managed under anesthesia using ultrasound guidance. The most common complications are skin ulceration (10–15%), local extravasation, and long-term contractures and motor dysfunction. More dreaded complications include compartment syndrome, painful neuropathy, and digital ischemia ( Fig. 37.9 ). Large VMs of the axilla and arm contain anomalous veins too large for sclerotherapy; these should be embolized.
Ethanol is very toxic and results in the most complications in the upper limb. It should be avoided for sclerotherapy of venous (and lymphatic) malformations, especially in the distal limb where vital structures are in tight quarters.
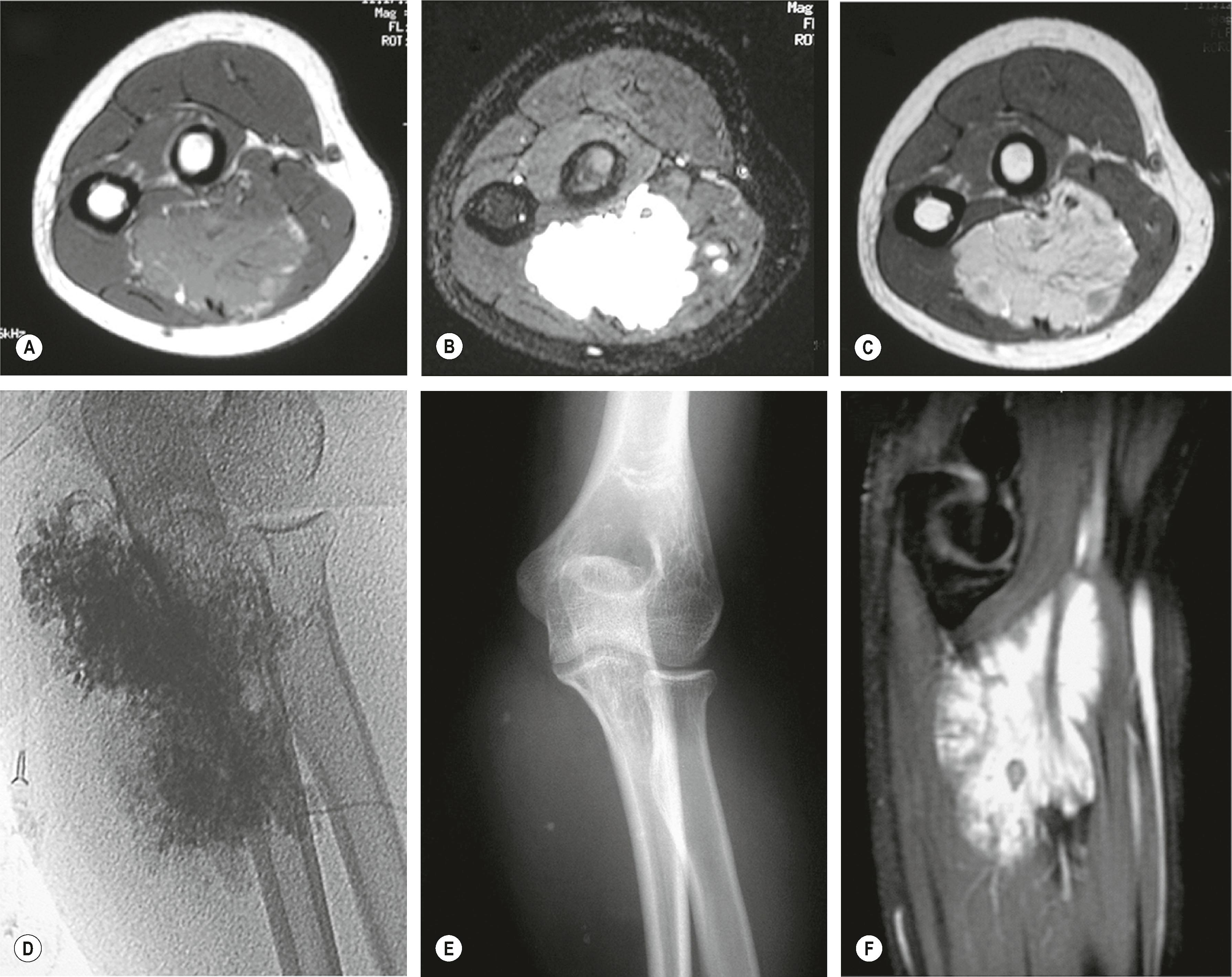
Sclerotherapy is most effective for small and large VM in the axilla, arm, elbow, and proximal forearm where muscle may be directly involved ( Fig. 37.10 ![]() ). Surgical resection is preferred in the distal forearm, wrist, thumb, and digits, where there is little muscle tissue and important neurovascular structures are near ( Fig. 37.11 ). The secondary scar generated by sclerotherapy in these regions is detrimental to nerve function, tendon gliding, and joint motion. Small nests or clusters of VM respond better to surgery than sclerotherapy. We prefer to avoid sclerotherapy prior to resection, as it can cause scarring and complicate the dissection field.
). Surgical resection is preferred in the distal forearm, wrist, thumb, and digits, where there is little muscle tissue and important neurovascular structures are near ( Fig. 37.11 ). The secondary scar generated by sclerotherapy in these regions is detrimental to nerve function, tendon gliding, and joint motion. Small nests or clusters of VM respond better to surgery than sclerotherapy. We prefer to avoid sclerotherapy prior to resection, as it can cause scarring and complicate the dissection field.
Large-sac venous malformations lend themselves well to preoperative percutaneous embolization with a glue-like agent prior to surgical excision. Our center has used n-butyl cyanoacrylate; the channels are drained of blood and filled with this material. Subsequent surgery is simplified because the lesion remains tented and does not bleed.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here