Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Severe coagulation deficiencies, or coagulopathies, are typically characterized by spontaneous or provoked bleeding, such as during surgery or trauma, and may result in life- or limb-threatening complications. By contrast, moderate and mild coagulopathies may remain clinically silent until they are detected coincidentally on routine laboratory screening tests (e.g., prothrombin time [PT], activated partial thromboplastin time [aPTT]), or when these tests are ordered to evaluate the cause of abnormal bleeding or bruising.
In general, coagulopathies may result from inadequate synthesis of coagulation factor proteins or from inhibition of activated factor proteins by acquired antibodies or anticoagulant medications. Qualitative defects, either congenital or acquired, may also result in bleeding.
Much of the morbidity of coagulopathies can be minimized or avoided altogether by prophylactic replacement of the deficient clotting factor proteins. In contrast to the lifelong clinical manifestations of hereditary or congenital coagulopathies, acquired deficiencies usually appear acutely in previously asymptomatic individuals; they may not be suspected and often remit spontaneously or after eradication of an inciting disease state or withdrawal of an offending medication. Acquired coagulation disorders may be associated with more severe bleeding than congenital disorders, in part because of the delay in diagnosis.
Hemophilia A and hemophilia B are caused by deficiencies or defects in clotting factor VIII (antihemophilic factor) and factor IX (antihemophilic factor B, or Christmas factor), respectively. A deficiency of either of these intrinsic coagulation pathway proteins results in inadequate formation of thrombin at sites of vascular injury.
Hemophilia A and B are sex-linked recessive disorders estimated to occur in 1 in 5000 and 1 in 30,000 male births, respectively. The higher incidence of hemophilia A may be due to the greater amount of DNA at risk for mutation in the large factor VIII gene (186,000 base pairs) compared with the factor IX gene (34,000 base pairs). Hemophilia A and B are observed in all racial and ethnic groups, and more than 20,000 individuals are affected in the United States. A substantial proportion (30%) of hemophilia cases arise as new, spontaneous mutations. Overall, the hemophilias are much more common than the autosomal recessive coagulation disorders (see later), which often affect progeny from consanguineous relationships and require the inheritance of two defective alleles for the bleeding manifestations to become evident.
As with other sex-linked recessive diseases, the genes for factor VIII and factor IX are located on the long arm of the X chromosome. Males with a defective allele on their single X chromosome transmit this gene to all their daughters, who are then obligate carriers, but to none of their sons. Because the offspring of female carriers inherit one affected X chromosome, half of their sons develop the coagulation disorder, and half of their daughters are obligate carriers. Female carriers may manifest bleeding symptoms, particularly postpartum bleeding, if the alleles on the X chromosome are unequally inactivated (lyonization), if the defective hemophilic allele is expressed in preference to the normal allele, and if plasma factor VIII levels are below 50%. Female hemophilia may arise as a result of mating between a hemophilic male and a female carrier (homozygous for the defective factor VIII or IX gene) or in carrier females who have the 45,XO karyotype (Turner syndrome) and are hemizygous for the defective hemophilia gene.
No single mutation is responsible for the hemophilias. Many missense and nonsense point mutations, deletions, and inversions have been described. Severe molecular defects predominate, with 40 to 50% of all cases of severe hemophilia A evolving from a unique inversion of intron 22 (the largest of the factor VIII introns). This inversion results from the recombination and translocation of DNA within intron 22 of the factor VIII gene, with areas of extragenic but homologous “nonfunctional” DNA located at a distance from intron 22. Other less common severe molecular defects include large gene deletions (5 to 10% of cases) and nonsense mutations (10 to 15% of cases). The encoded proteins resulting from these mutations are defective and do not express any factor VIII activity. Mild or moderate hemophilia A is commonly associated with point mutations and deletions. In contrast, factor IX mutations are more diverse, and severe hemophilia B is more likely caused by large deletions. Mutated clotting factor genes responsible for the hemophilias may also encode for the production of defective nonfunctional proteins that circulate in the plasma and are detected at normal quantitative levels by immunoassays but not by functional activity assays. A listing of the mutations that cause the hemophilias can be accessed via the Human Gene Mutation Database ( www.hgmd.org ).
Hemophilia A and hemophilia B are clinically similar, with clinical severity corresponding inversely to the circulating levels of plasma coagulant factor VIII or IX activity. However, hemophilia A is associated with a higher frequency of bleeding, a greater need for factor replacement therapy, and more frequent hospitalizations, thereby suggesting greater clinical severity than in hemophilia B. Individuals with less than 1% of normal factor VIII or IX activity are classified as having “severe” disease, which is characterized by frequent spontaneous bleeding events in joints (hemarthrosis) and soft tissues and by profuse hemorrhage with trauma or surgery. Although spontaneous bleeding is uncommon in mild deficiencies (>5% normal activity), excess bleeding typically occurs with trauma or surgery. A moderate clinical course is associated with factor VIII or IX levels between 1 and 5%. Approximately 60% of all cases of hemophilia A are clinically severe, whereas only 20 to 45% of cases of hemophilia B are severe.
Severe hemophilia is typically suspected and diagnosed during infancy in the absence of a family history. Among newborns, intracranial hemorrhage is the leading cause of morbidity and mortality, with a cumulative incidence of about 4%. Intracranial hemorrhage does not appear to be related to the mode of delivery, although half of such hemorrhages occur in the newborn period. Circumcision within days after birth is accompanied by excessive bleeding in less than half of severely affected boys. The first spontaneous hemarthrosis in severely affected hemophilic boys usually occurs between 9 and 18 months of age, when ambulation begins. In moderately affected individuals, it generally does not occur until 2 to 5 years of age. The knees are the most prominent sites of spontaneous bleeds, followed by the elbows, ankles, shoulders, and hips; wrists are less commonly involved. Female carriers may also sustain joint bleeding and hemarthrosis, often unrecognized until years later, with sports and/or trauma.
Acute hemarthroses ( Fig. 160-1 ) originate from the subsynovial venous plexus underlying the joint capsule and produce a tingling or burning sensation, followed by the onset of intense pain and swelling. On physical examination, the joint is swollen, hot, and tender to palpation, with erythema of the overlying skin. Joint mobility is compromised by pain and stiffness, and the joint is usually maintained in a flexed position.
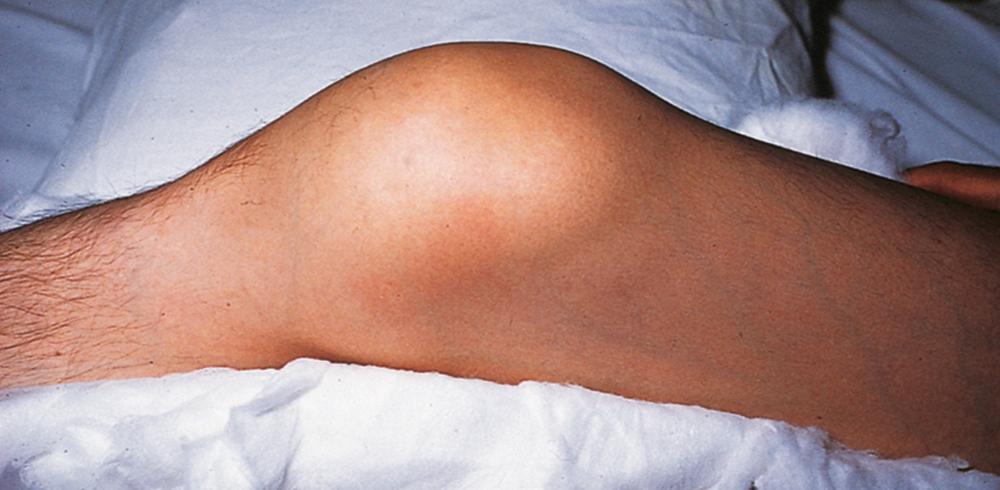
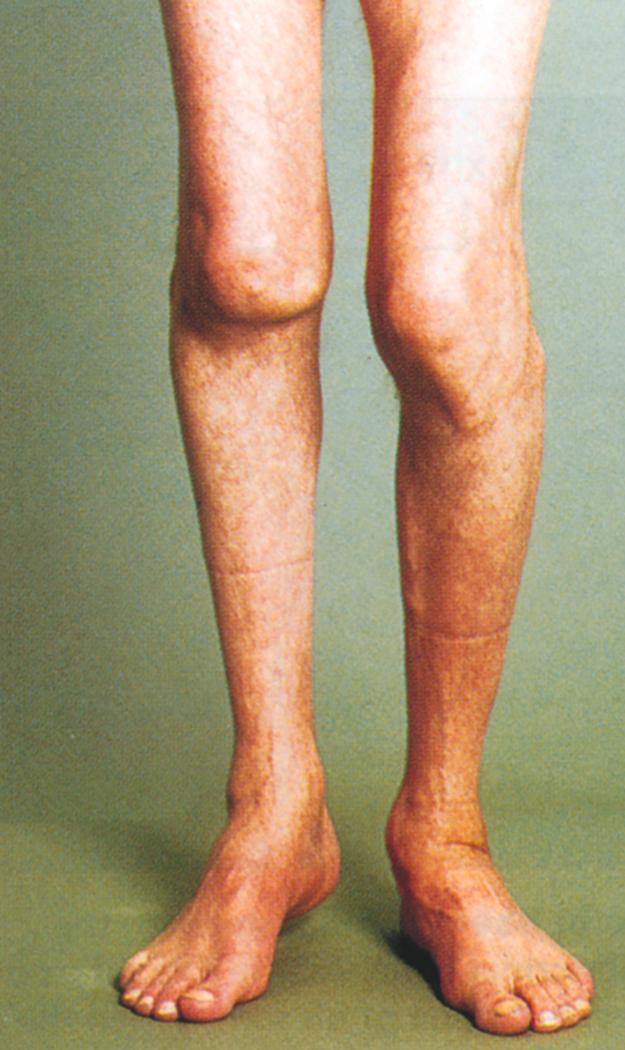
Recurrent or untreated bleeds result in chronic synovial hypertrophy and, eventually, damage to the underlying cartilage, with subsequent subchondral bone cyst formation, bony erosion, and flexion contractures. Abnormal mechanical forces from weight bearing can produce subluxation, misalignment, loss of mobility, and permanent deformities of the lower extremities ( Fig. 160-2 ). These changes are accompanied by chronic pain, swelling, arthritis, and disability. Plain radiographs and clinical examination of chronic hemarthroses often underestimate the extent of bone and joint damage; serial magnetic resonance imaging is the most sensitive technique to detect and monitor early and progressive disease, and early musculoskeletal ultrasound, performed in the office setting, may detect early joint disease even before symptoms occur.
Intramuscular hematomas account for about 30% of hemophilia-related bleeding events and are rarely life-threatening. Hematomas are usually precipitated by physical or iatrogenic trauma (e.g., after intramuscular injections) and may compromise sensory and motor function or arterial circulation if they entrap and compress vital structures in closed fascial compartments. The latter occurrence, termed compartment syndrome , presents with the rapid onset of swelling and severe pain in an extremity, unrelieved by factor infusion and standard analgesics, and is considered a medical emergency that may require fasciotomy to preserve tissue viability and provide pain relief. Retroperitoneal hematomas may be confused clinically with appendicitis or hip bleeds but should be suspected in a patient who is unable to stand erect due to the pain of muscle extension in the presence of hematoma. Immediate and aggressive treatment is required to prevent permanent anatomic deformity, such as flexion contracture, nerve damage, or pseudotumor formation (expanding hematomas that erode and destroy adjacent skeletal structures).
Bleeding from mucous membranes is not uncommon and when it involves the tongue or the retropharyngeal space, may threaten the airway. Gastrointestinal hemorrhage typically originates from anatomic lesions proximal to the ligament of Treitz and may be exacerbated by esophageal varices and/or thrombocytopenia secondary to cirrhosis and portal hypertension, or by the use of nonsteroidal anti-inflammatory drugs (NSAIDs) for the treatment of hemarthroses. Spontaneous bleeding in the genitourinary tract may arise from ureteral clots, renal stones, and/or less commonly, infection.
Intracranial bleeds occur in 10% of patients with a risk of 2% per year; they may be spontaneous or traumatic and are fatal in 30% of cases. Neuromuscular defects, seizure disorders, and intellectual deficits may ensue.
The diagnosis of hemophilia is suspected on the basis of a family and personal bleeding history and laboratory detection of prolongation of the aPTT (with normal PT). It is confirmed by significantly reduced plasma factor VIII or IX activity. As noted in Chapter 157 , the aPTT is not a sufficiently sensitive screening test to be prolonged in mild hemophilia, where the factor VIII level is sometimes greater than 30% of normal. Severe hemophilia is usually recognized in infancy, with circumcision bleeding; by contrast, moderate or mild disease is recognized later in life after trauma or surgery. Hemophilia can be distinguished from von Willebrand disease (VWD; Chapter 159 ) by normal ristocetin cofactor and von Willebrand factor antigen levels. In type 2 N VWD, the factor VIII level is significantly lower than ristocetin cofactor and von Willebrand factor antigen levels due to reduced factor VIII binding; this variant of VWD may require genotyping to distinguish it from hemophilia A. Other congenital intrinsic factor deficiencies (e.g., factor XI and XII) can be determined by coagulation factor–specific assays. Vitamin K deficiency (see later and Chapter 161 ) can be detected by the associated PT prolongation; deficiencies of factors II, VII, IX, and X; and resolution of the coagulation defect with vitamin K. The presence of heparin can be confirmed by correction of the aPTT after running the sample over a heparin absorption column. Failure of the aPTT to correct in a 1:1 mix with normal plasma suggests the presence of an inhibitor; specific inhibitors are associated with a single decreased factor level (usually factor VIII; see Alloantibody Inhibitors to Factors VIII and IX under Treatment), whereas blocking inhibitors cause nonspecific factor level changes associated with a positive hexagonal lipid assay (see Antiphospholipid Syndrome and Lupus Anticoagulant).
Individuals with hemophilia cared for at hemophilia treatment centers have lower mortality and reduced costs of care compared with those receiving treatment elsewhere. The chronic care model practiced at hemophilia treatment centers emphasizes prevention to reduce joint disease and complications, optimization of factor dosing, counseling regarding safe sports, and the avoidance of aspirin and other drugs that inhibit platelet function, as well as management of surgical procedures and chronic transfusion-transmitted infections, including hepatitis C and HIV infection.
Treatment and prevention of acute bleeding events in hemophilia A and B are based on replacement of the missing or deficient clotting factor protein to restore adequate hemostasis ( Table 160-1 ). Use of factor on a prophylactic basis several times weekly to prevent bleeds can reduce or prevent joint bleeds and reduce joint arthropathy. All clotting factor concentrates available in the United States, whether plasma-derived or genetically engineered, are considered extremely safe; none has been implicated in the transmission of bloodborne viral pathogens or prions.
| COAGULATION PROTEIN DEFICIENCY | INHERITANCE PATTERN | PREVALENCE | MINIMUM HEMOSTATIC LEVEL | REPLACEMENT SOURCES |
|---|---|---|---|---|
| Factor I (fibrinogen) | 50-100 mg/dL | Cryoprecipitate, FFP, fibrinogen concentrate | ||
| Afibrinogenemia | Autosomal recessive | Rare (<300 families) | ||
| Dysfibrinogenemia | Autosomal dominant or recessive | Rare (>300 variants) | ||
| Factor II (prothrombin) | Autosomal dominant or recessive | Rare (25 kindreds) | 30% of normal | FFP, factor IX complex concentrates |
| Factor V (labile factor) | Autosomal recessive | 1 in 1 million births | 25% of normal | FFP |
| Factor VII | Autosomal recessive | 1 in 500,000 births | 25% of normal | Recombinant factor VIIa (15-20 µg/kg), FFP, factor IX complex concentrates |
| Factor VIII (antihemophilic factor) | X-linked recessive | 1 in 5000 male births | 80-100% for surgery/life-threatening bleeds, 50% for serious bleeds, 25-30% for minor bleeds | Factor VIII concentrates (recombinant and recombinant extended-half-life factor VIII preferred) |
| Von Willebrand disease (also see Chapter 159 ) | >50% VWF antigen and ristocetin cofactor activity | DDAVP for mild to moderate disease (except type 2B; variable response in 2A); cryoprecipitate and FFP (not preferred, except in emergencies); plasma-derived factor VIII/VWF concentrate, viral attenuated, intermediate purity; recombinant VWF concentrate (the latter two proteins preferred for surgery, for disease unresponsive to DDAVP and for type 3) | ||
| Type 1 and 2 variants | Usually autosomal dominant | 1% prevalence | ||
| Type 3 | Autosomal recessive | 1 in 1 million births | ||
| Factor IX (Christmas factor) | X-linked recessive | 1 in 30,000 male births | 25-50% of normal, depending on extent of bleeding, surgery | Factor IX concentrates (recombinant and recombinant extended half-life factor IX preferred); FFP not preferred except in dire emergencies |
| Factor X (Stuart-Prower factor) | Autosomal recessive | 1 in 500,000 births | 10-25% of normal | Plasma-derived factor X concentrate, FFP, or factor IX complex concentrates |
| Factor XI (hemophilia C) | Autosomal dominant; severe type is recessive | 4% of Ashkenazi Jews; 1 in 1 million general population | 20-40% of normal | FFP or factor XI concentrate |
| Factor XII (Hageman factor), prekallikrein, high-molecular-weight kininogen | Autosomal recessive | Not available | No treatment necessary | — |
| Factor XIII (fibrin stabilizing factor) | Autosomal recessive | 1 in 3 million births | 5% of normal | Recombinant FXIII concentrate, FFP, cryoprecipitate, or viral-attenuated factor XIII concentrate |
The goal of replacement therapy (see Table 160-1 ) is to achieve plasma factor VIII or IX activity levels that are sufficient to prevent spontaneous bleeds. As these events are individualized and vary by age, level of activity, and pharmacokinetics, the optimal level of treatment may vary among affected patients. In general, however, activity levels of 15% are sufficient to prevent spontaneous bleeding, whereas factor activity levels of 25 to 30% are recommended to treat minor spontaneous or traumatic bleeds (e.g., hemarthroses, persistent hematuria). At least 50% clotting factor activity levels are recommended for severe bleeds (e.g., major dental surgery, maintenance replacement therapy after major surgery or trauma), and 80 to 100% activity for any life-threatening or limb-threatening hemorrhagic event (e.g., major surgery, trauma). After major trauma or if visceral or intracranial bleeding is suspected, replacement therapy adequate to achieve 100% clotting factor activity should be administered immediately (within 5 to 10 minutes) before diagnostic procedures are initiated.
To calculate the initial dose of replacement factor, plasma factor VIII activity generally increases about 2% (0.02 IU/mL) for each unit of factor VIII administered per kilogram of body weight, and factor IX activity increases about 1% (0.01 IU/mL) for each unit of factor IX administered per kilogram of body weight. Therefore, a 70-kg individual with severe hemophilia A or B (factor VIII or IX activity <1% of normal) who requires replacement to 100% activity for major surgery should initially receive 3500 IU of factor VIII or 7000 IU of factor IX concentrate. The circulating half-lives of factors VIII and IX require subsequent dosing at half the initial dose every 8 to 12 hours for factor VIII and every 18 to 24 hours for factor IX, respectively. However, this empirical dosing should be individualized according to the patient’s peak recovery and trough activity levels, using population pharmacokinetic modeling. The frequency of repeat dosing is also determined by the rapidity of pain relief, recovery of joint function, and resolution of active bleeding. Heterogeneity in the half-life of factor VIII concentrates is due to variability in clearance of von Willebrand factor.
Replacement is usually maintained for at least 10 to 14 days after major surgery to allow proper wound healing and to avoid postoperative bleeding. In the management of bleeds or surgical procedures in patients with hemophilia, either repetitive bolus injections, as described earlier, or continuous infusion can be used for delivery of clotting factor replacement. The latter can provide constant levels of the deficient coagulation factor and can be cost-effective. Extended half-life recombinant factor VIII and IX (including pegylated, albumin-fusion, nonacog alfa [factor IX], or Fc-fusion [Fc domain of immunoglobulin G 1 , IgG 1 ]) are also available for prophylaxis or treatment, as they are safe and effective in reducing bleed frequency and prolong half-life 1.5- to 2-fold for factor VIII and 2.5- to 5.0-fold for factor IX. These agents have also been used successfully in surgery, but they are costly and may not be available at many hospitals. The typical dose frequency for extended half-life recombinant factor VIII is twice weekly at a dose of 50 to 65 IU/kg; for extended half-life recombinant factor IX, the dose is once every 1 to 2 weeks at a dose of 100 IU/kg.
Nonfactor therapeutics given by subcutaneous administration also can be used as prophylaxis to prevent clinical bleeding. Emicizumab, which is the only such agent currently FDA-approved, is a bispecific monoclonal antibody that bridges activated factor IX and factor X to replace the function of missing activated factor VIII, whether due to a factor VIII inhibitor or factor VIII deficiency in hemophilia A. Emicizumab prophylaxis (1.5 mg/kg subcutaneously weekly or 3.0 mg/kg every 2 weeks, or 6.0 mg/kg every 4 weeks) is effective in reducing clinical bleeding in hemophilia A among patients of all ages, with or without inhibitors. However, emicizumab is effective only for prevention but not for the treatment of acute bleeds. For acute treatment of bleeds or surgery in patients receiving emicizumab, factor replacement is required urgently. A novel intravenous factor VIII fusion protein, efanesoctocog alfa, in which recombinant factor VIIIFc is fused to von Willebrand factor, results in a four-fold increase in the half-life of factor VIII, thereby suggesting the possibility of weekly intravenous dosing.
For hemarthroses, immediate or early replacement of the deficient clotting factor to normal hemostatic levels rapidly reverses the pain, whereas delayed treatment results in excess pain, morbidity, and joint damage. Optimal management once a bleed occurs includes rest, ice, (factor) concentrate, and elevation, as well as protective mechanotherapy for balanced and incremental rehabilitation of injured tissues.
The moderate or severe pain that accompanies acute hemarthroses responds to immediate analgesic relief. Narcotic analgesics, such as codeine or synthetic derivatives of codeine, are avoided because of their addictive potential, and early consultation with pain service physicians is recommended. Chronic acetaminophen analgesia, although helpful in low doses, may be associated with hepatic toxicity as higher doses are used, especially in patients with chronic hepatitis. For some patients compliant with prophylaxis, nonsteroidal anti-inflammatory drugs (NSAIDs; Table 26-4 ) may be used provided that bleeding does not occur. Aspirin (ASA) is generally contraindicated as it irreversibly inhibits platelet aggregation.
Over 75% of children and about 50% of adults engage in prophylaxis (preventive) factor infusions several times a week to maintain factor VIII or IX levels above 1% to prevent joint bleeds and the development of chronic hemophilic arthropathy. However, prophylaxis must be initiated before 4 years of age, the earlier the better, and in the absence of obesity to preserve joint motion. For optimal outcomes, the patient and/or parent should be involved in decision making and the treatment plan.
Although prophylaxis is the optimal strategy to prevent pain, reduced motion, and the disability of end-stage joint destruction, when prophylaxis has failed or was never initiated, surgery may be recommended. Synovectomy through open surgery or arthroscopy removes the inflamed tissue and may decrease pain and reduce recurrent bleeding, but usually only temporarily. Nonsurgical synovectomy (synoviorthosis), which involves the intra-articular administration of a radioisotope, may be considered in high-risk patients and for patients who have alloantibody inhibitors to factor VIII or IX (see later). Neither synovectomy nor synoviorthosis reverses joint damage, but both procedures may delay its progression. Non–weight-bearing exercises, such as swimming and isometrics, are important for periarticular muscle development and maintenance of joint stability for ambulation. Intractable pain and severe joint destruction secondary to repeated hemorrhage require prosthetic replacement. Chronic ankle pain responds best to open surgical or arthroscopic fixation and fusion (arthrodesis).
For genitourinary bleeding, clotting factor, fluid replacement, and, in children, a short course of steroids may be helpful to hasten resolution. However, genitourinary tract bleeding in patients with hemophilia may be worsened by the use of antifibrinolytic agents.
Alloantibodies to infused factor VIII or, less frequently, to factor IX are usually detected in childhood after a median of 9 to 12 exposures to clotting factor. The prevalence of factor VIII alloantibodies among hemophilia A patients is 15 to 25%, whereas the prevalence of factor IX alloantibodies among hemophilia B patients is 1.5 to 3%.
Inhibitors are associated with hospitalization, cost, and early death. Inhibitors occur preferentially in patients who have a family history of inhibitors, patients who have large multidomain factor VIII and factor IX gene deletions, and in Black and Hispanic patients, but it is difficult to predict in whom inhibitors will develop. Among Black patients, gene sequence data suggest that mismatched factor VIII transfusions may account for the high risk of developing inhibitors. Nongenetic risk factors include early high-intensity or frequent daily exposure to factors to treat a bleed, trauma, or surgery. In previously untreated patients, the risk of developing inhibitors appears to be higher with recombinant than with plasma-derived factor.
Factor VIII–specific IgG1 antibodies appear first, followed by IgG3 and IgG4 antibodies. However, the appearance of factor VIII–specific IgG3 antibodies is always associated with the subsequent diagnosis of factor VIII inhibitors.
The development of an alloantibody inhibitor is suspected when replacement therapy is ineffective in controlling bleeding symptoms. These antibodies completely neutralize clotting factor activity and prevent or reduce any increment in factor VIII or IX levels following bolus infusions of concentrate. Inhibitors are also associated with anaphylaxis and nephrotic syndrome.
Characterized as time and temperature dependent, inhibitors are quantitated in Bethesda units (BU). By definition, 1 BU is the amount of inhibitor that neutralizes 50% of the specific clotting factor activity in normal plasma. High responders, or patients with high-titer inhibitors (≥5 BU), mount an anamnestic antibody response to factor VIII clotting factor protein, usually within 5 to 7 days after subsequent exposure, and are no longer responsive to infused factor VIII. By contrast, low responders, or patients with low-titer inhibitors (<5 BU), do not manifest anamnesis, and such low-titer inhibitors can readily be overwhelmed by large amounts of human factor VIII or factor IX concentrate and can be successfully treated with three to four times the usual factor dose.
Management of patients with high-titer inhibitors requires both assurance of hemostasis using bypass therapy and eradication of inhibitor formation (see Table 160-1 ). Bypass is typically accomplished with factor VIII inhibitor bypass activity (FEIBA; a plasma concentrate containing activated proenzymes of the prothrombin complex factors, prothrombin, and factors V11, IX and X). The usual treatment consists of one dose of FEIBA (75 IU/kg) or two doses of recombinant factor VIIa (90 to 270 µg/kg), although some patients may respond better to one agent than the other. Sequential use of FEIBA and recombinant factor VIIa (e.g., alternating every 6 hours) can improve hemostasis over single-agent treatment, but careful monitoring for thrombosis is necessary. The total dose of FEIBA should not exceed 20,000 IU in 24 hours.
Secondary prophylaxis in adults, with either recombinant factor VIIa, 90 µg/kg daily, 270 µg/kg daily, or FEIBA 85 IU/kg weekly, may significantly reduce bleeding rates as compared with on-demand treatment. Because neither agent is very effective alone and is limited by either total dose or dose frequency (VIIa must be given every 2 to 3 hours), these bypass agents may be alternated in selected patients (without cardiovascular risk, obesity, hypertension) every 3 hours for severe bleeds, surgery, or trauma. Alternatively, emicizumab prophylaxis (1.5 mg/kg subcutaneously weekly) may reduce bleeding events and improve quality of life compared with no prophylaxis in adults and children who have hemophilia A and have inhibitors. , By avoiding the need for factor VIII, inhibitor titers often decline. Whether emicizumab and extended half-life agents such as recombinant factor VIIIFc will shorten the time to tolerance is currently uncertain. FEIBA should be avoided in patients who are receiving emicizumab because the combination increases the risk of thrombosis and thrombotic microangiopathy.
Eradication of inhibitors with immune tolerance induction is effective in about 75% of patients if initiated within 12 months of the detection of inhibitors. Immune tolerance induction regimens consist of daily factor concentrates (e.g., 200 IU/kg daily) to accomplish desensitization to infused factor. Immune tolerance induction is initiated as soon as the inhibitor is detected. Maintenance of tolerance following induction usually, but not always, requires factor VIII or IX prophylaxis. Single or combination immunosuppressive agents (e.g., rituximab, mycophenolate, or cyclosporine) are variably effective. Other novel agents that promote thrombin generation by alternate pathways, including siRNA-antithrombin (fitusiran), can markedly reduce bleeding frequency and factor use in patients with inhibitors.
The prevalence of hepatitis C virus infection is about 90% in patients who were transfused from the late 1970s through the mid-1980s, and the prevalence of human immunodeficiency virus (HIV) infection is about 80% in patients who were transfused with factor VIII products and 50% in patients who were transfused with factor IX products from 1978 through the mid-1980s. Patients with HIV now can be controlled with highly active antiretroviral therapy ( Chapter 357 ). Chronic hepatitis C infection ( Chapter 135 ) remains the leading cause of death in hemophilia, in part because antiviral therapy does not prevent hepatocellular cancer in patients who have had long-standing chronic infection.
Ancillary treatment strategies for the hemophilias include antifibrinolytic agents, such as ε-aminocaproic acid (50 mg/kg 3 to 4 times daily) or tranexamic acid (3 or 4 g orally daily in divided doses), to minimize mucous membrane bleeding, particularly in the oral cavity, and the application of fibrin glue to bleeding sites. Desmopressin (DDAVP) can be administered by nasal insufflation 30 minutes before a scheduled surgical procedure (one spray per nostril, to provide a total dose of 300 µg; or, in patients weighing less than 50 kg, 150 µg administered as a single spray). DDAVP may also be administered intravenously (dissolved in 50 mL normal saline) over 30 minutes at a dose of 0.3 µg/kg. DDAVP is especially useful in patients with mild hemophilia A because an adequate incremental rise in factor VIII activity can circumvent the use of clotting factor concentrates, and is generally available in most emergency department settings. Oral and intravenous fluid must be restricted to less than 1000 mL in any 24-hour period that DDAVP is administered whether to avoid hyponatremia and hyponatremic seizures. Other complications of DDAVP include facial flushing and tachyphylaxis (loss of response after three consecutive daily doses).
The hereditary hemophilias are model diseases for gene therapy because they are caused by specific, well-defined gene mutations. A small, incremental rise in the synthesis of clotting factors can lead to substantially improved treatment and quality of life and inadvertent overexpression by successful gene transfer would not be detrimental. Various adeno-associated (AAV) viral vectors carrying the Padua (FIX R338L) factor IX gain-of-function transgene in hemophilia B patients can achieve factor IX expression of 14 to 76% for up to 10 years and can significantly reduce the risk of bleeding and the usage of factor IX therapy. In patients with severe hemophilia A, trials of AAV8-BDD-factor VIII and AAV5-BDD (B-domain deleted factor VIII ) have achieved factor VIII expression levels of 60% or higher for up to 3 years, but then waning to 20% or lower in some trials. These treatments resulted in a more than 90% reduction in annualized rate of bleeding events and use of factors in both hemophilia A and B. An immune response to the AAV capsid, associated with increases in liver aminotransferase levels, sometimes occurs but appears to be responsive to corticosteroids or possibly mycophenolate mofetil. Less immunogenic vectors, innovative delivery systems, and CRISPR gene editing are also being studied.
Carrier detection and prenatal diagnosis have become technically feasible, very sensitive, and widely available. Noninvasive prenatal diagnosis by use of microfluidic digital PCR of maternal plasma DNA is based on the relative proportion of mutated versus normal alleles; it can detect an affected fetus as early as the eighth week of gestation. Because prenatal diagnosis requires the mother to have a known mutation, mothers are increasingly being tested in a nationwide hemophilia genotype program, MLOF (My Life, Our Future). However, at best only two thirds of carrier mothers will be identified because one third of new hemophilia cases arise as spontaneous mutations.
Early prenatal diagnosis is important to reduce intracranial bleeds in an infant with anticipated severe hemophilia by recommended caesarean delivery and to reduce circumcision-related bleeding by preventive factor treatment. Guidelines suggest caesarean section delivery be considered for any known male infants with severe hemophilia A. Vacuum extraction may increase cephalohematoma formation and is discouraged.
Early diagnosis is also important for the mother to reduce peripartum bleeding and advocate for genetic counseling regarding family planning. Next-generation sequencing, carrier testing, and prenatal diagnosis with genetic counseling are widely available in the United States through the network of federally funded hemophilia treatment centers. Fecundity rates remain high, and few confirmed carriers elect to terminate their pregnancies, even if an affected fetus is detected in utero. These decisions are influenced by the wide availability of safe and effective coagulation factor replacement concentrates and by the prospect of an eventual cure for the hemophilias through gene transfer or gene editing.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here