Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Cholecystitis, a common condition usually resulting from complications of cholelithiasis, occurs in two forms: acute and chronic. Acute cholecystitis requires urgent intervention, typically with antibiotics and cholecystectomy. In the setting of acute cholecystitis, cholecystectomy is optimally performed within 72 hours of symptom onset. If urgent cholecystectomy is not feasible, cholecystectomy can be performed electively, provided symptoms have resolved with medical management. Chronic cholecystitis is the manifestation of ongoing, intermittent inflammation and biliary colic. Patients with this condition benefit from elective cholecystectomy. A less common version of cholecystitis is acute acalculous cholecystitis, which occurs most often in critically ill patients. Although gallstones are, by definition, absent in this condition, cholecystectomy specimens in patients with acute acalculous cholecystitis often reveal biliary sludge (see Chapter 33 ).
The cause of acute calculous cholecystitis is an impacted gallstone in the outlet of the gallbladder, either in the infundibulum or in the cystic duct. The impacted gallstone results in gallbladder distension and edema with acute inflammation, which eventually can result in venous stasis and obstruction, followed by thrombosis of the cystic artery. Ultimately, ischemia and necrosis of the gallbladder can occur. Because the fundus of the gallbladder is the greatest distance from the cystic arterial blood supply, it is more sensitive to ischemia and is the most common location for necrosis of the gallbladder. The acute inflammation of cholecystitis may be complicated by secondary biliary infection. Positive bile cultures are found in approximately 20% of patients with acute cholecystitis, the most common of which are gram-negative bacteria of gastrointestinal origin, such as Klebsiella spp. and Escherichia coli. The incidence of bactobilia has been reported to be as high as 60% in patients who have had endoscopic sphincterotomy or other biliary instrumentation (see Chapters 30 and 31 ).
Most patients with acute cholecystitis are seen with severe, constant, right upper quadrant abdominal or epigastric pain, sometimes with radiation to the subscapular area. This pain may be preceded by intermittent, self-limited bouts of abdominal pain from episodes of biliary colic. Acute cholecystitis is frequently associated with fever and leukocytosis, findings that are not present in cases of uncomplicated biliary colic. Patients also may have a Murphy’s sign (inspiratory arrest on palpation of the right upper quadrant of the abdomen). Other presenting symptoms include nausea, vomiting, and anorexia.
Several disease processes can present similarly to cholecystitis and should be considered in the differential diagnosis. These include peptic ulcer disease, gastritis and gastroenteritis, irritable bowel syndrome, inflammatory bowel disease, right lower lobe pneumonia, and biliary dyskinesia. An initial chest radiograph is generally sufficient to assess for a right lower lobe infiltrate. The other diagnoses should be entertained and worked up appropriately in symptomatic patients without gallstones on ultrasound (US).
The Tokyo Guidelines are also a useful tool to assess the likelihood of acute cholecystitis and can be used to assist in diagnosis. These guidelines are based on three clinical and diagnostic categories: local signs of inflammation (Murphy’s sign or right upper quadrant mass or right upper quadrant tenderness), systemic signs of inflammation (fever, elevated C-reactive protein, elevated white blood cell [WBC] count), and imaging findings suggestive of cholecystitis (pericholecystic fluid, gallbladder wall edema, luminal debris and stone impaction). For patients who present with one item from each category, validation studies demonstrate a guideline sensitivity and specificity of 91% and 97%, respectively. Additionally, the Tokyo Guidelines stratify presentations of acute cholecystitis according to risk of 30-day mortality: 1.1% for mild acute cholecystitis (Grade I), 0.8% for moderate acute cholecystitis (Grade II), and 5.4% for severe acute cholecystitis (Grade III; P < .0001). Online calculators allow for easy access and use of this tool.
Abdominal US (see Chapter 16 ) is useful for assessing patients suspected to have acute cholecystitis. Typical findings include gallstones, gallbladder wall thickening (>4 mm), and pericholecystic fluid ( Fig. 34.1 ). In addition, the sonographer can assess for pain and inspiratory arrest when the gallbladder is directly compressed by the US probe (sonographic Murphy’s sign). Typically, conventional grayscale imaging is used, which, together with clinical picture and sonographic Murphy’s sign, is sensitive and specific for diagnosing acute cholecystitis, with an overall accuracy of greater than 90% (Pinto et al., 2013). Other ultrasound techniques that assess blood flow, such as Doppler and color velocity imaging, may improve accuracy in selected cases.
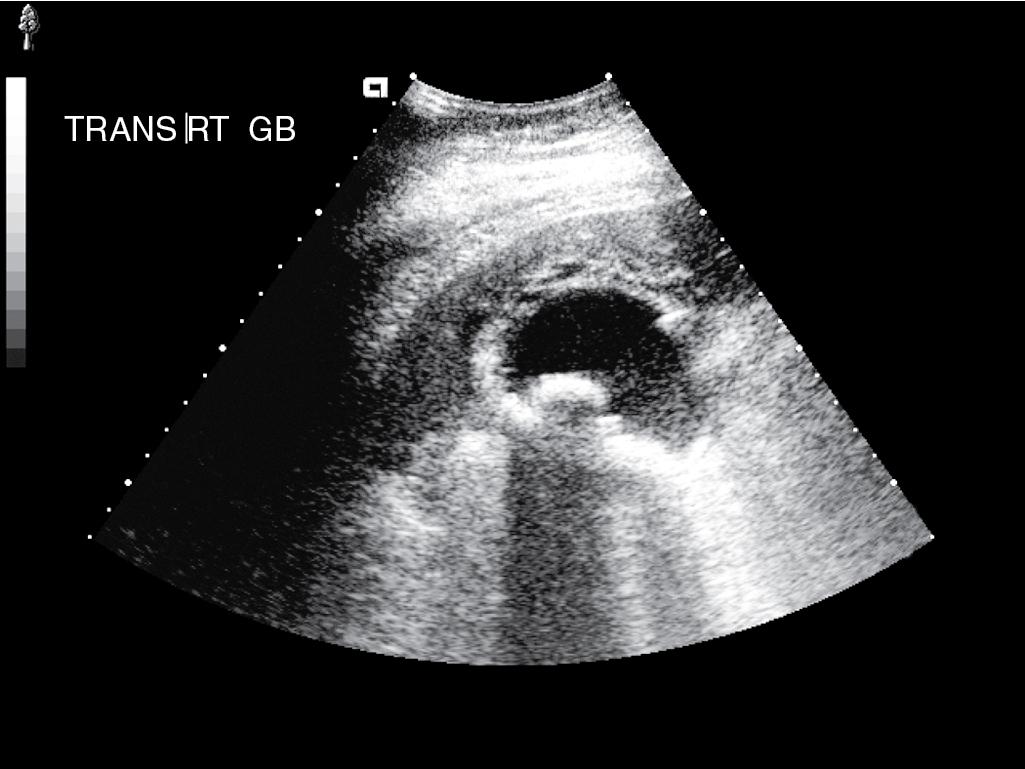
Hepatobiliary scintigraphy (see Chapter 18 ) is a useful study in selected patients when the diagnosis is uncertain. This nuclear medicine study is performed with derivatives of aminodiacetic acid (hepatoiminodiacetic acid, isopropylacetanilido iminodiacetic acid, or diisopropylacetanilido iminodiacetic acid), which are taken up by hepatocytes and secreted in bile. When the tracer is labeled with technetium, scintigraphy allows for visualization of the extrahepatic biliary system. A normal scan delineates the biliary tree, including the gallbladder, and shows prompt emptying of the agent into the duodenum. Nonvisualization of the gallbladder on scintigraphy implies obstruction of the cystic duct and is consistent with acute cholecystitis ( Fig. 34.2 ). Hepatobiliary scintigraphy is not useful in patients with reduced hepatic function because it depends on hepatic excretion of bile, but it is accurate in approximately 90% of patients with acute cholecystitis and may be more accurate than US alone in some presentations.

An added utility of scintigraphy is that in addition to gallbladder filling or nonfilling, it can also be used to quantify gallbladder emptying. Abnormal emptying is defined as an ejection fraction (EF) of 35% or less on scintigraphy, although cutoff values of between 35% and 40% have been reported. The evaluation of EF is potentially useful for patients with symptoms suggestive of biliary dyskinesia or chronic acalculous cholecystitis but usually unnecessary in cases of acute cholecystitis because 90% of acute cholecystitis is secondary to cholelithiasis obstruction of the cystic duct. Consensus on the utility of scintigraphy EF in diagnosing biliary dyskinesia or chronic cholecystitis remains debated. This is in part because of reliance on retrospective data limited by selection bias and because of inconsistent indications and variability in EF cutoff values. In a study of 141 symptomatic patients with normal US and scintigraphy demonstrating normal gallbladder filling and an EF of 35% or less who underwent cholecystectomy, 95% had symptom relief and 41% had cholecystitis on histologic evaluation of the gallbladder. A recent meta-analysis of studies from 1980 to 2016 ( n = 29 studies) examining the use of scintigraphy EF in the diagnosis of biliary dyskinesia or cholecystitis found only two randomized controlled trials (RCTs) and 27 observational studies. This meta-analysis demonstrated that of the patients who underwent cholecystectomy, the chance of symptom improvement was similar in patients with a low EF versus those with a normal EF (risk ratio [RR] 1.09, P = .07). However, they also found that in patients managed medically, symptom improvement was more likely in patients with normal EF than those with a low EF (RR 2.37, P < .0005). Because of the heterogeneity of studies and bias inherent in retrospective data, no definitive conclusions could be drawn regarding how to interpret a normal EF in the setting of biliary symptoms and unclear imaging findings. In the setting of biliary pain with unclear US findings, however, a low EF is considered a reliable indicator of a biliary etiology. ,
Hepatobiliary scintigraphy is more involved, more expensive, and requires a longer time than US; however, it should be considered in certain cases. Guidelines have suggested a diagnostic approach that starts with US for patients with biliary symptoms. If no gallstones are definitively identified, this should be followed by esophagogastroduodenoscopy to exclude alternative causes of symptoms, such as peptic ulcer disease or gastritis. If the endoscopy is negative, hepatobiliary scintigraphy should follow.
Computed tomography (CT; see Chapter 16 ) can also help diagnose acute cholecystitis and provides more detailed anatomic information than US. CT is particularly useful in patients whose symptoms suggest a complication such as pericholecystic abscess or an alternative diagnosis. The CT findings of acute cholecystitis are the same as those seen on US and include wall thickening, pericholecystic stranding or fluid, distension of the gallbladder, high-attenuation bile, and subserosal edema. CT is generally less sensitive than US for diagnosing acute cholecystitis, particularly early in the course, when the imaging findings may be subtle. ,
Initial treatment with antibiotics active against enteric bacteria should begin as soon as the patient is diagnosed with acute cholecystitis. Additionally, oral intake should be held, and appropriate intravenous (IV) fluid resuscitation should be started in preparation for surgery. Parenteral analgesics should be administered as needed.
The definitive treatment for acute cholecystitis is cholecystectomy (see Chapter 36 ). From the time this operation was first performed in 1882 by Langenbuch, open cholecystectomy has been the standard of care for patients with acute cholecystitis. With the advent of laparoscopic cholecystectomy in the 1980s, the standard approach has changed such that cholecystectomy is now routinely performed laparoscopically. The benefits of laparoscopic cholecystectomy are discussed in Chapter 36 , but they include a shorter postoperative stay and decreased analgesia requirements. Although the laparoscopic approach is now standard for most cases, it is interesting to note that two prospective randomized studies suggested little or no difference in intraoperative or postoperative complications or length of stay between laparoscopic versus small-incision open cholecystectomy , ; however, most of these patients underwent elective rather than urgent cholecystectomy.
Early analysis of the results of laparoscopic cholecystectomy in patients with acute versus chronic cholecystitis showed increased morbidity and mortality rates for patients with simple or complicated acute cholecystitis. Because of the increased morbidity and mortality, acute cholecystitis initially was considered a relative contraindication to laparoscopic cholecystectomy. Subsequent reports, however, have shown improved safety of this technique in the acute setting. The conversion rate to an open procedure is higher for patients with acute cholecystitis compared with patients undergoing elective cholecystectomy, but most patients with acute cholecystitis (>80%) can undergo successful laparoscopic cholecystectomy. Retrospective series have reported that risk factors for conversion to open cholecystectomy include obesity, elevated WBC count and elevated bilirubin, , previous surgery, and male gender.
Other novel surgical approaches to cholecystectomy have been proposed to treat patients with symptomatic gallstones, including mini-laparoscopic cholecystectomy (see Chapter 36 ), which uses 2- to 3-mm ports ; mini-cholecystectomy, in which a small (mean, 5.5 cm) incision is used to remove the gallbladder; single-incision laparoscopic cholecystectomy; and natural orifice transluminal endoscopic (NOTES) cholecystectomy with transvaginal extraction. Prospective randomized studies evaluating the safety of these techniques are lacking, but existing data suggest decreased postoperative pain and improved cosmesis at the expense of slightly longer operating times with these techniques.
Laparoscopic subtotal cholecystectomy (LSC) has also been evaluated as a means of decreasing the conversion rate to open procedure in patients with acute cholecystitis , (see Chapter 36 ). Subtotal cholecystectomy can be of two types: fenestrating or reconstituting (see Figs. 34.3 and 34.4 ). A subtotal fenestrating cholecystectomy involves excising the peritonealized gallbladder (anterior surface) and leaving the posterior wall of the gallbladder in situ. The remnant mucosa may be cauterized, any stone burden is evacuated, and the cut edge of the gallbladder can then be oversewn or cauterized. The cystic duct can also be sutured closed from the luminal/mucosal side to avoid injury to the common bile duct (see Chapter 36 ). In contrast, a reconstituting subtotal cholecystectomy involves excising the peritonealized gallbladder, extracting any stones, and closing (sewing or stapling) the inferior gallbladder in a way that preserves a small lumen and patent biliary drainage through the cystic duct. Drains are typically left after either approach.
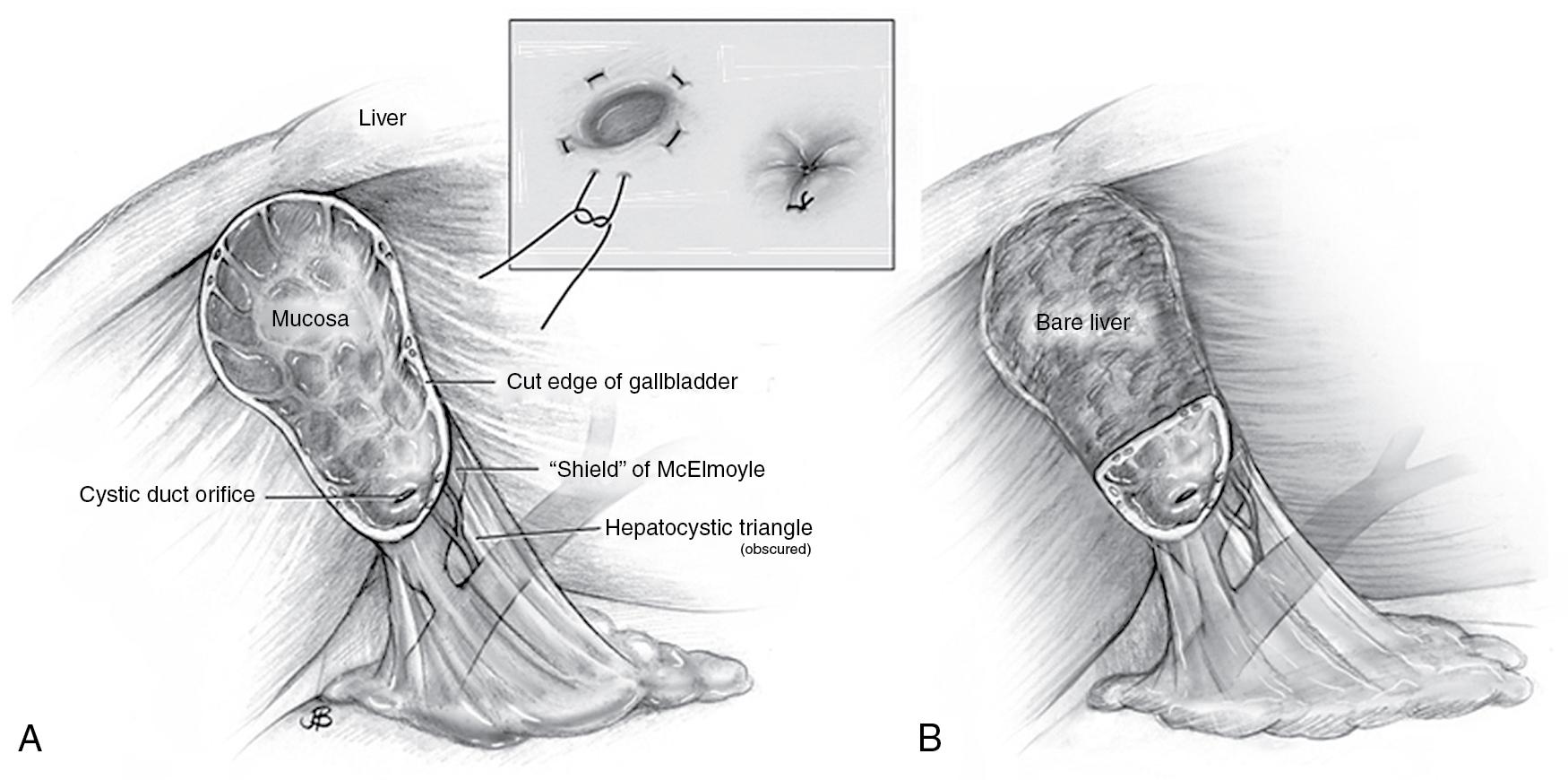
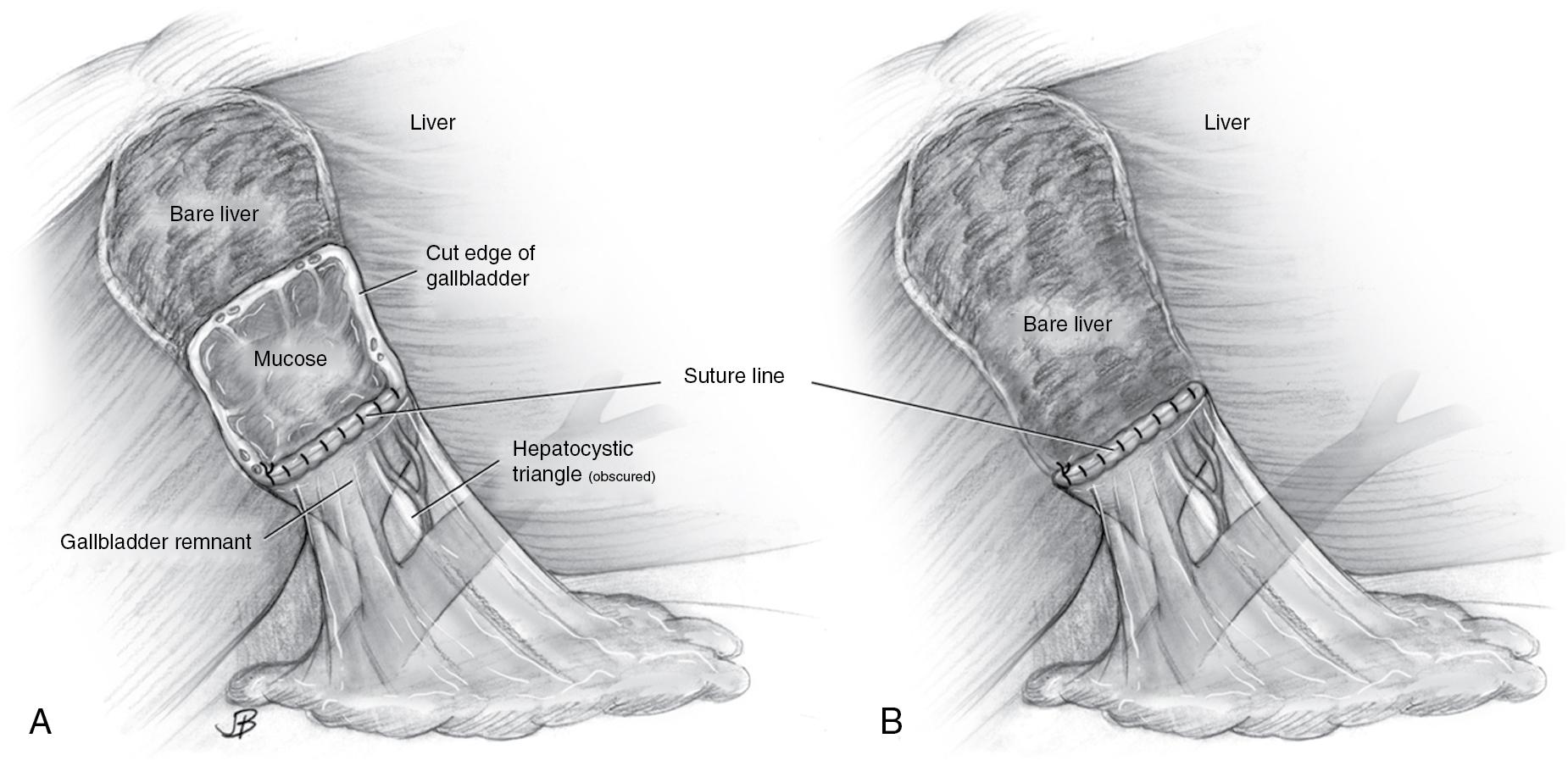
Each subtotal cholecystectomy technique has different advantages and disadvantages, and their feasibility depends on patient presentation and anatomy. In a retrospective multi-institutional study comparing outcomes after subtotal fenestration versus reconstituting cholecystectomy (median follow-up of six years, interquartile range [IQR] 5–10 years), fenestration was associated with a higher risk of bile leak (18% vs. 7%, P < .02), wound infection (11% vs. 3%, P < .02) and longer hospitalization (median 5 days, IQR 3–17 days vs. three days, IQR 2–6 days, P = .005). However, reconstitution was associated with an increased risk of recurrent biliary pathology (18% vs. 9%, P < .02). Interestingly, in this study patients who underwent subtotal fenestrating cholecystectomy had an increased risk of subsequent completion cholecystectomy (9% vs. 4%, P < .02) despite a lower risk of recurrent cholecystitis. This may be reflective of the fact that patients underwent completion cholecystectomy for various indications such as choledocholithiasis and biliary colic, in addition to recurrent cholecystitis. There was, however, no difference in reintervention rate (endoscopic retrograde cholangiopancreatography [ERCP] or completion cholecystectomy) between those patients who underwent subtotal fenestrating versus reconstituting cholecystectomy (32% vs. 26%, P = .21). Laparoscopic cholecystectomy remains the standard therapy for definitive treatment of patients with acute cholecystitis. Conversion to an open approach or performing either type of subtotal cholecystectomy is appropriate for high-risk cases where a laparoscopic approach may be unsafe.
In patients with a high perioperative risk related to sepsis, duration of presenting symptoms, or underlying medical comorbidities, initial treatment of acute cholecystitis with percutaneous cholecystostomy tube placement is preferred (see Chapter 35 ). The standard of care for this high-risk patient cohort is percutaneous cholecystostomy tube followed by interval cholecystectomy, which is typically performed at least six to eight weeks after tube placement. Percutaneous cholecystostomy is placed under US or CT guidance and decompresses the gallbladder by evacuating the infected bile and relieving the pain associated with gallbladder distension from outlet obstruction. Most patients (>80%) have immediate clinical improvement after biliary decompression.
The incidence and severity of complications after percutaneous cholecystostomy tube placement is low and relatively benign. , Approximately 33% of patients who undergo cholecystostomy tube for acute cholecystitis will experience tube-related complications. The most common complications include tube displacement, tube site skin infection, and tube site pain. ,
After resolution of the acute inflammatory process, the standard of care includes interval cholecystectomy in patients without contraindications to surgery. Laparoscopic cholecystectomy can often be performed successfully, but the conversion from a laparoscopic to open approach is relatively increased in this population. In patients with previous percutaneous cholecystostomy, the rate of conversion from a laparoscopic to open approach ranges from 14% to 32%. , This is in contrast to the relatively low conversion rates for elective cholecystectomy (5%) and emergent cholecystectomy performed for acute cholecystitis (6%). Overall, compared with patients who were treated with antibiotics and delayed cholecystectomy, patients who underwent percutaneous cholecystostomy tube followed by interval cholecystectomy had shorter overall hospital stay and decreased cost, although there is clearly selection bias, which can make these differences difficult to evaluate.
There is a subset of patients who are unable to undergo interval cholecystectomy, either because of prohibitive anticipated surgical morbidity and mortality or because of other considerations (i.e., noncurative cancer). These patients have two options for tube management: tube removal or indefinite tube continuation (sometimes referred to as a destination tube). The criteria for cholecystostomy tube removal include resolution of the obstructive inflammatory process and patency of the cystic and common bile ducts. Biliary patency can be assessed via a clamp trial where the cholecystostomy tube is clamped and the patient self-monitors for any recurrent symptoms or via cholecystography to confirm tube position and duct patency.
The most concerning complication after cholecystostomy tube removal in nonsurgical patients is recurrent cholecystitis. Recurrent cholecystitis after cholecystostomy tube removal is reported to be between 10% to 21%, although these numbers are skewed by the selection bias of retrospective research and the high mortality (43%) of nonsurgical patients. , Risk factors for recurrent cholecystitis after cholecystostomy tube removal include tube removal within 44 days of percutaneous placement (OR 5.6; 95% confidence interval [CI]: 1.25–23.2; P = .02) and history of choledocholithiasis (OR 24.4; 95% CI: 2.7–220.7; P = .005). Patients who underwent a successful clamping trial before tube removal had lower rates of recurrent cholecystitis (OR 0.10; 95% CI 0.01–0.8; P = .03).
Advancements in endoscopic technologies and techniques have led to expanded options for definitive nonoperative management of acute cholecystitis. Lumen-apposing self-expandable metallic stents (LASEMS) have historically been used for transgastric drainage of pancreatic pseudocyst or walled off necrosis (see Chapter 56 ). They have more recently, however, been adapted to offer transduodenal or transgastric gallbladder drainage in patients unfit for cholecystectomy. One such stent is the AXIOS stent (AXIOS; Xlumena Inc, Mountain View, CA), a fully covered self-expandable nitinol-based stent with anchoring flanges on opposing ends that inhibit post-placement migration. Its use in cholecystitis as a means of achieving biliary decompression has only been described in case reports of anywhere from one to 30 patients. A systematic review of 11 studies (78 patients total) found that AXIOS/LASEMS placement from duodenum or stomach to the gallbladder was successfully achieved in 97% of patients and relieved symptoms in 99.6% of patients. Minor procedure-associated complications included transient fever ( n = 1), hematochezia ( n = 1), and pain ( n = 1). There were no procedure-associated major complications and no reports of stent migration. Stents were removed within one to two weeks in 10 out of the 11 studies, after establishment of a fistulous tract between the gallbladder and either duodenum or stomach. Only one study ( n = 27 patients) examined long-term outcomes (three months): three patients developed mucosal ingrowth and two patients developed recurrent cholecystitis from an obstructed stent. Any consideration of LASEMS should occur at initial diagnosis and treatment planning as LASEMS are not compatible with other biliary drainage approaches (i.e., percutaneous cholecystostomy tube). LASEMS could offer a nonoperative management strategy for acute cholecystitis in patients unfit for surgery; however, multi-institutional RCTs are needed to definitively understand the short- and long-term risks and benefits of this approach and the most appropriate population for its application.
The optimal interval of time between the diagnosis of acute cholecystitis and definitive treatment with cholecystectomy has been the subject of many prospective randomized trials, with nine evaluating open cholecystectomy and five evaluating laparoscopic cholecystectomy. , The concern in operating on patients with early cholecystitis (typically defined as <72 hours) is the fear of increased operative complications, including common bile duct injury (see Chapter 36 ). The downside of performing delayed cholecystectomy (weeks after the diagnosis of cholecystitis) is that a subset of patients will develop recurrence cholecystitis before cholecystectomy, leading to readmission and urgent surgery. In multiple randomized prospective trials evaluating the timing of open cholecystectomy, patients undergoing early operation did not experience any increase in perioperative morbidity or mortality and had a shorter total length of hospital stay compared with patients undergoing delayed operation. , In addition, a meta-analysis of these trials demonstrated that more than 20% of patients did not respond to medical management while awaiting definitive treatment, and approximately half of these patients required urgent surgical treatment as a result. Additionally, no increase in morbidity was seen in patients undergoing early (<72 hours from symptom onset) versus late (>72 hours from symptom onset) cholecystectomy with either laparoscopic ( P = .6) or open ( P = .2) approach. However, patients undergoing delayed cholecystectomy had significantly prolonged total hospitalization and higher cost of care compared with patients who underwent early cholecystectomy.
Injury to the common bile duct (see Chapters 36 and 42 ) is a feared complication of any cholecystectomy but particularly for those performed in the setting of acute cholecystitis. Acute severe inflammation can obscure biliary anatomy and predispose to biliary injuries and complications. Multiple prospective randomized trials have demonstrated ( Table 34.1 ) that although early cholecystectomy is associated with a significant increase in operation time compared with delayed cholecystectomy ( P = .002), there is no significant difference in postoperative morbidity or mortality, including the incidence of common bile duct injury.
| REFERENCE | N | DEFINITION | MORBIDITY RATE | LENGTH OF STAY (DAYS) | CONVERSION TO OPEN CHOLECYSTECTOMY |
|---|---|---|---|---|---|
| Ozkardes et al., 2014 | 60 | Early: <24 hours | 27% | 5 | 13% |
| Late: 6–8 weeks | 0 | 8 | 0% | ||
| Saber & Hokkam, 2014 | 120 | Early: <72 hours | NR | 2 | 5% |
| Late: 6–8 weeks | NR | 6 | 2% | ||
| Gutt et al., 2013 | 618 | Early: <24 hours | 12% | 5 | 10% |
| Late: 7–45 days | 34% | 10 | 12% | ||
| Macafee et al., 2009 | 72 | Early: <72 hours | 22% | 6 | 3% |
| Late: 3 months | 11% | 6 | 3% | ||
| Kolla et al., 2004 | 40 | Early: <4 days | 20% | 4 | 25% |
| Late: 6–12 weeks | 15% | 10 | 25% | ||
| Johansson et al., 2003 | 145 | Early: <7 days | 18% | 5 | 31% |
| Late: 6–8 weeks | 10% | 8 | 21% | ||
| Lai et al., 1998 | 104 | Early: <24 hours | 9% | 8 | 24% |
| Late: 6–8 weeks | 8% | 12 | 8% | ||
| Lo et al., 1998 | 99 | Early: <3 days | 29% | 6 | 11% |
| Late: 6–8 weeks | 13% | 11 | 23% |
No significant difference has been found in the conversion rate (laparoscopic to open approach) in early versus delayed cholecystectomy. Nevertheless, conversion to open surgery was higher (20%–30%) in patients with acute cholecystitis compared with patients undergoing elective laparoscopic cholecystectomy in the nonacute setting. Perhaps the most important finding was that in all but one study, patients randomly assigned to delayed cholecystectomy did not respond to medical management (supportive care and antibiotics) in 15% to 30% of cases. Although patients in the early surgery group generally experienced a longer postoperative hospital stay ( P = .004), most of these trials demonstrated a decrease in overall length of hospital stay (surgical admission plus readmission) in the early compared with the delayed group (cumulative P < .001). , Early cholecystectomy has also been demonstrated to be more cost effective than delayed cholecystectomy. This was illustrated in a meta-analysis of studies of various designs performed by Lau and colleagues, which concluded that early surgery was more cost effective because of its associated reduced overall length of hospital stay and avoidance of readmissions for recurrent symptoms. Early laparoscopic cholecystectomy is therefore the preferred surgical technique for patients with acute cholecystitis.
Catena and colleagues have proposed the use of a harmonic scalpel for improved hemostasis and biliostasis in laparoscopic cholecystectomy, and preliminary data suggested it may decrease the conversion rate to open procedure in patients undergoing laparoscopic cholecystectomy for acute cholecystitis. A prospective RCT subsequently confirmed these findings.
The majority of trials examining early versus delayed laparoscopic cholecystectomy define “early” as within 72 hours of symptom onset, but the impact of the time from symptom onset to cholecystectomy on outcomes has also been examined in more detail. A nonrandomized prospective study by Tzovaras and colleagues assessed 129 patients undergoing laparoscopic cholecystectomy for acute cholecystitis. Patients were divided into three groups according to the time from symptom onset to cholecystectomy: less than three days, between four and seven days, and greater than seven days. This study found no significant difference in conversion rate (laparoscopic to open), morbidity, or postoperative hospital stay among these groups and suggested that cholecystectomy may be safe even if performed up to or after seven days from symptom onset. The results of this study, however, should be interpreted in the context of its inherent selection bias because the timing of surgery was determined by surgeon discretion rather than randomization of clinically similar groups.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here