Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Duplex ultrasound (DU) refers to two modes of ultrasound, namely B mode that obtains gray-scale imaging, and Doppler that evaluates the velocity and direction of blood flow in the vessel. DU determines the degree of carotid artery stenosis based on gray-scale plaque estimation and Doppler spectral analysis of the peak systolic velocity (PSV) and end-diastolic velocity (EDV) in the common carotid artery (CCA), internal carotid artery (ICA) and external carotid artery (ECA), as well as calculation of the ICA to CCA PSV ratio (see Ch. 22 , Vascular Laboratory: Arterial Duplex Scanning). Based on these, consensus ultrasound criteria were developed to standardize carotid DU examinations and for diagnosing carotid artery stenosis severity ( Table 91.1 ). Criteria have been validated to diagnose a greater than 70% stenosis. DU remains the first-line imaging modality for identifying patients with ICA stenosis because it is noninvasive, has low cost, is readily available, and has high sensitivity and specificity ( Table 91.2 ), with now the vast majority of carotid endarterectomy (CEA) procedures performed on the basis of DU alone.
| Degree of Stenosis (%) | PRIMARY PARAMETERS | ADDITIONAL PARAMETERS | ||
|---|---|---|---|---|
| ICA PSV (cm/s) | Plaque Estimate (%) a | ICA/CCA PSV (ratio) | ICA EDV (cm/s) | |
| Normal | <125 | None | <2.0 | <40 |
| <50 | <125 | <50 | <2.0 | <40 |
| 50–69 | 125–230 | ≥50 | 2.0–4.0 | 40–100 |
| >70 but less than near occlusion | >230 | ≥50 | >4.0 | >100 |
| Near occlusion | High, low or undetectable | Visible | Variable | Variable |
| Total occlusion | Undetectable | Visible, no detectable lumen | Not applicable | Not applicable |
a Plaque estimate (diameter reduction) with gray-scale and color Doppler US.
| Stenosis Group | Imaging | Sensitivity (95% CI) | Specificity (95% CI) |
|---|---|---|---|
| 70%–99% (0.77–0.89) | US | 0.89 (0.85–0.92) | 0.84 |
| (0.91–0.97) | CTA | 0.77 (0.68–0.84) | 0.95 |
| (0.76–0.90) | MRA | 0.88 (0.82–0.92) | 0.84 |
| (0.89–0.96) | CEMRA | 0.94 (0.88–0.97) | 0.93 |
| 50%–69% (0.87–0.94) | US | 0.36 (0.25–0.49) | 0.91 |
| (0.63–0.89) | CTA | 0.67 (0.30–0.90) | 0.79 |
| (0.78–0.97) | MRA | 0.37 (0.26–0.49) | 0.91 |
| (0.93–0.99) | CEMRA | 0.77 (0.59–0.89) | 0.97 |
| 0%–49%, 100% (0.62–0.95) | US | 0.83 (0.73–0.90) | 0.84 |
| (0.74–0.98) | CTA | 0.81 (0.70–0.88) | 0.91 |
| (0.76–0.95) | MRA | 0.81 (0.70–0.88) | 0.88 |
| (0.90–0.99) | CEMRA | 0.96 (0.90–0.99) | 0.96 |
However, there are certain limitations when interpreting DU findings. These include inability to image below the clavicle or above the mandible, overestimation of ipsilateral stenosis in the presence of contralateral severe carotid stenosis or occlusion, , inability to obtain accurate velocity measurements in presence of excessive calcification due to acoustic shadowing, and difficulty interpreting carotid subocclusion versus a so-called “string sign.” Additionally, due to decreased vessel wall compliance, carotid stents will increase peak systolic velocities. If this is not taken into account for post-carotid stent DU surveillance, the degree of in-stent restenosis will be overestimated. Therefore, consensus criteria have been adjusted to account for this ( Table 91.3 ).
| Stenosis % | Stented Carotid Artery | Native Carotid Artery |
|---|---|---|
| 0%–19% | PSV <150 cm/s and ICA/CCA ratio <2.15 |
PSV <130 cm/s |
| 20%–49% | PSV 150–219 cm/s | PSV 130–189 cm/s |
| 50%–79% | PSV 220–339 cm/s and ICA/CCA ratio ≥2.7 | PSV 190–249 cm/s and EDV <120 cm/s |
| 80%–99% | PSV ≥340 cm/s and ICA/CCA ratio ≥4.15 | PSV ≥250 cm/s and EDV ≥120 cm/s, or ICA/CCA ratio ≥3.2 |
Finally, extracranial DU offers limited information about flow dynamics in the vertebral arteries (VAs). DU is useful for demonstrating antegrade and/or retrograde flow, suggesting proximal subclavian or innominate artery disease. However, it is not always possible to image VA origins and only those sections running between the transverse processes can be imaged distally. Accordingly, if a patient presents with suspected vertebrobasilar symptoms, DU assessment can exclude co-existent carotid or arch vessel disease, but further diagnostic imaging is mandatory.
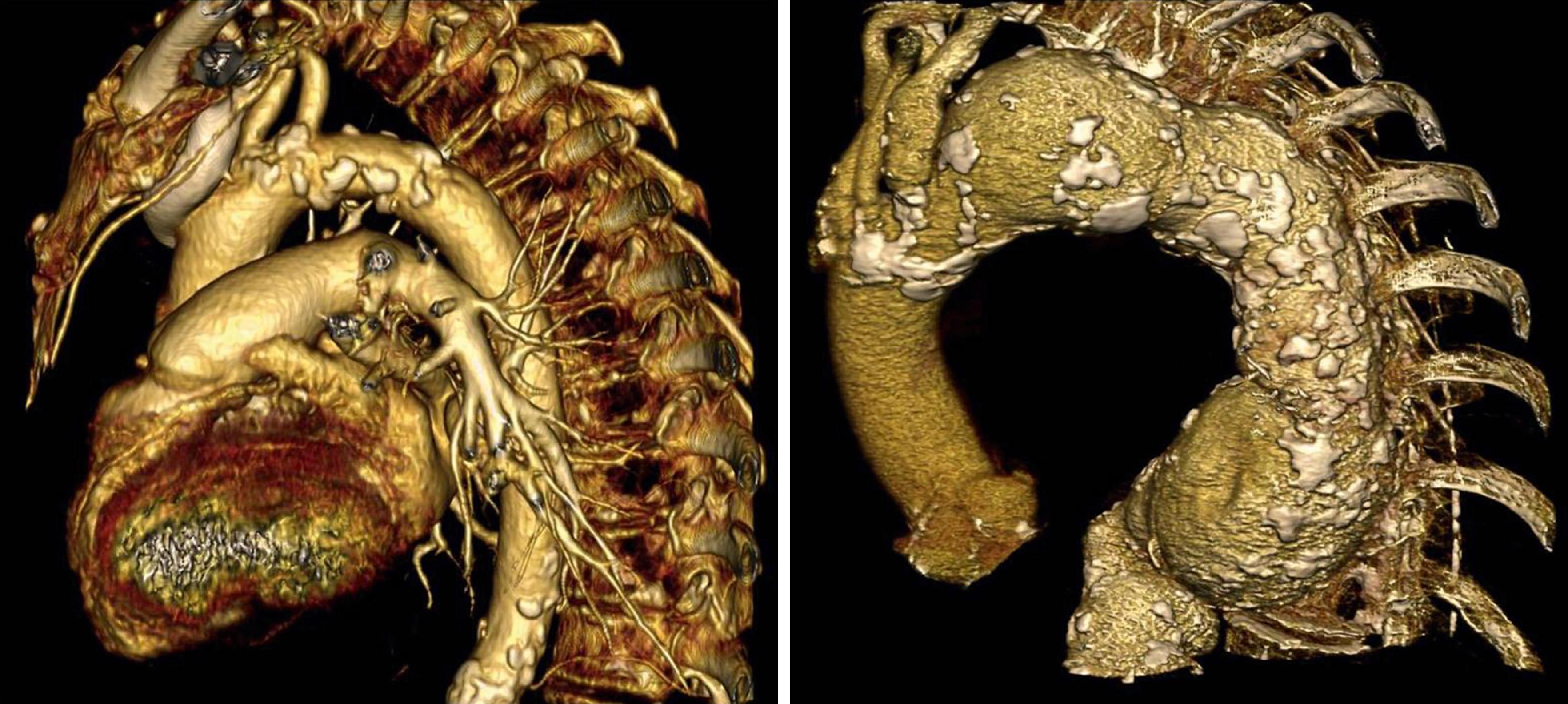
Multidetector CT angiogram (MDCTA) has revolutionized the role of CT in carotid artery disease. MDCTA is fast, offers excellent submillimeter spatial resolution and can visualize soft tissue, bone and vessel at the same time. It can rapidly demonstrate vascular anomalies, even if occluded ( Fig. 91.1 ), and can provide data regarding the extent of vessel calcification, especially in the aortic arch ( Fig. 91.2 ). Additionally, by utilizing rapid advances in software and hardware, combined with bolus tracking, imaging can now be achieved with a lower radiation dose and with reduced contrast than conventional CT (see Ch. 29 , Computed Tomography).
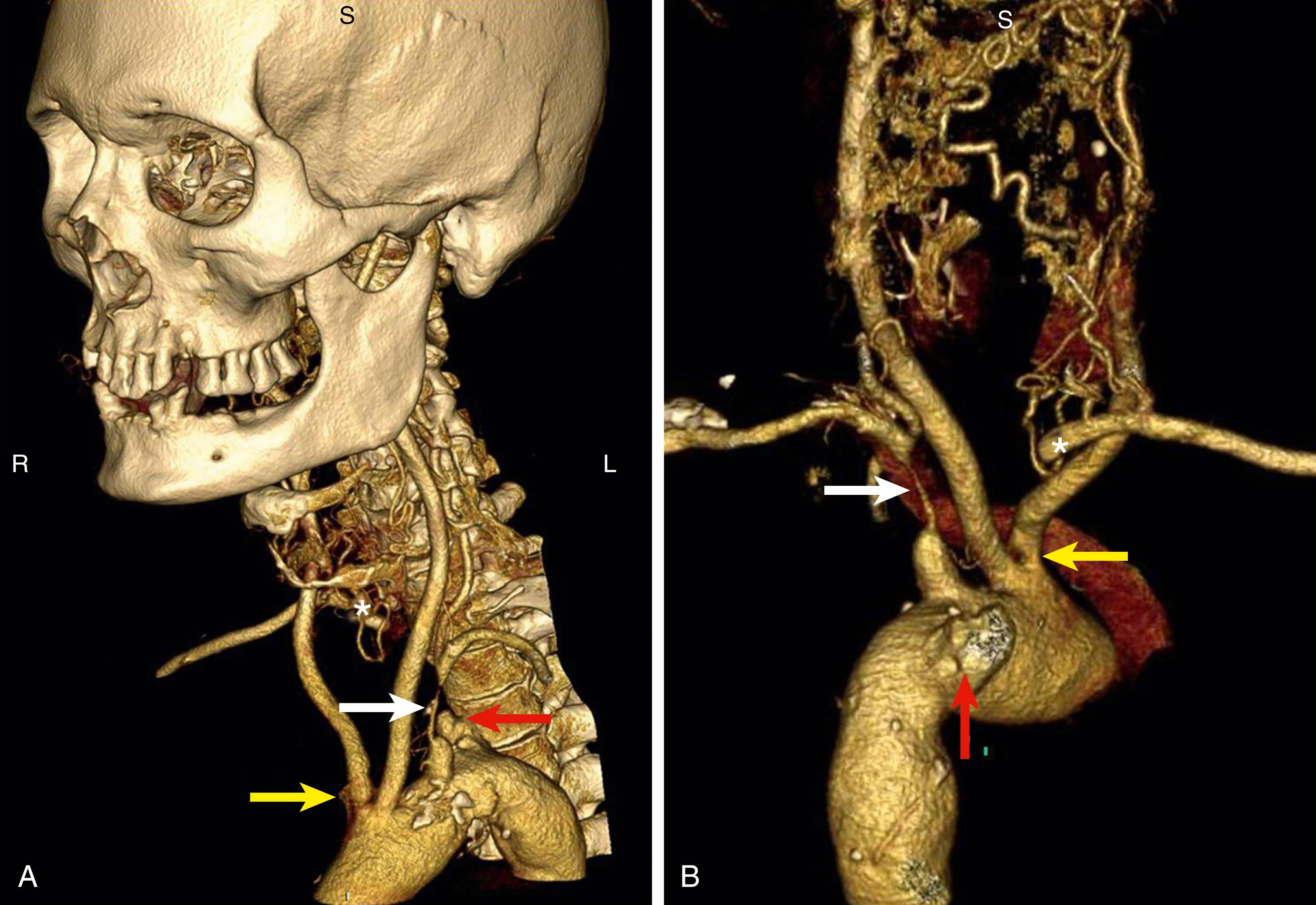
MDCTA is particularly useful for patients with 50%–69% stenosis where DU has been found to be less sensitive and specific, or when further anatomic evaluation of proximal or distal arteries is needed. Additionally, it is indicated for patients undergoing evaluation for carotid artery stenting (CAS) to delineate relevant anatomy necessary for procedural success. Given the limitations of DU, MDCTA is mandatory for diagnostic evaluation of vertebrobasilar disease as it allows for accurate visualization of the origins of these vessels as they pass through bony structures. Vessel tortuosity is easily demonstrated, and there is no problem with artifact at the VA origins.
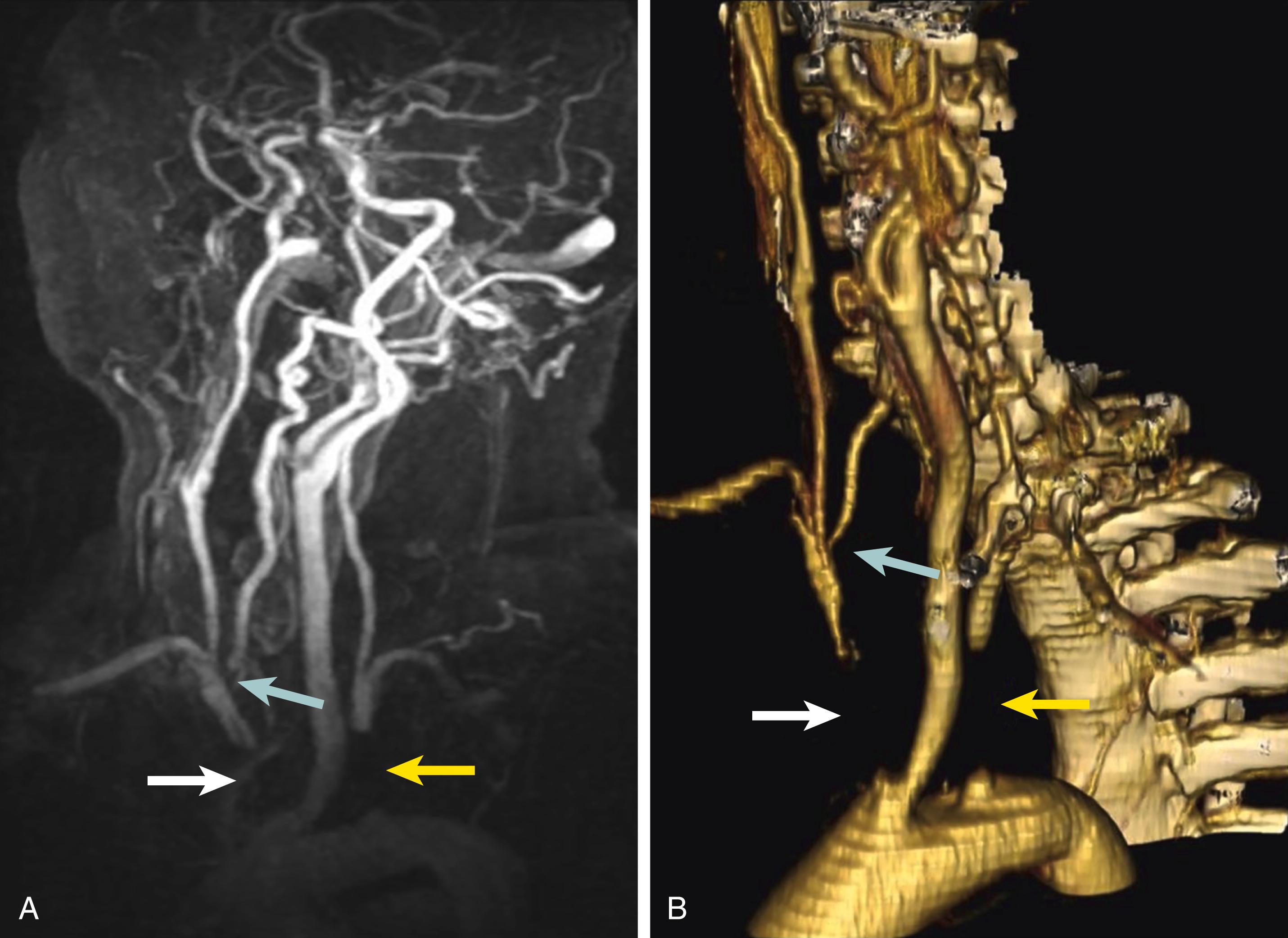
MDCTA is limited by the need for intravenous contrast, a risk of contrast-related nephropathy, not being able to provide information on flow directionality, and the exposure to radiation. Additionally, a large calcium burden can limit the ability to distinguish contrast from calcium during post-processing imaging. Finally, there are occasional situations where MDCTA and contrast-enhanced magnetic resonance angiography (CEMRA) (discussed below) cannot reliably provide accurate and diagnostic imaging information in selected complex patients, especially those with tandem syphon disease or stenoses within coiled segments of the ICA ( Fig. 91.3 ). In these cases, high quality selective carotid digital subtraction angiography (DSA) imaging provides valuable additional information.
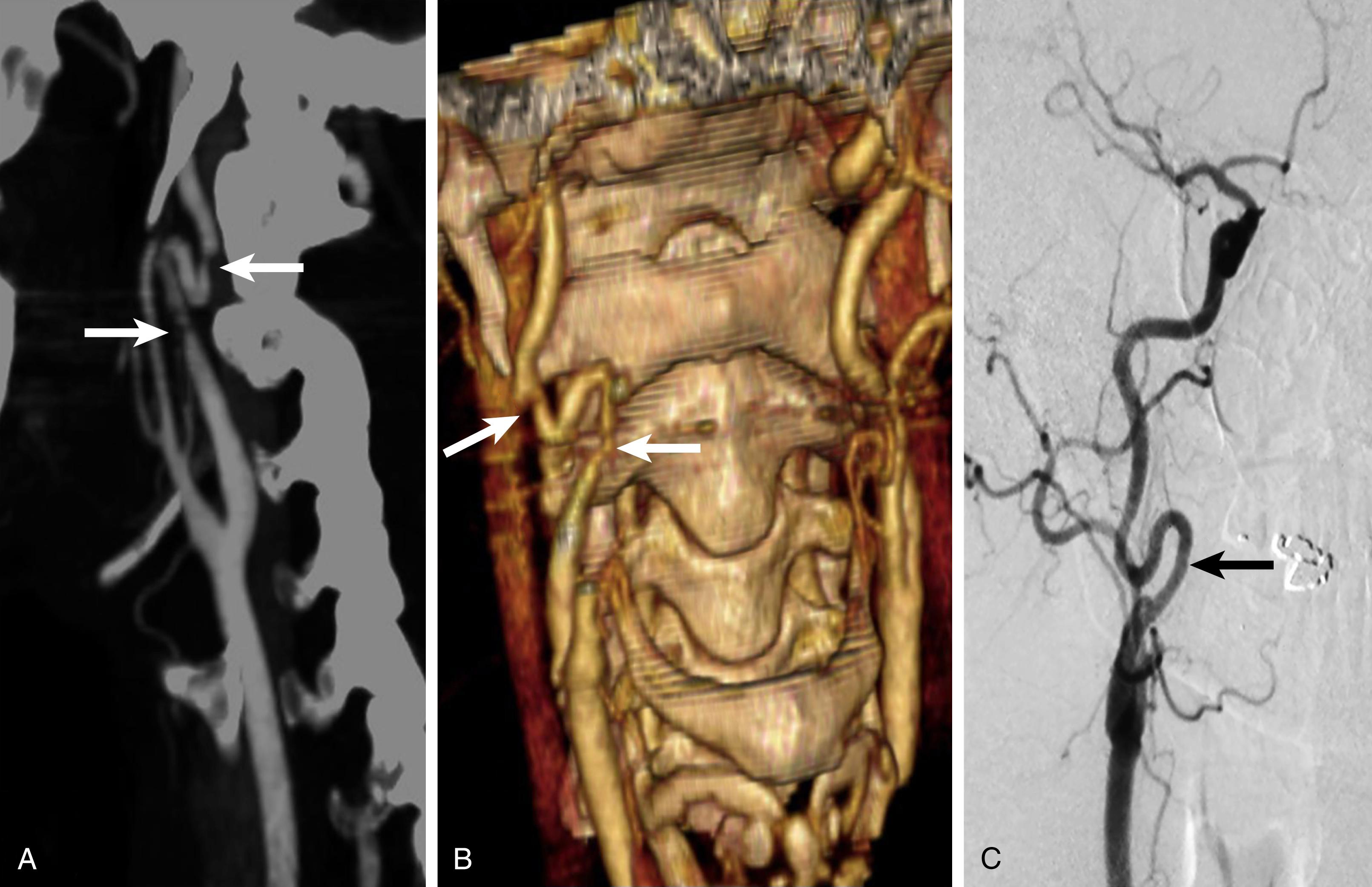
Digital subtraction angiography (DSA) remains the gold standard for carotid imaging; however, given advances in DU and MDCTA, it is rarely required prior to surgery. DSA is performed by placing an injection catheter, usually through a femoral approach, into first the aortic arch and then individually selecting the common carotid arteries, followed by power injections of iodinated contrast (see Ch. 27 , Arteriography). The degree of ICA stenosis is measured using the North American Symptomatic Carotid Endarterectomy Trial (NASCET) method, which is the calculated ratio between the residual luminal surface at the level of the ICA stenosis and the luminal surface of the distal normal ICA where there is no stenosis. Of note, DSA usually requires imaging in more than one plane to accurately identify lesion severity. DSA provides high quality imaging that is accurate, objective, and easy to interpret. It can identify lesions from the aortic arch to the intracranial vessels. It is particularly useful for diagnosis when there is discordant imaging findings between DU and MDCTA and/or CEMRA. Additionally, DSA is useful in situations where MDCTA can provide misleading information in patients with severe distal ICA disease ( Fig. 91.4 ) and in patients where there is marked coiling of the ICA in the upper reaches of the neck (see Fig. 91.3 ). Most importantly, DSA is both a diagnostic and therapeutic tool thus making it particularly useful in acute situations such as trauma or the postoperative management of stroke after surgery.
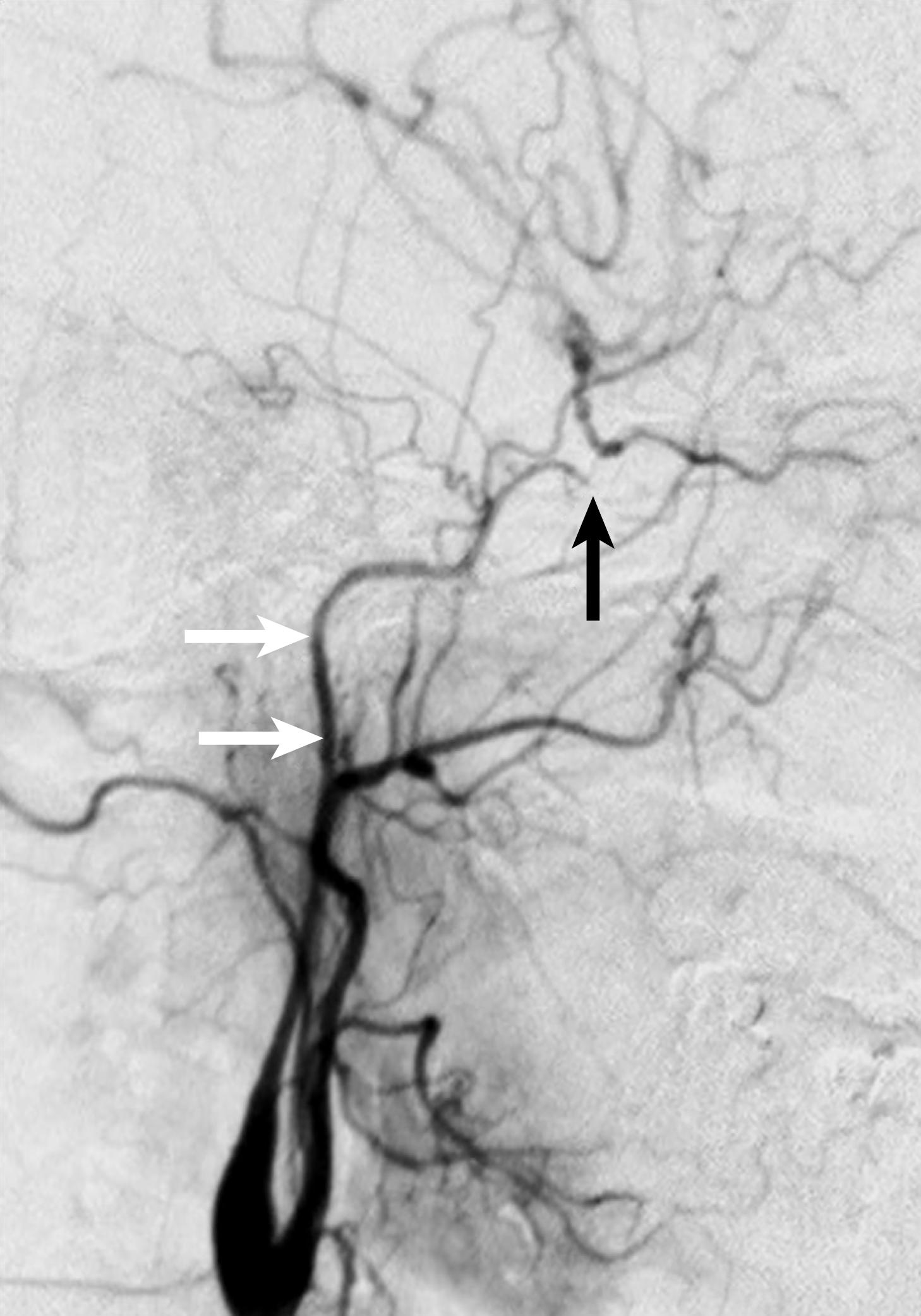
However, the performance of angiography is not without potential risks secondary to the invasive nature of the procedure and the use of contrast, stroke being the most devastating of those complications. In the Asymptomatic Carotid Atherosclerosis Study (ACAS), angiographic related stroke accounted for 50% of the procedural complications after CEA. In a review of complications following 23,416 carotid angiograms, groin hematomas occurred after 4% of procedures, while 2.6% of patients suffered a transient or permanent neurologic deficit. As a result, there is no role for routine catheter angiography in modern cerebrovascular practice.
Magnetic resonance angiography (MRA) in the evaluation of extracranial carotid disease utilizes either contrast-enhanced MRA (CEMRA) or time of flight (TOF) MRA. Both have the advantage of being noninvasive, do not require ionizing radiation, and provide an unlimited number of projections of the carotid lumen from a single acquisition. It can also assess intrathoracic and intracranial lesions that are not amenable to DU interrogation. When dedicated protocols are used, MRA also can demonstrate specific plaque components, including calcium, lipid, fibrocellular element, or thrombus within the plaques. Table 91.2 summarizes sensitivity analyses for CEMRA and TOF MRA (see Ch. 30 , Magnetic Resonance Imaging and Arteriography).
CEMRA uses gadolinium as the contrast agent, and compared to MDCTA has fewer flow-related artifacts and provides a much greater field of view that enables high-resolution imaging from the aortic arch ( Fig. 91.5 ) up to the circle of Willis (COW) while retaining the ability to evaluate flow directionality. However, unlike MDCTA, adjacent soft tissue structures are not well visualized unless unsubtracted or additional MR imaging is performed, and calcium within a plaque is not well defined, and as a result the degree of stenosis is often overestimated. Additionally, CEMRA confers the risk of nephrogenic systemic fibrosis in patients with renal insufficiency exposed to the gadolinium contrast agent. Unlike iodinated contrast-induced nephropathy, following which commonly patients recover kidney function, this is irreversible leading to permanent renal failure, along with fibrosis of the skin and joints. To avoid the risk of this devastating complication, TOF MRA is particularly useful as it does not use any contrast, provides strong vascular signals, even when flow is low, and additional three-dimensional TOF MRA can offer better spatial resolution for measuring stenosis severity and enabling assessment of flow directionality in steal phenomena. However, in practice, the lack of availability and the need for specific sequences and hardware to enable dedicated plaque imaging, along with the rare but devastating complication of nephrogenic systemic fibrosis, have left now little role for MRA.
Transcranial Doppler (TCD) ultrasound provides rapid, noninvasive, real-time assessment of cerebrovascular function. TCD can be used to measure flow velocity in the basal arteries of the brain including the anterior cerebral artery (ACA), middle cerebral artery (MCA) and posterior cerebral arteries (PCA); and to assess relative changes in flow, diagnose focal vascular stenosis, or to detect embolic signals within these arteries. While there is no routine role for TCD in evaluating patients with carotid artery disease, especially given its lack of availability in most vascular labs and clinical settings, its utility in diagnosing spontaneous in vivo embolization can be useful. Embolization is more commonly encountered in the MCA ipsilateral to a symptomatic carotid stenosis, and this is indicative of an unstable plaque (see Ch. 89 , Cerebrovascular Disease: The Unstable Carotid Plaque). TCD-diagnosed spontaneous embolization may also assume an increasingly important role in identifying a high risk for stroke cohort of patients with asymptomatic carotid disease. A meta-analysis of six observational studies, including 1144 patients, with an asymptomatic 70%–99% stenosis observed that the presence of a single embolic signal during one hour of TCD monitoring was associated with a near eightfold increase in stroke during follow-up. Thus, in the rare situation where TCD is available, recognition of this phenomenon should prompt the surgeon to undertake an expedited CEA.
The Society for Vascular Surgery (SVS) clinical recommendations for carotid imaging recommendations state that DU in an Intersocietal Commission for the Accreditation of Vascular Laboratories (ICAVL) accredited vascular laboratory is the initial diagnostic imaging of choice for evaluating the severity of stenosis in both symptomatic and asymptomatic patients. These images should be reviewed by physicians experienced in vascular ultrasound interpretation. Unequivocal identification of stenosis of 50% to 99% in neurologically symptomatic patients or 70% to 99% in asymptomatic patients is sufficient to make a decision regarding intervention. When DU is nondiagnostic or suggests stenosis of intermediate severity (50% to 69%) in an asymptomatic patient, or when evaluation of the vessels proximal or distal to the cervical carotid arteries is needed for diagnosis or to plan therapy, additional imaging with MDCTA, CEMRA or DSA is required.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here