Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Cardiac involvement in systemic disease can broadly be divided into those conditions where the heart is involved in the disease process itself and those where a structural or functional cardiac abnormality is associated with other anomalies, usually in a recognizable syndrome. Many of the conditions in the latter group have received attention in the sections of this book dealing with etiology and genetics. They are not dealt with again here, although they may be mentioned or the discussion amplified as necessary. A vast number of systemic diseases, nonetheless, can involve the heart during childhood. Cardiac aspects of the latter diseases are the focus of this chapter.
Von Gierke disease is an autosomal recessive disorder. Clinical manifestations of type I glycogen storage disease are profound hypoglycemia associated with hyperlipidemia, hyperuricemia, and lactic acidosis. It presents in childhood and primarily involves the liver, kidneys, and small intestinal mucosa. Pulmonary hypertension in association with type Ia glycogen storage disease has been described; when present, the prognosis is very poor. Postulated mechanisms include chronic stimulation of the smooth muscle of the pulmonary arterioles by the persistent hepatic metabolism of circulating catecholamines such as serotonin. Indeed, serotonin levels have been shown to be markedly elevated in some patients with glycogen storage disease type I. However, elevated serotonin levels alone do not appear to confer pulmonary vascular disease on these patients. Rather, it is hypothesized that other mediating factors in concert with persistently elevated serotonin levels increase the risk for pulmonary vascular changes. Gene therapies are being developed to treat glycogen storage disease type I and initial results appear promising.
Pompe disease is a generalized glycogen storage disease in which glycogen of normal structure accumulates in the myocardium, skeletal muscle, and liver. The disease is progressive and is associated with deficiency of lysosomal α-1,4-glucosidase. There are four subtypes based on age at onset of clinical symptoms: infantile (classic Pompe disease), childhood, juvenile, and adult. The age at onset correlates inversely with the measured activity of lysosomal α-1,4-glucosidase in muscle cells or fibroblasts. In the infantile form, which generally has more severe cardiac involvement than the forms with later onset, there is generalized accumulation of glycogen in the heart (including in the conduction tissues), in skeletal muscle (notably the tongue and diaphragm), and in the liver. Central and peripheral neurons and smooth muscle are also affected. The results are cardiomegaly, hepatomegaly, a thickened diaphragm, and macroglossia. In the heart, the glycogen is deposited mainly in ventricular muscle. There is gross thickening of the ventricular walls, with impairment of both diastolic and systolic performance. The infants typically appear normal at birth, although cases of severe neonatal ventricular hypertrophy have been reported. The median age at onset of clinical symptoms is 1.6 months. Muscle weakness and hypotonia along with loss of motor milestones are noted during the first 6 months of life, and signs of CHF become evident. Although there is excess glycogen in the liver, hepatomegaly is not commonly present until heart failure is apparent. The disease is progressive, and most affected babies die before the age of 1 year. The clinical course may be complicated by arrhythmias. Since patients with Pompe disease appear very sensitive to digoxin, this drug must be used with extreme caution. Irritability and poor feeding often draw attention to the disease. The cardiac physical signs are not characteristic, with variable murmurs being heard. Unexplained cardiomegaly and CHF in a generally “floppy” baby should suggest the diagnosis.
The chest radiograph may be normal at birth, but in all affected infants the heart becomes enlarged within a few weeks. There is no specific pattern to the cardiac silhouette but rather a generalized smooth enlargement of the contour. The characteristic electrocardiographic features are a short PR interval, wider than normal QRS complexes, and voltage evidence of left or biventricular hypertrophy, which can be severe ( Fig. 59.1 ). In addition, in the majority there are Q waves and inverted T waves in leads I, II, and the left chest leads. Electrophysiologic studies have shown a short A-H interval. Both M-mode and cross-sectional echocardiography demonstrate gross increase in the thickness of the ventricular free walls and the ventricular septum ( Fig. 59.2 ). Impaired diastolic filling is frequent, together with reduction of the rate and extent of systolic shortening. Cardiac catheterization is rarely performed as it adds little to the diagnosis or management.
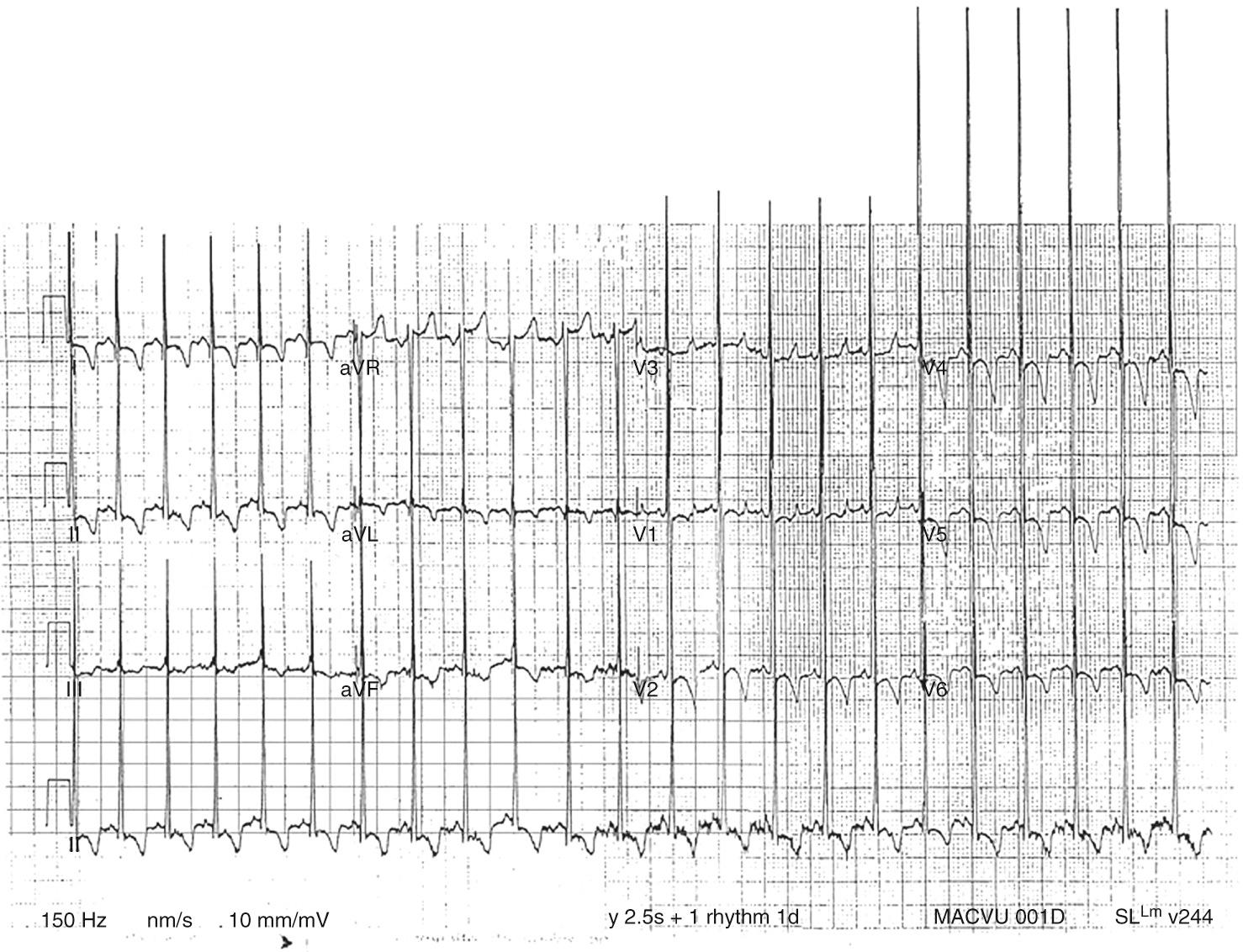
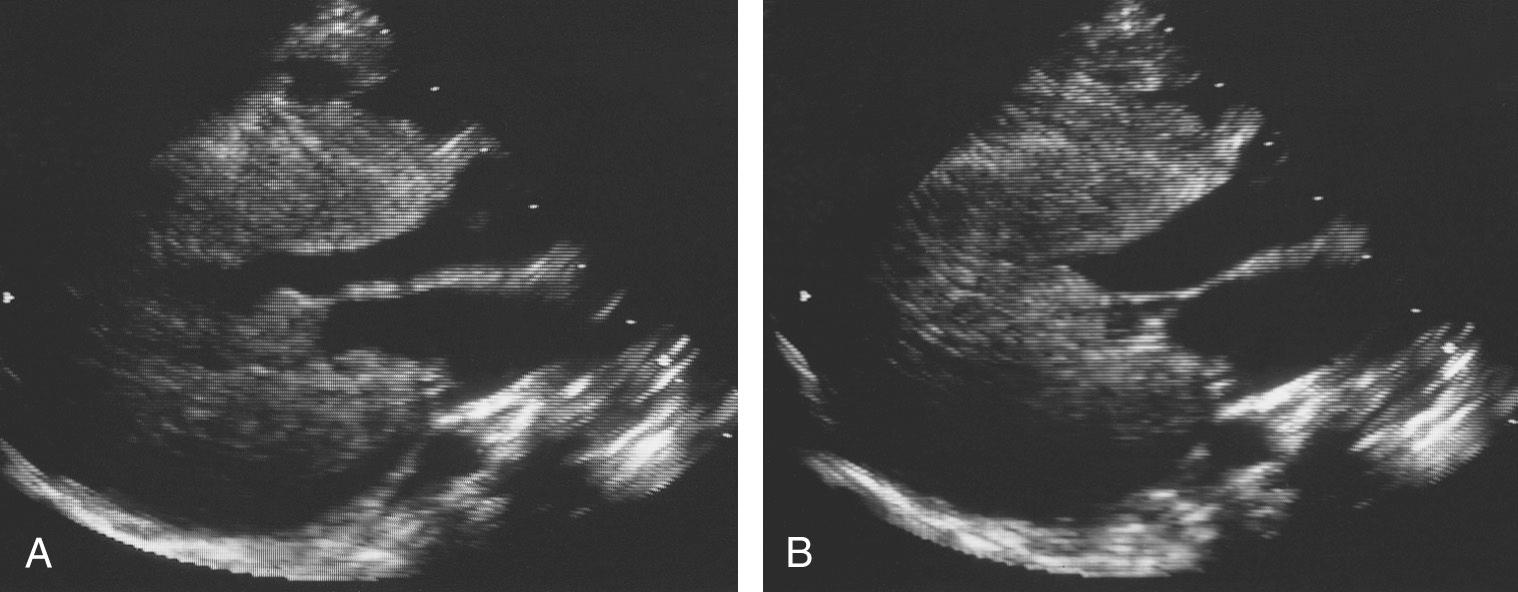
The complete clinical picture together with the characteristic electrocardiographic and echocardiographic findings will lead immediately to the definitive diagnostic investigation. This is the demonstration of deficiency of lysosomal α-1,4-glucosidase in fibroblasts grown from a skin biopsy. Sometimes the skeletal muscle abnormalities are less evident. The presentation is then as a cardiomyopathy alone. Pompe disease should be considered in any such case and skin biopsy performed. Until recently there was no specific treatment available, and supportive and decongestive measures failed to improve outcomes. However, recent studies using recombinant human lysosomal acid α-glucosidase show promise in improving survival. Early diagnosis, typically via standardized newborn screening in developed countries, and early initiation of enzyme replacement therapy shows the most benefit. Since the disease appears to be inherited in an autosomal recessive fashion, parents should be advised of the availability of prenatal diagnosis via culture of amniocytes obtained by amniocentesis.
Danon disease is included in this section because it was previously considered to be a variant of Pompe disease known as glycogen storage disease type IIb, with normal acid maltase. The disease is due to a deficiency of lysosome-associated protein 2 (lamp-2) and manifests as a progressive hypertrophic cardiomyopathy with skeletal myopathy. Other similar diseases in this family of autophagic vacuolar myopathies are still being studied. Some demonstrate autosomal recessive inheritance, whereas others are x-linked, and the degree of cardiac and skeletal involvement is variable.
In Cori disease, an autosomal recessive condition, glycogen accumulates in skeletal muscle, the liver, and cardiac muscle due to a deficiency of amylo-1,6-glucosidase, the enzyme necessary for breaking down branch points in glycogen chains. There are three subtypes, which are dependent on the primary site of abnormal glycogen storage (IIIa, liver and muscle; IIIb, liver; IIIc, muscle). A fourth subtype (IIId) involves normal debrancher enzyme activity but a deficiency of debrancher enzyme transferase activity. Patients with types IIIa and c have a tendency to develop skeletal muscle weakness and left ventricular hypertrophy, which is progressive. A study found that 58% of patients with glycogen storage disease IIIa had some degree of ventricular hypertrophy. However, the clinical course appears to be less severe, with fewer symptoms, as compared with hypertrophic obstructive cardiomyopathy.
Andersen disease is a rare heterogeneous glycogen storage disease characterized by the deposition of glycogen of abnormal structure in the liver, leading to cirrhosis. There may also be deposition of glycogen in the heart. Consequently, although liver dysfunction is the most common clinical manifestation, the disease can rarely present with dilated cardiomyopathy, which is typically severe.
McArdle disease is an autosomal recessive condition that results from a deficiency of muscle glycogen phosphorylase. It is often not diagnosed until adolescence or adult life and is commonly misdiagnosed in childhood. Its main clinical features are muscle fatigability, muscle cramps, and myoglobinuria. Rare lethal variants have been reported in infants, but the heart is typically spared. This may be due to activity of a distinct cardiac phosphorylase isozyme that retains activity in patients with deficiency of the skeletal isozyme. No clinical cardiac manifestations have been reported, but on occasion the electrocardiogram (ECG) has features similar to those seen in Pompe disease.
Hers disease involves both x-linked and autosomal recessive modes of inheritance and results from deficiency of liver phosphorylase. Both have involvement of the liver in childhood, whereas involvement of muscles occurs in young adults with the autosomal recessive form of the disease. Rare forms of phosphorylase b kinase deficiency have been described, in which deposition of glycogen is limited to the heart.
Tarui disease is a rare form of glycogen storage disease that presents in early childhood or adult life with fatigability, muscular weakness, which can be progressive, muscle cramps, and myoglobinuria. Typically the heart is spared. However, an infantile form of the disease has been described in the members of one family. Cardiomyopathy occurred in addition to the progressive muscular weakness, and abnormal deposition of glycogen was noted in the cardiac muscle at autopsy. Progressive cardiomyopathy has also been reported in an adult.
The mucopolysaccharidoses result from deficiency of lysosomal enzymes involved in the degradation of mucopolysaccharides. The incompletely degraded mucopolysaccharides then accumulate in the tissues. The substances accumulated are dermatan sulfate, heparan sulfate, or keratan sulfate. They can accumulate alone or in combination. There is skeletal involvement in all forms. In most, there is glaucoma and corneal clouding. Retinal pigmentation frequently occurs. Deafness is a feature of all types. In most, there is hepatosplenomegaly. Involvement of the central nervous system is common, usually with cervical myelopathy as a consequence of pachymeningitis or atlanto-occipital subluxation.
Cardiovascular involvement is a feature of all types. The mucopolysaccharides are deposited in arterial walls, including the coronary arteries, producing lesions similar to atherosclerosis. A dilated aortic root is also frequently seen. Deposition in cardiac valves leads to valvar stenosis and/or regurgitation. The various forms of these diseases are brought about by deficiencies of 10 identifiable lysosomal enzymes. Specific deficiencies can be demonstrated in cultured fibroblasts, and prenatal diagnosis from culture of amniocytes is possible. The availability of such diagnosis is important, since there is genetic variability within different forms of mucopolysaccharidoses.
The three major clinical forms of α- l -iduronidase deficiency are Hurler syndrome, Scheie syndrome, and a syndrome intermediate between the two, Hurler-Scheie syndrome. These diseases are due to defects in the gene encoding α- l -iduronidase, and multiple defects have been elucidated. These include nonsense, missense, insertional, deletional, and splice-type gene defects. It was previously thought that the clinical severity of the disease was related to the level of de novo enzyme activity, but no biochemical differences have been identified to distinguish the subtypes.
The defect in Hurler syndrome results in a virtual absence of lysosomal α- l -iduronidase. This enzyme is responsible for the breakdown of heparan sulfate and dermatan sulfate to heparan and hyaluronic acid, respectively. The enzyme is completely absent in fibroblasts but some activity is present in the liver. Consequently traces of the breakdown products of heparan and dermatan may be found in the urine. As a consequence of this enzyme deficiency, both heparan and dermatan sulfates accumulate in the lysosomes of many tissues. When in neurons, the lesions bear some resemblance to those found in Tay-Sachs disease. Deposition in the arterial walls is associated with proliferation of smooth muscle cells, and the lesions are described as “pseudoatheromatous.” There is proliferation of both elastic fibers and collagen accompanying the lysosomal accumulation of mucopolysaccharides.
The babies seem to be normal at birth, the clinical features appearing after the age of 1 year, when the facial features become coarse. Premature closure of the skull sutures and hydrocephalus as a consequence of pachymeningitis lead to cranial deformities. The characteristic lumbar lordosis develops because of stiff joints. Growth retardation then becomes evident after the age of 2 or 3 years; deafness, corneal clouding, and (sometimes) glaucoma subsequently develop. The liver and spleen are always enlarged. Although the heart is rarely spared, clinical evidence of cardiac involvement is seen only in half the patients. Angina pectoris is an occasional symptom because of coronary artery involvement, but more frequently attention is drawn by the finding of a cardiac murmur or systemic hypertension. The murmurs are variable and usually not loud. Rarely, the murmur of aortic or mitral insufficiency may be heard. Cardiac failure as the presenting feature associated with endocardial fibroelastosis has been reported.
There are typical skeletal radiologic features. The clavicles have wide medial ends. The lower thoracic and upper lumbar vertebrae have a flared and hook-shaped appearance. There are also changes in the skull and long bones, the latter being more severely affected in the upper limbs. The heart is usually enlarged but with no specific silhouette, although left atrial enlargement will occur with severe mitral regurgitation. Similarly, there are no specific electrocardiographic features, although combined ventricular hypertrophy is frequent. A long QT interval has been reported in some patients.
Pathologic findings in the heart include deposition of mucopolysaccharide in structures such as the sinus and atrioventricular nodes as well as in the myocardium and endocardium. The coronary arteries often demonstrate severe luminal narrowing, and care must be taken whenever these patients are subjected to general anesthesia or sedation, as hypotension can lead to coronary ischemia and death. The mitral valve is most frequently involved, followed by the aortic and tricuspid valves. Pulmonary valve involvement is only rarely reported. Valvar changes include nodular thickening along the free edges, which may lead to stenosis or regurgitation. Evidence suggests that the accumulation of dermatan sulfate leads to impaired elastogenesis, which may lead to some of the characteristic arterial and valvar deformities.
Thickening of the valve leaflets is characteristically seen echocardiographically. Left ventricular cavity enlargement will reflect the volume load resulting from valvar regurgitation, and left ventricular mass may be increased as a result of compensatory hypertrophy and deposition of mucopolysaccharide in the myocardium. Cardiac catheterization and angiocardiography add little to the diagnostic findings, which include systemic and mild pulmonary hypertension. If angiography is performed, the hemodynamics will reflect the severity of valvar insufficiency. The disease progresses inexorably, death occurring by the age of 10 years from heart failure, sudden death, or from chest infection. Hematopoietic stem cell transplantation has been beneficial in selected patients for many aspects of the disease. However, the valvar lesions remain progressive. Enzyme replacement therapy with human recombinant α- l -iduronidase has also proved beneficial, but, as with stem cell transplantation, the valvar lesions appear to remain and even progress.
Patients with Scheie syndrome are less severely affected and have normal stature and intellect. They also have a near normal life span. The most striking features are corneal clouding and stiff joints. Typical cardiac manifestations are aortic stenosis and regurgitation or mitral regurgitation. These should be managed in a similar fashion to that employed in otherwise normal subjects. Scheie syndrome is inherited in autosomal recessive fashion.
Hurler-Scheie syndrome falls in severity between the two extremes of α- l -iduronidase deficiency. The patients have short stature with mental retardation and multiple bony defects. There is clouding of the cornea and stiff joints, claw-hand being particularly common. Aortic and mitral valve involvement is the primary manifestation, but asymmetric septal hypertrophy has also been reported. The clinical course is intermediate between Hurler and Scheie syndromes, patients living into adolescence or even to the third decade.
Deficiency of iduronate sulfate results in blocked degradation of dermatan sulfate. The difference in clinical profile between this and Hurler and Scheie syndromes (e.g., the absence of corneal clouding in Hunter syndrome) may result from specific variability in the degree of blockage of degradation of the mucopolysaccharide. Furthermore, it may be that the block to degradation caused by the accumulation of iduronate sulfate may be bypassed by hyaluronidase. The severe and mild forms of Hunter syndrome both have total (or near total) deficiency of iduronate sulfatase. However, as with mucopolysaccharidosis type I, the clinical phenotype may be representative of the degree of residual enzyme activity specific to certain gene mutations, of which more than 300 have been found for Hunter syndrome.
The condition can occur with a wide variation in severity, which tends to run true in any given family. Apart from the extreme rarity of corneal clouding in Hunter syndrome and the presence of hearing loss, the clinical features are those of Hurler syndrome, although usually less severe. A positive distinguishing physical sign pointed out by Hunter himself (1916) is the occurrence of pebble-like ivory-colored skin lesions. These are seen over the scapulae and occasionally on the pectoral regions.
Cardiac involvement produces all the manifestations so far mentioned, namely aortic and mitral regurgitation or stenosis, ischemic changes, and evidence of myocardial deposition and dysfunction. Echocardiography is a useful method for evaluating cardiac involvement in Hunter syndrome. The clinical course is extremely variable. Severely affected individuals may die in adolescence. At the opposite end of the spectrum, however, survival beyond the sixth decade has been reported. Death in younger patients is usually associated with progressive neurologic deterioration. The disease is inherited as an X-linked recessive trait, although cases in females have been reported. Since the reproductive fitness of the Hunter gene is low, a large proportion of cases result from new mutations.
Recently a new treatment for Hunter syndrome has emerged with the development of recombinant human iduronate-2-sulfatase. This is well tolerated and associated with improvement in several outcome parameters, including forced vital capacity, urinary excretion of glycosaminoglycans, liver and spleen volume, and 6-minute walk distance. However, the effect of enzyme therapy on the cardiac lesions remains to be determined.
The degradation of heparan sulfate and N -sulfated or N -acetylated α-linked glucosamine requires five enzymes: N -sulfoglucosamine sulfohydrolase (sulfamidase), α-2-acetamido-2-deoxy- d -glucoside acetamidodeoxyglucohydrolase (α- N -acetylglucosaminidase), heparan acetyl CoA:α-glucosaminide N -acetyltransferase, N -acetylglucosamine-6-sulfatase, and N -glucosamine-3-O-sulfatase. Deficiency of one of the five enzymes required for this degradation results in the Sanfilippo syndrome, which is an autosomal recessive disorder. Consequently there are five biochemically distinct types of the disease (designated a to e ), although they all present the same clinical features. Incidentally, type e has yet to be uncovered in humans, although it exists in animal models.
The onset is usually evident in the first few years of life with “behavioral” problems. Mental and neurologic deterioration are severe and lead to death in the first two decades. Bone, joint, and cardiac involvement is generally less severe than in Hurler syndrome. Corneal clouding is never seen.
There is wide variation in the severity and age at death in all four forms, but type a is likely to be the most severe. Inheritance is in autosomal recessive fashion. Cardiac involvement is similar to that of other mucopolysaccharidoses, with a number of patients reported to have mitral valve involvement. Although treatment is primarily supportive, animal studies have been undertaken to assess enzyme replacement therapy in a mouse model with mucopolysaccharidosis type III-b.
Morquio syndrome results from defective degradation of keratan sulfate. It occurs in two biochemically distinct forms. So-called type a is due to a deficiency of n -acetylgalactosamine-6-sulfate sulfatase, whereas type b results from deficiency of β-galactosidase. The two types have similar clinical features, but type b is less severe, sometimes being called the “long-legged” variant. Despite the generally increased severity of features with type a, more mild forms of type a can occur. Keratan sulfate is excreted in the urine in type a, but this is less evident in type b.
Keratan sulfate is found in cartilage, intervertebral discs, and the cornea. Thus skeletal involvement with dwarfism, pectus excavatum, and bowed legs are the most obvious manifestations. Corneal clouding is common. In contrast to the mucopolysaccharidoses described earlier, the joints in patients with Morquio syndrome are hyperextensible. Absence or severe hypoplasia of the odontoid process, together with laxity of its associated ligaments, leads to atlanto-occipital subluxation and consequent cervical myelopathy. Sinus tachycardia is a common feature of this disease, but the mechanism is unclear. Valves of the heart are often involved, with thickening of mitral and aortic leaflets, although significant valve dysfunction is less common. Aortic root dilation, concentric left ventricular hypertrophy and, rarely, asymmetric septal hypertrophy have all been described. Survival beyond the third or fourth decade is not unusual. The effects of the cervical myelopathy and respiratory problems are the usual cause of death. Experimentation with enzyme replacement therapy in animals has been undertaken and holds some promise.
Deficiency of arylsulfatase b results in an inability to hydrolyze the sulfate groups in dermatan sulfate. The clinical picture is similar to that of Hurler syndrome, but normal intelligence is usual. Although severe in its classical form, milder variations exist. Affected infants can present acutely with cardiomyopathy. The mitral and aortic valves are frequently involved and the disease is typically progressive. Regurgitation is the primary valve disorder, but stenosis or a combination or stenosis and regurgitation will become more common over time. Valvar dysfunction severe enough to necessitate replacement has been noted in young adults. Left ventricular aneurysm has also been reported. Death usually occurs in the third decade. The condition is inherited in an autosomal recessive fashion, although some cases have been presumed to be X-linked. Enzyme replacement therapy with human recombinant arylsulfatase b has been studied and found to be effective in terms of arresting the progression of cardiac valve disease when started late in life, but it is postulated that it may have even better efficacy if started in early infancy.
Deficiency of β-glucuronidase results in a clinical syndrome of extremely variable severity. Included in the features are coarse facies, corneal clouding, abdominal and inguinal hernias, puffy hands and feet, hepatosplenomegaly, and a small thoracolumbar hump. Cardiovascular manifestations include hypertension, aortic aneurysm, valve thickening, aortic regurgitation, obstructive arterial disease including coronary involvement, and cardiomyopathy. Fetal hydrops has also been reported. This extremely rare condition is inherited in autosomal recessive fashion. Duration of survival varies widely and depends on the severity of the disease. Death as early as 30 months has occurred in one child with severe disease. Animal studies involving enzyme replacement therapy have been performed and are encouraging in terms of improving the cardiovascular changes associated with this disease.
The mucolipidoses present with clinical features similar to the mucopolysaccharidoses but are biochemically distinct. Leroy and Demars observed inclusions in cultured fibroblasts that occupied the whole cytoplasmic space apart from the Golgi apparatus. It was because of this that the name inclusion-cell, or i-cell, disease was coined. The cause of the lysosomal storage defect is deficiency of several acid hydrolases in the lysosome, but this is not the primary problem, since the plasma abounds in these acid hydrolases (albeit in unstable forms). The problem is failure to locate the hydrolases within the lysosome. Failure of phosphorylation of mannose residues of the hydrolases is the primary defect. Hydrolases without mannose 6-phosphate components are then not recognized by the lysosome and are not transported across the lysosomal membrane, particularly in connective tissue. In this way, inclusion-cell disease and pseudo-Hurler polydystrophy differ from sialidosis (previously called mucolipidosis type I), where there is a single lysosomal enzyme defect. Mucolipidoses types II (inclusion-cell disease) and III (pseudo-Hurler polydystrophy) result from a deficiency of uridine diphosphate (udp)-n-acetylglucosamine:lysosomal enzyme n-acetylglucosamine-1-phosphotransferase. The degree to which this enzyme is deficient determines the ultimate phenotype. Diagnosis is suggested by clinical features resembling mucopolysaccharidoses but without their biochemical abnormalities. Findings of high serum levels of β-hexosaminidases, iduronate sulfatase, and arylsulfatase are diagnostic. The characteristic enzymatic deficiencies in fibroblasts can be identified in cultured cells.
Inclusion-cell disease is an autosomal recessive condition that results from a severe deficiency of the phosphotransferase enzyme due to specific gene mutations that result in a marked reduction in enzyme activity. Various defects in the gene encoding this enzyme have been discovered in patients with this disorder. Patients with inclusion-cell disease present with clinical features very similar to those of Hurler syndrome. Hepatosplenomegaly is not so obvious, whereas striking gingival hypertrophy is a feature not encountered in Hurler syndrome. Furthermore, the disease becomes evident earlier than does Hurler syndrome. Corneal clouding is the rule. The skeletal and joint abnormalities, together with myocardial infiltration, usually lead to death by the age of 5 years either from respiratory causes or cardiac failure. All children have cardiac involvement, frequently with thickening and insufficiency of the mitral valve and, less frequently, the aortic valve. Asymmetric septal hypertrophy has been reported. Treatment of the cardiac manifestations is usually supportive, although surgical management of valve involvement has been reported. Also, success with allogeneic stem cell transplantation in terms of disease progression has been reported in a small number of cases.
Mucolipidosis type III is an autosomal recessive condition that is less severe, and also less common, than type II and is due to a deficiency of the same phosphotransferase enzyme. However, in this type the enzyme activity is less severely reduced and the manifestations are less severe. There is significant variability in the clinical severity of this disease. This is likely due to various genetic defects leading to different levels of enzyme activity. Patients are usually spared the joint manifestations early in life (unlike those with inclusion-cell disease) and often present with joint stiffness at the age of 4 or 5 years. Growth is moderately retarded and corneal clouding is present by the age of 7 or 8 years. The patients are disabled by carpal tunnel syndrome and destruction of the hip joints. Cardiac involvement, typically gradual thickening and eventual regurgitation of the mitral and aortic valves, does occur but is usually not sufficiently severe to cause clinical problems. Patients with pseudo-Hurler polydystrophy generally survive into the fourth decade. More recently, with advances in understanding of the genetics of the disease, mucolipidosis has been subcategorized into type III α/β and type III gamma as the deficient enzyme is the product of two genes. The first encodes the α and β subunits and the second encodes the gamma subunit. Despite this, the two subtypes have similar manifestations.
Specific lysosomal enzymatic deficiencies result in failure of degradation of glycoproteins with consequent accumulation of glycoproteins in many tissues, especially the nervous system. They became recognized when patients with presentations similar to those with the mucopolysaccharidoses were found to have biochemically distinct diseases.
The five primary disorders of glycoprotein degradation, mannosidosis, fucosidosis, sialidosis, galactosialidosis, and aspartylglycosaminuria, can all be diagnosed by demonstration of the enzyme defect in cultured fibroblasts. Prenatal diagnosis is often possible.
Deficiency of α-mannosidase is a rare, autosomal recessive disorder that results in the accumulation of oligosaccharides, as their degradation is dependent on lysosomal activity of this enzyme. Oligosaccharides are excreted in the urine. Several defects in the gene encoding α-mannosidase have been discovered. The specific defect may result in decreased enzyme synthesis, decreased enzyme activity within the lysosomal environment, decreased localization of the enzyme within the lysosome, or faulty posttranslational modification of the enzyme. The patients present with features suggestive of mucopolysaccharidosis but have an increased susceptibility to infections. Progressive mental retardation is typical. Early onset of the disease categorized as type I is associated with increased severity. Death occurs between 3 and 10 years of age. Late-onset disease (type II) runs a more benign course. Cardiac manifestations are not frequently reported. However, a short PR interval has been reported in several patients. The mechanism for this is unknown. Treatment has been attempted with bone marrow transplantation, and strategies currently under investigation include various forms of enzyme replacement therapy.
Deficiency of α- l -fucosidase results in the accumulation of fucosylated oligosaccharides and glycolipids. Two clinical types are recognized. The first type presents in infancy with coarse facies, growth retardation, mental retardation, and neurologic deterioration. Convulsions and respiratory infections often occur. The second type has a more benign course and a later onset. Cardiomegaly, probably as part of a generalized visceromegaly, is the most common cardiac feature but is benign. These two types probably represent both ends of a continuum that is dictated by a patient's specific enzyme activity as determined by his or her specific gene defect. Bone marrow transplantation has been performed with good results, and further experimentation with enzyme replacement therapy is ongoing.
The basic defect in sialidosis is deficiency of α-neuraminidase, with accumulation of sialoglycoconjugates. Two forms exist. The first is of late onset and patients are of normal appearance but develop the cherry-red spot myoclonus syndrome. Decreased visual acuity is associated with a cherry-red spot in the macular region. Neurologic (and occasional renal) manifestations dominate the clinical picture. The second type has an early onset, even on occasion being obvious at birth. The patients have coarse features and enlargement of various organs including the heart. Echocardiography has shown an increased left ventricular wall thickness along with thickening of the mitral valve. There is great variability in the spectrum of severity, even in the group with early onset. Survival beyond 20 years is rare; occasionally, however, affected subjects are stillborn. Fetal hydrops has been reported as a presenting feature. Sialidosis is inherited in autosomal recessive fashion. Enzyme replacement therapy and gene transfer treatments are under investigation.
In galactosialidosis, patients have a defect in the production of lysosomal protective protein/cathepsin A, which helps to form a stable and activated complex with β-galactosidase and α-neuraminidase. Thus the symptoms are a combination of those seen in sialidosis and Morquio syndrome, the severity of which is likely determined by the specific genetic defect and its overall effect on the production of functional levels of the protein. An infantile form has been described, and there appears to be a fair amount of clinical variation even in those diagnosed as infants. Structural congenital heart disease has been reported in patients with galactosialidosis, but the cardiac manifestations are usually similar to those seen in Morquio syndrome and sialidosis, with aortic and mitral valve thickening, which is progressive. Recently the role of these enzymes and protective protein/cathepsin A in elastogenesis has begun to be unraveled, helping to further explain the phenotype associated with these genetic disorders. Although no specific treatment yet exists, early work has begun on therapies involving enzyme replacement or gene transfer.
Aspartylglycosaminuria is a lysosomal storage disease due to a defective or deficient glycosylasparaginase. This enzyme is required for complete breakdown of asparagine-linked glycoproteins within the lysosome. Accumulation of these glycoprotein residues leads to severe and progressive neurologic impairment. It is associated with coarse features, joint laxity, and early rapid somatic growth followed by a reduced adolescent growth spurt leading ultimately to short stature and mental retardation. Animal models demonstrate residue accumulation within the heart, but clinical cardiac involvement does not appear to predominate. Therapy via enzyme replacement is currently being studied in animal models.
Lysosomal acid lipase is necessary for the cleavage of triglycerides and cholesterol esters from lipoproteins delivered to the lysosome. Complete or partial deficiency of lysosomal acid lipase results in accumulation of cholesterol in most tissues of the body. The disease occurs in two forms. Wolman disease, with complete absence of enzymatic activity, is a disease of infancy presenting with vomiting, diarrhea, hepatosplenomegaly, failure to thrive, anemia, and calcification of the adrenal glands. Cardiac manifestations are not usually evident, but microscopic examination of the arteries shows excess fatty deposits. Hepatomegaly is frequently the only sign in the milder form of the disease, cholesterol ester storage disease, although premature atherosclerosis is also seen. The diagnosis of Wolman disease is suggested by the association of hepatosplenomegaly with adrenal calcification. Definitive diagnosis of either disease can be made by assessing acid lipase activity in cultured skin fibroblasts. The disease is inherited in autosomal recessive fashion. Successful treatment with hematopoietic stem cell, bone marrow, and cord blood transplantation has been reported.
In Niemann-Pick disease, there is accumulation of sphingomyelin in the cells as a result of deficiency of sphingomyelinase (type a and b). A second distinct form (type c) was recently discovered, which arises from the defective function of cholesterol transport. The primary cells affected are those of monocyte-macrophage lineage as they are frequently employed in the metabolic turnover of these substances. In type a, patients exhibit hepatosplenomegaly in infancy and profound central nervous system involvement. These individuals rarely survive beyond 3 years of age. Type b patients also have hepatosplenomegaly along with pathologic alterations in their lungs, but there are usually no central nervous system signs. In type c, psychosis predominates initially. Many of these patients are of Ashkenazi Jewish heritage. The disease is characterized by hepatosplenomegaly and the occurrence of foam storage cells in many tissues. The heart is not usually affected, but one infant with acute neuronopathic disease had endocardial fibroelastosis. Since there were no storage cells in the heart, however, this may have been a chance association. Lipid profile abnormalities have also been described in children with Niemann-Pick disease types a and b, possibly leading to premature atherosclerosis. Low levels of high-density lipoprotein cholesterol was the most consistent finding, whereas elevated triglycerides and levels of low-density lipoprotein cholesterol were seen in approximately two-thirds. These abnormalities were noted at an early age and may reflect deranged cholesterol metabolism in these cells as a result of sphingomyelin accumulation. Niemann-Pick disease in most of its forms is inherited in an autosomal recessive fashion.
Gaucher disease is the most common inherited disorder of glycolipid metabolism. In his original description, Phillippe Gaucher ascribed the changes to a primary epithelioma of the spleen. There is excessive accumulation of glucosylceramide in cells of the reticuloendothelial system in organs throughout the body, resulting from deficiency of the enzyme glucocerebrosidase, which cleaves glucose from glucocerebroside. Although over 150 different mutations of the gene encoding glucocerebrosidase have been described, the disease occurs in three varieties based on the presence or absence of neurologic manifestations and their rate of progression. The first (type I, the chronic nonneuronopathic form) can be diagnosed at any age and is the most common form. It is characterized by hypersplenism, hepatomegaly (with evidence of abnormal liver function), and skeletal lesions (including aseptic necrosis of the femoral head). Other long bones and vertebrae may also be eroded. In patients with this type of disease, cardiac involvement may be seen, with myocardial infiltration or restrictive pericardial disease. The most frequently encountered cardiac problem, however, is cor pulmonale secondary to pulmonary involvement. Mitral and aortic stenosis and insufficiency can also be seen, and severe valvar and aortic arch calcification has been reported. The course is variable. Death may occur in early childhood or, particularly when the onset is late, there may be a normal life expectancy. Further variability is apparently the consequence of the nonneuronopathic form at onset changing to one of the other forms with poorer prognosis.
The acute neuronopathic form (type II) is usually recognized within the second half of the first year of life. Neurologic involvement is evident early, afflicting particularly the cranial nerves and extrapyramidal tracts. The mechanism of death is usually a respiratory infection, since—owing to incoordination of the nasopharynx—aspiration is common.
The subacute neuronopathic form (type III) falls between the acute and chronic forms. The neurologic involvement renders it less benign than the chronic variant, but its course usually stretches over many years.
Although describing Gaucher disease in terms of three distinct phenotypes is convenient, the actual observed behavior of this disease is much less well defined. Patients with the same genotype can have widely differing phenotypes, and patients even within a particular type can have markedly differing clinical courses. Thus, although the genetic defects leading to Gaucher disease are being elucidated and include over 150 specific mutations already identified, the genotype-phenotype link is still quite unclear.
The diagnosis of Gaucher disease is confirmed by the finding of typical storage cells in the bone marrow or by liver biopsy. The Gaucher cell is large and lipid-laden. The cytoplasm is described as having a “wrinkled tissue paper” or “crumpled silk” appearance. The nucleus is eccentric. These cells must be differentiated from cells found in multiple myeloma, leukemia, thalassemia, and congenital dyserythropoietic anemia. Demonstration of the enzymic deficiency in cultured skin fibroblasts or in leukocytes confirms the diagnosis. Treatment options include bone marrow transplantation and gene therapy in rare cases, but enzyme replacement therapy has become the standard of care in the majority of cases. In fact, type I Gaucher disease was the first lysosomal storage disorder for which, in 1991, an effective enzyme replacement therapy was developed. Improvement in the visceral organ involvement is common, but neurologic damage is generally not responsive to exogenous enzyme therapy. All three variants are inherited as autosomal recessive traits. Intrauterine diagnosis is available, and heterozygotes can be identified at least for the acute and chronic types.
Deficiency of α-galactosidase, a lysosomal enzyme, results in the accumulation of phosphosphingolipids in the lysosomes of many tissues and also in the body fluids. The most frequently affected tissue is the vascular endothelium. The disease is of X-linked inheritance, but heterozygous women can show severe manifestations of the disease. The gene locus for the enzyme is on the long arm of the X-chromosome.
The disease usually presents in childhood in the male homozygote, often with periodic crises of severe pain of burning character, which usually starts in the hands and feet. Crises occur most usually in the afternoon. Such crises, which become less frequent and severe with time, may, however, be followed by eruption of skin lesions, angiokeratomas, and typical opacities of the cornea and the lens. The angiokeratomas are clusters of dark-red to purple punctate lesions, which are usually flat or slightly raised. They occur most frequently between the umbilicus and the knees and do not blanch on pressure. Hyperkeratosis and hypohydrosis usually accompany the angiokeratomas. Ocular lesions include typical creamy whorl-like opacities in the cornea. They are frequently found in the female heterozygote as well as the male homozygote. Cardiac disease is manifest with increasing age. Myocardial ischemia and infarction are common and are secondary to the vascular lesions. Mitral regurgitation and aortic stenosis are the most frequently encountered valvar lesions. Infiltration of the conduction tissues also occurs. This results in progressive shortening of the PR interval, as in other storage diseases that affect the specialized atrioventricular conduction axis. Myocardial deposition can be detected echocardiographically by the demonstration of septal and left ventricular wall thickening. Progressive deposition of glycosphingolipid means that the cardiac problems themselves are also progressive. Since concomitant renal involvement occurs, the cardiac effects are exacerbated by, for example, renal hypertension. The clinical course in the male homozygote is one of steady deterioration during early adult life, death being due to cardiac or renal disease. The heterozygote female experiences little limitation of style and length of life. The diagnosis can be confirmed (and heterozygotes identified) by demonstrating the enzymic deficiency in leukocytes and by an abnormally high content of accumulated substrates in tears or urinary sediment. Prenatal diagnosis is available. Enzyme replacement therapy was first reported in 2002, and positive effects on cardiac involvement are evident. Fabry disease should be considered whenever unexplained left ventricular hypertrophy is discovered, as early diagnosis and initiation of enzyme replacement therapy has shown clear benefit.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here