Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
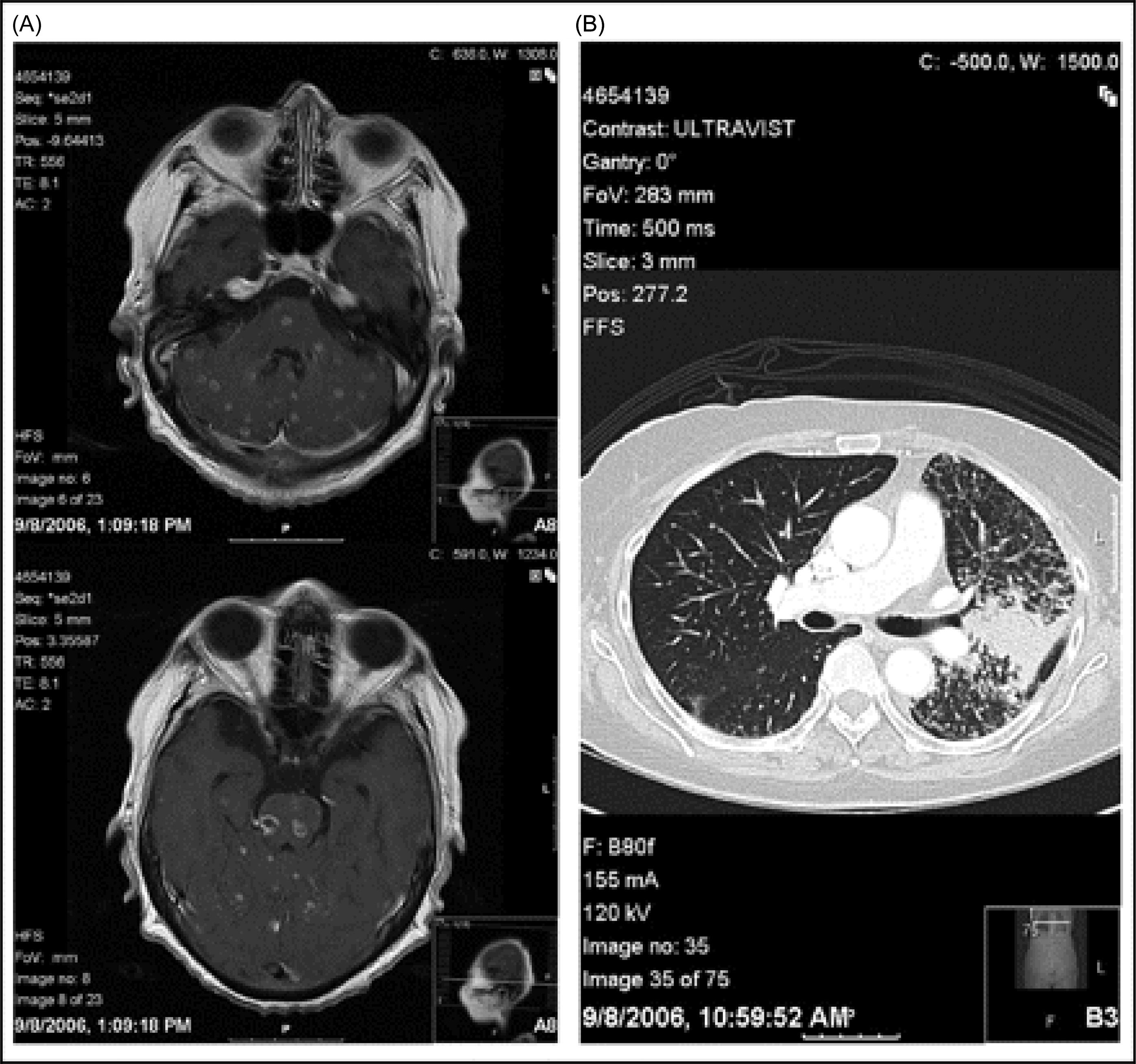
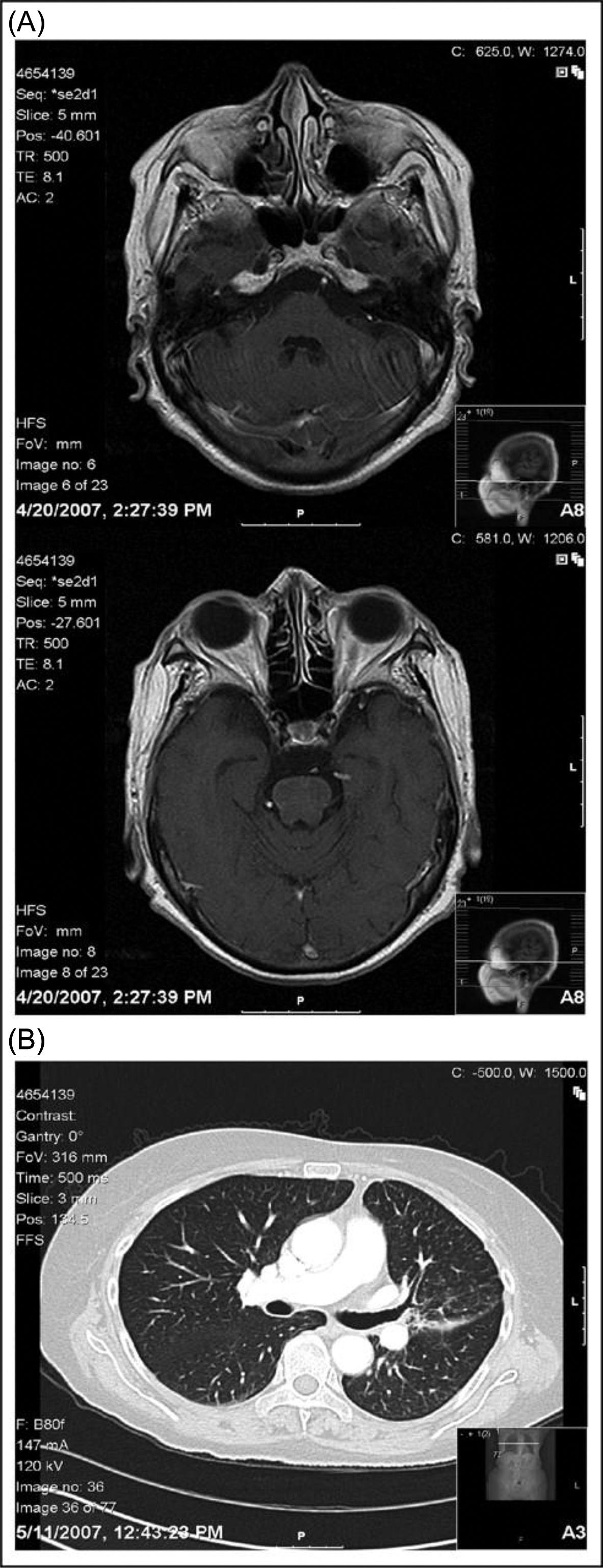
A 60-year old never-smoker woman presents with left-sided weakness and ataxia due to innumerable metastatic lesions within the brain originating from an advanced adenocarcinoma of the lung. She receives whole brain radiotherapy (WBRT) and systemic chemotherapy and her symptoms temporarily improve. A few months later her disease recurs both clinically and radiographically within the brain. Institution of erlotinib, a small molecule inhibitor of the epidermal growth factor receptor (EGFR) tyrosine kinase, results in a complete radiographic resolution of brain lesions, improvement in clinical symptoms and performance status, and partial response of the primary lung tumor ( Figures 14.1 and 14.2 ) ( ).
Metastatic tumors are among the most common mass lesions in the brain. In the USA, up to 200 000 people per year, or approximately 40% of patients diagnosed with an invasive malignancy, will develop brain metastases during the course of their illness ( ). Such lesions adversely affect the quality of life as well as the overall survival duration. The incidence of metastatic neoplasms involving the brain is greater than primary central nervous system (CNS) neoplasms. The majority of patients are greater than 60 years old, which is consistent with the median age of most invasive neoplasms. While metastatic disease to the brain may arise from any primary site, the most common sites of origin are the lung, followed by the breast, skin (overwhelmingly, melanomas) and the gut.
Approximately 85% of metastases are supratentorial, while only 1–3% are to the brainstem ( ). Approximately 10% of patients will have an unknown primary site and, in 9% with a known source, the brain is the sole site of metastases. Primary lung cancers make up half of all metastatic brain tumors. Of those patients with lung cancer who survive for at least 2 years, 80% will have brain metastases. However, even within lung cancers, there is a differential propensity for the development of brain metastases based upon histology: small cell lung cancers comprise only 15–20% of all lung cancers, but lead to half of all brain metastases from lung cancers. Notably, about 30–40% of patients with non-small cell lung cancer (NSCLC) may develop clinically apparent metastatic disease to the brain; an early autopsy series noted that 55% of patients with NSCLC were found to have brain metastases ( ).
A considerable body of work has been dedicated to find effective and safe treatments to improve survival of patients with brain metastases. These include surgery or stereotactic radiosurgery for oligometastatic disease, whole brain radiotherapy, and chemotherapy, either alone or as combined-modality treatments. Most of the larger reports, series or trials of localized treatments of brain metastases include a variety of tumor histologies. More importantly, the natural history of the underlying systemic disease often impacts the overall survival and, in some cases, the clinical benefit of any aggressive treatment specifically directed to the brain metastases. One of the largest retrospective analyses of patients with NSCLC and brain metastases was reported from the Cleveland Clinic ( ). The goal was to evaluate the outcome of treatment in patients with newly diagnosed NSCLC who also had an isolated, single, synchronous brain metastasis, with the brain being the sole metastatic site. Over a 14-year period (1982–1996) a total of 219 patients with lung cancer and synchronous brain metastases were identified; of these, only 33 patients had a solitary metastasis, and were further studied. The majority of those patients (64%) were treated with surgical resection followed by whole brain radiotherapy, and the remaining patients were treated with surgery alone or some form of radiotherapy, either as whole brain or stereotactic radiotherapy. The disease in the CNS was controlled in 31 patients (94%). The median disease-free survival (DFS) was 3.3 months, with only nine patients suffering brain recurrence. The median overall survival (OS) was 6.9 months, though patients who received whole brain radiotherapy had a markedly prolonged OS compared to those who did not receive WBRT, 9.1 months versus 2.9 months ( p =0.002). Most of the patients died from systemic disease progression; control of extracranial disease was associated with a markedly prolonged median OS of 20.1 months. While these results are encouraging, there are several notable factors: this was a very small and highly selected patient population; 85% of the patients in the original cohort had multiple brain metastases, where a surgical resection or stereotactic radiosurgery may not be practical or of benefit. Furthermore, the main cause of death in the studied population was still systemic disease progression, though 27% also had intracranial relapse. The low radiotolerance of normal brain tissue limits the cumulative dose of whole brain radiotherapy. With these factors in mind, it is clear that alternative agents must be developed to treat intracranial metastases from NSCLC.
Cranial radiotherapy (RT) is most frequently used to control neurologic symptoms caused by brain metastases. An important role for RT, that is a sensitizing effect for epidermal growth factor receptor tyrosine kinase inhibitors (EGFR-TKIs), has also been proposed ( ). However, the efficacy of RT is influenced by patient performance status, the number of metastatic lesions in the brain and extracranial tumor histology. Furthermore, RT has acute and chronic side effects which could impact overall quality of life. Overall, WBRT may extend survival by 4–6 months.
Surgery is another option to treat brain metastases but is limited to single lesions (oligometastases) with accessible locations. When combined with subsequent RT, surgery improves survival compared to radiotherapy alone in lung cancer patients with oligometastatic disease ( ). However, as many patients with NSCLC and brain metastases have multiple brain lesions, this option is not widely used.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here