Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Bone marrow examination is an important part of the workup of patients with lymphoma. It is often performed for staging purposes as well as for follow-up to evaluate for response to therapy or recurrence. In some patients, a diagnosis of lymphoma is initially made on a bone marrow biopsy obtained for evaluation of unexplained cytopenias, fevers of unknown origin, unexplained organomegaly, or mass lesions difficult to access for biopsy.
Evaluation of bone marrow involvement by lymphoma can be challenging, particularly in patients without a prior diagnosis at an extramedullary site. For example, benign lymphoid infiltrates are frequently encountered in the bone marrow core biopsies in older individuals or patients with autoimmune diseases and can be difficult to distinguish from lymphoma, even with the help of ancillary techniques. In patients with an established diagnosis of lymphoma, the assessment of bone marrow involvement can be complicated by situations such as a discordant histologic type in the bone marrow compared with the extramedullary lesion. In addition, subclassification of lymphoma based on the bone marrow findings is not always straightforward and requires not only the knowledge of the diagnostic features of various lymphoma subtypes but also the appropriate use of ancillary techniques. It is also important to recognize the limitations of classification of lymphoma based on bone marrow findings alone; biopsy of extramedullary lesions may be required.
The core biopsy is usually the most informative in evaluating the bone marrow for lymphoma. However, peripheral blood smears, bone marrow aspirate smears, particle clot sections, and touch imprints also provide valuable complementary information and may be diagnostic in themselves. Therefore, all these preparations should be examined together and the findings correlated. An adequate bone marrow sampling is important to ensure the detection of focal lymphomatous infiltrates. It is recommended that the bone marrow core biopsy specimen be at least 1.5 to 2 cm long; the yield of lymphoma detection is significantly higher with bilateral than with unilateral core biopsies ( Fig. 56-1 ). In addition, sections from multiple levels of the core biopsy should be examined to increase the detection rate.
Flow cytometric immunophenotyping, immunohistochemistry, cytogenetic analysis, and molecular testing are important in evaluating bone marrow specimens for lymphoma and should be used in a logical and cost-effective way based on the morphologic findings and clinical setting. Whenever ancillary techniques are used, the results should be correlated with one another and with the morphologic findings. Knowledge of each technique, including its limitations, is important in effective use of these techniques in bone marrow evaluation of lymphoma.
This chapter focuses on the distinction between benign and lymphomatous infiltrates and morphologic features of various subtypes of lymphoma in the bone marrow. In addition, non-lymphoid lesions mimicking lymphoma in the bone marrow are discussed.
Benign lymphoid aggregates frequently occur in the bone marrow in older individuals as well as in various reactive conditions, such as systemic autoimmune disorders (rheumatoid arthritis, lupus, autoimmune hemolytic anemia, idiopathic thrombocytopenia, Hashimoto's thyroiditis), aplastic anemia, viral infection (human immunodeficiency virus infection, hepatitis), and myeloproliferative neoplasms and myelodysplastic syndromes.
The assessment of lymphoid infiltrates in the bone marrow is first based on the morphologic findings; however, other studies, such as immunohistochemistry, flow cytometric immunophenotyping, or molecular analysis, may be required for further evaluation. Although these techniques are usually informative, the nature of the lymphoid infiltrate may remain unknown in some cases. The extent of testing to analyze a lymphoid infiltrate in the bone marrow depends on the clinical setting and the degree of suspicion for lymphoma. Several morphologic features can be used in distinguishing benign lymphoid aggregates from lymphoma ( Table 56-1 ).
| Benign | Malignant |
|---|---|
| Aggregates are few in number | Aggregates are variable in number and may be multiple |
| Aggregates with random distribution | Paratrabecular aggregates may be present |
| Aggregates are usually round, well circumscribed | Aggregates are often irregularly shaped, with infiltrative borders |
| Polymorphous cellular compositions | Usually homogeneous cellular composition except in some peripheral T-cell lymphomas and Hodgkin's lymphoma; atypical cytologic features may be present |
| Intrasinusoidal infiltrates usually absent | Intrasinusoidal infiltrates may be present |
| Vascularity is often prominent | Vascularity is usually not prominent except in peripheral T-cell lymphomas |
| Benign germinal centers are occasionally present | Benign germinal centers are not present except in some marginal zone lymphomas |
| No morphologically abnormal lymphocytes in bone marrow smears or imprints | Morphologically abnormal lymphocytes may be present in bone marrow smears or imprints |
| Immunostains show a mixture of B and T cells, often with T-cell predominance (exceptions occur) | Immunostains showing a predominance of B cells, aberrant phenotype, or monoclonal plasma cells suggest B-cell lymphoma; an aberrant T-cell phenotype suggests T-cell lymphoma |
| No monotypic B-cell population or phenotypically abnormal T-cell population identified by flow cytometry (exceptions occur) | Immunoglobulin light chain restriction or aberrant T-cell phenotype by flow cytometry (exceptions occur) |
| No monoclonal IGH or TCR gene rearrangement by molecular analysis (exceptions occur) | Monoclonal IGH or TCR gene rearrangements are often identified by molecular analysis (exceptions occur) |
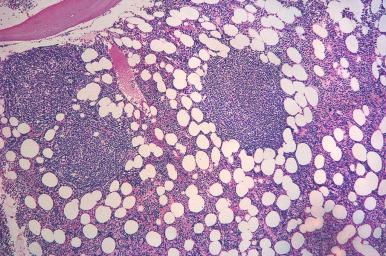
Benign lymphoid aggregates are usually single or few in number, small, well circumscribed, random, and non-paratrabecular ( Fig. 56-2 ). The cells within the aggregates are often polymorphous and may include plasma cells and histiocytes. Reactive germinal centers may be present ( Fig. 56-3 ) and are more commonly seen in patients with autoimmune diseases. However, germinal centers are not exclusively seen in benign lymphoid infiltrates; non-Hodgkin's lymphomas, particularly splenic marginal zone lymphoma, may also contain reactive germinal centers in the bone marrow.
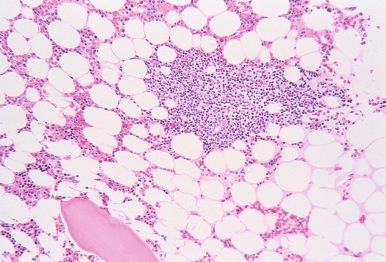
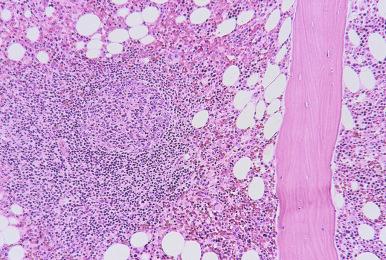
In contrast to benign lymphoid aggregates, lymphomatous aggregates are frequently multiple and large, and often have infiltrative borders. Paratrabecular lymphoid infiltrates are almost always neoplastic and most frequently associated with follicular lymphoma. The presence of distinct intrasinusoidal lymphoid infiltrates is usually an indication of a neoplastic process and most commonly seen in intravascular large B-cell lymphoma, hepatosplenic T-cell lymphoma, splenic marginal zone lymphoma, and splenic diffuse red pulp small B-cell lymphoma. Neoplastic lymphoid infiltrates generally exhibit a more homogeneous cellular composition than benign lymphoid infiltrates. However, a polymorphous infiltrate also characterizes some subtypes of lymphomas, most notably peripheral T-cell lymphoma and Hodgkin's lymphoma. Importantly, the presence of morphologically abnormal lymphocytes and architectural features in the bone marrow core biopsy, such as paratrabecular localization and infiltrative borders, should raise the suspicion of a neoplastic process.
A panel of immunohistochemical stains on the bone marrow core biopsy or particle clot section is often required to assist in the distinction between benign and neoplastic lymphoid infiltrates. Immunohistochemical stains for B-cell markers (e.g., CD20, PAX5) and T-cell markers (e.g., CD3) are often used to determine the proportions of B and T cells within the lymphoid aggregates. Benign lymphoid aggregates usually have a mixture of B and T cells, and T cells frequently predominate ( Fig. 56-4 ), whereas lymphoid infiltrates composed primarily of B cells, especially if multiple, are often neoplastic ( Fig. 56-5 ). However, some B-cell lymphomas may be accompanied by a significant number of reactive T cells, such as follicular lymphoma and T-cell/histiocyte-rich large B-cell lymphoma. Therefore, a mixture of B and T cells or a predominance of T cells in the lymphoid infiltrate does not completely exclude a diagnosis of B-cell lymphoma.
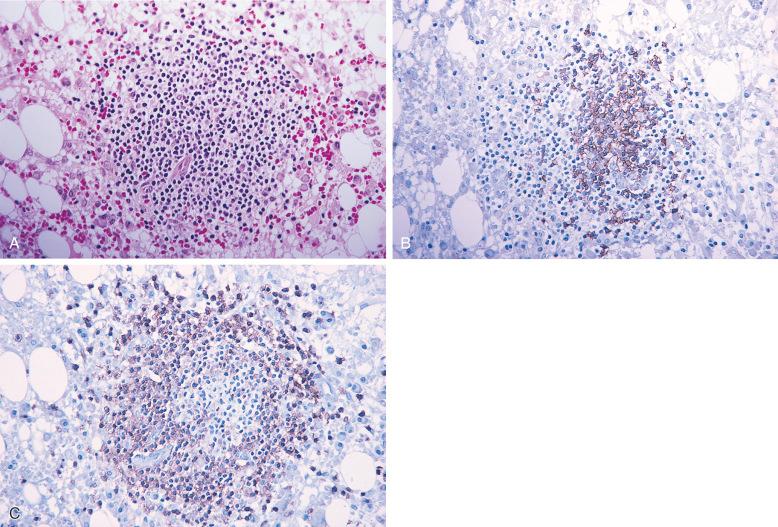
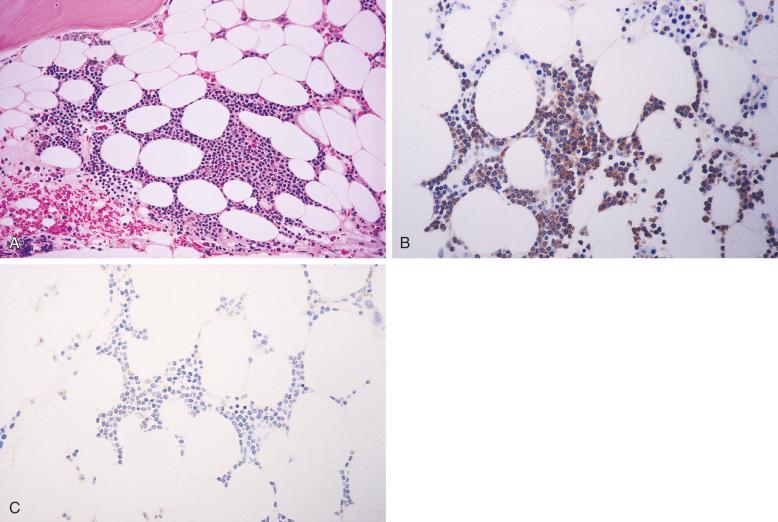
Similar to lymph nodes, benign germinal centers in the bone marrow are BCL2 negative. CD10 is typically negative in benign lymphoid infiltrates but positive in about 50% of follicular lymphomas in the bone marrow. Similar to the extramedullary lymphomas, demonstration of an aberrant B- or T-cell phenotype by immunohistochemistry, such as expression of CD5 in B cells or loss of pan–T-cell antigens in T cells, supports a diagnosis of B- or T-cell lymphoma, respectively. Demonstration of immunoglobulin light chain restriction in the plasma cells associated with an atypical lymphoid infiltrate supports a diagnosis of B-cell lymphoma with plasmacytic differentiation, such as marginal zone lymphoma and lymphoplasmacytic lymphoma. Immunohistochemical stains for proteins associated with specific lymphoma subtypes, such as BCL1/cyclin D1 in mantle cell lymphoma and ALK1 in anaplastic large cell lymphoma, help not only with diagnosis but also with subclassification of lymphoma.
Special consideration should be given to patients who have been treated with anti-CD20 agents and continue to exhibit lymphoid infiltrates in the bone marrow. In some cases, the lesions represent residual lymphoma; in others, the lymphoma has been eliminated by the therapy, and the lymphoid infiltrates are composed entirely of CD3 + T cells. These aggregates may mimic residual lymphoma because they can be large, multiple, or even paratrabecular ( Fig. 56-6 ). When residual B-cell lymphoma remains in the bone marrow of patients treated with anti-CD20 therapy, the lymphoma cells may be CD20 − . For all these reasons, immunostains or flow cytometric immunophenotyping with B-cell antigens other than CD20 (e.g., CD79a, PAX5) is often necessary to evaluate post-therapy bone marrow.
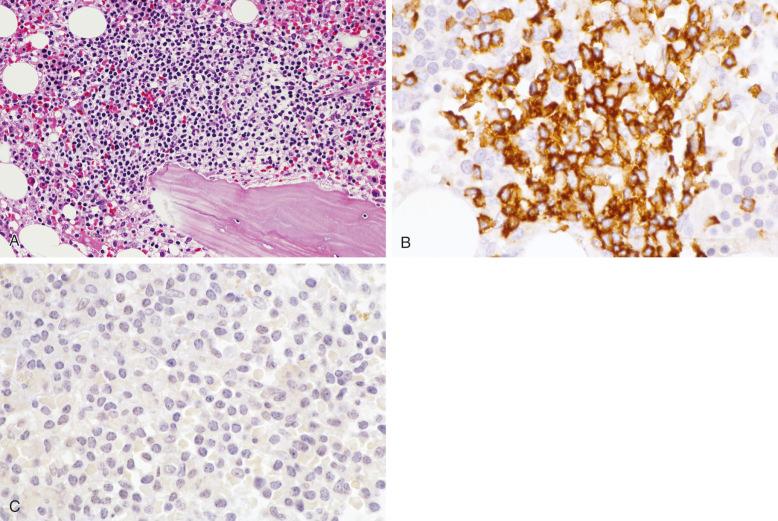
In the early course of granulocyte colony-stimulating factor (G-CSF) therapy, the bone marrow frequently shows a marked myeloid shift to immaturity and contains sheets of promyelocytes, often in a paratrabecular location. The findings may mimic large cell lymphoma ( Fig. 56-7 ). Knowledge of the history of G-CSF therapy can aid in the distinction from lymphoma. If the history is not provided, characteristic changes associated with G-CSF therapy, such as severe toxic changes and a shift to immaturity in granulocytes in the blood and bone marrow aspirate, can also alert one to this possibility. Well-prepared histologic sections that allow observation of the morphologic features of promyelocytes are essential. Immunostains for myeloperoxidase, CD20, or CD3 may be required to make the distinction ( Fig. 56-7 ).
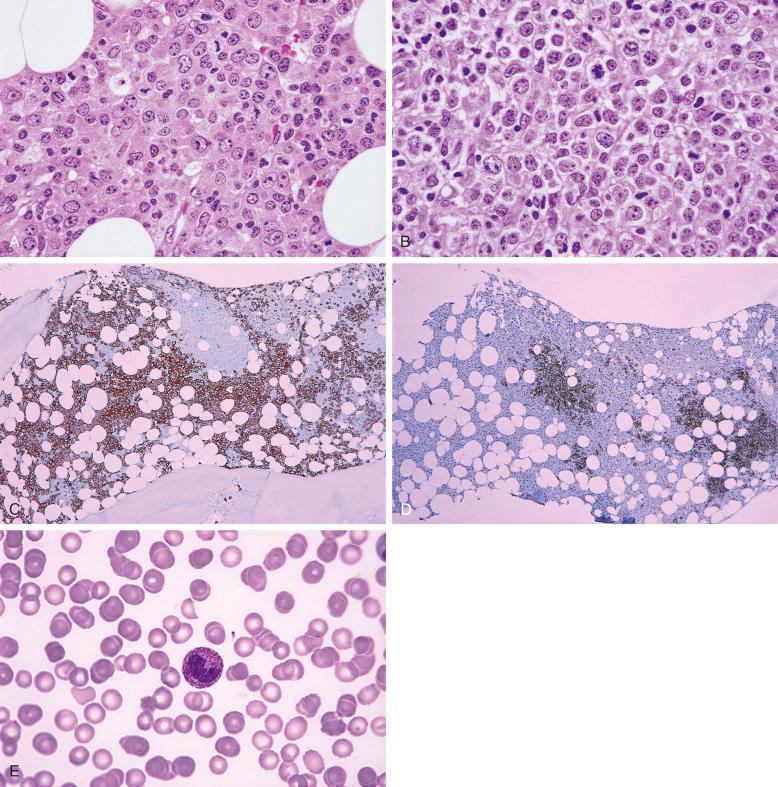
Flow cytometric immunophenotyping performed on the bone marrow aspirate or peripheral blood is an important part of workup for bone marrow involvement by lymphoma, and the correlation with morphology is usually excellent. However, discrepancy between the bone marrow core biopsy and the aspirate may occur because of small, focal lesions or bone marrow fibrosis preventing aspiration of cells; thus, a negative flow cytometric result does not exclude the possibility of lymphoma. Flow cytometric immunophenotyping can be performed on a peripheral blood specimen when a bone marrow aspirate is not available, especially if the patient is known to have a lymphoma associated with a higher incidence of peripheral blood involvement, such as chronic lymphocytic leukemia/small lymphocytic lymphoma, splenic marginal zone lymphoma, and lymphoplasmacytic lymphoma. On occasion, monotypic B cells are identified by flow cytometry without morphologic evidence of lymphoma. In these cases, it is prudent to evaluate whether the biopsy is of adequate size and quality and to examine multiple levels of sections ( Fig. 56-8 ). It may also be appropriate to perform immunohistochemical stains to ensure that a subtle abnormal lymphoid infiltrate is not missed.
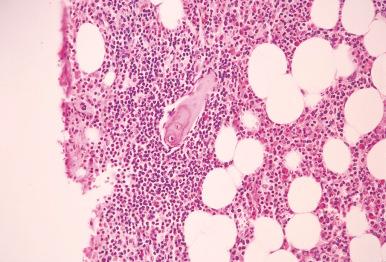
Low levels of monoclonal B cells (<5.0 × 10 9 /L) can be identified in the blood in up to 7% of older individuals who are otherwise healthy and without clinical or morphologic evidence of lymphoma. This finding is termed monoclonal B lymphocytosis (MBL). MBL usually exhibits a chronic lymphocytic leukemia–like phenotype (CD5 + , CD23 + ). Most patients with MBL also have lymphoid aggregates in the bone marrow, but they are not regarded as diagnostic for lymphoma. Small monoclonal B-cell populations can also be initially identified in the bone marrow aspirate in patients without evidence of lymphoma, and these monoclonal B-cell populations are more commonly CD5 − than MBL in the peripheral blood. In these CD5 − cases as well as in cases with mantle cell lymphoma–like phenotype, clinical and laboratory workup may be indicated to rule out lymphoma. On occasion, aberrant T-cell populations of unclear significance can also be identified by flow cytometry in patients without evidence of lymphoma. These findings emphasize the importance of correlating immunophenotypic results with the clinical and morphologic findings in bone marrow evaluation.
Molecular analysis is not typically a routine part of bone marrow evaluation for lymphoma but may play an important adjunctive role, especially in cases in which morphologic and immunohistochemical findings are uncertain. Polymerase chain reaction analysis for immunoglobulin heavy chain (IGH) or T-cell receptor (TCR) gene rearrangements on paraffin-embedded bone marrow tissue or fresh aspirate is typically used.
Clonal IGH or TCR gene rearrangements have been reported in about 65% to 85% and 60% to 70% of bone marrow specimens with morphologic and immunohistochemical evidence of B- or T-cell lymphoma, respectively. Clonal IGH gene rearrangements have also been identified in about 15% to 65% of cases interpreted as suspicious for lymphoma and 10% to 40% of cases without morphologic evidence of lymphoma. Of importance, clonal TCR gene rearrangements were reportedly identified in morphologically negative bone marrow in more than half of patients diagnosed with T-cell lymphomas. Interestingly, one study reported that clonal T-cell populations were identified in about 13% of B-cell lymphomas and clonal B-cell populations in about 3% of T-cell lymphomas in the bone marrow. It is also important to be aware that clonal B- or T-cell populations may be present in various reactive conditions, such as inflammatory diseases, autoimmune disorders, and after stem cell transplantation. Therefore, the results of molecular studies should be interpreted in the clinical context and correlated with additional information because monoclonality is not synonymous with malignancy, and failure to detect monoclonality does not rule out a lymphoma.
Compared with typical benign lymphoid infiltrate, differentiation of unusual reactive lymphoid proliferations from lymphoma is often more problematic.
Systemic polyclonal immunoblastic proliferation is a rare reactive lymphoplasmacytic proliferation often encountered in the setting of an acute immune disorder. This unusual reactive process involves the peripheral blood, bone marrow, and lymph node; other organs, such as the liver and spleen, are often involved as well. The leukocyte count is usually elevated with an absolute lymphocytosis that includes reactive lymphocytes, immunoblasts, and plasma cells; neutrophilia with a shift to immaturity may be present ( Fig. 56-9 ). Anemia and thrombocytopenia are almost always present, and the anemia is frequently immune mediated, with a positive direct antiglobulin test result. In addition, patients often have polyclonal hypergammaglobulinemia.
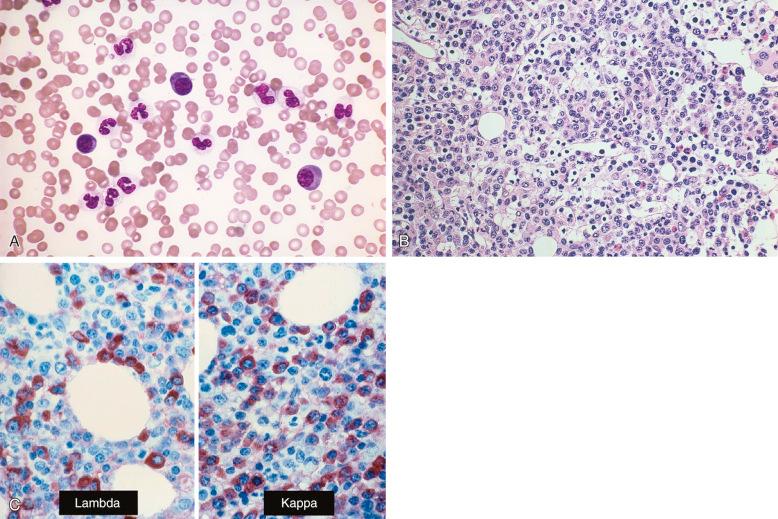
Bone marrow aspirate smears and core biopsy show numerous lymphocytes, immunoblasts, and plasma cells. Focal lymphocytic aggregates are characteristically present in the biopsy sections; they may be inconspicuous or large. The plasma cells and immunoblasts in the infiltrate show a polyclonal immunoglobulin light chain staining pattern ( Fig. 56-9 ). Flow cytometry reveals polyclonal B cells, and molecular analysis is negative for clonal IGH and TCR gene rearrangement or, rarely, oligoclonal B- and T-cell populations. Although the cause of this disorder is unknown, clonal cytogenetic abnormalities have been found in a subset of patients, raising the possibility of a cryptic neoplastic proliferation. The clinical behavior is variable; some patients respond to steroid therapy, but others require chemotherapy. In the small number of reported cases, the mortality rate during the acute phase of the illness was high, about 50%. The majority of patients who recover do not experience recurrence or relapse.
It is important to rule out an underlying lymphoma before arriving at a diagnosis of systemic polyclonal immunoblastic proliferation. Some peripheral T-cell lymphomas, particularly angioimmunoblastic T-cell lymphoma and other Epstein-Barr (EBV)–associated lymphoproliferative disorders, may exhibit similar clinical and morphologic features.
Reactive polymorphous lymphohistiocytic lesions are composed of heterogeneous cellular infiltrates of lymphocytes, including small, mature-appearing and large transformed lymphocytes, often with admixed plasma cells, eosinophils, mast cells, and epithelioid histiocytes, some arranged in poorly formed granulomas. These lesions in the marrow are often larger than typical benign lymphoid aggregates; they may be multiple, situated adjacent to bone trabeculae, and have irregular borders ( Fig. 56-10 ), features that are difficult to differentiate from lymphomas. Polymorphous lymphohistiocytic proliferations are most commonly found in patients with immunodeficiency, such as patients with AIDS. However, they may also be seen in patients with autoimmune diseases such as rheumatoid arthritis. A diagnosis of non-Hodgkin's lymphoma on a bone marrow biopsy in immunodeficient patients should be made with caution, especially in the absence of confirmation from an extramedullary site or supporting evidence from pertinent ancillary studies.
Bone marrow involvement by lymphoma represents stage IV disease based on the Ann Arbor lymphoma staging system. The overall incidence of lymphoma involving the bone marrow is 35% to 50%. However, there is considerable variability for different lymphoma subtypes. In general, indolent lymphoma, highly aggressive lymphoma, and the majority of peripheral T-cell lymphomas involve the bone marrow with high frequency. For example, follicular lymphoma, chronic lymphocytic leukemia/small lymphocytic lymphoma, and mantle cell lymphoma involve the bone marrow in up to 60%, 85%, and 90% of cases, respectively ; Burkitt's lymphoma, in about 30% to 60% of cases ; and diffuse large B-cell lymphomas, in 20% to 30% of cases.
The incidence of bone marrow involvement by specific subtypes of peripheral T-cell lymphomas has a wide range. For example, virtually all hepatosplenic T-cell lymphomas and up to 70% of angioimmunoblastic T-cell lymphomas involve the bone marrow. Anaplastic large cell lymphoma and nasal-type natural killer (NK)/T-cell lymphoma involve the bone marrow in approximately 10% to 30% and 10% to 20% of cases, respectively. Primary cutaneous T-cell lymphomas, such as mycosis fungoides, are generally considered to spare the bone marrow until advanced stage. However, careful examination identifies marrow involvement in approximately 22% of patients with cutaneous T-cell lymphoma at diagnosis.
Non-Hodgkin's lymphomas infiltrate the bone marrow in a variety of architectural patterns ( Table 56-2 ), and more than one pattern is often seen in an individual patient. Knowledge of these features is helpful in identifying neoplastic lymphoid infiltrates and in some cases facilitates lymphoma classification.
| Type of Lymphoma | Incidence of Involvement (%) | Pattern of Involvement * | Cytology | Comments |
|---|---|---|---|---|
| Chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL) | 85 (SLL) 100 (CLL) |
Focal random Diffuse Interstitial |
Small, mature lymphocytes; proliferation centers may be present | Paratrabecular infiltrates indicate lymphoma of non-CLL/SLL type; LEF1 positivity distinguishes CLL/SLL from other small B-cell lymphomas |
| Lymphoplasmacytic lymphoma | 80-100 | Focal random Focal paratrabecular Interstitial Diffuse |
Spectrum of cells from small lymphocytes to plasma cells; immunoblasts may be present; Dutcher bodies are often present; increased mast cells | Paratrabecular infiltrates may be present; majority of cases have MYD88 L265P mutation |
| Mantle cell lymphoma | 60-90 | Focal random Interstitial Diffuse Focal paratrabecular Rarely intrasinusoidal |
Small lymphocytes with irregular nuclei; may be blastoid; rare cells with prominent nucleoli | Paratrabecular infiltrates may be present; circulating lymphoma cells are common; BCL1 and SOX11 are positive |
| Follicular lymphoma | Grade 1-2: 50-70 Grade 3: 15-25 |
Paratrabecular Focal random Diffuse Interstitial |
Small cleaved lymphocytes usually predominate; large cleaved or non-cleaved cells may be present | Distinct paratrabecular infiltrates are common; neoplastic follicles may be present; CD10 and BCL6 positivity in the lymphoma cells is lower in the bone marrow than in the lymph node; large B-cell lymphoma may have discordant, low-grade follicular lymphoma in the bone marrow |
| Splenic marginal zone lymphoma | 100 | Intrasinusoidal Interstitial Focal random Diffuse |
Small lymphocytes with slightly irregular nuclei, condensed chromatin, and moderate amounts of cytoplasm | Intrasinusoidal infiltration is common; reactive germinal centers may be present; “villous lymphocytes” may be present in the blood smears |
| Low-grade extranodal marginal zone lymphoma | 5-44 | Focal random Focal paratrabecular Interstitial Intrasinusoidal |
Small cells with condensed chromatin and scant to moderate amounts of cytoplasm; occasional scattered large cells may be present | Extent of bone marrow infiltration usually minimal; overt blood involvement is uncommon |
| Nodal marginal zone lymphoma | 30-50 | Focal random Interstitial Focal paratrabecular |
Small cells with condensed chromatin and scant to moderate amounts of cytoplasm | |
| Diffuse large B-cell lymphoma | Common type: 10-30 Double-hit: 40-90 |
Focal random Paratrabecular Diffuse |
Large cells with irregular nuclei, distinct or prominent nucleoli; may have discordant histology subtype in the bone marrow such as a low-grade B-cell lymphoma | Lymphomatous infiltrates can be heterogeneous with prominent T cells (T-cell/histiocyte-rich large B-cell lymphoma); immunohistochemistry for B-cell antigens is essential in identifying the large B-cell component; rare cases of large cell lymphoma are intravascular |
| Burkitt's lymphoma | 30-60 | Interstitial Diffuse |
Medium-sized cells with reticular chromatin, multiple small nucleoli, and basophilic cytoplasm; cytoplasmic vacuoles are common | MYC gene rearrangement positive; “starry sky” pattern may be present; frequent mitoses and necrosis are common |
| Peripheral T-cell lymphoma, not otherwise specified | 20-40 | Focal random Diffuse |
Polymorphous lymphoid population; nuclei are often hyperchromatic and irregular; large cells with nucleoli may be present; prominent reactive cell component often intermixed with lymphoma cells | Vascularity and reticulin fibrosis are often prominent; infiltrate is less well demarcated than most B-cell lymphomas |
| Anaplastic large cell lymphoma | 10-30 | Focal random Interstitial with scattered cells Diffuse |
Large cells with horseshoe-shaped nuclei and abundant cytoplasm (hallmark cells) | Detection rate is higher with immunostaining for CD30 or ALK1 |
| Hepatosplenic T-cell lymphoma | 100 | Intrasinusoidal Interstitial |
Variable; often medium-sized lymphocytes with cytologic atypia; may have dispersed chromatin resembling blasts | Lymphomatous infiltrates may be subtle, and immunostaining for CD3 and cytotoxic proteins is helpful; typically, TCR γδ + /CD4 − /CD8 − isochromosome 7q is common |
| Angioimmunoblastic T-cell lymphoma | 50-80 | Focal random | Polymorphous infiltrates; neoplastic cells with clear cytoplasm are uncommon | Positivity for CD10 and CXCR13 in the lymphoma cells is less common in the bone marrow; primary diagnosis based on the bone marrow findings alone is challenging |
| NK/T-cell lymphoma, nasal type | 10-20 | Interstitial, often with scattered single cells | Variably sized; small, medium, to large cells, or a mixture | In situ hybridization for EBER may be necessary to identify subtle infiltrate in the bone marrow |
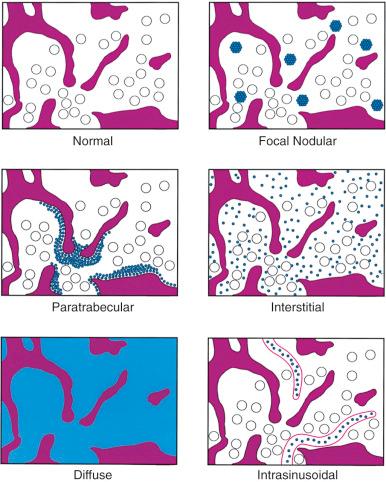
Lymphomatous infiltrates in the bone marrow can occur in five different patterns: focal random (nodular), focal paratrabecular, interstitial, diffuse, and intrasinusoidal ( Fig. 56-11 ). Focal infiltrates, the most common pattern, are characterized by discrete collections of neoplastic lymphocytes. Even though they focally displace bone marrow and fat cells, they are usually associated with considerable sparing of normal hematopoietic tissue. Focal infiltrates are present in either random or paratrabecular locations. Focal random lymphoid infiltrates occupy space away from the bone trabeculae, whereas paratrabecular infiltrates preferentially grow along and “hug” the bone trabeculae. Random lymphoid infiltrates that expand and focally touch the bone are not considered paratrabecular. In interstitial infiltrates, the neoplastic lymphocytes infiltrate between normal hematopoietic cells without significantly disrupting the bone marrow architecture. They usually do not replace large amounts of bone marrow tissue, even though there is generally widespread bone marrow involvement. Diffuse infiltrates completely replace the hematopoietic elements between the bone trabeculae in a portion or all of the bone marrow core biopsy section. Intrasinusoidal infiltration is characterized by collections of neoplastic lymphocytes within the sinusoids; these infiltrates are typically subtle and difficult to appreciate on hematoxylin and eosin–stained section but can be highlighted by immunohistochemical stains.
This section describes the characteristics of each type of B-cell lymphoma in the bone marrow, and the morphologic features specific to bone marrow involvement are emphasized. Other features, such as immunophenotype and genetic characteristics, are briefly discussed, and more details can be found in other chapters.
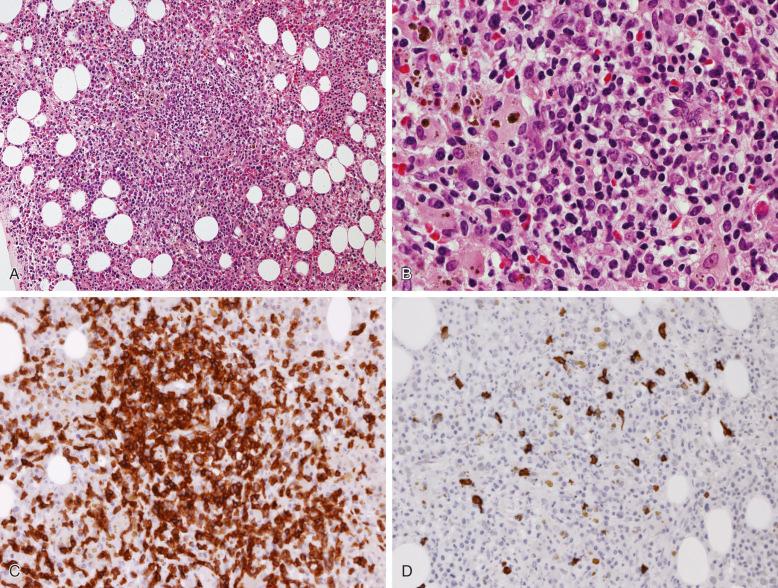
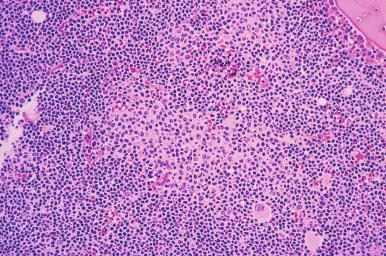
Chronic lymphocytic leukemia (CLL) is the leukemic manifestation, and small lymphocytic lymphoma (SLL) is the tissue counterpart of the same disease process. About 85% of patients with SLL have bone marrow involvement at diagnosis; rarely, patients may have isolated bone marrow involvement. Although an absolute lymphocytosis is usually absent in SLL, a small population of circulating monoclonal B cells can be frequently detected by flow cytometry. Approximately 15% of patients who are seen with SLL later have overt lymphocytosis. The patterns in which CLL/SLL infiltrates the bone marrow are focal random ( Fig. 56-12 ), diffuse, interstitial, or mixed. Although focal random infiltrates can expand to touch the bone trabeculae, distinctly paratrabecular infiltrates are absent. When paratrabecular infiltrates are observed, the diagnosis of CLL/SLL should be questioned, and other types of lymphoma, such as follicular lymphoma and mantle cell lymphoma, should be considered in the differential diagnosis. The neoplastic lymphocytes are small with round nuclei, condensed chromatin, and scant cytoplasm. Proliferation centers, the characteristic morphologic finding of CLL/SLL in lymph node tissue, are occasionally encountered in the bone marrow core biopsy ( Fig. 56-13 ).
The typical immunophenotype of CLL/SLL includes dim CD20 + , CD5 + , CD10 − , CD23 + , FMC7 − /dim + , CD79b − /dim + , and dim surface immunoglobulin light chain restriction. Some cases of CLL/SLL may have overlapping morphologic and immunophenotypic features with mantle cell lymphoma; however, a negative immunohistochemical staining for BCL1/cyclin D1 essentially rules out the latter possibility.
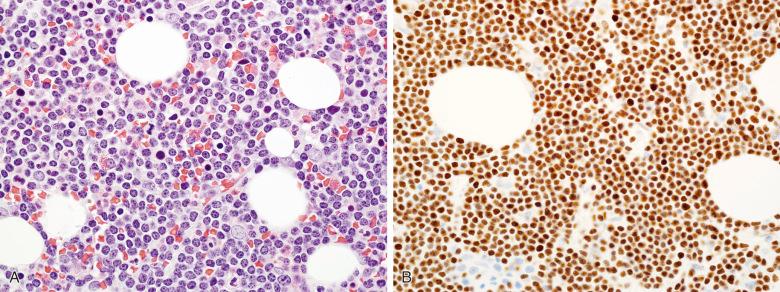
Newer markers for CLL/SLL have been identified. CD200, an immunoglobulin superfamily membrane glycoprotein, is found to be highly expressed in virtually all cases of CLL/SLL but absent or expressed at a low level in mantle cell lymphoma; thus, immunohistochemical or flow cytometric analysis of CD200 is helpful in distinction of the two diseases. CD200 is also expressed in lymphoplasmacytic lymphoma, hairy cell leukemia, and B-lymphoblastic leukemia; thus, the results of flow cytometric and immunohistochemical studies should be interpreted in clinical and morphologic context. Recently, nuclear overexpression of lymphoid enhancer-binding factor 1 (LEF1) has been found to be highly associated with CLL/SLL among small B-cell lymphomas and serves as a reliable immunohistochemical and flow cytometric marker to differentiate CLL/SLL from other small B-cell lymphomas, including mantle cell lymphoma and CD5 + marginal zone lymphoma ( Fig. 56-14 ). Care must be taken in interpreting cases with frequently admixed T cells as LEF1 is normally expressed in T cells.
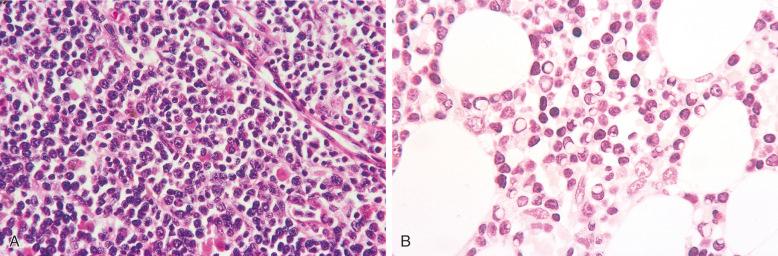
Lymphoplasmacytic lymphoma (LPL) is a B-cell neoplasm that commonly involves the bone marrow and, less frequently, the lymph nodes. Most LPLs in the bone marrow represent Waldenström's macroglobulinemia (WM), which is defined as LPL with bone marrow involvement and a serum immunoglobulin M (IgM) monoclonal protein of any concentration. LPL infiltrates in the bone marrow are focal random, focal paratrabecular, interstitial, or diffuse; mixed patterns are common. The infiltrates of LPL/WM consist of small lymphocytes, plasmacytoid lymphocytes, and plasma cells in variable proportions. The plasmacytoid lymphocytes are usually in the minority and may be inconspicuous. The number of plasma cells varies considerably; they are usually a minor component of the infiltrate but can predominate in some cases. The plasma cells are often admixed within the neoplastic lymphoid infiltrate but are occasionally present as small clusters away from the lymphoid aggregates. Intranuclear inclusions (Dutcher bodies) are often identified in the plasma cells ( Fig. 56-15 ). Transformed lymphocytes with distinct nucleoli may be present but are usually low in number. In addition, mast cells and histiocytes are often increased in the bone marrow involved by LPL. The bone marrow aspirate contains lymphoma cells similar to those in the bone marrow core biopsy; peripheral blood may also be involved, but an absolute lymphocytosis is uncommon.
The characteristic immunophenotype of LPL is CD20 + , CD23 − , CD5 − , CD10 − , CD103 − , surface IgM + , and surface immunoglobulin light chain restricted. Expression of CD5 or CD23 may be observed in some cases.
LPL/WM should be distinguished from other B-cell neoplasms with plasmacytic differentiation, such as splenic marginal zone lymphoma. LPL may also mimic plasma cell myeloma when the plasma cell component predominates. The presence of both monotypic B cells and plasma cells with the same light chain restriction and other clinical and laboratory features, such as an IgM serum paraprotein with hyperviscosity, lack of lytic bone lesions, and presence of lymphadenopathy, help distinguish LPL from plasma cell myeloma. The distinction between LPL and splenic marginal zone lymphoma in the bone marrow can be problematic because they share morphologic and phenotypic features and occasionally also clinical features. LPL predominantly involves the bone marrow but may also involve lymph node or spleen in some cases. Splenic marginal zone lymphoma typically exhibits prominent splenomegaly and almost always involves blood and bone marrow, but peripheral lymph nodes are typically not involved. The most distinguishing features of LPL from splenic marginal zone lymphoma in the bone marrow have been reported to include paratrabecular involvement, presence of lymphoplasmacytoid cells, Dutcher bodies, and increased numbers of mast cells, whereas prominent intrasinusoidal infiltration and reactive germinal centers are more characteristic for splenic marginal zone lymphoma.
There are no specific chromosomal abnormalities recognized in LPL. Recently, MYD88 L265P mutation has been identified in more than 90% of patients with LPL/WM; this mutation is uncommon in other B-cell lymphomas, including splenic marginal zone lymphoma. Although it is not entirely specific, demonstration of MYD88 L265P mutation is a valuable adjunct in the diagnosis of LPL/WM.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here