Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Osteoma cutis represents a reactive, metaplastic, lamellar bone formation in the dermis and/or subcutis
Primary and secondary forms of osteoma cutis can be separated on the basis of the absence or presence of an underlying skin disorder
Primary osteoma cutis develops in normal skin or in the absence of an identifiable skin disorder (idiopathic)
Secondary osteoma cutis always arises in previously damaged skin (e.g., in abnormal skin)
Primary osteoma cutis can be isolated (e.g., nonsyndromic) or associated with various syndromes, including myositis ossificans progressiva, progressive osseous heteroplasia, fibrodysplasia ossificans progressiva, Albright hereditary osteodystrophy, and platelike osteoma cutis, or metabolic disorders like pseudohypoparathyroidism and pseudopseudohypoparathyroidism
Secondary osteoma cutis develops in the background of a preexisting inflammatory skin condition (acne, folliculitis, localized scleroderma, dermatomyositis, scars, infections, chronic venous insufficiency, among others), neoplasia (pilomatricoma, chondroid syringoma, melanocytic nevus, basal cell carcinoma, among others), or other conditions (trauma, Becker nevus, among others)
Primary osteoma cutis represents about 15% to 30% and secondary forms represent about 70% to 85% of cases
Plaquelike osteoma cutis
Congenital or within the first year of life
Acquired lesions presenting in adults appear to be more common in males, with predilection for the head, and especially the face
Multiple miliary osteomas
Middle-aged Caucasian women
History of severe acne in about 55%, also reported after bisphosphonate (alendronate) therapy for osteoporosis, artificial tanning, severe blistering secondary to sunburn, and exogenous ochronosis
Other types of secondary osteoma cutis
No sex, age, or site predilection
Asymptomatic, skin-colored papule(s), plaque(s), or nodule(s) of hard consistency
Primary osteoma cutis
Solitary, plaquelike, multiple, or widespread (disseminated)
Can be the presenting sign of an underlying syndrome, especially in infancy
Plaquelike osteoma cutis
Reddish or skin-colored plaque of hard consistency
Size usually less than 15 cm
Predilection for scalp, face, and limbs, but any site can be affected
Criteria for the diagnosis include
Presence at birth or during first year of life
At least one bony plate with/without other cutaneous osteomas
No evidence of abnormalities in calcium or phosphorous metabolism
No history of infection or trauma or any other predisposing event(s)
Secondary osteoma cutis
Solitary or multiple
Multiple miliary osteomas
Numerous skin papules
Progressive increase in numbers and peripheral spread frequently reported
Bluish hue of the papules, especially when history of minocycline treatment
Size from 1 to 3 mm
Scalp and face, followed by trunk, breast, extremities, and buttocks
Multiple lesions and plaquelike growth can be cosmetically disturbing
Topical tretinoin therapy, surgical excision, needle mini-incision followed by osteoma extraction, laser treatment, dermabrasion
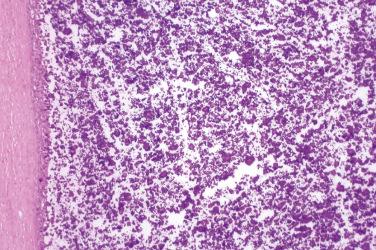
Lamellar mineralized bone, typically arranged concentrically in the dermis, subcutis, or both
Osteocytes and osteoblasts embedded within the bone, most frequently at the periphery, whereas osteoclasts are much more infrequent
Medullary spaces containing adipose tissue and variably abundant bone marrow (usually absent)
Rim of fibroblasts frequently surrounds lamellar bone
Transepidermal elimination of bony spicules occasionally present
GNAS gene mutations encoding G-protein alpha-stimulatory subunit in patients with progressive osseous heteroplasia, Albright hereditary osteodystrophy, platelike osteoma cutis, and pseudohypoparathyroidism type 1a
ACVR1 gene mutations encoding receptors for bone morphogenetic proteins in patients with fibrodysplasia ossificans progressiva
Osteochondroma (benign cartilaginous exostosis)
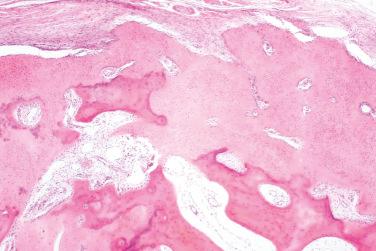
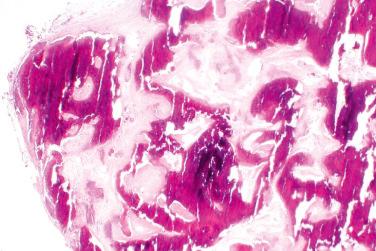
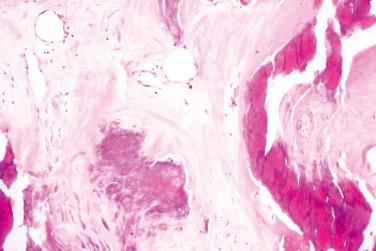
The entity is characterized by calcium salt deposition in different periarticular soft tissues
Although the term tumoral calcinosis has been used inconsistently in the literature for a variety of unrelated conditions having in common periarticular calcifications, including primary and secondary tumoral calcinosis, it should be restricted for hereditary conditions only (e.g., primary tumoral calcinosis)
Two types of primary tumoral calcinosis have been recognized: (1) hyperphosphatemic type with familial basis caused by mutations in N-acetylgalactosaminyltransferase 3 ( GALNT3 ), KLOTHO or fibroblast growth factor 23 ( FGF23 ) genes and (2) normophosphatemic type with likely familial basis caused by mutation in the sterile alpha motif domain containing 9 (SAMD9) gene
Significant higher incidence in patients of African descent
No sex predominance
Usually starts in the first and second decade of life
Local, painless swelling progressing to firm mass with reduced joint mobility
Ulceration of the overlying skin with secondary inflection not uncommon
Pain inconsistent, usually associated with compression of the local nerve
Predilection for large joints of the hips, shoulders, and elbows, less often hand and wrist
Usually lobular/multilobular, densely calcified mass in the periarticular soft tissue, most commonly localized at the extensor surface of joints along the bursa
Ocular involvement ranging from angioid streaks to corneal calcification deposits occasionally present
Early surgical excision to prevent joint limitation(s), but associated with high recurrence rate
Surgical excision combined with phosphate deprivation (aluminum hydroxide) in conjunction with acetazolamide has proven the most effective
Histological features depend on the stage of evolution
Early lesions
Multiple pseudocystic spaces filled with eosinophilic debris undergoing calcification
Pseudocystic spaces lined by epithelioid macrophages and multinucleated giant cells
Advanced lesions
Densely calcified material
Hyalinized connective tissue
Not contributory
Calcinosis of chronic renal failure
Calcinosis universalis (typically involving multiple tissue planes like subcutis, muscles, and fascia, frequently associated with connective tissue diseases)
Calcinosis circumscripta (dermal and/or subcutaneous nodules at diverse sites, often in the context of connective tissue diseases)
Calcific tendonitis
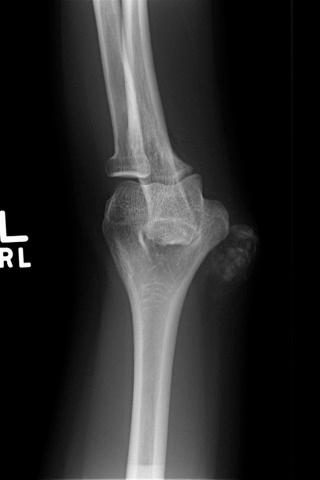
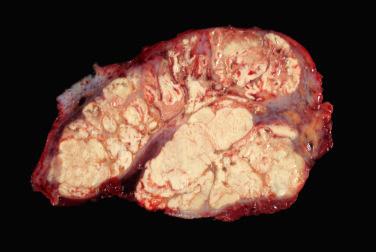
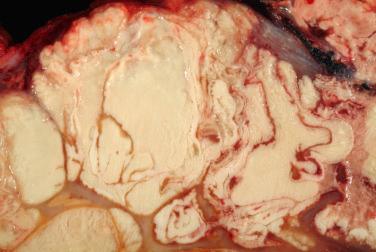
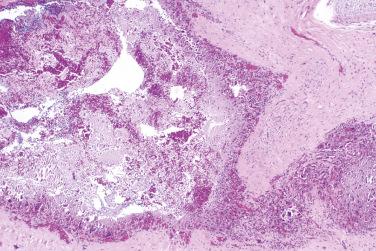
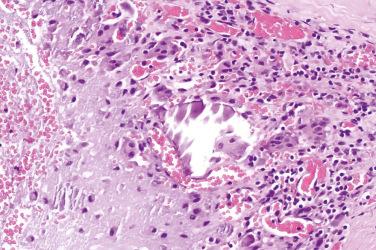
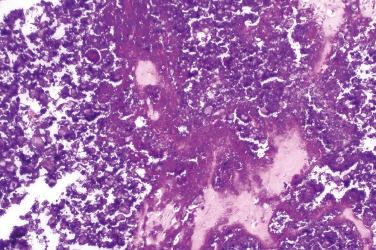
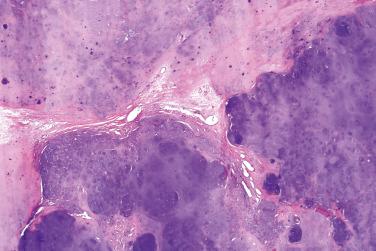
A benign, cartilage-forming tumor composed of lobules of mature hyaline cartilage occurring outside the skeletal system in the superficial or deep soft tissue(s)
Male predominance
Most common in patients between 30 and 60 years of age
Familial and congenital occurrence exceptional
Slowly growing, well-demarcated, asymptomatic mass or nodule
Solitary lesions by far predominate
Occasionally painful
Lesions localized at the ungual/periungual site (e.g., nail matrix, nail bed) frequently associated with nail dystrophy/deformity
Less than 2 cm in the majority of cases
Closely associated with the tendon, tendon sheath, joint capsule, or periosteum
Strong predilection for extremities (over 90%), in particular, hands and feet, with fingers being the most common single site
Exceptional sites include dura, spinal canal, oral cavity, pharynx, larynx, skin, fallopian tube, and parotid gland
Uniformly benign clinical course; no malignant transformation has been reported
Complete excision curative
Recurrences after marginal/incomplete excision in roughly 10% to 15%
Well-circumscribed, lobular/multilobular proliferation in the dermis and/or subcutis
Mature adult-type hyaline cartilage is the principal, often exclusive, component of the lesion
Mature chondrocytes, typically arranged in cords/rows
Absent or mild nuclear pleomorphism
Absent or a few normal mitoses
Occasional binucleated cells (binucleated lacunae)
Abundant chondroid matrix
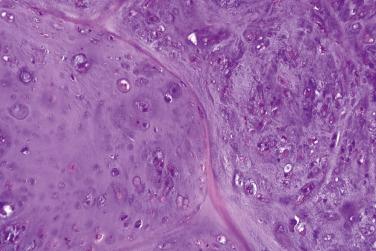
Chondroblastic variant
Hypercellular proliferation in comparison with adult-type cartilage
Immature cells, resembling chondroblasts
Plump, rounded or spindled nuclei
Prominent eosinophilic cytoplasm
Considerable variation in size and shape (e.g., nuclear and cytological atypia)
Normal mitoses can be present, but atypical mitoses absent
The presence of immature cells is not related to biological potential
Richly vascularized stroma
Secondary changes (can be extensive)
Calcifications, outlining lacunae in a “lacelike” or “chicken-wire” pattern, or more extensive (e.g., “tumoral calcinosis–like”)
Metaplastic ossification, especially at the periphery of the lobules
Myxoid change
Cystic degeneration
Hemorrhage
Focal granuloma–like proliferation of histiocytes and osteoclast-like giant cells present in about 10% of the lesions, usually at the periphery of the tumor lobules
S100-protein positive
Markers of epithelial differentiation negative
Cytogenetic analysis performed on limited numbers of soft tissue chondromas detected clonal chromosomal abnormalities in a subset of cases, none of them consistent, including monosomy of chromosome 6, rearrangements of chromosome 11, supernumerary rings derived from chromosome 12, and rearrangements of chromosome 12q13–q15
Activating IDH1 mutations are not seen
Enchondroma
Synovial chondromatosis
Skeletal chondrosarcoma extending into the surrounding soft tissue
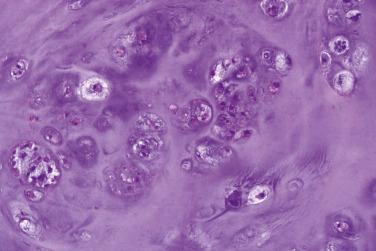
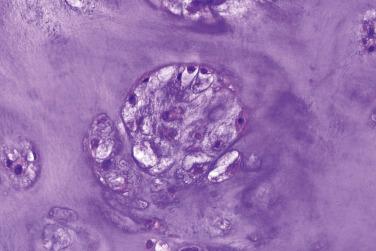
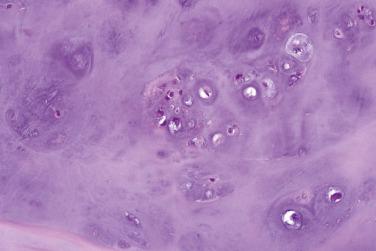
A rapidly growing, bone-forming, reactive lesion that occurs almost exclusively on the digits and is thought to be triggered by trauma
Rare
Wide age range with predilection for young adults
Males more commonly affected
Rapidly growing
Small lesion
Nonspecific painful swelling
Fingers more commonly involved than toes
Predilection for proximal phalanx
Lesions not attached to bone
Simple excision
Local recurrences are exceptional
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here