Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The aggressiveness of the lesions described in this chapter ranges between purely benign and frankly malignant. Although these lesions frequently are treated satisfactorily with intralesional procedures, such as curettage, they are sometimes very aggressive locally and require marginal or wide resection. Systemic involvement, although rare, must be evaluated and treated. Giant cell tumors and chondroblastomas can develop pulmonary metastases and in rare cases can be fatal. Langerhans cell histiocytosis (LCH) can involve multiple organ systems in addition to bone involvement and likewise in rare cases can be fatal. This chapter briefly describes the clinical, radiographic, and pathologic features of these lesions. A summary table is provided for quick reference ( Table 26.1 ).
| tumor | Age (yr) | Demographics | Site | Presentation | Imaging | Histology | Treatment | Comments |
|---|---|---|---|---|---|---|---|---|
| Giant cell tumor | 20–40 | Slight female predominance | Distal femur Proximal tibia Distal radius |
Pain Pathologic fracture (10%–30%) |
Eccentrically located in epiphysis Purely radiolucent (no matrix formation) Usually no rim of reactive bone Abuts subchondral bone May exhibit cortical destruction with soft-tissue extension Metaphyseal in skeletally immature patients |
Multinucleated giant cells in sea of mononuclear cells Nuclei of mononuclear cells identical to nuclei of giant cells |
Extended curettage Resection if residual bone stock inadequate Consider radiation for spinal/sacral tumors Resection of pulmonary metastases |
3% incidence of benign pulmonary metastases |
| Chondroblastoma | 10–25 | Male:female 2:1 | Distal femur Proximal tibia Proximal humerus |
Pain Symptoms can mimic chronic synovitis |
Well-circumscribed lesion in epiphysis or apophysis May cross an open physis Frequently with rim of bone, 30%–50% with matrix calcification |
Sheets of chondroblasts (polygonal cells with distinct cytoplasmic outlines) “chicken wire” calcification Multinucleated giant cells Secondary aneurysmal bone cyst in 20% |
Extended curettage Resection of pulmonary metastases |
1% incidence of benign pulmonary metastases |
| Chondromyxoid fibroma | 10–30 | Slight male predominance | Proximal tibia | Pain Can present with painless mass in hands and feet |
Well-circumscribed bubbly lesion Thin rim of reactive bone (appearance similar to nonossifying fibroma) |
Lobules of hypocellular myxoid cartilaginous tissue Lobules separated by cellular fibrous tissue |
Extended curettage | Important to distinguish from chondrosarcoma |
| Osteoblastoma | 10–30 | Male:female 3:1 | Posterior elements of spine Any bone |
Pain Painful scoliosis Neurologic symptoms |
Bone-forming lesion in posterior elements of spine Variable/nonspecific radiographic appearance outside of spine |
Fibrovascular stroma Osteoid/woven bone Osteoblastic rimming Histologic appearance similar to osteoid osteoma |
Extended curettage or resection Might require spinal stabilization |
Important to distinguish from low-grade osteosarcoma |
| Langerhans cell histiocytosis | <20 | Male:female 2:1 | Vertebral bodies Flat bones Diaphysis of long bones |
Bone lesions may be painful or asymptomatic Can mimic osteomyelitis (pain, fever, local signs) |
Vertebra plana “hole within a hole” appearance in flat bones Aggressive, permeative appearance with periosteal reaction in long bones Radiographic appearance varies from very benign to very aggressive Can be multifocal Bone scan sometimes falsely negative |
Large histiocytic cells with indented nucleus and abundant cytoplasm S-100 positive Clusters of eosinophils Birbeck granule seen on electron microscopy |
Observation of asymptomatic lesions (usually resolve) Steroid injection for symptomatic lesions Curettage/grafting for impending fractures Chemotherapy for systemic disease |
Hand-Schüller-Christian disease triad of skull lesions, exophthalmos, and diabetes insipidus Letterer-Siwe disease—fever, lymphadenopathy, hepatosplenomegaly, and multiple bone lesions |
According to a Mayo Clinic series, giant cell tumors represent 5% of neoplasms of bone. They typically occur in patients 20 to 40 years old, and there is a slight female predominance. The most common location for this tumor is the distal femur, followed closely by the proximal tibia. In the distal radius (the third most common location), these tumors frequently are more aggressive. Spinal involvement, other than the sacrum, is rare.
Giant cell tumors usually are solitary lesions; however, 1% to 2% may be synchronously or metachronously multicentric. It is unclear whether multicentric disease represents multiple primary lesions or simply bone metastases from a single primary lesion. Although these tumors typically are benign, pulmonary metastases occur in approximately 3% of patients. Some patients with pulmonary metastases have spontaneous regression or remain asymptomatic for many years. Others may have progressive pulmonary lesions, however, that lead to death despite the tumors remaining histologically benign. The overall mortality rate from disease for patients with pulmonary metastases is approximately 15%. Patients with recurrent lesions or primary lesions that appear aggressive radiographically (stage 3) are at higher risk for pulmonary metastases.
Malignant giant cell tumors represent less than 5% of cases and are classified as primary or secondary. Primary malignant giant cell tumors are extremely rare and are defined as sarcomas that occur within lesions that otherwise are typical of benign giant cell tumors. Secondary malignant giant cell tumors are sarcomas that occur at the sites of giant cell tumors that have been treated, usually with radiation.
Most patients with giant cell tumors have progressive pain that often is related to activity initially and only later becomes evident at rest. The pain is rarely severe, unless a pathologic fracture has occurred. In 10% to 30% of patients, pathologic fractures are evident at initial examination.
Radiographic findings often are diagnostic. The lesions are eccentrically located in the epiphyses of long bones and usually abut the subchondral bone. Although rare in skeletally immature patients, giant cell tumors arise in the metaphysis in this patient population. One theory suggests that these tumors originate in the metaphysis and later extend into the epiphysis after closure of the physes. Radiographically, the lesions are purely lytic. The zone of transition can be poorly defined on plain radiographs. In less aggressive tumors, a partial rim of reactive bone may be present. The lesion frequently expands or breaks through the cortex; however, intraarticular extension is rare because the subchondral bone usually remains intact. Matrix production usually is not evident within the bone but often is evident if there is soft-tissue extension, soft-tissue recurrence, or pulmonary metastases. Magnetic resonance imaging (MRI) is useful to determine the extent of the lesion within the bone and in the soft tissue. On MRI, the lesion usually is dark on T1-weighted images and bright on T2-weighted images. MRI also may reveal fluid-fluid levels typical of a secondary aneurysmal bone cyst, which occurs in 20% of patients.
Microscopically, giant cell tumors are composed of many multinucleated giant cells (typically 40 to 60 nuclei per cell) in a sea of mononuclear stromal cells. The nuclei of the mononuclear cells are identical to the nuclei of the giant cells, a feature that helps distinguish giant cell tumors from other tumors that may contain many giant cells. Areas of storiform spindle cell formation, reactive bone formation, or foamy macrophages may be seen. Secondary aneurysmal bone cysts also may be present. Many authors have attempted to grade these tumors histologically, but no grading system has proved to be of prognostic significance.
Giant cell tumors frequently are locally aggressive. Most manifest as stage 2 or stage 3 lesions. Historically, treatment consisted of simple curettage; however, subsequent recurrence rates were greater than 50%. Now, most published series document recurrence rates of 5% to 15%. The decrease in recurrence rates probably can be attributed to several factors. MRI now allows for more accurate assessment of the extent of lesions, and the technique of curettage has improved. It is important to create a cortical window that is at least as large as the lesion to prevent leaving residual tumor cells “around the corner” adjacent to the near-side cortex. Also, use of a power burr to enlarge the cavity 1 to 2 cm in all directions is now considered standard. Care should be taken, however, to avoid perforation through the subchondral bone into the joint. The use of adjuvants, such as liquid nitrogen, phenol, bone cement, electrocautery, or an argon beam coagulator, theoretically helps kill any remaining tumor cells. Also, preliminary studies suggest that bisphosphonates (administered systemically or locally) might help prevent recurrence.
To fill the defect after curettage, the surgeon has several options, including autograft bone, allograft bone, an artificial bone graft substitute, or methyl methacrylate bone cement. If an autograft is to be harvested from another site, separate gloves and instruments should be used because cross-contamination could lead to transplantation of tumor cells to the harvest site. A bone graft (or artificial substitute) has the theoretical advantage of restoring normal biomechanics to the joint surface to prevent future degenerative joint disease and restoring bone stock, which may help if future procedures are necessary. There are two main disadvantages, however, to using bone grafts: (1) the joint must be protected for an extended time to prevent a pathologic fracture and (2) tumor recurrence often is difficult or impossible to distinguish from graft resorption. These disadvantages may be overcome with the use of bone cement as a filling agent. Bone cement provides immediate stability, which aids in quicker rehabilitation; allows easier detection of recurrence, which is evident as an expanding radiolucency adjacent to the cement mantle; and may kill residual tumor cells through the heat of polymerization.
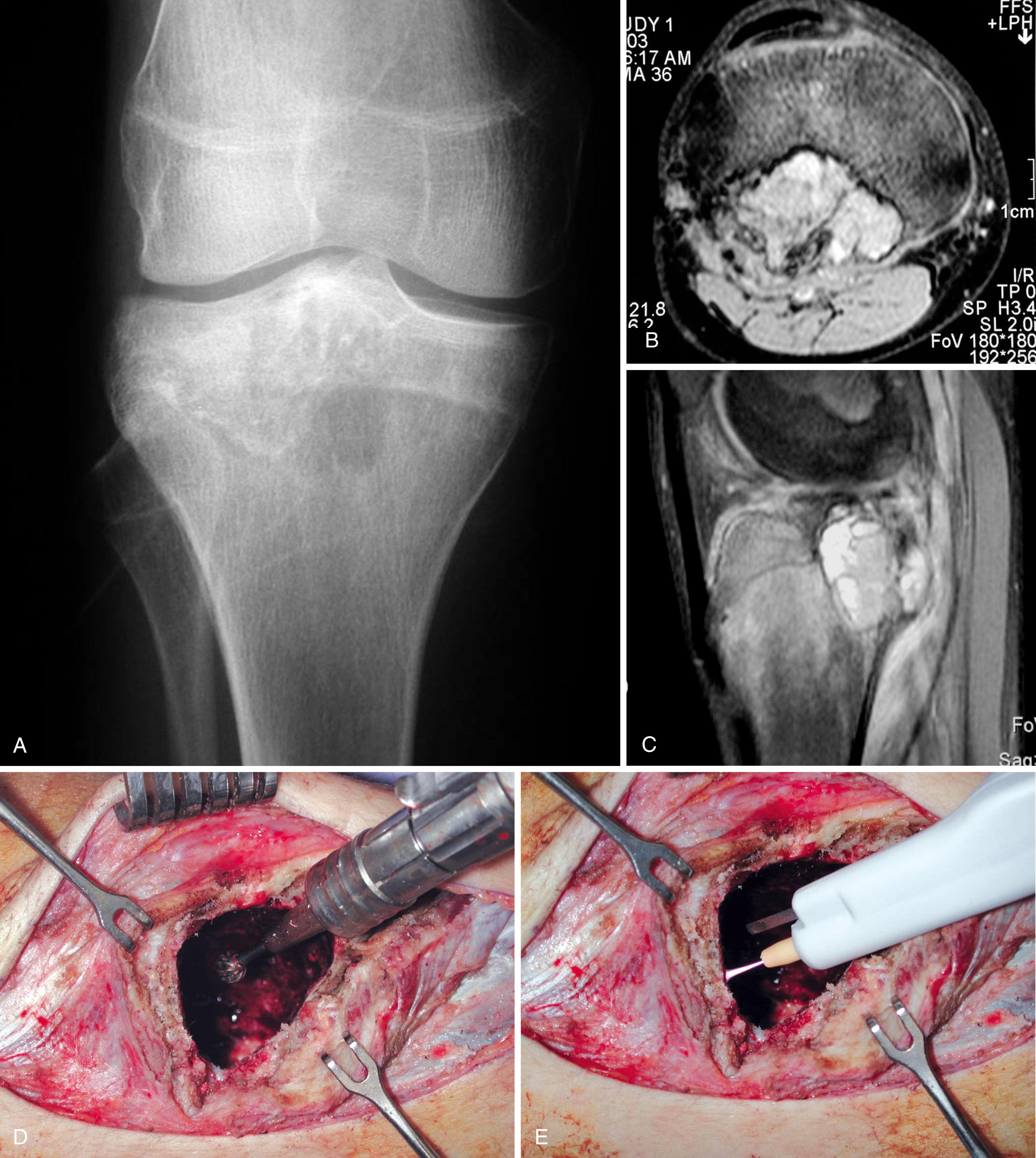
Our institution treats most giant cell tumors with aggressive, extended curettage followed by argon beam coagulation, which is easy to use, effective, and associated with few complications. Phenol or liquid nitrogen is not used as adjuvant treatment because of potential complications, such as pathologic fracture, wound healing problems, and nerve injury. Bone cement is chosen to fill the cavity because of its ease of application, immediate structural support, and ease with which local recurrence can be detected adjacent to the cement mantle. Screws placed in a crossed ( Fig. 26.1 ) or divergent ( Fig. 26.2 ) pattern are used to augment the cement mantle. Biomechanical studies performed at our institution have shown this method to significantly increase the strength of the reconstruction.
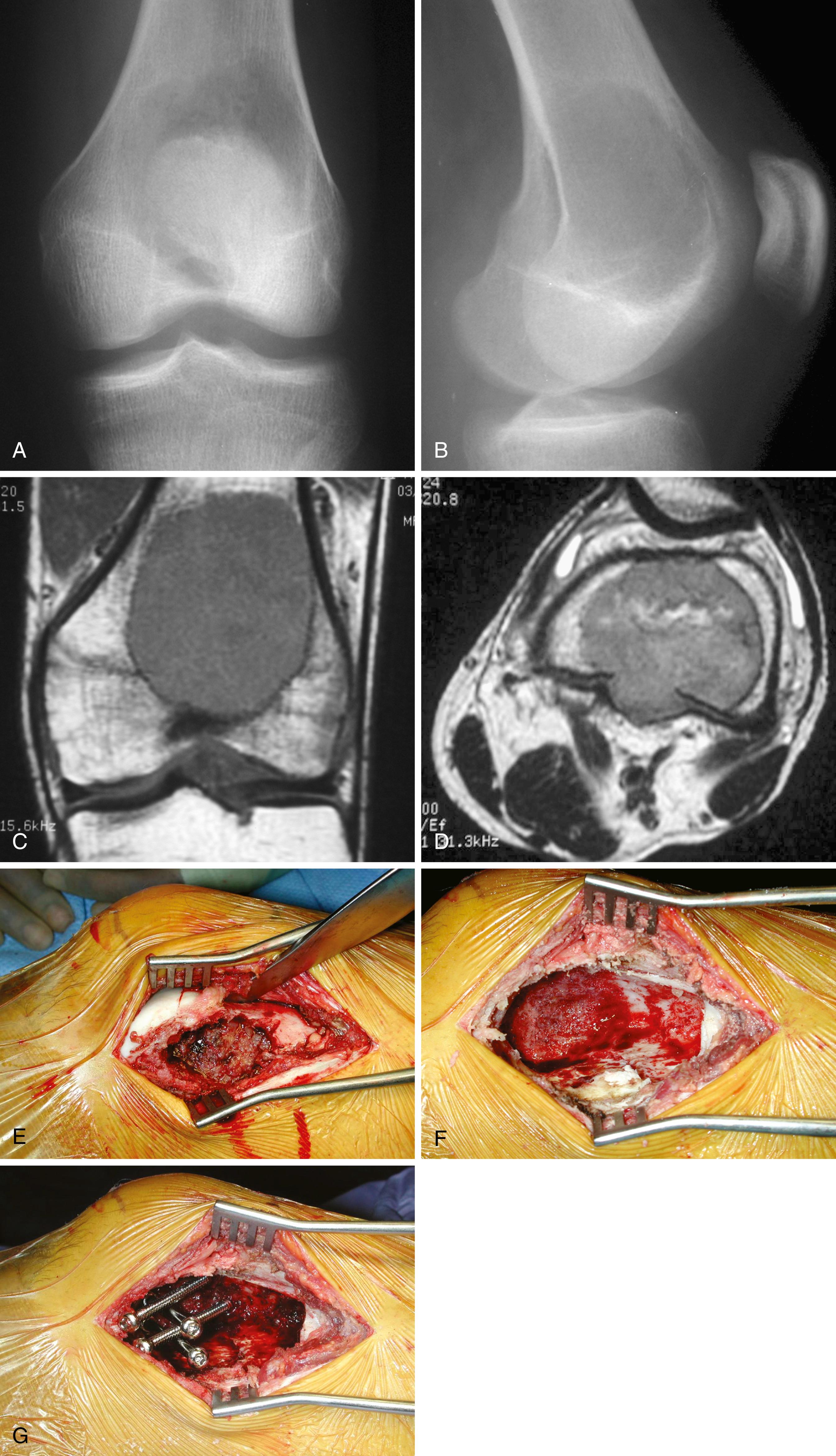
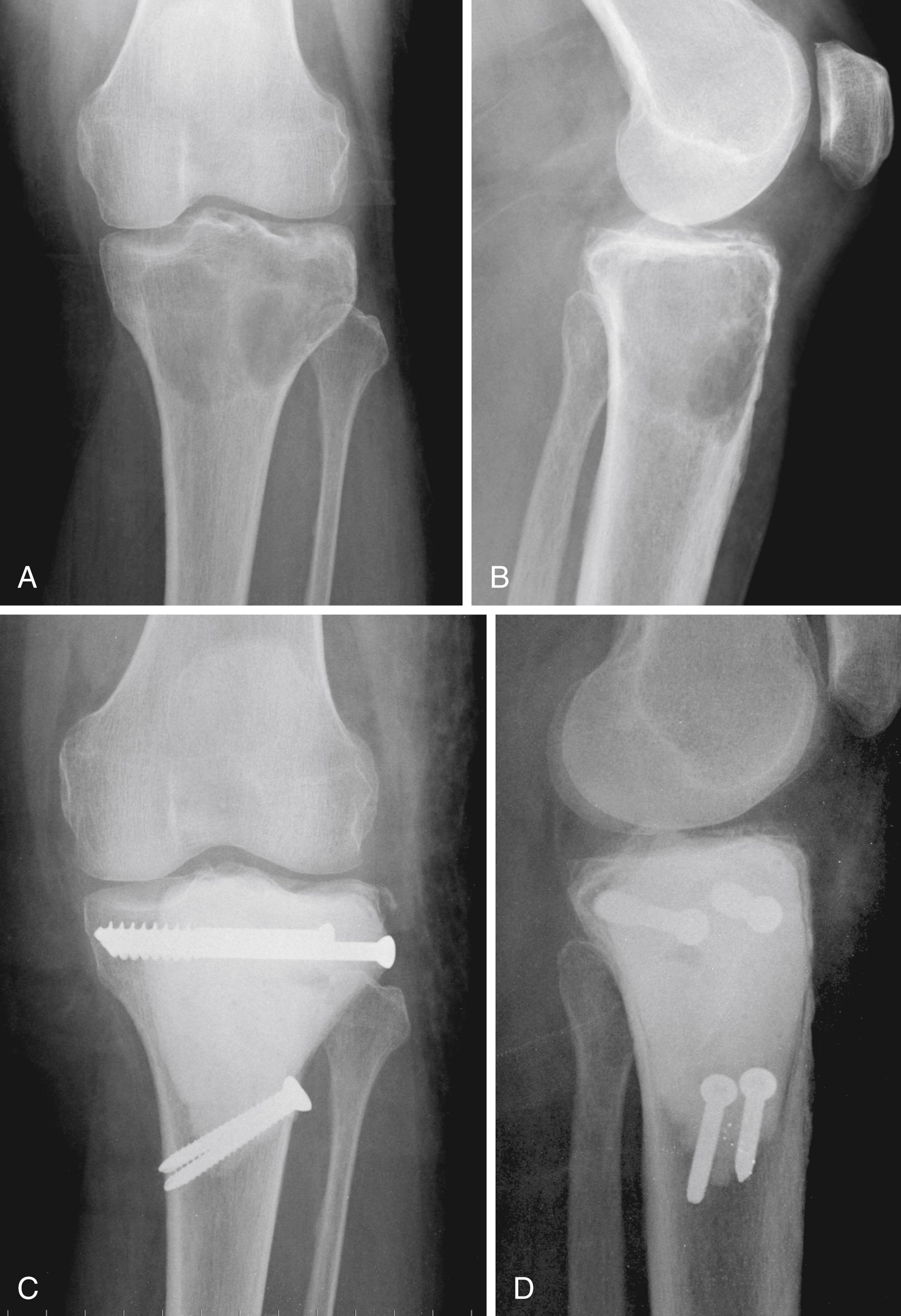
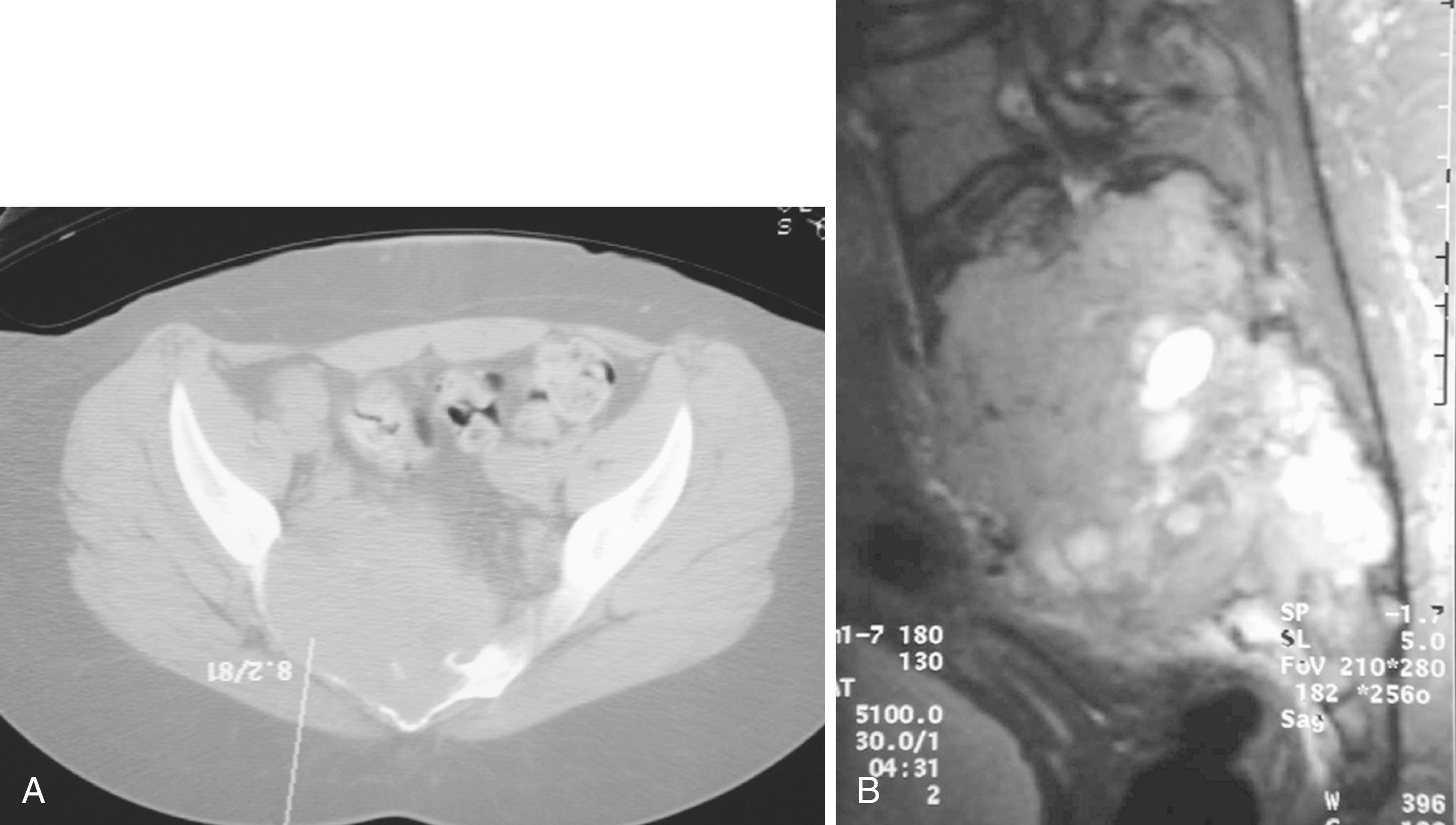
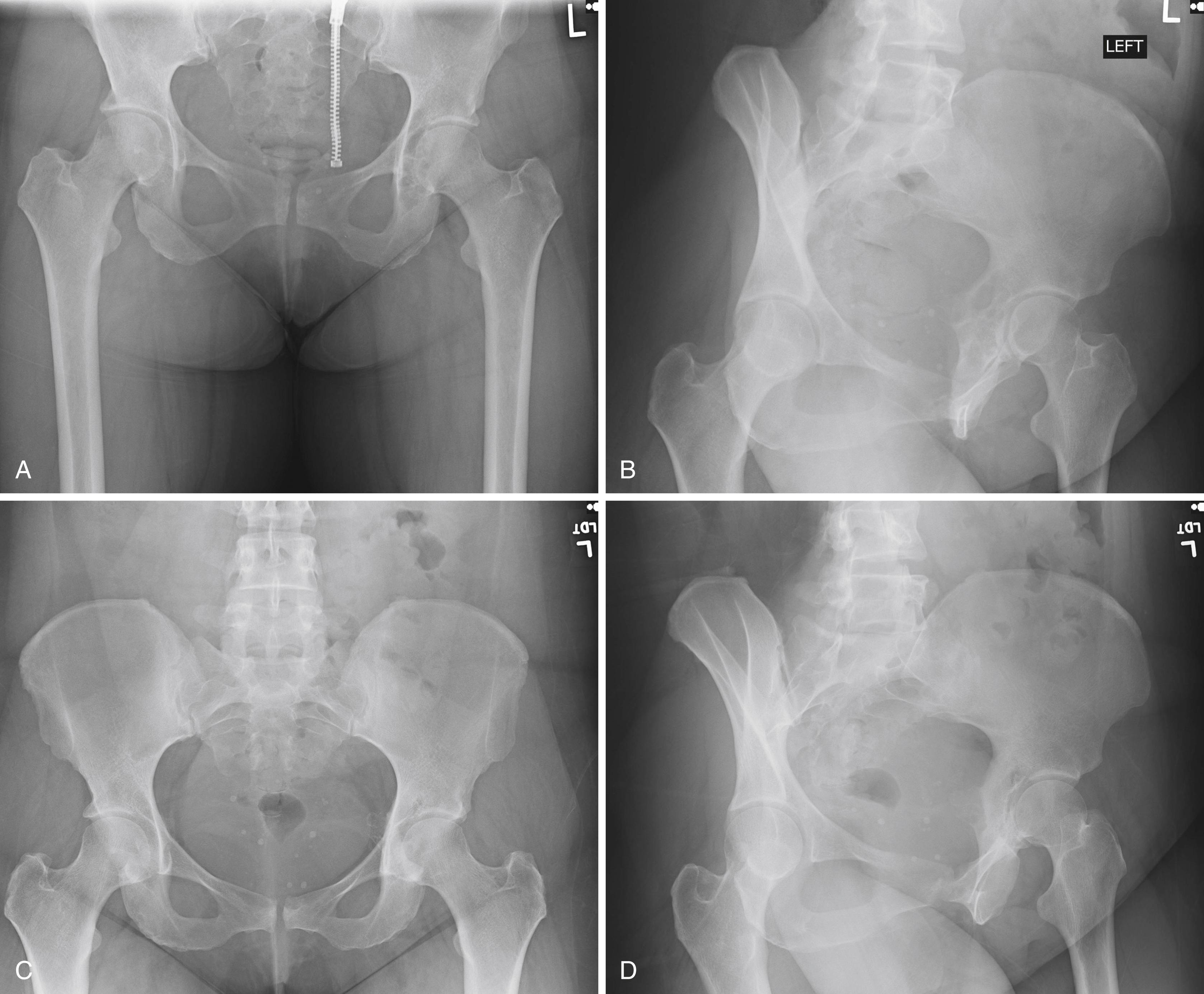
En bloc wide resection may be required in some stage 3 tumors, cases of local recurrence, and tumors recalcitrant to other methods of intervention. Around the knee, a hemicondylar osteoarticular allograft reconstruction or a rotating hinge endoprosthesis may be necessary ( Fig. 26.3 ). For aggressive lesions of the distal radius, primary resection and reconstruction with a proximal fibular autograft (either as an arthroplasty or as an arthrodesis) may be indicated ( Fig. 26.4 ). For lesions in expendable bones (e.g., the distal ulna, clavicle, or proximal fibula), primary resection without reconstruction may be indicated. For inoperable lesions in the spine or pelvis, irradiation or embolization (or both) may be used ( Fig. 26.5 ); however, caution is advised because of the risk of sarcomatous change in patients treated with irradiation. In patients with pulmonary metastases, resection should be attempted. Chemotherapy has limited success, and irradiation should be reserved for symptomatic inoperable lesions.
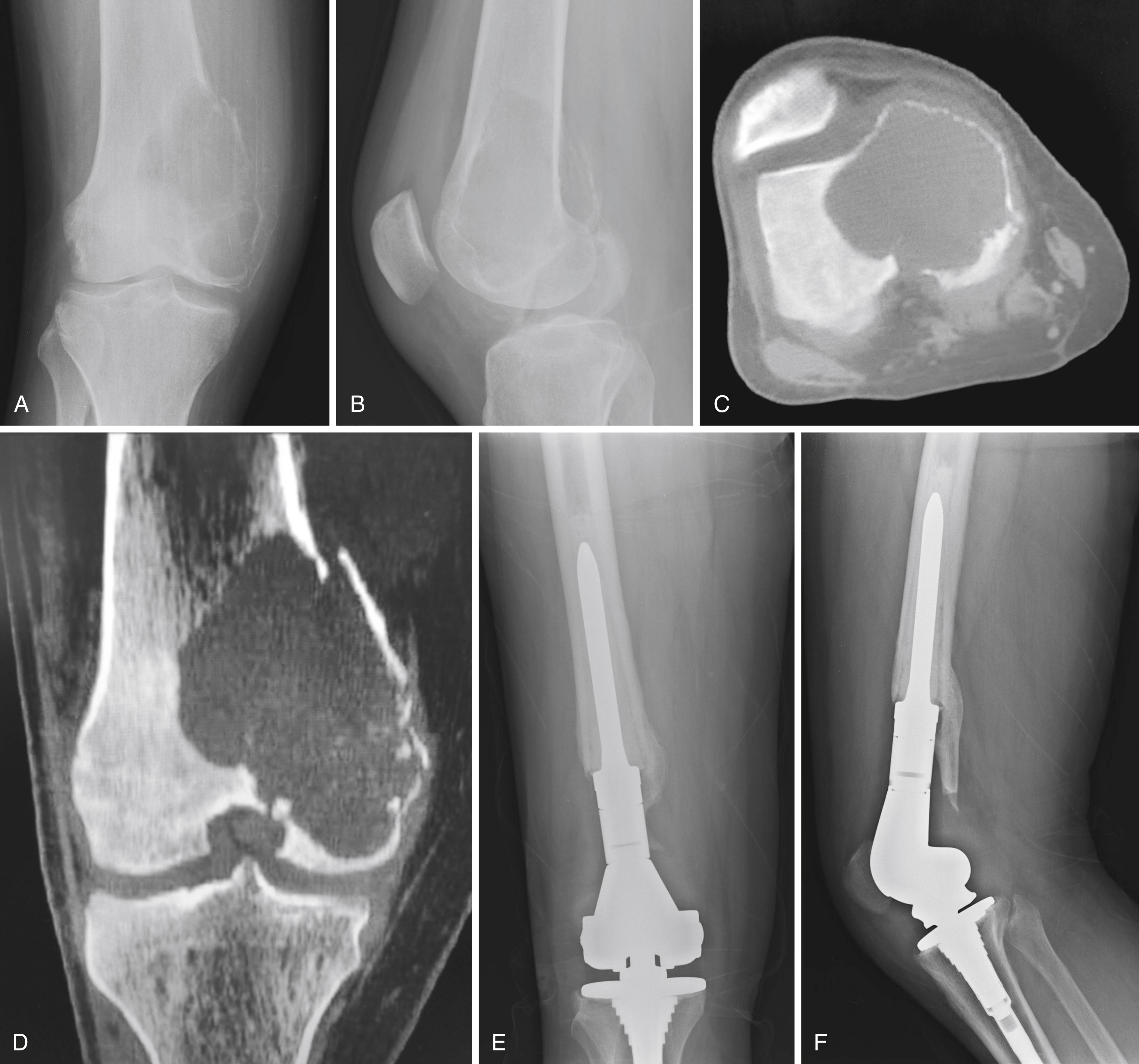
There is clinical rationale for the use of bisphosphonates in treating giant cell tumor of bone, as these drugs inhibit osteoclastic activity and promote osteoclast apoptosis. Studies using systemic zoledronic acid in inoperable tumors have reported stabilization of both local and metastatic disease. Bisphosphonates have been proposed for use as a surgical adjuvant or as an option in unresectable tumors; however, high-level evidence is still lacking, and further investigation is required to validate its use.
Another new treatment under examination is the systemic administration of denosumab. Denosumab is a fully human monoclonal antibody that inhibits normal and tumor-associated bone lysis by limiting osteoclastic maturation (i.e., prevents activation of receptor activator of nuclear factor-kB [RANK]). This drug has approval from the US Food and Drug Administration for use in adults and skeletally mature patients who have an unresectable giant cell tumor or a condition in which surgical resection would result in severe morbidity. Early results are promising, with radiographic studies showing sclerosis and reconstitution of cortical bone with a subsequent decrease in clinical symptoms such as pain. Longer term data are still being collected.
Patients diagnosed with giant cell tumors require long-term follow-up. Most local recurrences and pulmonary metastases occur within 3 years but have been reported to occur 20 years later. Chest radiographs should be obtained at the time of diagnosis to stage the lesion. We routinely obtain a chest computed tomography (CT) scan as a baseline reference at this time. At minimum, patients should have radiographs of the primary tumor site and the chest at 3- to 4-month intervals for 2 years, at 6-month intervals for the following year, and annually thereafter. An abnormality on the chest radiograph should be evaluated further with CT. Bone recurrence usually is evident as an expanding lucency on the radiograph. Soft-tissue recurrences may be apparent as ossification or may be evident only as a palpable mass, in which case MRI is indicated.
Treatment of recurrent lesions is the same as for primary lesions. If biopsy shows that the tumor is still benign, repeat curettage or resection should be performed.
Chondroblastoma, a rare neoplasm, typically occurs in patients 10 to 25 years old, with a 2:1 male predominance. According to the Mayo Clinic series, this tumor represents 1% of all primary bone tumors. Because of its rarity, no prospective and few retrospective studies have been reported in the literature. This tumor has a predilection for the epiphyses or apophyses of long tubular bones (e.g., distal femur, proximal humerus, and proximal tibia). Less frequently, chondroblastoma tends to occur in flat bones in older patients. Multicentric disease is exceedingly rare. Most patients complain of progressive pain that may mimic a chronic synovitis or other intraarticular pathologic conditions.
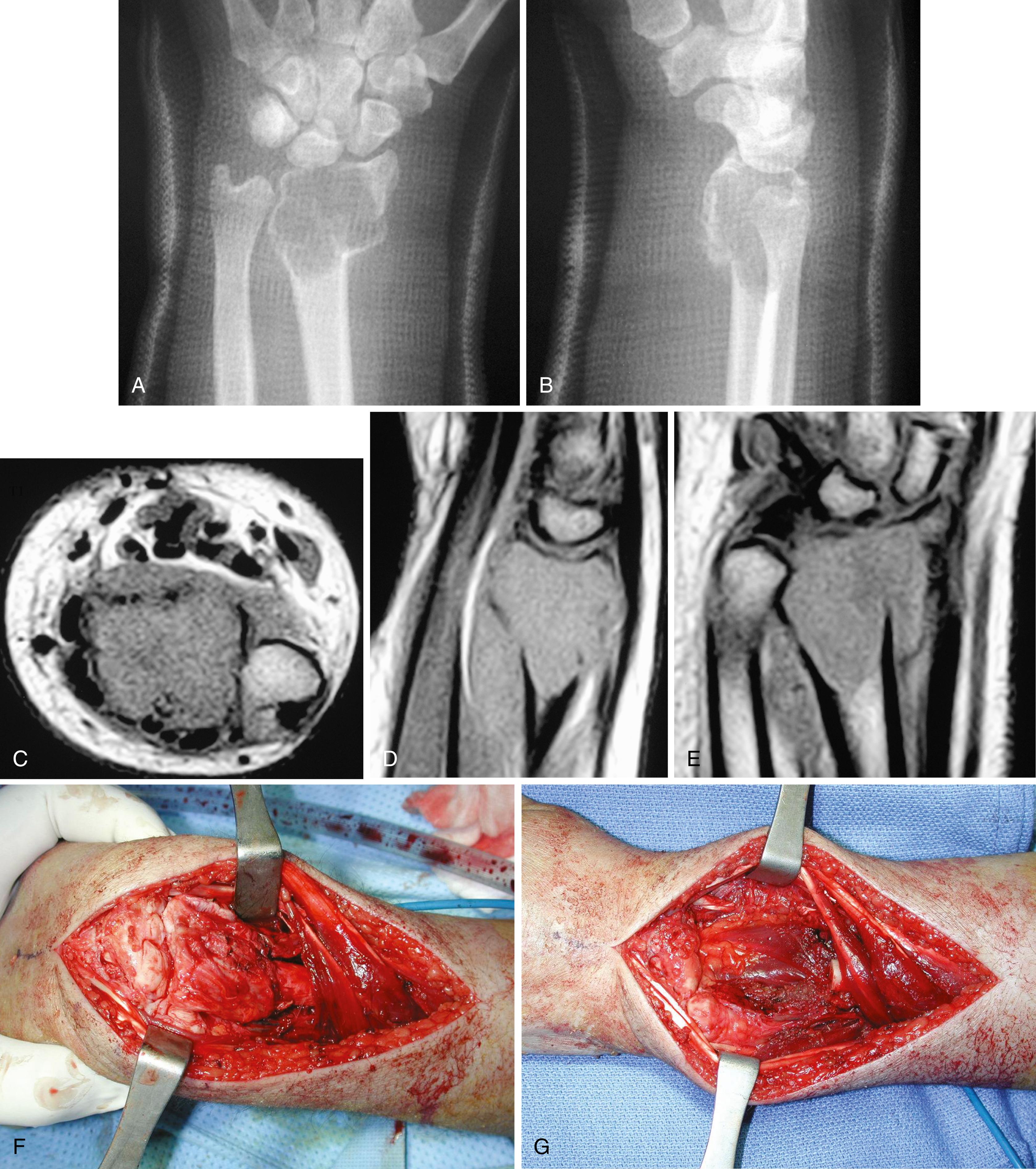
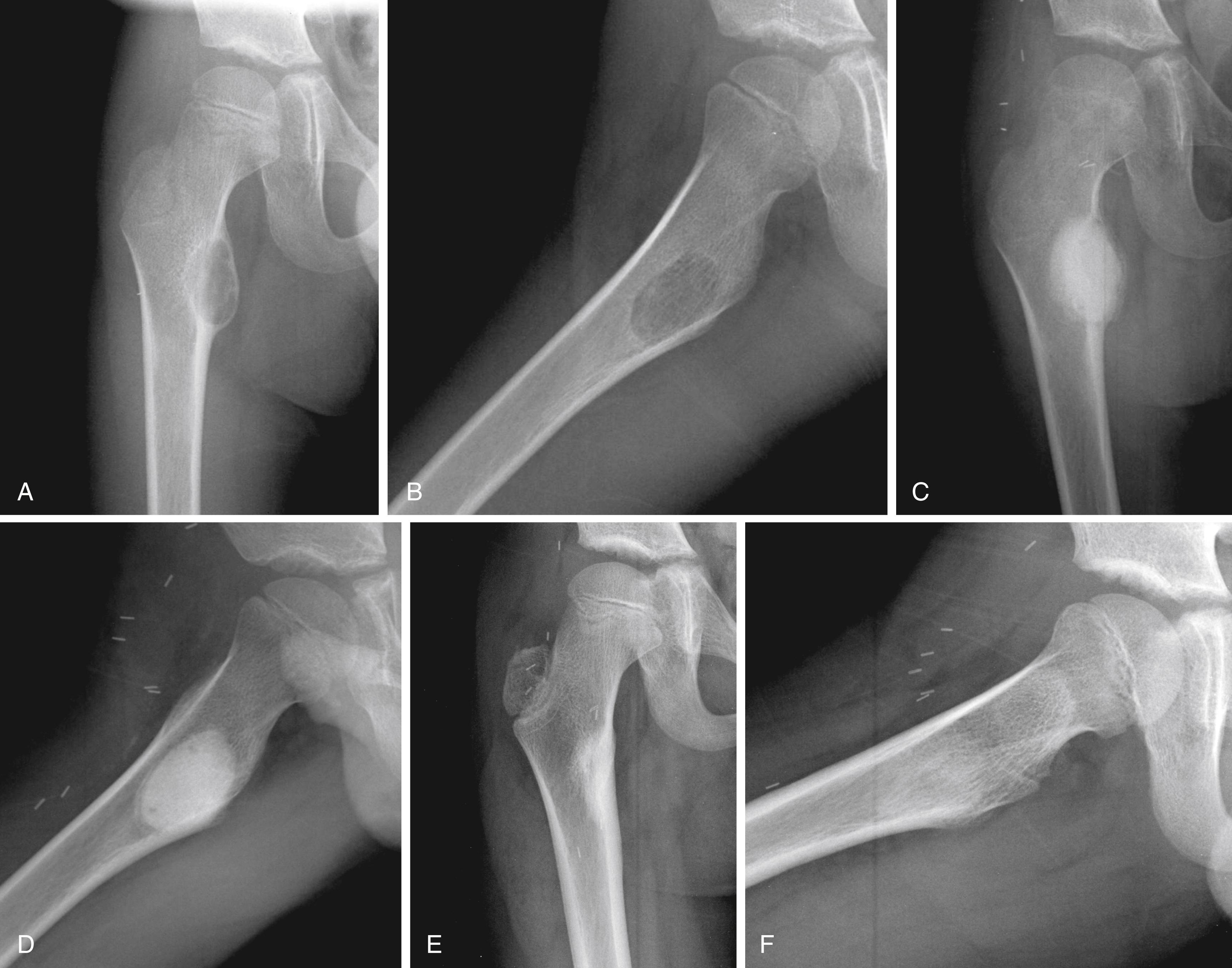
Radiographic findings usually are characteristic. This well-circumscribed lesion is usually centered in an epiphysis of a long bone; however, it may also be located in an apophysis, such as the greater tuberosity ( Fig. 26.6 ) or the greater trochanter ( Fig. 26.7 ). Often it has a surrounding rim of reactive bone ( Fig. 26.8 ), and 30% to 50% exhibit matrix calcification. CT can be helpful in detecting subtle areas of calcification that may or may not be detectable on plain radiographs. MRI frequently demonstrates abundant surrounding edema. Soft-tissue extension is extremely rare. In children, a well-circumscribed epiphyseal lesion that crosses an open physis is highly suggestive of chondroblastoma but could also represent an infectious process. For adults, differential diagnoses for an epiphyseal lesion include giant cell tumor and clear cell chondrosarcoma. In contrast to chondroblastomas, however, giant cell tumors usually do not have a rim of sclerotic bone or intralesional calcification and may have a soft-tissue component.
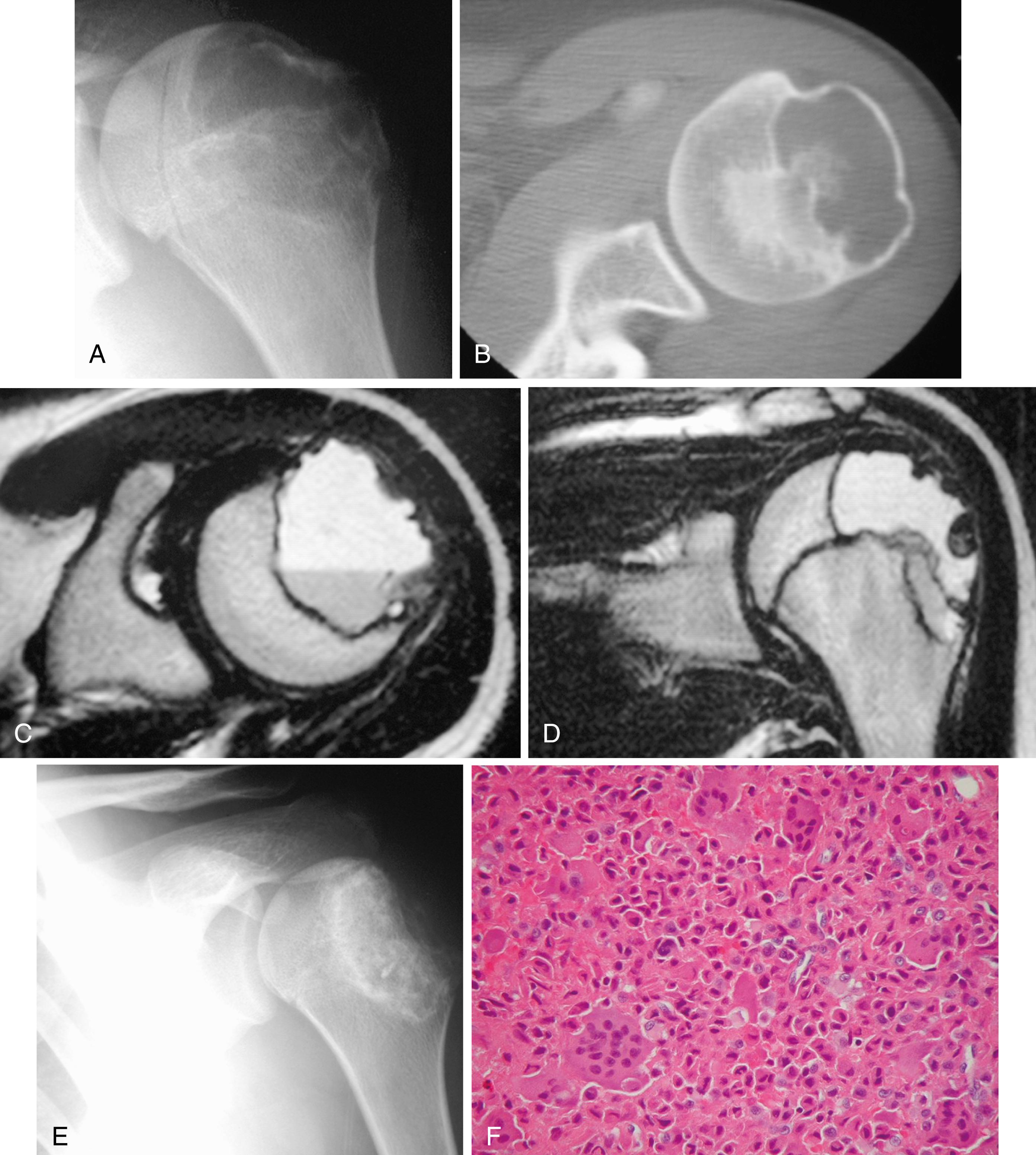
Microscopically, chondroblastoma consists of sheets of chondroblasts usually with a background of chondroid matrix. The cells are round to polygonal with distinct cytoplasmic outlines. Dystrophic calcification is frequently present and may surround individual cells, giving the classic “chicken wire” appearance. Multinucleated giant cells are abundant, and secondary aneurysmal bone cysts are present in 20% of patients. Histologic grading is of no prognostic significance.
Chondroblastomas usually present as stage 2 and, more rarely, as stage 3 lesions. Although they typically are not as aggressive as giant cell tumors, surgical management is warranted for almost all chondroblastomas owing to the slow progressive nature of the disease. Following biopsy to establish the diagnosis, treatment consists of extended curettage and bone grafting or placement of bone cement. This technique is associated with high chance of joint preservation and good functional outcomes. Adequate curettage always should take precedence over sparing the physis ( Fig. 26.9 ). In cases where physeal growth arrest has been reported, it is not known whether the insult is from the chondroblastoma itself or the surgical intervention.
Radiographs of the primary site and of the chest should be obtained every 6 months for at least 3 years and annually thereafter. Recurrence occurs in 10% to 20% of patients and can be treated similar to a primary lesion. Benign pulmonary metastases occur in approximately 1% of patients and should be treated by resection. Malignant transformation of a chondroblastoma is extremely rare.
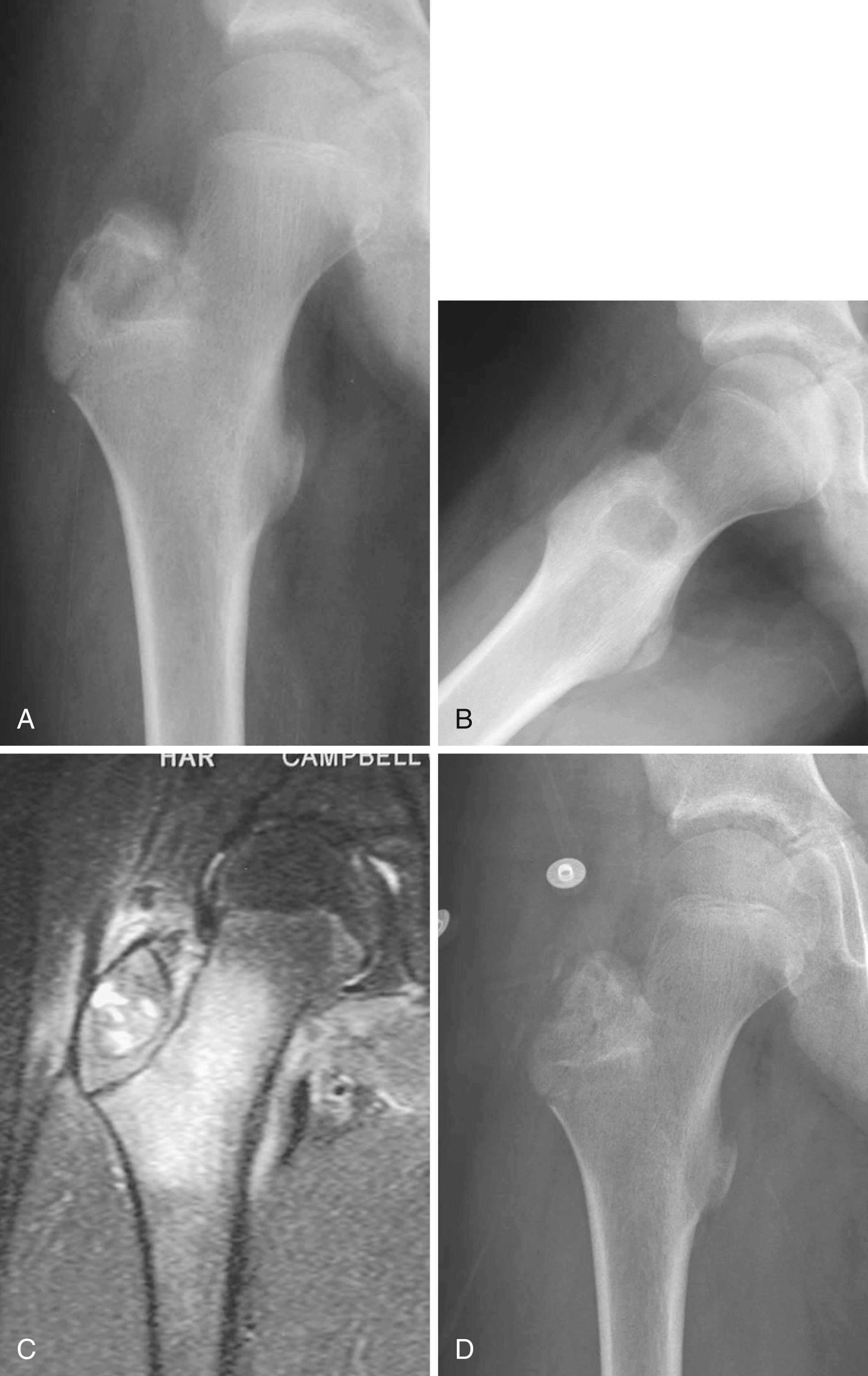
Chondromyxoid fibroma is a rare lesion of cartilaginous origin, representing less than 0.5% of all bone tumors according to the Mayo Clinic series. Although chondromyxoid fibromas may occur at any age, most occur in patients 10 to 30 years old. Any bone may be involved, but the proximal tibia is the most common location ( Figs. 26.10 and 26.11 ). Although patients typically complain of pain, if the tumor is in the hands or feet, a painless mass or swelling may be the chief complaint.
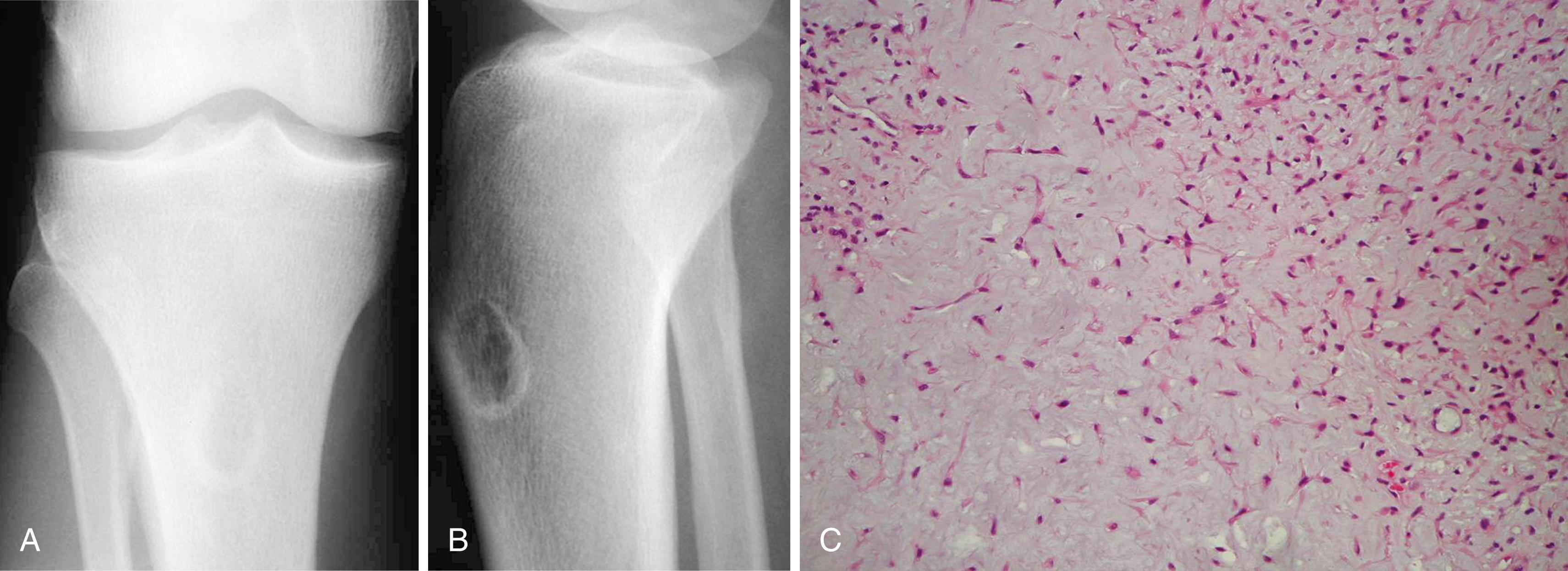
The radiographic appearance is that of a benign neoplasm. It usually is a well-circumscribed lesion with a rim of sclerosis in the metaphysis of a long bone and may have a bubbly appearance mimicking a nonossifying fibroma. In contrast to other cartilaginous lesions, radiographic evidence of intralesional calcification usually is absent (except in the rare instance of a surface lesion in which calcification may be abundant). Chondromyxoid fibroma rarely is included in the radiographic differential diagnosis of a lesion, unless it is in the proximal tibial metaphysis. Other diagnoses to include in the differential are chondrosarcoma, chondroblastoma, fibrous dysplasia, nonossifying fibroma, giant cell tumor, aneurysmal bone cyst, and simple bone cyst.
Microscopically, chondromyxoid fibroma appears lobulated. The center of the lobules contains loose myxoid tissue, and the periphery contains a more cellular fibrous tissue. The background often appears chondroid, although distinct areas of hyaline cartilage are rare. Microscopic calcification may be present. The lesion may contain areas with atypical pleomorphic hyperchromatic nuclei, but this should not lead to the erroneous diagnosis of chondrosarcoma if the lesion is otherwise radiographically and histologically consistent with chondromyxoid fibroma.
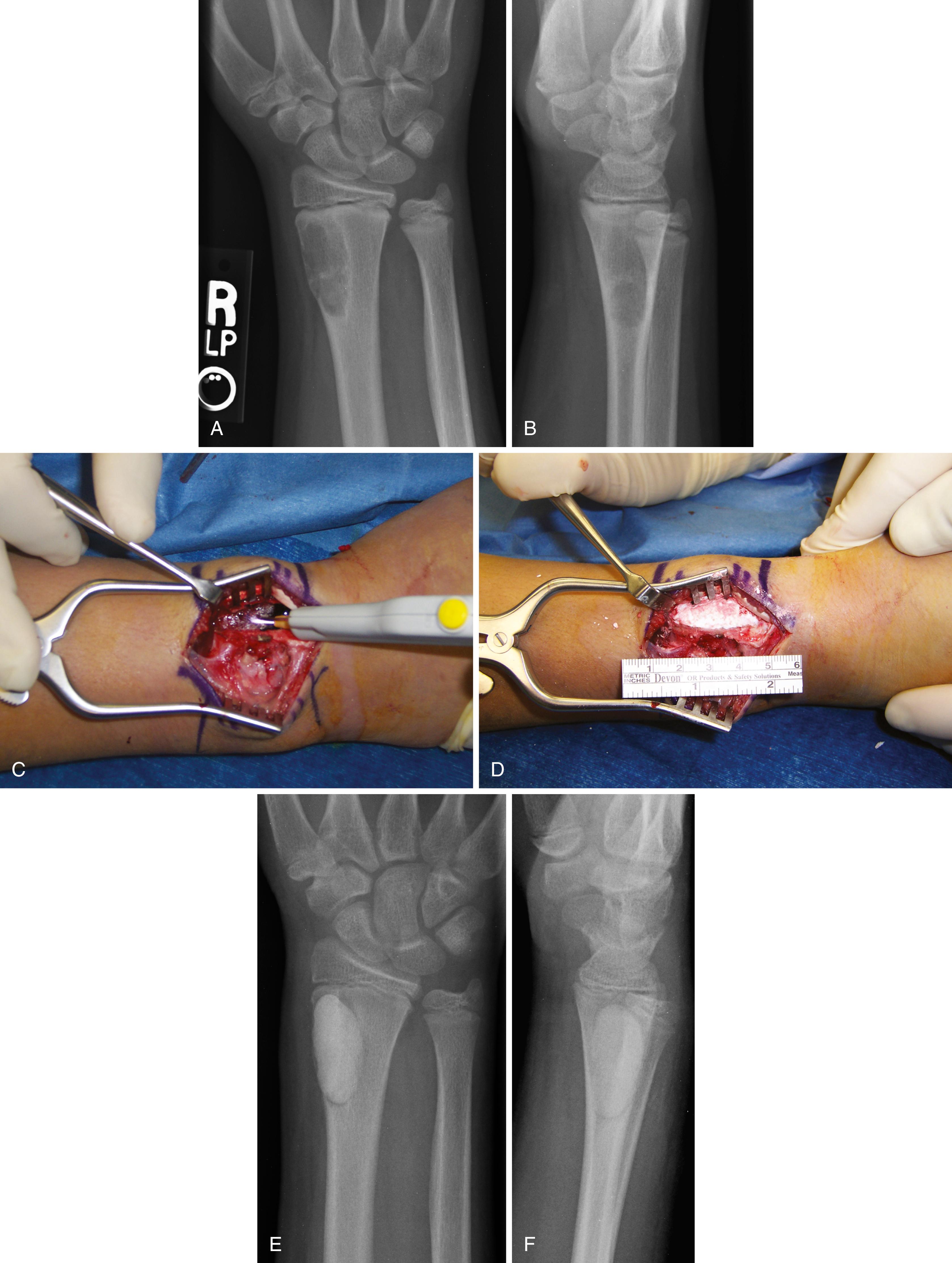
Treatment consists of resection or extended curettage with bone grafting ( Fig. 26.12 ). Wide resection is associated with a lower risk of recurrence than is intralesional curettage but results in a potentially larger functional deficit. Local recurrence occurs in about 20% of patients and is treated with repeat surgery. Malignant degeneration is rare.
Osteoblastoma is a rare bone-forming neoplasm that represents less than 1% of bone tumors in the Mayo Clinic series. Most patients with osteoblastoma are 10 to 30 years old. There is a male predominance of 3:1. Although any bone may be involved, 40% to 50% of the lesions are in the spine. Pain, which is the most common symptom, may be similar to that produced by an osteoid osteoma (i.e., worse at night and relieved by nonsteroidal antiinflammatory drugs). In the spine, painful scoliosis or neurologic deficit may be present. In the lumbar spine, signs and symptoms of nerve root compression may be evident, whereas in the thoracic spine, cord compression is more common. The tumor usually is slow growing, and symptoms may be present for 1 to 2 years before a diagnosis is made.
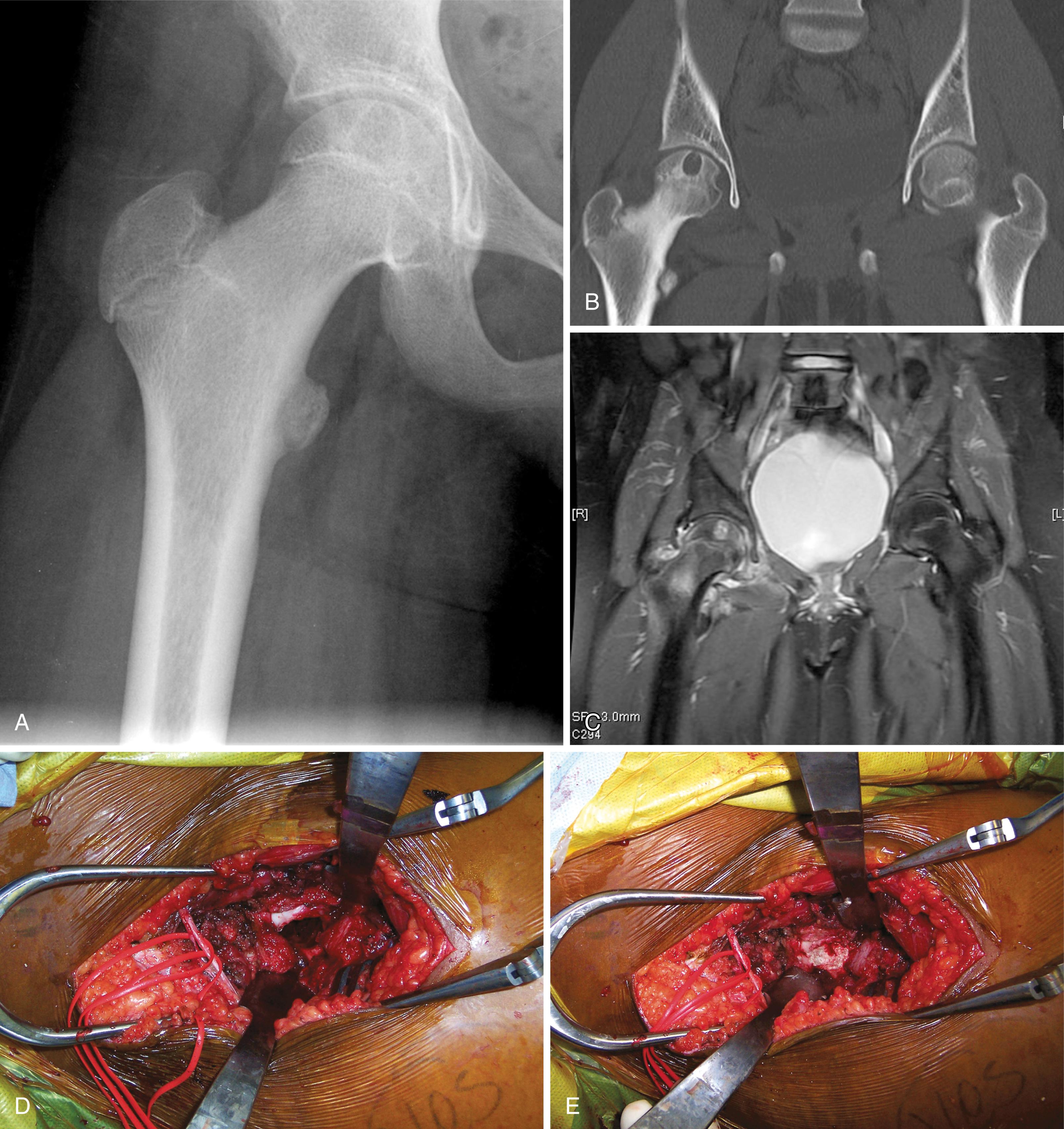
The most common radiographic appearance is that of a bone-forming neoplasm in the posterior elements of the spine in a young patient ( Figs. 26.13 and 26.14 ). The differential diagnoses include aneurysmal bone cyst and osteoid osteoma. Outside the spine, however, the radiographic appearance rarely reveals the diagnosis. The classic appearance of a mineralized central nidus with a surrounding radiolucent halo and reactive sclerosis is seen only occasionally ( Fig. 26.15 ), in which case the differentiation of osteoblastoma from the more common osteoid osteoma is based on size because the nidus of an osteoid osteoma is less than 1.5 cm. More often, the radiographic appearance is nonspecific. The lesion may be purely radiolucent ( Fig. 26.16 ), sclerotic, or mixed. Lesions may be diaphyseal or metaphyseal, and they may be primarily cortical or intramedullary. In some cases, they may have a frankly malignant radiographic appearance. Extension into the soft tissue is rare except in the spine, where soft-tissue extension is common.
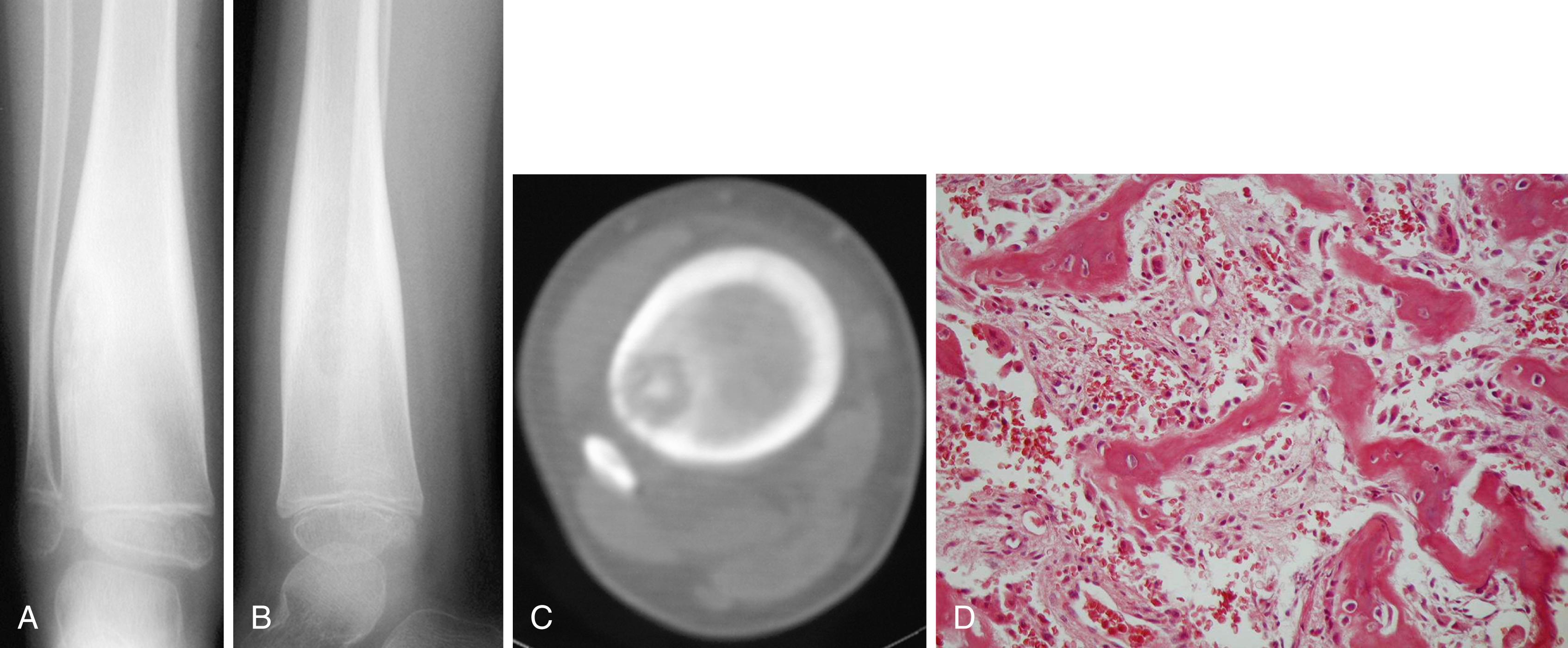
Microscopically, the lesion resembles an osteoid osteoma. It contains a fibrovascular stroma with the production of osteoid and primitive woven bone. Bony trabeculae are lined by a single layer of osteoblasts, which may be important in differentiating osteoblastoma from osteosarcoma. Other features favoring an osteoblastoma include sharp circumscription and a loose arrangement of the tissue. Features favoring a malignant diagnosis include permeation of surrounding tissue and sheets of osteoblasts without bone production.
Treatment consists of extended curettage or resection. Bone grafting of the defect may be necessary. In the spine, instrumented fusion may be necessary if resection causes instability. Some authors recommend adjuvant radiation therapy for spinal lesions because revision surgery for recurrences in this area is difficult. Other authors have noted that some incompletely removed lesions subsequently have remained quiescent. Most authors do not recommend radiation therapy unless absolutely necessary for symptomatic inoperable lesions.
Sarcomatous degeneration has been reported and may be more common in lesions previously treated with radiation. Many of these cases likely represent initial misdiagnoses of low-grade osteosarcomas. Regardless, it is apparent that some cases initially diagnosed as osteoblastoma have behaved aggressively later and occasionally have led to the death of the patients. As follow-up care, patients should have serial radiographs of the primary site and of the chest.
Langerhans cell histiocytosis (LCH), previously called histiocytosis X because of its unknown etiology, refers to a group of diseases with similar pathologic features. Because the disease may affect virtually any organ system in the body, there is a wide range of clinical presentations. Whether LCH represents a reactive or a neoplastic process is still a matter of debate. The term eosinophilic granuloma refers to isolated bone lesions, and Hand-Schüller-Christian disease classically refers to the clinical triad of skull lesions, exophthalmos, and diabetes insipidus. Currently, however, some authors use the term Hand-Schüller-Christian disease simply to refer to any case of multisystemic LCH. Letterer-Siwe disease, another variation, usually has its onset before 3 years of age and is characterized by fever, lymphadenopathy, hepatosplenomegaly, and multiple bone lesions. Letterer-Siwe disease frequently is rapidly fatal. Whether these terms represent different diseases or simply different manifestations of the same pathologic process is debatable. Because the pathogenesis is poorly understood, there is currently no optimal treatment protocol for multifocal involvement, and the reactivation rate is high. Diverse therapeutic modalities may be considered, depending on the organ system involved, including surgery, radiation, and chemotherapy. Risk factors for succumbing to LCH include multisystem involvement and diagnosis at an age younger than 2 years.
The orthopaedic surgeon is primarily concerned with eosinophilic granuloma of bone. Patients usually are 5 to 20 years old and usually have progressive pain. The clinical picture may be similar to that produced by osteomyelitis with pain at rest (and at night), fever, and local signs of inflammation. Any bone may be affected, but the most common locations are the vertebral bodies, the flat bones, and the diaphyses of long bones.
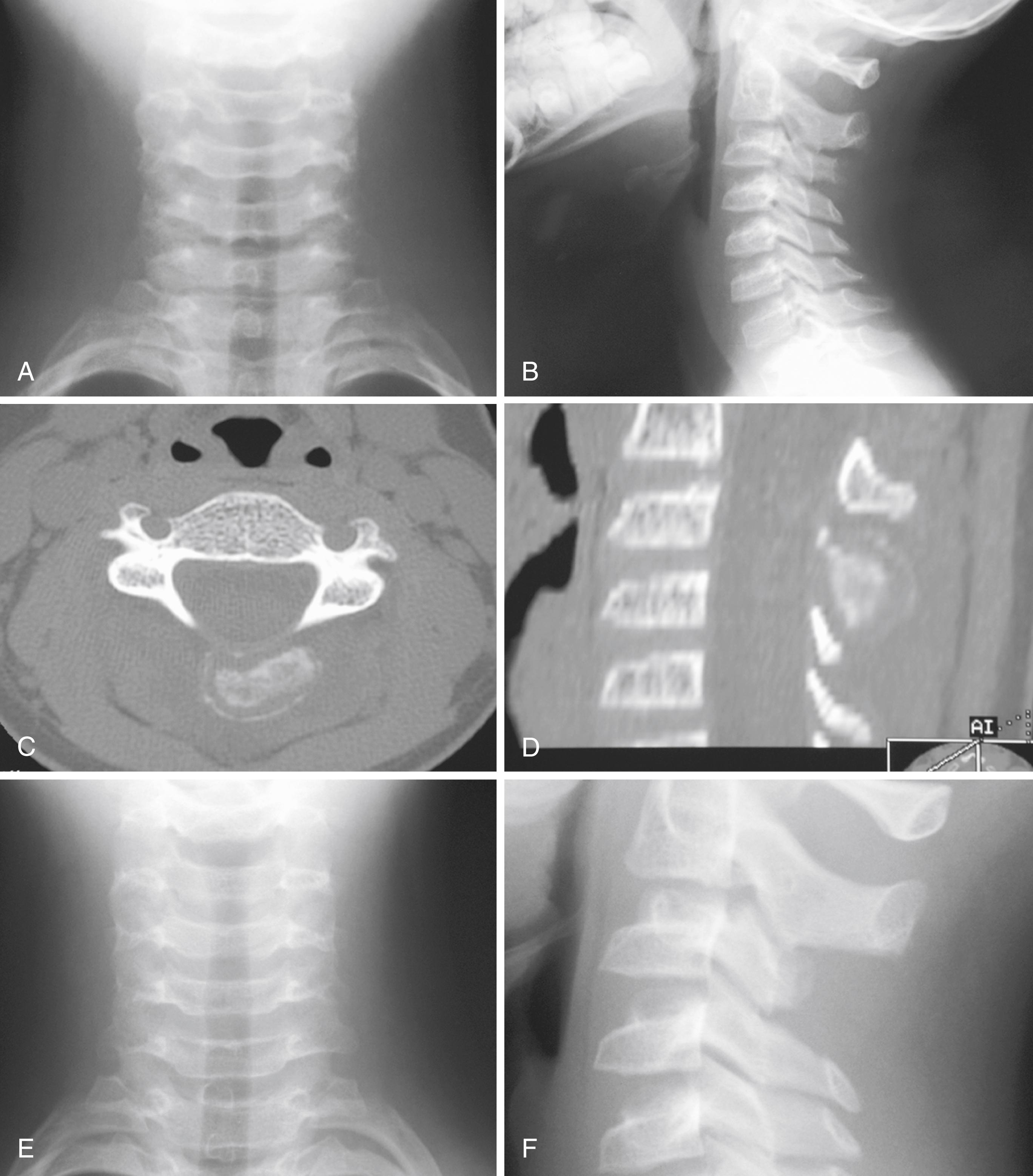
Radiographically, LCH can have various appearances. Marked flattening of the vertebral body, or vertebra plana, is a common manifestation. Although LCH is the most common cause of vertebra plana ( Fig. 26.17 ), other disease processes should be considered in the differential diagnosis if the clinical situation warrants. Other causes of vertebra plana include Ewing sarcoma, lymphoma, leukemia, Gaucher disease, aneurysmal bone cyst, and infection. In flat bones, the lesions usually are well circumscribed, “punched-out,” purely lytic lesions. The lesions may have a “hole within a hole” appearance because of different involvement of the two tables. In the diaphyses of long bones, the lesions may have an aggressive permeative appearance with periosteal reactive bone formation ( Fig. 26.18 ). This appearance may resemble Ewing sarcoma, infection, or lymphoma. A bone scan may help identify additional lesions, but approximately 30% of scans may be falsely negative. A skeletal survey is more effective for this purpose.
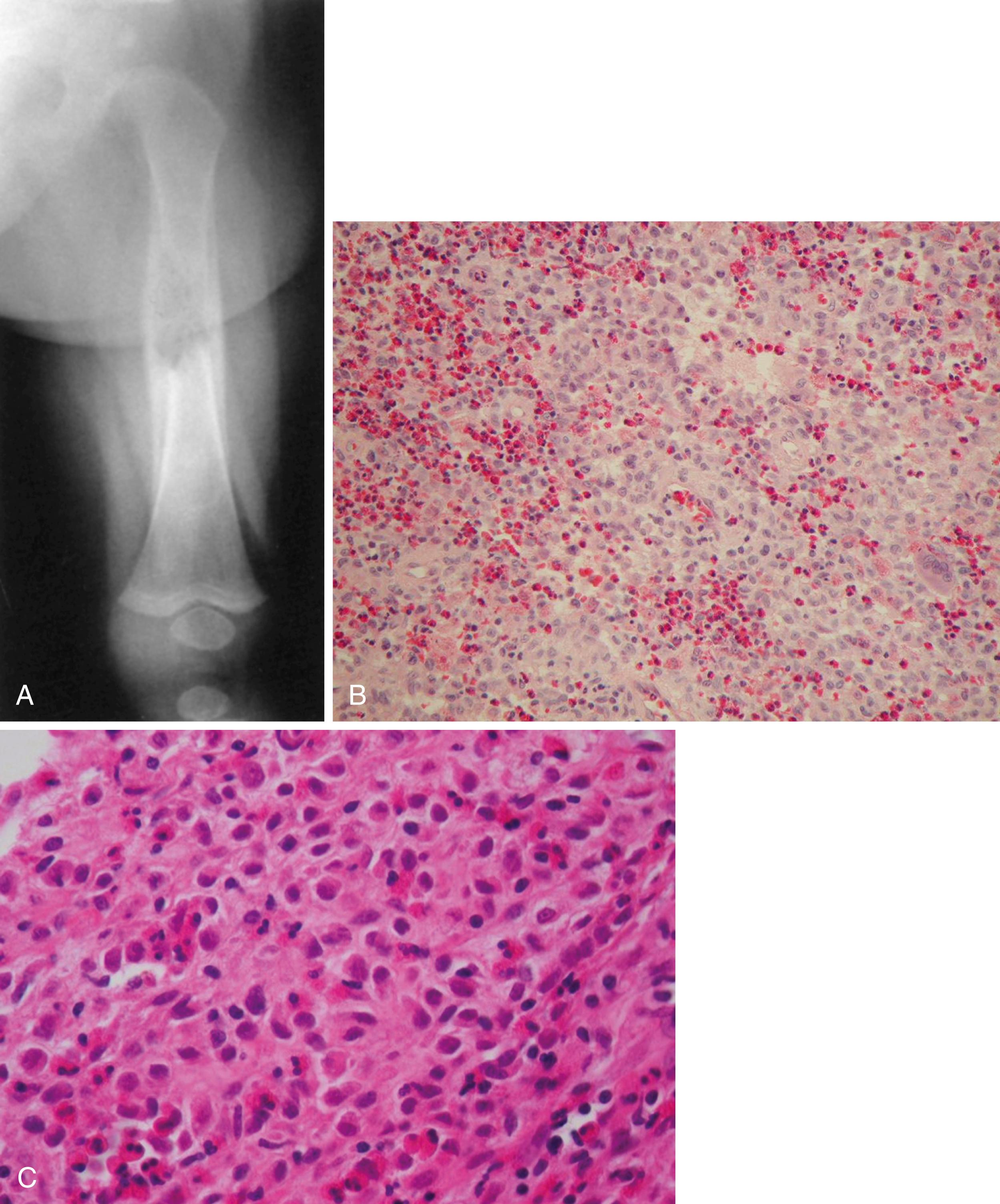
Microscopically, the diagnosis is made by the identification of Langerhans cells. The Langerhans cell is a large histiocytic cell with an indented nucleus, a crisp nuclear membrane, and abundant eosinophilic cytoplasm. The cells stain positively for S-100 protein. The lesion also contains multinucleated giant cells and other inflammatory cells, including clusters of eosinophils. Electron microscopy may identify characteristic organelles in the Langerhans cell cytoplasm called Birbeck granules.
Biopsy is required to make the diagnosis. When the diagnosis is established, most of the orthopaedic manifestations of LCH can be treated conservatively. Simple skeletal lesions tend to resolve spontaneously over a period of months to years, and healing may be initiated from the biopsy itself. Other recommended treatments have included corticosteroid injections, radiation therapy, and curettage with or without bone grafting. A few case reports have suggested that intravenous zoledronic acid may help resolve pain in symptomatic lesions, but the long-term sequelae are not well defined. If a lesion is asymptomatic, no treatment is necessary because lesions have been noted to regress spontaneously. Indications for treatment include pain, restriction of mobility, impending pathologic fractures, and nerve compression (spinal or optic nerve). Intralesional injection of steroids is a relatively safe and effective treatment modality. Low-dose radiotherapy, however, should be reserved for involvement of spinal or optic nerve compression. If the diagnosis is established by open biopsy, the lesion can be curetted during the same procedure. Care must be taken, however, to rule out infection before placing corticosteroids or bone graft into a lesion.
Vertebra plana likewise may be treated conservatively because most lesions spontaneously regress. Vertebral height typically is partially restored with growth in skeletally immature patients. Temporary bracing may help relieve symptoms. Irradiation may be indicated for treatment of mild neurologic signs. Surgical decompression and fusion with instrumentation is indicated for rapidly progressive neurologic signs or cord compression that is not responsive to radiation therapy.
The overall prognosis for skeletal lesions is excellent with a very low rate of local recurrence and few complications. Systemic disease may be progressive, however, and may require chemotherapy.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here