Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Abnormal uterine bleeding affects up to one-third of reproductive-age women and accounts for a third of all outpatient gynecologic visits.
The Federation Internationale de Gynecologie et d’Obstetrique (FIGO) has created a classification of causes of abnormal uterine bleeding, PALM COEIN, which has helped to organize the approach to treatment.
Abnormal uterine bleeding is a common cause of anemia and can be acute or chronic.
The presence of anemia in a reproductive-age woman should prompt investigation of reproductive dysfunction.
The pathophysiology of abnormal uterine bleeding depends on the cause of the bleeding and this also dictates treatment options.
Abnormal uterine bleeding (AUB) affects up to one-third of reproductive-age women who are not pregnant and can occur in conjunction with the benign pathologic processes discussed later in this chapter (fibroids, adenomyosis, endometrial polyps), endometrial hyperplasia or cancer, or in the absence of specific pathologies. AUB accounts for around one-third of all outpatient gynecologic visits. In the United States, AUB (previously called dysfunctional uterine bleeding) is commonly equated with anovulatory bleeding, whereas in Europe it is a diagnosis of exclusion of excessive bleeding not due to demonstrable pelvic disease, complications of pregnancy, or systemic disease.
Etiologies are different for premenarchal, reproductive age, and postmenopausal patients. Premenarchal bleeding is typically caused by immaturity of the gonadal axis, infection, foreign body, gynecologic malignancy, or trauma and is beyond the scope of this section. In reproductive-age women, the possibility of pregnancy must always be considered. All patients with postmenopausal bleeding should undergo evaluation for endometrial cancer. This section will focus on bleeding in nonpregnant women who are menstruating, the traditional definition of AUB.
There has been a push towards changing and standardizing terminology for AUB as confusing and conflicting terminology has made research collaborations and interpretation of clinical trials difficult. , Terms like “menorrhagia,” “metrorrhagia,” and “dysfunctional uterine bleeding” are now less favored than heavy menstrual bleeding or irregular menstrual bleeding. To standardize communication, the Federation Internationale de Gynecologie et d’Obstetrique (FIGO) created a universal classification of causes of abnormal uterine bleeding for women in their reproductive years and updated the classification in 2018. Normal frequency of menses is defined as 24 to 38 days; the normal duration of menses is less than or equal to 8 days. Regularity is defined as less than or equal to 7 days difference for patients aged 26 to 41 or less than or equal to 9 days for patients 18-25 years of age or 42 to 45 years of age. Heavy menstrual bleeding is no longer defined by a quantitative amount, but rather by any amount of bleeding that interferes with a woman’s quality of life.
FIGO’s “PALM-COEIN” nomenclature for AUB has four categories that are defined by structural criteria. These are bleeding due to P olyps, A denomyosis, L eiomyomas and M alignancy or hyperplasia, known by the acronym PALM. The other four are related to nonstructural abnormalities and include bleeding due to C oagulopathy, O vulatory disorders, E ndometrial causes, I atrogenic, and pathologies N ot otherwise classified, termed COEIN (pronounced like coin). It is hoped that this classification system should facilitate collaborations to study AUB; however, the ease of use has to be demonstrated. The three nonmalignant structural causes of AUB will be discussed in this chapter.
When considering abnormal uterine bleeding, it is important to determine and report the pattern and if it is heavy, cyclic, or intermenstrual. Heavy menstrual bleeding (HMB), defined by a large amount or long duration of bleeding, may occur in 10% to 30% of women and in up to 50% of women in perimenopause. , HMB is the type of abnormal bleeding frequently associated with benign uterine pathology, including uterine fibroids, adenomyosis, Cesarean scar defect, or bleeding disorder. Other potential etiologies include endometritis, pelvic inflammatory disease (PID), levonorgestrel-releasing intrauterine devices (IUDs), and uterine arteriovenous malformations. , HMB occurs with menses and may be associated with anemia. When considering etiology, it is also important to consider whether the bleeding is intermenstrual, which is often lighter bleeding and may be associated with endometrial polyps, malignancy, contraception, endometritis, or PID. Noncyclic bleeding is often associated with anovulation and may be heavy or light depending on the etiology of anovulation, as described below. If there is a change in pattern or amount of bleeding, or if bleeding occurs less than every 24 days, it is important to rule out malignancy, especially in patients over 45 or younger if there are risk factors (obesity and chronic anovulation).
Around 13% of women with HMB have some form of coagulopathy. Systemic disease as a cause of HMB should especially be suspected in adolescents where coagulopathies including thrombocytopenia, von Willebrand disease, or other coagulopathies may be the underlying cause. , Additionally, coagulopathies in the presence of structural abnormalities can synergize resulting in a severe phenotype of HMB.
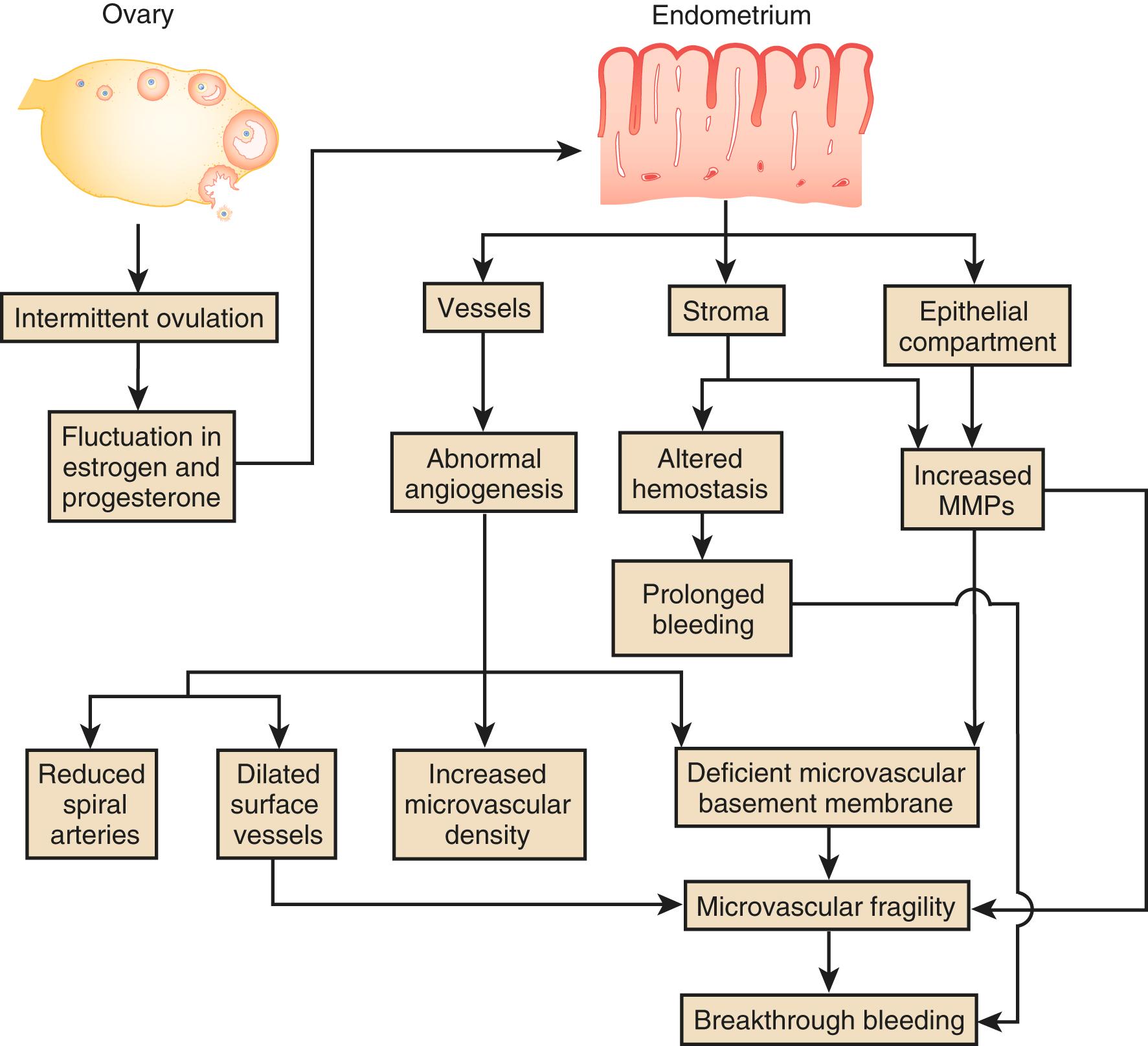
A variety of ovulatory disorders can lead to AUB. Anovulatory irregular uterine bleeding is characterized by irregular and prolonged bleeding secondary to disturbances in the hypothalamic-pituitary-ovarian axis and often occurs without molimina. It is most common in the extremes of reproductive life and in association with polycystic ovary syndrome. The unopposed action of estrogen on the uterus, resulting in dilated veins and the lack of suppression of spiral arteriole development, may represent the underlying pathophysiology. , , Large, thin-walled tortuous vessels can be demonstrated on the surface of the hyperplastic endometrium. Unopposed estrogen reduces vascular tone either through direct effects of estrogen on vascular smooth muscle cells or increased production of nitric oxide, leading to vasodilatation. The endometrium often breaks down unevenly in these circumstances. Scattered patches of thrombotic foci and necrotic degeneration are found adjacent to abnormally proliferated endometrium. This uneven endometrial breakdown often leads to noncyclic and HMB may also be seen after a pregnancy loss or after delivery.
Ovulatory irregular uterine bleeding is characterized by regular episodes of heavy menstrual flow, usually with the heaviest loss during the first three days of menstruation. Though many ovulatory disorders can be traced back to endocrinopathies, the underlying abnormality appears to be defects in processes that regulate the loss of blood during menstruation, primarily angiogenesis, vasoconstriction, and hemostasis. In contrast to anovulatory irregular uterine bleeding, the surface vessels of the endometrium appear to be grossly normal and only minor abnormalities have been described in endometrial and myometrial veins like venule ectasia. Thyroid function and pituitary function should be evaluated as treating these etiologies may result in the resumption of normal ovulation and cyclic bleeding patterns. Obesity has also been linked to irregular uterine bleeding as adipose tissue produces estrone, which causes endometrial proliferation.
Endometrial causes of AUB are similar in pathogenesis to ovulatory disorders. Hypoxia, inflammation, hemostasis, and angiogenesis all play critical roles in the shedding and subsequent scarless repair of the functional upper layer of the endometrium. A recent publication from a murine model showed that hypoxia-inducible factor 1 alpha (HIF-1α) plays an important role in endometrial repair and may be a target for therapies in women with heavy menstrual bleeding. Perturbation of local glucocorticoid metabolism, aberrant prostaglandin synthesis, and excessive plasminogen have all been implicated in AUB-E. , The disruption of local endometrial hemostasis processes can be due to deficient production of vasoconstrictors like endothelin-1 and prostaglandin F2α, increased production of vasodilators such as prostaglandin E2 and prostacyclin, and excessive breakdown of clot in the endometrium by abnormal production of plasminogen activator. In addition, vascular smooth muscle cell proliferation is reduced in the spiral arterioles in the mid- and late secretory stages in women with menorrhagia, possibly contributing to vessel instability. Endometrial bleeding-associated factor (EBAF) (a.k.a. TGF-β4 ), a member of the TGF-β family of growth factors, suppresses production of collagen and promotes expression of collagenolytic and elastinolytic enzymes by antagonizing the normal signaling pathway activated by TGF-β growth factors. Abnormal expression of EBAF , which occurs only in the late secretory and menstrual phases of a normal cycle, has been reported in the endometrium of women with HMB. Angiopoietin 1 &2 ( Ang-1 and Ang-2 ) may also be involved in the pathogenesis of HMB. Ang-1 promotes vascular maturation, while Ang-2 destabilizes vessels and initiates neovascularization. An altered ratio of Ang-1 to Ang-2 in the endometrium due to downregulation of Ang-1 expression is also associated with HMB. ,
AUB due to iatrogenic causes (AUB-I) usually includes the use of exogenous steroid therapy in form of combined estrogen and progestin pill, patch or ring, subdermal implant, and levonorgestrel-releasing intrauterine device or in nonmedical forms (supplements or cosmetics). AUB-I also includes bleeding due to medications that cause coagulation disorders and ovulatory dysfunction. Bleeding secondary to iatrogenic causes may be treated in the short term with doxycycline as it has antiinflammatory properties on the endometrium or addition of norethindrone acetate or medroxyprogesterone acetate. The “not classified” category (AUB-N) includes a number of entities that do not fall into any group; for example, AUB due to uterine arteriovenous malformation. Furthermore, this category has been left for new entities that may cause AUB that have not been yet discovered.
It is important to note that a woman can have AUB due to more than one cause; for example, a woman with HMB due to submucosal fibroid (AUB-L) can also have concomitant anovulation if she has PCOS and would have AUB-O type of bleeding as well.
Evaluation for abnormal uterine bleeding includes a thorough history outlining the bleeding patterns and amount as previously described, and physical examination to quantify the amount of current bleeding and identify any contributory findings such as an enlarged uterus, cervical polyp, vaginal mass or laceration, foreign body, or adnexal mass or tenderness. Laboratory workup may include a complete blood count, hCG to exclude pregnancy, thyroid function tests, prolactin level, FSH, estradiol, androgen levels, coagulation testing, and infectious disease testing. Endometrial sampling may be warranted, as discussed previously. Pelvic ultrasound, saline infusion sonogram, or hysteroscopy may be warranted to evaluate the anatomy of the uterus depending on availability of office evaluation.
These pathophysiological mechanisms proposed to underlie irregular uterine bleeding are targets for novel therapeutic interventions ( Fig. 26.1 ). The use of nonhormonal oral antifibrinolytic medication called tranexamic acid has shown a 66% response rate for HMB and has shown to be extremely helpful in patients with AUB. , While tranexamic acid has been widely used internationally for many years, its recent FDA approval is leading to increasing use in the United States. To choose an appropriate treatment, it is important to identify the etiology of abnormal uterine bleeding. Although it is often chronic, it can acutely worsen or reach a threshold requiring emergent evaluation.
Acutely worsening bleeding or prolonged chronic unrecognized blood loss can result in anemia and may require immediate and long-term treatment. In this case, high-dose estrogen may be used to treat bleeding in the acute phase. Historically, intravenous administration of equine estrogens was used; currently, daily use of multiple oral contraceptive (OC) tablets is more common. Estrogen allows the endometrial lining to grow over the denuded basalis and cover the friable bleeding surface. An antiemetic may need to be given with high-dose estrogen. Tranexamic may also be used in this context. Many patients have relative contraindications to the use of high-dose estrogen, as they increase the risk of venous thromboembolism and stroke. Contraindications may include age older than 35, tobacco use, history of thromboembolism, or obesity. In these cases, high-dose progestins such as depot medroxyprogesterone acetate or norethindrone can be used in the acute phase.
Nonacute options include the use of contraceptive steroids in the form of pill, ring, patch, or progestin-containing IUDs and oral GnRH antagonists (discussed in detail in the leiomyoma section). Selective progesterone receptor modulators, where they are available, can be used to suppress endometrial growth and stabilize the endometrial vasculature; MMP inhibitors to prevent extracellular matrix catabolism, including the matrix of the vessel walls; and NSAIDs or selective COX-2 inhibitors to suppress prostanoid synthesis. In select cases, GnRH agonists can be used to suppress menses for a limited period of time but are not used long term due to hypoestrogenic side effects.
For the patient who does not desire fertility, surgical destruction or removal of endometrium via endometrial ablation with newer nonresectoscopic, operator-friendly devices has shown impressive success rates and are also shown to be safe. Of note, endometrial ablation failure requiring retreatment or hysterectomy for pain is more likely in patients younger than 45, with parity greater than 5, prior tubal ligation, and history of dysmenorrhea. Additionally, endometrial ablation does not function as a contraceptive, so both extrauterine and abnormal intrauterine pregnancies are possible. Hysterectomy is a definitive but less desirable option because of its invasiveness and long-term sequelae for AUB patients that are not concerned about fertility preservation.
Uterine leiomyomas are very common benign clonal smooth muscle cell tumors that have increased incidence, severity, and burden in Black women.
Gonadal steroids, growth factors, stem cells, and key driver mutations play a role in the pathogenesis and growth of uterine leiomyomas.
Therapeutic options for uterine leiomyomas can range between observational, medical, interventional, or surgical management. Despite multiple options, there is little evidence to guide treatment.
The introduction of oral GnRH antagonists with steroidal add-back provides the promise of long-term medical therapy for leiomyomas.
Since fibroids are the leading indication for hysterectomy, new data that there are long-term health risks to hysterectomy even with ovarian conservation will likely guide future treatment ( Fig. 26.2 ).
Uterine leiomyomas, also termed myomas or fibroids, are benign clonal smooth muscle cell neoplasms clinically appreciated in approximately 25% of all women. Black women appear to have a three-fold increased incidence and relative risk. , A systematic review examining epidemiologic risk factors for uterine fibroids showed that in prospective cohort studies Black race was the only ubiquitous risk factor with 1.69 or higher risk than White women. There is mixed data regarding fibroid risk in Latina women compared to other races. , , Careful pathologic study of surgical specimens suggests that > 80% of Black and 70% of White women have detectable leiomyomas, which parallels the lifetime incidence of the clinical disease. , Thus, there appears to be little occult disease in Black women. This suggests that growth acceleration of transformed myocytes into clinical fibroids may be ubiquitous in this group. , Black women are not only significantly more likely to have leiomyomas than White women but are also likely to be younger at the time of diagnosis and hysterectomy. , They also have more severe diseases , , and are two to three times more likely to undergo hysterectomy for leiomyomas and six times more likely to require myomectomy , ( Table 26.1 ). There are also data showing regional differences that may reflect racial differences such as the increased risk of hysterectomy in the southern United States. Although most studies showing this racial disparity are from within the United States, a study out of South Africa also found a higher prevalence of hysterectomy for fibroids in Black women.
| Fibroid Characteristic | Black vs. Caucasian Women | Reference Number |
|---|---|---|
| Incidence of uterine fibroids | Threefold increase | 2 |
| Relative risk | Threefold increase | 2 |
| Age at diagnosis | 3-5 years younger | 8 |
| Severity of disease | Fivefold increase | 8 |
| Fibroid growth at older age (≥ 45 years) | Sevenfold to Eightfold increase | 57 |
| Myomectomy risk | Sixfold increase | 10 |
| Hysterectomy risk | Twofold to threefold increase | 11 |
Known risk factors do not adequately explain this racial disparity, with modifiable and genetic factors both thought to play a role. , Studies on Black women correlate fibroid risk to polycystic ovarian syndrome, history of physical and sexual abuse, self-reported experience of racism, diet, and environmental exposures. Understanding the unique genetic and environmental factors leading to increased risk for Black women is a key research agenda for leiomyomas.
Reproductive factors affect the risk of leiomyomas. Numerous studies have shown that parity is associated with a significantly decreased fibroid risk of up to 5 fold, while time greater than 5 years since last birth increases risk. , , , A family history of fibroids and increasing age up to the age of menopause are associated with increased fibroid risk. OCs and injectable progestin contraceptives appear to protect against clinically evident fibroids, thus making them doubly useful to the woman at risk for fibroids who desires contraception. , Recent work has proposed an increase in vitamin D levels in women using OCs as a mechanism for protection against leiomyomas because low vitamin D levels also appear related to fibroid risk. , Postmenopausal hormone replacement therapy (HRT) has been associated with a higher likelihood of having fibroids on pathology after surgery in one report, although this may have been due to bias, since HRT may have inhibited normal postmenopausal fibroid regression and/or resulted in bleeding that led to hysterectomy. Early menarche is also associated with an increased risk of developing fibroids and may partially explain earlier disease in Black women who have earlier menarche than White women.
Comorbidities, as well as environmental and dietary habits, also appear to influence the risk of myoma formation. Patients with obesity and hypertension have an increased incidence of fibroids. Smoking in women with a low BMI, which has an antiestrogenic effect, seems to also have a protective effect; however, other deleterious effects outweigh any benefit of smoking. , , Decreased vegetable and fruit intake, increased dietary long-chain omega-3 fatty acids, significant consumption of red meats, foods with a high glycemic index or preservatives, and soybean milk consumption as associated with increased relative risk of fibroids, while consumption of vitamin A decreases the risk of myomas. , Caffeine consumption and dietary carotenoids have not shown a difference in risk. Reduced dairy consumption, use of hair relaxer, and increased intake of alcohol (especially beer) appear to increase risk in Black women. , However, no one has demonstrated that dietary intervention leads to changes in fibroid incidence, symptomatology, or regression.
The pathogenesis of fibroids continues to be an area of study. Genetic changes (primarily somatic mutations but also including a rare but important germline mutation), gonadal steroids, stem cells, and growth factors all play a role in transformation, clonal expansion, and growth. Growth and regression are variable, with a median 9% increase in volume over 6 months (range −89% to +138%), and often higher rates in Black women.
Leiomyomas are monoclonal and each fibroid is an independent clonal event, although there are a variety of genetic pathways involved in fibroid pathogenesis. , Thus there can be multiple fibroid genotypes within the same uterus. Both polymorphisms of G6PD and the androgen receptor show this clonality. ,
While twin studies, , familial clustering, , and cytogenetic changes were the initial window into fibroid genetics, it is now believed there are four key driver mutations: the mediator complex subunit 12 (MED12) group, the high mobility AT-hook2 (HMG2) group , the fumarate hydratase (FH) group, and a rare group associated with deletion of collagen IV α5 (COL4A5) and COL4A6. , There are also rare fibroids that have none of these mutations. It also appears that chromothripsis, a global event causing multiple chromosome rearrangements at once, may also play a role in leiomyomas.
MED12 , which accounts for over 50% of fibroids in most series, is a transcriptional factor and a mediator of both global and specific gene transcription located on chromosome Xq13. , , MED12 was altered in 70% of tumors from 80 patients studied in this report from Scandinavia; pathway analysis suggested that ECM receptor interaction, Wingless family ( Wnt) signaling, and focal adhesion pathways were altered by this change. Whole genome sequencing has shown MED12 mutations to be frequently present in fibroids in American women of racially and ethnically diverse backgrounds, confirming its importance as a key molecule in fibroid pathobiology. Additionally, a murine model suggests that the MED12 mutation is a gain-of-function mutation sufficient to stimulate leiomyoma formation and recapitulate the genetic instability seen in the disease. There is increasing evidence that the downstream Wnt/β-catenin signaling pathway may also play a key role in fibroid pathogenesis. ,
High mobility group protein A2 ( HMGA2 , formerly called HMGI-C ) is an architectural transcription factor located on chromosome 12 involved in the pathogenesis of fibroids via activation of PLAG1 through (12;14) translocations. , , , The dysregulation of HMGA2 might be associated with fibroid growth by the increased expression of CDKN2A (which encodes ARK[p14]). Intact ARF (p14) maintains senescence in fibroids.
While rare, FH mutations are significant. They are most commonly autosomal dominant germline mutations, although somatic mutations have been reported. , The germline FH mutation is also the cause of a syndrome termed Hereditary Leiomyomatosis and Renal Cell Cancer (HLRCC) (Mendelian Inheritance in Man [MIM] 605839). Affected families manifest cutaneous leiomyomas and papillary renal cell carcinoma (RCC) with variable penetrance. It is currently uncertain whether affected women have an increased risk of uterine sarcomas or just unusual pathology that can be confused with malignant disease.
The uterine disease and RCC are atypical in their presentation compared to their sporadic counterparts; uterine fibroids can appear in young premenopausal women, and the papillary RCC is often metastatic at presentation and more likely to be seen in adult women. Two other syndromes had described only the association of cutaneous and uterine leiomyomas, but lessons from molecular genetics suggest these are incomplete forms of the HLRCC syndrome and should be of historical interest only. ,
FH is an enzyme that is part of the Krebs tricarboxylic acid cycle found at 1q 42-43. Germline mutations appear to result in absent or nonfunctional proteins, and thus, FH appears to act as a tumor suppressor. , Although work on elucidating the pathogenesis of HLRCC syndrome continues, FH mutations appear to induce a change toward a hypoxic phenotype. , Thus, the hypothesized relationship between hypoxia and myoma pathogenesis appears linked to this subset of leiomyomas. Finally, immunohistochemistry looking for both absent FH staining and increased staining for succinate dehydrogenase (SDH)—the substrate of the FH enzyme—have raised suspicion of this syndrome based on pathologic exam of uterine and cutaneous leiomyomas and broadening our understanding of the phenotypic spectrum.
Identifying women at higher risk of malignancy due to HLRCC syndrome is just as important as identifying women whose families carry the BReast CAncer gene (BRCA) mutations or have Lynch syndrome. However, in the future, individualized therapy will likely be possible based on genotype and underlying predisposition genes.
Finally, deletions of collagen genes COL4A5 and COL4A6 are also shown to be associated with a familial syndrome known as diffuse leiomyomatosis with Alport syndrome and, rarely, with nonsyndromic fibroids. There may be a synergistic effect between the inactivation of COL4A5, COL4A6, and insulin receptor substrate 4 (IRS4) underlying the pathogenesis of Alport syndrome.
The mutation of tumor repressor REST and activation of the mTOR signaling pathway may also play a role. The Eker Rat model for leiomyomas has a germline defect in the tuberous sclerosis complex 2 ( Tsc-2 ) tumor suppressor gene. Tsc-2 also activates mTOR, which makes the latter a particularly interesting gene in the pathogenesis of leiomyomas. ,
Epidermal growth factor (EFG) is a growth factor mitogenic for smooth muscle cells and EGF mRNA is upregulated in leiomyomas only in the secretory phase of the cycle. , Receptor levels appear to be similar in leiomyomas and myometrium. The latest work concentrates on the role of nicotinamide adenine dinucleotide phosphate (NADPH) oxidase-derived reactive oxygen species (ROS) for signaling EGF and platelet-derived growth factor (PDGF) signaling pathway, leading to myoma cell proliferation.
Heparin-binding growth factors are important biological regulators in leiomyomas since they can be secreted and bound to the reservoir of heparin sulfate proteoglycans, filling the leiomyomatous ECM. Heparin-binding epidermal growth factor ( HBEGF ), vascular endothelial growth factor ( VEGF ), platelet-derived growth factor ( PDGF ), hepatoma-derived growth factor ( HDGF ), and the previously described basic FGF are all found in myomas. Many have also been documented to be stored in the ECM.
Insulin-like growth factors (IGFs) can act as smooth muscle cell mitogens and were originally shown to have increased binding to leiomyomas compared to myometrium. However, assessment of mRNA levels suggests that gene expression differed among studies. Later studies suggested specific modulation of the IGF-binding proteins. Recent work has shown the role of activation of tyrosine kinases and especially the IGF-1 signaling pathway in fibroids. , Regulation of these factors following GnRH-agonist treatment has also been reported. There may also be increased prevalence of leiomyomas in women with acromegaly.
Prolactin also appears to play an important role in myoma pathogenesis. In vitro studies suggest that it is mitogenic for leiomyoma and myometrial smooth muscle cells and that the prolactin receptor is present in these tissues, setting up an autocrine or local endocrine system. Additionally, agents that appear to cause clinical regression of uterine leiomyoma also appear to decrease prolactin production in vitro . ,
The resident immune cells also appear to influence leiomyoma biology. Mast cells have been implicated in leiomyoma pathobiology given that they are generally uniformly distributed in myometrium but highly variable in leiomyomas. , Recent work has suggested a correlation of mast cell number with vasculature. A number of cytokines have also been shown to be differentially regulated in leiomyomas and myometrium. Interleukin 8 (IL8) has decreased expression of both the ligand and its receptor in myometrium compared to leiomyomas. The functional significance of this is shown by the fact that neutralizing antibodies to IL8 decreases cellular proliferation in vitro . Monocyte chemotactic protein-1 (mcp1) is largely undetectable in normal samples of leiomyoma and myometrium but is increased significantly following GnRH-agonist therapy. ,
Wnt 7a, the human homolog of the wingless Drosophila genes involved in anteroposterior (AP) axis formation and smooth muscle cell patterning, appears to be suppressed in leiomyomas compared to normal myometrium and to be inversely related to ER-α expression. In contrast, secreted frizzled-related protein 1 ( sFRP1 ), a modulator of Wnt signaling, is increased in leiomyomas (particularly in the late proliferative phase) and increased by estradiol treatment and hypoxia. HOX gene expression does not appear to differ between leiomyomas and myometrium. The mRNA for proto-oncogenes cfos and cjun are also overexpressed in leiomyomas compared to normal myometrium.
Parathyroid hormone-related peptide (PTHrP) mRNA is also overexpressed in leiomyomas compared with normal myometrium. , Serum overexpression of this protein originating from a fibroid simulating the hypercalcemia of malignancy has been reported in the literature.
Micro-RNAs (mi-RNA) are small noncoding RNAs that generally inhibit gene expression and appear to have a key role in leiomyoma pathogenesis. While early studies showed there was differential expression of specific miRNAs between leiomyomas and normal myometrium and association of key miRNAs with leiomyoma size and patient race, more recent studies have started to define the key regulatory pathways they influence.
There are substantial in vitro data supporting major roles for both estrogen and progesterone in the biology of uterine leiomyomas, with progesterone being the most influential. The role of progesterone on myoma growth has moved beyond the simplistic concept of increasing mitosis to include inhibiting the apoptosis pathway via β-cell lymphoma 2 (Bcl 2) induction. The apoptotic inhibitor Bcl2 is present in leiomyomas but is largely undetectable in the myometrium. The role of Kruppel-like transcription factor 11 ( KLF11 ) in integrating progesterone-mediated myoma cell signaling and proliferation has been shown. , Likewise, regarding estrogen action, local action is likely key via upregulation of the enzyme aromatase P450 and its gene CYP19, expressed in higher concentrations in African American patients . Other elements of estrogenic response in myomas can also come into play as myoma cells have a modest increase in type I isotype of 17β hydroxysteroid dehydrogenase .
Modulation of steroid receptors is also important. Leiomyomas have increased amounts of both estrogen and progesterone receptor (ER and PR) messenger ribonucleic acid (mRNA) compared with normal myometrial tissue. Both the A- and the N-terminally truncated B-isoforms of the PR appear to be present in both leiomyomas and myometrium; however, the A-isoform predominates. , Similarly, ER-α rather than ER-β appears to be the predominant form in leiomyomas.
In addition to the direct action of ovarian steroids on the uterus, it is possible that the reproductive axis may also influence uterine metabolism through the direct action of pituitary gonadotropins on the uterus. Gonadotropin-releasing hormone (GnRH), which is clinically used to reduce myoma size, abolishes gene expression differences between normal myometrium and myomas. The placental glycoprotein chorionic gonadotropin (hCG) has been shown by several laboratories to have direct actions on myometrial metabolism. Additionally, work has shown that follicle-stimulating hormone (FSH), luteinizing hormone (LH), thyroid-stimulating hormone (TSH), and their common α-subunit can all have stimulatory effects on uterine prolactin production. , There appears to be a variant LH/hCG receptor present in human uterine tissue that may modulate this action. , LH has also been associated with myoma formation but not growth independent of the patient’s age. Genome-wide microarray studies have shown a strong role of glucocorticoids in the pathogenesis of fibroids. There are new data to suggest that the Nr4A nuclear receptor, which does not have a ligand, may be playing a role in fibroid pathogenesis. This receptor family is important because of its regulation of the profibrotic/extracellular matrix pathways. Finally, there is increasing evidence that stem cells may be playing a key regulatory role in leiomyoma biology. , ,
Leiomyomas can also be viewed as fibrotic tumors with a dynamic extracellular matrix (ECM) playing an important role in pathophysiology ( Fig. 26.3 ). This hypothesis dates back to the 1990s when experiments demonstrated that the ECM characterizing fibroids contains significant amounts of collagen types I and III protein, and upregulation of mRNA levels occurs during the proliferative phase of the menstrual cycle in leiomyomas but not myometrium. Other matrix components including matrix metalloprotease stromelysin 3 ( MMP 11 ) and dermatopontin (a collagen-binding protein also with decreased expression in keloid scars) have also been shown to be dysregulated in leiomyomas. , Morphologic arrangement of extracellular proteins is also abnormal in myomas and increased stiffness of these ECM alterations leads to altered gene expression through solid-state signaling. These modified mechanical stresses on cells lead to activation of Rho-dependent signaling. Activation of this solid-state signaling and altered state of stress may also contribute to fibroid growth.
The transforming growth factor-β ( TGF-β ) system also appears to be involved in the pathophysiology of leiomyomas, as in other fibrotic processes. A complete review of this topic is beyond the scope of this chapter. Leiomyomas appear to have higher levels of TGF-β and particularly TGF-β3 mRNA and protein and this in turn affects cellular proliferation. , , Vitamin D supplementation has shown reversal of TGF-β3 induced fibrosis in fibroids. Additionally, Granulocyte-macrophage colony-stimulating factor ( GM-CSF ), connective tissue growth factor ( CTGF ), TGF-β4 (also known as lefty or ebaf , endometrial bleeding-associated factor ), the sma-and mad-related (SMAD) family of transcriptions factors, and the mitogen-activated protein kinase (MAPK) signaling pathway appear to be part of the fibrotic pathway dysregulated in myomas or the myometrium or endometrium of the uterus in women with leiomyomas or abnormal uterine bleeding. ,
Angiogenesis, the formation of new blood vessels, is physiologic in the female reproductive tract as opposed to most other tissues where it is pathologic. Abnormalities in uterine blood vessels and angiogenic growth factors also appear to play a role in the pathobiology of myomas. The myomatous uterus shows increased numbers of arterioles and venules as well as venule ectasia. These changes are not confined to the leiomyoma itself but also involve the myometrium and the endometrium. Although such venous abnormalities were originally postulated to be the result of physical compression of the vascular structures by bulky myomas, it is likely that molecular alterations are actually responsible for increased vessel number or abnormal function. , Menstruation-induced state of hypoxia may promote the initial transformation of myocytes to abnormal myocytes.
The process of angiogenesis involves interactions with specific components of the ECM that are dysregulated in fibroids such as collagens type I and III. There are also conflicting data regarding whether the resident immune cells (especially mast cells) contribute to myoma physiology by modulating angiogenesis.
The basic fibroblast growth factor ( bFGF ) receptor/ligand system appears to be a significant factor in leiomyoma pathophysiology. In addition to promoting angiogenesis, bFGF is a smooth muscle cell mitogen and acts similarly to estradiol on leiomyoma smooth muscle cells. Leiomyomas have increased levels of bFGF mRNA compared to matched myometrium, a reservoir of bFGF protein in the ECM, and dysregulation of the endometrial type I bFGF receptor. ,
Uterine leiomyomas do not always necessitate treatment. Generally, expectant management is appropriate until the woman develops symptoms that she determines warrant treatment. Symptoms are usually thought of in two categories, HMB and bulk symptoms, that can exist simultaneously and are dependent on the number, size, and location of the fibroid. FIGO also has a classification system for myoma location that has been developed to help define the location of fibroids in relation to the myometrium. The fibroid types are as follows: Type 0 to 2 rare submucosal fibroids ranging from most intracavitary to least. Types 3, 4, and 5 similarly represent intramural, with types 6 and 7 as serosal and type 8 as cervical.
While we classically think of HMB as related to submucosal fibroids, other contributing factors include abnormal uterine vasculature, impaired endometrial hemostasis, or abnormal contractility near fibroids. This multifactorial etiology is highlighted by a study that showed endometrial changes in leiomyomatous uteri in areas unaffected by fibroids. , ,
Bulk symptoms occur when fibroids become so numerous and/or so bulky that they enlarge the entire uterus, which compresses surrounding structures, or the fibroid itself compresses surrounding structures. Fibroids can be single or many, microscopic or tens of centimeters, and can cause the uterus to be as large as a 20-week uterus. These symptoms include increased frequency of urination, difficulty emptying or even obstruction, changes in bowel habits such as constipation, pelvic pain or pressure, back pain or pressure, or venous compression leading to increased risk of venous thromboembolism. , Pelvic pressure may be described as chronic, intermittent, pressure, or dull pain. In rare cases of large fibroids or uteri, fibroids can cause hydronephrosis. Rarely, fibroids may cause dyspareunia with deep penetration, most often when anterior or fundal in location. Fibroids may outgrow their blood supply and degenerate or if pedunculated may torse, which can cause degeneration of the fibroid and associated pain. Commonly, the pain is associated with low-grade fever, uterine tenderness on palpation, elevated WBC, and peritoneal signs lasting from days to weeks.
In some cases, fibroids can also contribute to infertility, most often if in contact with the endometrial cavity or greater than 4 centimeters, or with obstetric complications. Fibroids have not been shown to cause miscarriage and in a recent study that used multivariable analysis, increasing age appeared to be the driving factor that has led people to conclude fibroids are linked to miscarriage. Adverse pregnancy outcomes include growth restriction, malpresentation, preterm labor and birth, and obstruction of labor.
Lastly, fibroids may uncommonly cause increased erythropoietin production resulting in polycythemia, autonomous production of parathyroid hormone-related protein resulting in hypercalcemia, and hyperprolactinemia. ,
Symptoms often cause women to seek diagnosis; however, both clinically significant and insignificant fibroids are also commonly found incidentally on pelvic exams or imaging performed for other purposes. An abdominal or pelvic exam may reveal an enlarged, mobile uterus with an irregular shape.
There is no laboratory testing that aids in the diagnosis of fibroids. Pelvic ultrasound, with a 95% sensitivity rate for uteri less than 10 weeks, is an ideal diagnostic imaging study and can identify location and relation to some surrounding structures. The fibroid will appear as a hypoechoic well-circumscribed round mass, with shadowing, and may be hyperechoic if highly cellular or have calcifications if degenerating. While expert sonographers can distinguish adenomyomas from fibroids and raise the suspicion of other pathology, if further delineation, characterization, and mapping are needed for diagnosis or treatment planning, magnetic resonance imaging (MRI) provides the greatest level of detail. , For fibroids affecting the endometrial cavity, saline-infused sonograms or hysteroscopy may be helpful. Of note, fibroids may show uptake on PET CT scan performed for other purposes, but CT scan is not helpful for the evaluation of fibroids.
The current evidence is that fibroids do not evolve into sarcomas. However, since both leiomyomas and sarcomas can present as a myometrial mass, assessing a woman’s risk factors for sarcoma is an essential part of choosing a therapy. Sarcoma incidence increases with increasing age and after menopause. Neither being of Black race nor having “rapid” clinical growth increases the risk of sarcomas. While no test has a good positive predictive value for sarcomas, in large part because of their rarity, magnetic resonance imaging especially with diffusion weighting has a good predictive value for ordinary fibroids and endometrial biopsies can diagnose some sarcomas in contact with the uterine cavity and other reasons for AUB.
Sarcomas have been inadvertently treated as leiomyomas with both medical and interventional/surgical therapies; in general, failure to respond to conservative therapy raises additional suspicion of sarcomas. In the past decade, this issue was brought to the forefront in controversy regarding morcellation, using electronic instruments to break leiomyomas into smaller pieces for removal through minimally invasive surgeries. With morcellation, there can be inadvertent dissemination of sarcomas throughout the peritoneum, which worsens prognosis.
However, given the high prevalence of fibroids, assuming every uterine mass is cancer and every presumed cancer requires open surgery to remove leads many women to have unnecessary surgery or increased surgical morbidity. While additional work is ongoing to identify biomarkers of sarcomas, currently assessing clinical risk factors, utilizing endometrial biopsies and MRIs to clarify risk, and utilizing shared decision making with patients as they decide about treatment is standard.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here