Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Aspergillosis is a disease caused by one or more of the species of the genus Aspergillus . Sporelike structures called conidia are aerosolized from the mold form of the organism growing in the environment. When conidia reach tissue, they germinate to form invasive filaments called hyphae .
The most common species infecting humans are Aspergillus fumigatus, Aspergillus flavus, Aspergillus terreus , and Aspergillus niger. Aspergillus species are usually identified in culture by characteristic microscopic features of hyphae and the structures producing conidia. When some species are not readily identifiable, they may be reported by the clinical laboratory as “ Aspergillus species” or “ Aspergillus sp.” A. fumigatus may be reported as “ A. fumigatus species complex.” A. terreus is resistant to amphotericin B. Aspergilli within tissue appear as dichotomously branched (Y-shaped) septate hyphae. Scedosporium and Fusarium species also may produce septate hyphae in tissue. The presence of septa and dichotomous branching differentiates Aspergillus species from the Mucorales, which are the causative organisms of mucormycosis ( Chapter 312 ).
Aspergillus species are ubiquitous organisms in the external environment, including soil, decaying matter, and air, in temperatures as high as 40 to 50° C. Aspergilli are easily isolated from houses, particularly from basements, crawl spaces, bedding, humidifiers, ventilation ducts, potted plants, dust, condiments (e.g., pepper), and marijuana samples. Aspergilli cause abortion in cattle and are important pathogens of marine organisms, insects, and domesticated and wild birds. Aflatoxin, which is one of the most potent carcinogens known, is produced by strains of A. flavus at ambient temperature on stored grain, spices, and nuts. Foodborne ingestion of preformed aflatoxin may cause hepatic necrosis or hepatocellular carcinoma ( Chapter 181 ) in animals and humans.
Aspergillus species may be acquired from airborne conidia in inpatient and outpatient health care settings. Nosocomial aspergillosis is associated with building renovation, new construction, unfiltered air, contaminated ventilation systems, and fireproofing materials. Hospital water, which may become aerosolized during activities such as showering, also can be a potential source of aspergilli. As human pathogens, Aspergillus species may cause acute invasive disease, chronic infection, or allergic symptoms ( Table 311-1 ).
| CATEGORY | SPECIFIC FORMS OF ASPERGILLOSIS |
|---|---|
| Acute invasive aspergillosis | Invasive pulmonary aspergillosis
Extrapulmonary aspergillosis
Disseminated aspergillosis |
| Chronic aspergillosis | Aspergilloma Chronic necrotizing pulmonary aspergillosis Chronic cavitary pulmonary aspergillosis Aspergillus otomycosis |
| Allergic forms of aspergillosis | Allergic bronchopulmonary aspergillosis Extrinsic allergic alveolitis Allergic Aspergillus sinusitis |
Acute invasive aspergillosis develops in immunocompromised patient populations, particularly patients with severe neutropenia ( Chapter 153 ), hematologic malignancies, hematopoietic stem cell transplantation ( Chapter 163 ), severe aplastic anemia ( Chapter 151 ), primary immunodeficiencies ( Chapter 231 ), and solid organ transplantation, especially of heart, lung, and liver. Genetic deficiency of the soluble pattern-recognition receptor called PTX3 (long pentraxin 3) caused by homozygous haplotype (h2/h2) in the PTX3 gene of donor cells leads to impaired neutrophilic antifungal capacity and an increased risk for invasive aspergillosis in recipients of stem cell transplants. Persistent neutropenia, corticosteroids, other immunosuppressive agents, graft-versus-host disease (GVHD), and cytomegalovirus disease are the most frequently observed clinical risk factors. Patients who have influenza ( Chapter 332 ) and immunosuppressed patients with coronavirus disease 2019 (COVID-19; Chapter 336 ) also are at risk for pulmonary aspergillosis. ,
Invasive aspergillosis complicates the care of up to 13% of immunocompromised patients. Invasive aspergillosis is associated with increased hospital mortality and 30-day readmission rates, as well as excess duration of hospitalization and costs.
Conidia of Aspergillus spp. are the key infectious propagules that are acquired by humans from the inanimate environment via organisms that grow in decaying organic matter. These asexual, sporelike structures are 3-5 μ in diameter and have an outer surface covered by rodlets composed of hydrophobin proteins that confer hydrophobicity and adherence to mammalian cell surfaces. The hydrophobins and dihydroxynaphthalene melanins of conidia protect the fungus against host defenses. Inhaled conidia are mechanically cleared by the mucociliary escalator of the pseudostratified columnar respiratory epithelium. Conidia that reach the alveolar space adhere to pulmonary alveolar macrophages via lectin-like attachment sites, including dectin-1, DC-SIGN, and pentraxin 3. When (1→3)-β- d -glucans of swollen conidia are recognized by dectin-1, macrophages are activated to release cytokines and chemokines. Conidia are normally engulfed within phagosomes and destroyed via nonoxidative phagolysosomal mechanisms. Conidia that are not destroyed may germinate to form hyphae, which are then damaged by neutrophils.
These innate host defenses are abrogated in immunocompromised patients. Corticosteroids suppress phagocytosis and nonoxidative killing by macrophages. The recruitment of neutrophils is dependent on the release of chemokines by the lung epithelial cells and CARD 9 signaling. Defects in CARD9 signaling can lead to increased host susceptibility to Aspergillus . Defects in the genes encoding NADPH oxidase lead to chronic granulomatous disease, which strongly predisposes to invasive aspergillosis. Neutropenia and neutrophil dysfunction allow hyphae to invade the respiratory epithelium and vascular endothelium, thereby leading to thrombosis, infarction, and hemorrhage. Among the putative virulence factors of A. fumigatus , serine proteases, elastases, phospholipases, and gliotoxin further contribute to tissue injury, invasion, and dissemination.
This capacity to invade blood vessel walls (angioinvasion) results in hemorrhagic infarction. These processes lead to formation of a necrotic center surrounded by a ring of hemorrhage and edema, which correlates with a “halo sign” surrounding the nodular density. Galactomannan, which is a heteropolysaccharide of the Aspergillus cell wall, is a useful biomarker that is released into the circulation and alveolar spaces during invasive pulmonary aspergillosis. (1→3)-β- d -glucan is another Aspergillus cell wall polysaccharide that is detected in serum during invasive disease.
An aspergilloma consists of a fungal ball composed of matted hyphae and debris in a preformed cavity from previous pulmonary tuberculosis, histoplasmosis, or fibrocystic sarcoidosis. Chronic necrotizing pulmonary aspergillosis characteristically causes a slowly progressive inflammatory destruction of lung tissue superimposed on chronic lung disease. Chronic cavitary pulmonary aspergillosis is defined as the presence of multiple Aspergillus -related cavities, which may or may not contain an aspergilloma. Patients with chronic cavitary pulmonary aspergillosis may have genetically mediated deficits in innate host defenses.
Allergic bronchopulmonary aspergillosis results in a hypersensitivity response that develops in the chronic settings of asthma ( Chapter 75 ) and cystic fibrosis ( Chapter 77 ) with a type I hypersensitivity response that results in elevated IgE to Aspergillus antigens, mast cell degranulation, and bronchoconstriction. Type III hypersensitivity reactions in allergic bronchopulmonary aspergillosis result in elevated interleukin (IL)-4 and IL-5, the latter contributing to eosinophilia.
The classic clinical manifestations of invasive pulmonary aspergillosis in immunocompromised hosts are fever and focal pulmonary infiltrates, nodules, or wedge-shaped densities resembling infarcts. Cough, pleuritic pain, and hemoptysis also may be present. Focal pulmonary infiltrates may progress to a cavity on recovery from neutropenia. Pulmonary infiltrates may also present as bronchopneumonia in an immunosuppressed patient. Concomitant pleural effusion may develop and represent Aspergillus empyema. Tracheobronchial aspergillosis in immunocompromised patients presents as ulcerative, pseudomembranous, or plaquelike large airway disease that may presage pulmonary parenchymal invasion. This invasive form has been seen in mechanically ventilated patients, including patients with COVID-19 ( Chapter 336 ).
Acute Aspergillus sinusitis may occur concomitantly or independently of invasive pulmonary aspergillosis. Although symptoms may include fever, localized pressure, and pain, they may be absent in severely immunocompromised patients. Eschar on the nasal septum and turbinates may be observed by speculum examination or endoscopy. Acute Aspergillus sinusitis of the ethmoid and sphenoid sinuses may progress to cavernous sinus thrombosis with symptoms referable to cranial nerves III, IV, V 1,2 , and VI. A. flavus has a high propensity for causing acute sinus infection.
The tissue targets of extrapulmonary and disseminated aspergillosis most commonly include the CNS, where abscesses and infarcts are characteristic. Patients with CNS aspergillosis present with focal paresis, cranial nerve deficits, and seizures. The glucose level in cerebrospinal fluid (CSF) is usually normal, and cultures of CSF are negative. Other extrapulmonary manifestations include endophthalmitis, myocardial infarction, gastrointestinal disease, renal infarction, cutaneous lesions, and Budd-Chiari syndrome. Esophageal ulcers and mesenteric thrombosis may produce gastrointestinal bleeding. Renal infection may present as flank pain and hematuria.
Aspergillus endocarditis usually begins as an isolated infection in intravenous drug users or after cardiac valvular surgery. Aspergillus endocarditis most commonly presents as major arterial emboli to brain, lung, kidney, and bone. Blood cultures, which are seldom positive, may be delayed in growth by as much as 14 to 21 days. Aspergillus pericarditis may arise from contiguous pulmonary lesions or through transmural infection from endocardial infection.
Locally invasive aspergillosis usually develops in immunocompromised patients as cutaneous ulcers, focal rhinitis, osteomyelitis, and septic arthritis. Cutaneous ulcers have been associated with use of contaminated adhesive tape and arm boards. Keratitis, endophthalmitis, and infection of burn wounds may develop from traumatic inoculation in otherwise immunocompetent patients.
An aspergilloma appears on chest radiograph as a fungus ball in a cavity ( Fig. 311-1 ). Symptomatic patients present with cough, hemoptysis, dyspnea, weight loss, fatigue, chest pain, or fever. Sputum culture is typically positive for Aspergillus species, particularly A. niger . Pleural aspergillosis may complicate surgical resection of aspergilloma or develop spontaneously as a bronchopleural fistula or concomitantly with tuberculosis.
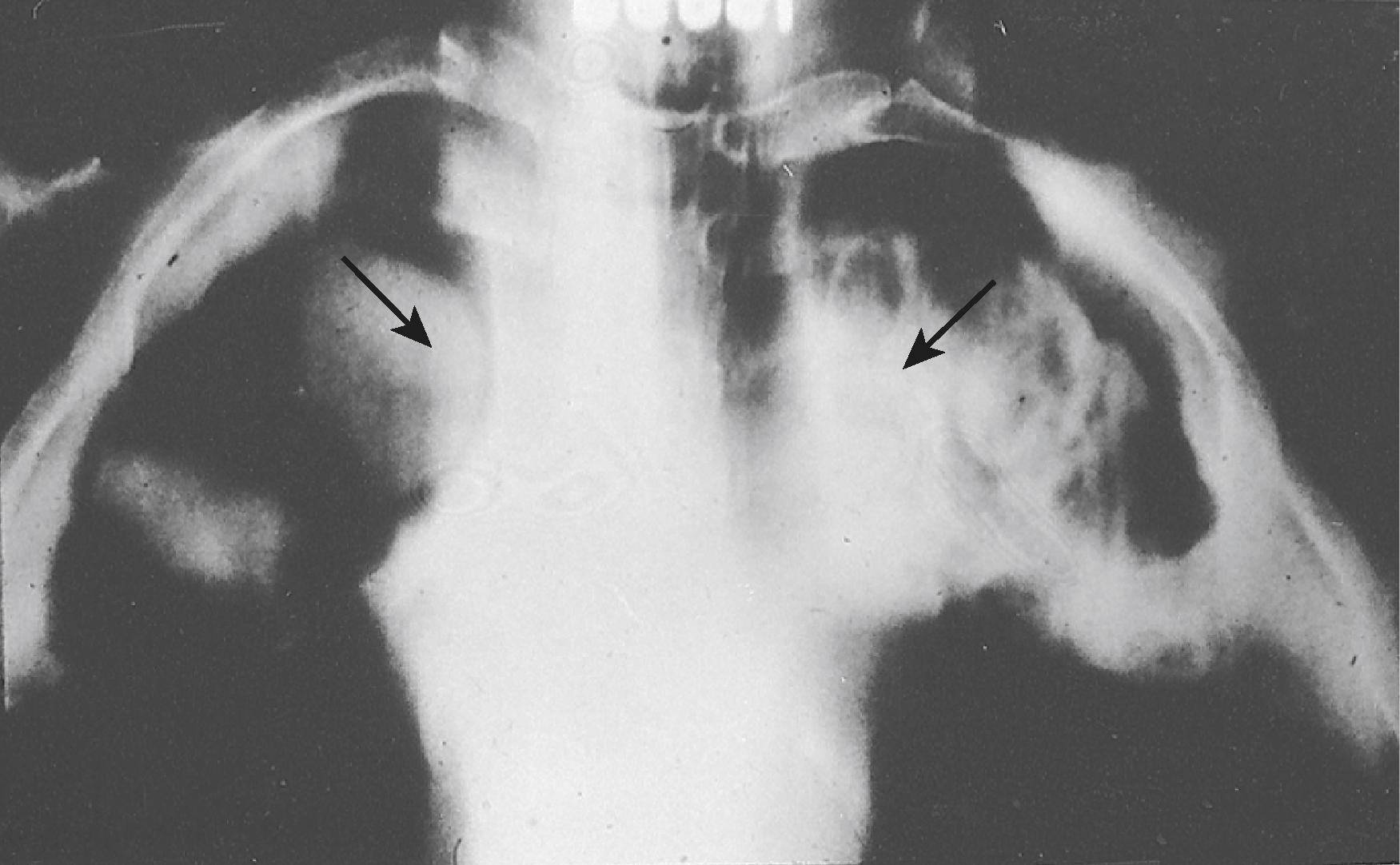
As a stage in the repair process of infarcted lung tissue in neutropenic patients, one or more apparent “aspergillomas” may develop in consolidated lesions during recovery from neutropenia. These apparent aspergillomas do not develop in preexisting cavities and create an “air-crescent sign,” or Monod sign, during their formation.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here