Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
In 1963, idoxuridine became the first antiviral compound to be licensed by the US Food and Drug Administration (FDA) for the topical treatment of herpes simplex virus (HSV) keratitis. This was followed shortly by licensure of amantadine in 1966 as the first systemic antiviral compound for the treatment of influenza A infection. During the 1970s, only vidarabine (1976) received licensure for the systemic treatment of HSV central nervous system (CNS) infections. The licensure of acyclovir in 1982 opened the field of clinical antiviral drug intervention and heralded the era of rapid development of new drugs.
In addition to antiretroviral drugs for the treatment of the human immunodeficiency virus (HIV), three additional non-HIV antiviral drugs were licensed in the 1980s: trifluridine (1980), ribavirin (1985), and interferon (1986). Ten non-HIV antiviral drugs were licensed in the 1990s: foscarnet (1991), rimantadine (1993), ganciclovir (1994), famciclovir (1994), valacyclovir (1995), topical penciclovir (1996), cidofovir (1996), palivizumab (1998), zanamivir (1999), and oseltamivir (1999). Lamivudine, which was approved originally as an HIV medication, was licensed for the treatment of chronic hepatitis B virus (HBV) infection in 1998.
Fewer new antiviral agents were brought to market in the first decade of the 21st century, and all except valganciclovir and remdesivir were for the treatment of hepatitis B: valganciclovir (2001), tenofovir disoproxil fumarate (TDF; 2001), adefovir dipivoxil (2002), pegylated interferon (2003), entecavir (2005), and telbivudine (2006). Since 2010, 18 non-HIV antiviral drugs have been licensed for use in the US, 15 of which were for the treatment of hepatitis C: boceprevir (2011), daclatasvir (2015), dasabuvir (2014), elbasvir (2016), glecaprevir (2017), grazoprevir (2016), ledipasvir (2014), ombitasvir (2014), paritaprevir (2014), pibrentasvir (2017), simeprevir (2013), sofosbuvir (2014), telaprevir (2011), velpatasvir (2016), voxilaprevir (2017) for hepatitis C; baloxavir (2018) and peramivir (2014) for influenza; letermovir (2017) for cytomegalovirus (CMV), and remdesivir (2021) for SARS-CoV-2.
Many of the hepatitis C drugs are used in fixed combinations (e.g., elbasvir and grazoprevir [Zepatier]; glecaprevir and pibrentasvir [Mavyret]; ombitasvir, paritaprevir, and ritonavir copackaged with dasabuvir [Viekira Pak]; sofosbuvir and ledipasvir [Harvoni]); sofosbuvir and velpatasvir [Epclusa]; sofosbuvir, velpatasvir, and voxilaprevir [Vosevi]), and those with a pediatric indication are discussed as such in this chapter. The pace of development of more potent hepatitis C antiviral agents has been rapid and has rendered first-generation products obsolete.
Approved antiviral agents have demonstrated efficacy in the treatment of infections caused by HSV, CMV, varicella-zoster virus (VZV), HIV, respiratory syncytial virus (RSV), influenza A and B, hepatitis B and C, human papillomavirus (HPV), and Lassa virus. In addition, in October 2020, following an initial Emergency Use Authorization, the FDA approved the first anti-coronavirus drug remdesivir in May 2020. Major sites of action of antiviral agents are listed in Table 295.1 , with selected antiviral drugs for specific viruses given in Table 295.2 . Mechanisms of antiviral resistance are presented in Table 295.3 .
| Major Site of Action | Antiviral Agent |
|---|---|
| Viral entry, adsorption, penetration, or uncoating | Amantadine Rimantadine |
| Transcription or replication of viral genome | Acyclovir or valacyclovir Adefovir dipivoxil Cidofovir Dasabuvir Entecavir Famciclovir or penciclovir Foscarnet Ganciclovir or valganciclovir Lamivudine Paritaprevir Ribavirin Simeprevir Sofosbuvir Telbivudine Tenofovir Trifluridine Vidarabine |
| Viral assembly, release, or de-aggregation | Ledipasvir Ombitasvir Oseltamivir Peramivir Zanamivir |
| Virus | Antiviral Drugs Indicated for Management |
|---|---|
| Coronavirus | Remdesivir |
| Cytomegalovirus | Ganciclovir Valganciclovir Foscarnet Cidofovir Letermovir |
| Hepatitis B | Entecavir Tenofovir Lamivudine Telbivudine Adefovir |
| Hepatitis C | Sofosbuvir-ledipasvir Sofosbuvir-ribavirin Sofosbuvir/velpatasvir Glecaprevir/pibrentasvir |
| Herpes simplex virus | Acyclovir Valacyclovir Famciclovir Penciclovir Foscarnet Cidofovir |
| Influenza A and B | Oseltamivir Zanamivir Peramivir Baloxavir |
| Lassa virus | Ribavirin |
| Human papillomavirus | Cidofovir |
| Respiratory syncytial virus | Ribavirin |
| Varicella-zoster virus | Acyclovir Valacyclovir Famciclovir Penciclovir Foscarnet |
| Antiviral | Viruses | Mechanism of Resistance | Clinical Correlates of Resistance | Alternative Antiviral Agents |
|---|---|---|---|---|
| Acyclovir Valacyclovir | HSV, VZV | Usually due to mutations in thymidine kinase gene, resulting in absent or altered thymidine kinase; rarely due to mutation in DNA polymerase gene | Persistent or progressive infection due to resistant strains isolated from patients with severely compromised immunity (e.g., bone marrow transplant recipients, those with AIDS); isolates of HSV from healthy people described in those receiving long-term suppressive therapy | Foscarnet |
| Cidofovir | CMV, HSV | Mutation in DNA polymerase gene | Only rare clinical isolates reported | Foscarnet |
| Famciclovir Penciclovir | HSV, VZV | Usually due to mutations in thymidine kinase gene, resulting in absent or altered thymidine kinase; also, can result from mutation in DNA polymerase gene | Persistent or progressive infection due to resistant strains isolated from patients with severely compromised immunity (e.g., bone marrow transplant recipients those with AIDS); isolates of HSV from healthy people described in those receiving long-term suppressive therapy | Foscarnet Acyclovir |
| Foscarnet | HSV, VZV, CMV | Mutations in viral DNA polymerase gene | Only rare clinical isolates have been reported | Acyclovir Cidofovir |
| Ganciclovir Valganciclovir | CMV | Decreased intracellular phosphorylation due to mutations in the CMV UL97 gene with decreased expression of CMV phosphotransferase enzymes or mutation in viral DNA polymerase gene | Responsible for severe, rapidly progressive infection in patients with severely compromised immunity (e.g., bone marrow transplant recipients, those with AIDS) | Foscarnet CidofovirLetermovir |
| Lamivudine | HBV | Mutation in HBV polymerase gene | Reappearance of HBV DNA in serum after its initial disappearance, although most patients continue to have lower serum HBV DNA and ALT levels compared with pretreatment levels | Adefovir Entecavir |
Non-HIV antiviral small-molecule agents that are licensed by the FDA and are used in the treatment of children are the primary focus of this chapter. Interferons are a family of nonspecific regulatory proteins associated with a variety of antiviral, antiproliferative, and immunomodulating activities. , Their use largely is being supplanted by entecavir and tenofovir, and they have been completely replaced by small molecules in the management of hepatitis C. Interferons, therefore, are not addressed in detail. Monoclonal and polyclonal antibody preparations, also used in the treatment and prevention of viral infections, are discussed in pathogen-specific chapters and in Chapter 5 .
Acyclovir (Zovirax) (i.e., acycloguanosine [ACV]) is a deoxyguanosine analogue with an acyclic side chain that lacks the 3′-hydoxyl group of natural nucleosides. After preferential uptake by infected cells, acyclovir is monophosphorylated by virus-encoded thymidine kinase (TK). Subsequent diphosphorylation and triphosphorylation are catalyzed by host cell enzymes, resulting in acyclovir triphosphate concentrations that are 40–100 times higher in HSV-infected cells than in noninfected cells.
Acyclovir triphosphate prevents viral DNA synthesis by inhibiting the viral DNA polymerase. In vitro, acyclovir triphosphate competes with deoxyguanosine triphosphate as a substrate for viral DNA polymerase. Because acyclovir triphosphate lacks the 3′-hydroxyl group required for elongation of the DNA chain, the growing chain of DNA is terminated. In the presence of the deoxynucleoside triphosphate complementary to the next template position, the viral DNA polymerase is functionally inactivated. Acyclovir triphosphate is a much better substrate for the viral polymerase than for cellular DNA polymerase, resulting in little incorporation of acyclovir into cellular DNA. The higher concentration of the active triphosphate metabolite in infected cells plus the affinity for viral polymerases result in the low toxicity of acyclovir in noninfected host cells.
Acyclovir is most active in vitro against HSV, with activity against VZV being about 10-fold less but still substantial. Although Epstein-Barr virus (EBV) has only minimal TK activity, EBV DNA polymerase is susceptible to inhibition by acyclovir triphosphate. EBV is moderately susceptible to acyclovir in vitro. Activity against CMV is limited because CMV does not encode TK, and CMV DNA polymerase is poorly inhibited by acyclovir triphosphate. The relative activities of drugs used in the treatment of herpesvirus infections are summarized in Table 295.4 .
| Virus | Acyclovir | Penciclovir | Vidarabine | Foscarnet | Ganciclovir | Letermovir |
|---|---|---|---|---|---|---|
| HSV-1 | +++ | ++ | ++ | ++ | +++ | − |
| HSV-2 | +++ | ++ | ++ | ++ | +++ | − |
| VZV | +++ | +++ | ++ | ++ | ++ | − |
| CMV | +/− | +/− | +/− | ++ | ++ | +++ |
| EBV | + | − | − | + | ++ | − |
Resistance of HSV to acyclovir has long been recognized. In some referral centers, 5%–14% of HSV isolates from immunocompromised patients are resistant. Resistance can result from mutations in the viral TK gene or the viral DNA polymerase gene. Although these acyclovir-resistant isolates exhibit diminished virulence in animal models, they can cause severe, progressive, debilitating mucosal disease and, rarely, visceral dissemination in HIV-infected patients. , Acyclovir-resistant strains of HSV also have been recovered from patients receiving cancer chemotherapy, bone marrow and solid-organ transplant recipients, children with congenital immunodeficiency syndromes, and neonates. , Although it is uncommon, genital herpes caused by acyclovir-resistant isolates has been reported in immunocompetent hosts, who usually have received chronic acyclovir therapy.
Acyclovir-resistant strains of VZV are reported. Resistance is caused by the same mechanisms described for HSV isolates. Most acyclovir-resistant VZV strains have been isolated from HIV-infected children and adults who have had profound depletion of CD4 + cells (≤100/mm 3 ) and prolonged acyclovir therapy. Changes in the susceptibility of VZV isolates to acyclovir can develop after only 4–12 weeks. Foscarnet is the drug of choice for HSV and VZV infections caused by acyclovir-resistant strains.
Valacyclovir (Valtrex) is the l -valyl ester of acyclovir. After oral administration, valacyclovir is rapidly converted to acyclovir by first-pass metabolism in the liver. Its mechanism of action, antiviral spectrum, and resistance are the same as those of its parent drug, acyclovir.
Acyclovir is available as a topical formulation, in oral formulations (i.e., capsules, tablets, and suspension), and as a sterile powder for intravenous infusion. Only 15%–30% of the oral formulations of acyclovir is absorbed; peak concentrations of approximately 0.5 μg/mL are attained 1.5–2.5 hours after a 200-mg dose. , Steady-state concentrations of acyclovir after intravenous doses of 2.5–15 mg/kg range from 6.7 to 20.6 μg/mL.
Systemically administered acyclovir is distributed widely, attaining high concentrations in the kidneys, lung, liver, heart, and skin vesicles; concentration attained in the cerebrospinal fluid (CSF) is approximately 50% that of plasma. Acyclovir readily crosses the placenta and accumulates in breast milk. The rate of protein binding ranges from 9% to 33% and is independent of the plasma drug concentration. Less than 20% of acyclovir is metabolized to a biologically inactive metabolite; >60% of administered drug is excreted intact in the urine. The half-life of acyclovir is 2–3 hours in older children and adults and 2.5–5 hours in neonates with normal creatinine clearance (CrCl). Elimination of acyclovir is prolonged in patients with renal dysfunction, with a half-life of approximately 20 hours in those with end-stage renal disease. Dosage modifications are necessary for patients with CrCl values <50 mL/min.1.73 m 2 ( Table 295.5 ). Acyclovir is readily hemodialyzed. The mean plasma half-life in patients during hemodialysis is about 5 hours, requiring that patients receive an additional dose of acyclovir after each dialysis ( Table 295.6 ). The half-life of acyclovir is 13–18 hours during continuous ambulatory peritoneal dialysis (CAPD), and no supplemental dose is necessary after adjustment of the dosing interval.
| Creatinine Clearance (mL/min/1.73/m 2 ) | Suggested Modifications of Standard Intravenous Dose | Suggested Modification of Standard Oral Dose |
|---|---|---|
| >50 | No modification necessary | No modification necessary |
| 25–50 | Maintain unit dose, but increase the dosing interval by 50% | No modification necessary |
| 10–25 | Maintain unit dose, but double the dosing interval | Maintain unit dose, but double the dosing interval |
| 0–10 | Reduce dose by 50% and double the dosing interval | Maintain unit dose, but triple the dosing interval |
| Antiviral Agents | Creatinine Clearance–Necessitating Dose Adjustment (mL/min/1.73 m 2 ) | Need for Supplementary Dose After Hemodialysis |
|---|---|---|
| Acyclovir | <50 | Yes |
| Adefovir | <50 | No b |
| Amantadine | <80 | No |
| Entecavir | <50 | No b |
| Famciclovir (for VZV) | <60 | Yes |
| Famciclovir (for HSV) | <40 | Yes |
| Foscarnet | <100 | Yes |
| Ganciclovir | <80 | Yes |
| Lamivudine | <50 | Unknown |
| Oseltamivir | <30 | Unknown |
| Peramivir | <50 | Yes |
| Ribavirin | — | No |
| Rimantadine | — | No |
a Cidofovir dosing must be adjusted for renal dysfunction but is based on changes in the creatinine level from baseline.
b Administer recommended dose at the appropriate interval after hemodialysis.
After oral administration of valacyclovir, rapid and complete conversion to acyclovir occurs with first-pass intestinal and hepatic metabolism. The bioavailability of valacyclovir in adults exceeds 50%, which is three to five times greater than that of acyclovir. The peak serum concentration, which is attained about 1.5 hours after a dose has been given, is proportional to the amount of drug administered, ranging from 0.8 to 8.5 μg/mL for doses of 100–2000 mg. The area under the drug concentration–time curve (AUC) approximates that seen after intravenous acyclovir. All other pharmacokinetic characteristics are similar to those of acyclovir.
Valacyclovir oral suspension has been studied in children 1 month to 12 years of age. Bioavailability is estimated to be 45%–51% in all age groups except those 3–5 months of age, in whom the bioavailability is about 22%. Approximate dose proportionality in the maximal concentration observed (C max ) and AUC occurs across the 10- to 25-mg/kg dose range (i.e., dose normalized differences usually within ≤30%), with the exception of children 2–5 years of age, for whom a modest increase in dose from 20 to 25 mg/kg is associated with almost doubling the C max and AUC values.
Acyclovir consistently has demonstrated a favorable safety profile. The topical formulation can cause transient burning, especially when applied to ulcerated mucosal lesions. Oral acyclovir sometimes is associated with mild gastrointestinal upset, rash, and headache, but even long-term administration usually is well tolerated. Intravenous acyclovir usually is well tolerated. Because of its alkalinity (pH 9–11), extravasation of drug at the injection site can cause severe inflammation, phlebitis, and sometimes a vesicular eruption leading to cutaneous necrosis. Reversible nephrotoxicity occurs if acyclovir is given by rapid intravenous infusion or to patients who are poorly hydrated or who have preexisting renal compromise. Obstructive nephropathy results from the formation of acyclovir crystals that precipitate in renal tubules. In one study, acute renal insult occurred in 35% of patients beyond the neonatal period who were given acyclovir intravenously. Nephrotoxicity was associated with a dose exceeding 15 mg/kg, age >8 years, and BMI >19 kg/m 2 . Other reported side effects of intravenous acyclovir include rash, sweating, nausea, headache, hematuria, and hypotension. The use of high doses of intravenous acyclovir (60 mg/kg/day) for the treatment of neonatal disease has been associated with neutropenia in some patients.
The most serious side effect of acyclovir is neurotoxicity. When it occurs, lethargy, confusion, hallucinations, tremors, myoclonus, seizures, extrapyramidal signs, and changes in state of consciousness develop within the first few days of starting therapy. Neurotoxicity usually occurs in patients with compromised renal function who attain high serum concentrations of the drug. Neurotoxic manifestations usually resolve spontaneously within several days after the discontinuation of acyclovir; hemodialysis may be useful in severe cases associated with high serum concentrations of the drug.
Although acyclovir is mutagenic at high concentrations in some in vitro assays, it is not teratogenic in various animal models. Acyclovir use in pregnant women is not associated with congenital defects or other adverse gestational outcomes. Neutropenia occurs in approximately 20% of infants given acyclovir orally to suppress HSV reactivation after neonatal infection.
The profiles of adverse effects and potential drug interactions observed with valacyclovir therapy are the same as those observed with acyclovir. Neurotoxicity has not been reported in humans, although it has been observed in animal models. Manifestations resembling thrombotic microangiopathy have been described in patients with advanced HIV disease receiving high doses of valacyclovir, but the coadministration of multiple medications makes the establishment of a causal relationship difficult.
Acyclovir is effective for the treatment of infections caused by HSV and VZV in immunocompetent and immunocompromised hosts. Table 295.7 outlines the infections for which acyclovir is effective, the recommended dosage, and magnitude of anticipated benefits. Unless otherwise stated, the anticipated benefits of acyclovir therapy are based on the results of randomized, controlled trials.
| Infection | Dosage | Anticipated Benefits | Factors Favoring Beneficial Effect of Therapy | Strength of Indication |
|---|---|---|---|---|
| Initial genital HSV | 15 mg/kg/day IV in 3 divided doses for 5–7 days or 40–80 mg/kg/day (maximum: 1000 mg/day) PO in 3–4 divided doses for 5–10 days a | 4- to 12-day reduction in duration of viral shedding, 3- to 12-day reduction in time to healing, and 1- to 10-day reduction in duration of pain; therapy is not associated with reduced frequency of subsequent recurrences | Intravenous, rather than oral, therapy; therapy for primary, rather than less severe first episode, nonprimary infections | ++ |
| Recurrent genital HSV | 1000 mg/day in 5 divided doses for 5 days or 1600 mg/day in 2 divided doses for 5 days or 2400 mg/day in 3 divided or doses for 2 days a | 1- to 2-day reduction in duration of viral shedding and time to healing; no difference in duration of pain or frequency of subsequent recurrences , , | Initiation during prodromal phase of infection | +/− |
| Primary HSV labialis in adults | 2400 mg/m 2 /day PO in 4 divided doses for 10 days | 6-day reduction in the duration of viral shedding and 2- to 4-day reduction in the duration of drooling, gum swelling, and healing of oral and cutaneous lesions | Initiation within 4 days of onset of signs of infection | ++ |
| Recurrent HSV labialis in adults | 1000 mg/day PO in 5 divided doses for 5 days | 0.5- to 2-day reduction in the duration of pain and time to healing | Initiation during prodromal or erythema stage | +/− |
| Mucocutaneous HSV in compromised hosts | 30 mg/kg/day IV in 3 divided doses for 7–14 days | 60%–80% reduction in the duration of viral shedding, 30%–60% reduction in healing time and the duration of pain | Recovery of immune function | +++ |
| Viscerally disseminated HSV in pregnant women and transplant recipients | 30 mg/kg/day IV in 3 divided doses for 7–14 days | Improved likelihood of recovery based on anecdotal reports, rather than controlled trials , | — | +++ |
| HSV encephalitis | 30–45 mg/kg/day IV in 3 divided doses for 14–21 days | Reduced mortality and improved long-term neurologic outcome | High (>6) Glasgow Coma Score at time of initiation of therapy; young age (<30 years) | ++++ |
| Neonatal HSV | 60 mg/kg/day IV in 3 divided doses for 14 (SEM) to 21 (CNS, disseminated) days | Reduced mortality and improved long-term neurologic outcome , | Infection limited to the skin, eye, and mucosal membranes; absence of seizures at initiation of therapy for CNS infection | ++++ |
| Primary varicella in normal children, adolescents, and adults | 80 mg/kg/day (maximum: 3200 mg/day) PO in 4 divided doses for 5 days | 1- to 3-day reduction in new lesion formation and fever; reduced maximal number of lesions | Initiation of therapy within 24 h of onset of infection; older subjects and secondary cases in household benefit more than younger subjects and those with community-acquired infections | +/++ |
| Disseminated varicella in previously healthy subjects | 30 mg/kg/day IV in 3 divided doses for 7–10 days | Improved likelihood of recovery based on anecdotal reports rather than controlled trials | — | +++ |
| Zoster in normal hosts | 80 mg/kg/day (maximum: 4000 mg/day) PO in 5 divided doses for 5–7 days | Reduced time to healing of skin lesions, duration of acute pain, and duration of viral shedding , | Initiation of therapy within 24–48 h of onset of infection | + |
| Zoster ophthalmicus in normal hosts | 80 mg/kg/day (maximum: 4000 mg/day) PO in 5 divided doses for 5–7 days | Reduced incidence of keratitis and anterior uveitis | — | ++ |
| Primary varicella in compromised hosts | 30 mg/kg/day IV in 3 divided doses for 7–10 days | Reduced incidence of cutaneous and visceral dissemination; reduced duration of lesion formation and time to healing , | Initiation of therapy within 24–48 h of onset of infection and mild degree of immunologic dysfunction | ++++ |
| Zoster in compromised hosts | 30 mg/kg/day IV in 3 divided doses for 7–10 days | Reduced risk of cutaneous dissemination and visceral complications , | Initiation of therapy within 24–48 h of onset of infection and mild degree of immunologic dysfunction | ++++ |
a Topical acyclovir is not recommended because its effects on duration of viral shedding, pain, and lesions are much less than those of systemic therapy in patients with primary infections.
Therapy is indicated for life-threatening infections and for disease associated with severe morbidity and death, such as HSV encephalitis, neonatal HSV infections, and VZV infections in immunocompromised hosts. , Acyclovir therapy also is indicated for mucocutaneous HSV infections in immunocompromised hosts and for disseminated HSV and VZV infections in otherwise normal hosts, including pregnant women. Acyclovir is effective for the treatment of symptomatic primary genital HSV infections, reducing the median duration of virus shedding, pain, and length of time to complete healing.
Intravenous and oral acyclovir therapies are almost equally efficacious; topical treatment provides less benefit. Neither intravenous nor oral treatment with acyclovir for primary or recurrent genital HSV disease reduces the frequency of recurrences. Acyclovir probably reduces the ocular morbidity of zoster ophthalmicus. The advantages of treating previously healthy individuals with herpes labialis, , recurrent genital herpes, , , varicella, and herpes zoster , are less dramatic, and the treatment of these patients should be individualized.
Favorable effects of therapy are more likely when acyclovir is begun soon after the onset of infection. For example, previously healthy people with primary varicella may benefit only if acyclovir is begun within 24 hours of appearance of the first cutaneous lesions. Benefit, however, can be observed in the treatment of severe, life-threatening infections caused by HSV and VZV even if acyclovir is commenced several days after the onset of infection, provided that irreversible damage has not occurred.
Indications for prophylactic acyclovir therapy are summarized in Table 295.8 . Prophylactic acyclovir is almost exclusively used for the suppression of HSV infections; its efficacy for CMV infections is inferior to that of ganciclovir. The most frequent indication for long-term prophylactic therapy is in patients with frequently recurrent genital infections in whom daily administration of acyclovir reduces the frequency of recurrences by at least 75%. Acyclovir has been shown to maintain a high degree of efficacy and little toxicity even after >5 years of continuous suppressive therapy.
| Antiviral Agent | Clinical Indication | Expected Effect | Dosage |
|---|---|---|---|
| Acyclovir | Frequently recurrent genital HSV | 70%–90% reduction in frequency of clinical episodes of infection , , ; frequency of asymptomatic shedding also reduced, but not eliminated | Start with 1000 mg/day in divided doses and slowly reduce to minimum effective dose; attempt withdrawal after 6–12 months; if symptoms recur, restart therapy |
| Frequently recurrent HSV labialis | 53% reduction in number of clinical recurrences | 400 mg bid for 4 months | |
| Skiers with histories of sun-induced recurrences of HSV labialis | 73% reduction in risk of recurrence | 400 mg bid for 1 week | |
| Suppressive therapy after treatment of acute neonatal HSV disease | 30% reduction in cutaneous recurrence (all disease categories); improved neurodevelopmental outcomes (CNS category) | 300 mg/m 2 /dose tid for 6 months; monitor absolute neutrophil counts at 2 and 4 weeks, then monthly during therapy | |
| Reactivations of HSV associated with serious systemic complications such as erythema multiforme, aseptic meningitis, and eczema herpeticum | Reduction in the frequency of serious complications associated with HSV infection | Use is supported by theoretical benefit and anecdotal reports, rather than controlled trials; , dosage is empiric | |
| Dermabrasion of facial skin or surgery involving the trigeminal ganglion in patients previously infected with HSV-1 | Reduction in the frequency of postoperative outbreaks of herpes labialis , | Use is supported by theoretical benefit and anecdotal reports, rather than controlled trials; dosage is empiric | |
| HSV-seropositive persons undergoing bone marrow or solid-organ transplantation or induction chemotherapy for hematologic malignancy | 50%–60% decrease in number of herpetic recurrences during the time of drug administration | 500–750 mg/m 2 /day begun before transplantation or induction chemotherapy and continued for about 6 weeks thereafter; intravenous therapy initially and oral therapy when tolerated | |
| CMV-seropositive persons undergoing bone marrow or solid-organ transplantation | As much as 50% reduction in the frequency of CMV infection and disease in CMV-seropositive recipients and, to a lesser extent, in recipients at risk for primary infection; however, ganciclovir is drug of choice | 1500 mg/m 2 /day begun before transplantation and continued for 1–3 months thereafter; intravenous therapy initially, and oral therapy when tolerated | |
| Ganciclovir | CMV-seropositive solid-organ transplant recipients | Reduction in frequency of CMV disease | 10 mg/kg/day in 2 divided doses, started several days before transplantation and continued for 2–3 weeks after transplantation; 5 mg/kg/day in single dose thereafter |
| CMV-seropositive renal or liver transplant recipients beginning OKT3 suppressive therapy | Reduction in frequency of CMV disease | 10 mg/kg/day in 2 divided doses for 2–3 weeks after OKT3 initiated | |
| Bone marrow transplant recipients with positive CMV surveillance culture | Reduction in frequency of CMV disease | 10 mg/kg/day in 2 divided doses for 2–3 weeks after transplantation; 5 mg/kg/day in single dose thereafter |
The use of prophylactic acyclovir therapy in pregnant women has been explored in several small studies, but the numbers of patients evaluated are insufficient to prove definitively its safety or efficacy in this population. Neonatal HSV disease in babies born to women receiving antiviral suppression is reported. Brief courses of acyclovir can be given to HSV-1–infected patients to suppress reactivation of HSV that predictably follows stimuli such as exposure to ultraviolet radiation or facial surgery. Longer courses can be given to those with frequently recurrent oral HSV infections, , and to people after ocular HSV disease to prevent recurrence.
Prophylactic acyclovir also has proved to be a useful strategy for postponing the inevitable reactivation of HSV infection after severe immunosuppression of HSV-seropositive patients. Anecdotal data suggest that prophylactic acyclovir can reduce the frequency of attacks of erythema multiforme temporally related to HSV.
Oral acyclovir suppressive therapy following parenteral acyclovir therapy for acute neonatal HSV disease improves neurodevelopmental outcomes at 1 year of age of children with CNS involvement. Suppressive oral acyclovir therapy also delays the development of cutaneous recurrences in all categories of neonatal HSV disease.
The indications for valacyclovir therapy are the same as those for acyclovir. , Adult treatment doses are 1 g given orally three times daily for 7 days for herpes zoster in patients >50 years of age; 1 g given orally twice daily for 10 days for first-episode genital herpes; 500 mg given orally twice daily for 3 days for episodic treatment of recurrent genital HSV disease; 1 g given orally once daily for suppression of recurrent genital HSV; and 2 g given orally twice daily for 1 day for recurrent herpes labialis (≥12 years of age). The efficacy of valacyclovir has been demonstrated in the short-term treatment of recurrent herpes labialis (2 g PO bid for 1 day) and in the suppression of recurrent herpes labialis (500 mg once daily).
In children, valacyclovir dosing is 20 mg/kg per dose (dose not to exceed 1 g) administered 3 times daily (for VZV) or twice daily (for HSV). In the US, valacyclovir is licensed for the treatment of pediatric patients with orolabial HSV recurrences (≥12 years of age) and chickenpox (2–17 years of age). Although comparative data from controlled studies are limited, valacyclovir may be advantageous in treating infections caused by viruses that are less sensitive to acyclovir than HSV (i.e., VZV and CMV).
Cidofovir (Vistide), or (5)-1-(3-hydroxy-2-(phosphonylmethoxy)-propyl)cytosine (HPMPC), is a novel acyclic phosphonate nucleotide analogue. Cidofovir has a mechanism of action that is similar to that of the nucleoside analogues, such as acyclovir and ganciclovir. In its native form, however, cidofovir already has a single phosphate group attached, and viral enzymes are not required for the initial phosphorylation of the drug. Cellular kinases sequentially attach two additional phosphate groups, converting cidofovir to its most active form. The active compound is a competitive inhibitor of DNA polymerase. Although cidofovir is taken up by virus-infected and uninfected cells, the active form of the drug exhibits a 25-fold to 50-fold greater affinity for the viral DNA polymerase compared with the cellular DNA polymerase, selectively inhibiting viral replication. , Incorporation into the growing viral DNA chain results in reductions in the rate of DNA synthesis.
Cidofovir has activity against HSV and CMV. Because of its unique phosphorylation requirements for activation, the drug usually maintains activity against acyclovir- and foscarnet-resistant HSV isolates and against ganciclovir- and foscarnet-resistant CMV mutants. Cidofovir exhibits marked activity against CMV, with inhibitory concentrations of 0.1 μg/mL for susceptible clinical isolates. Although cidofovir is less potent in vitro against HSV than is acyclovir, its favorable pharmacokinetic profile increases its anti-HSV activity. Cidofovir also has in vitro activity against VZV, EBV, human herpesvirus 6, human herpesvirus 8, polyomaviruses, orthopoxviruses, adenovirus, BK virus, and HPV.
Only a small number of cidofovir-resistant CMV isolates have been described. Some also are resistant to ganciclovir because of mutations in the DNA polymerase gene, but they remain sensitive to foscarnet.
After intravenous administration, the plasma half-life is 2.6 hours, although cidofovir persists in cells for prolonged periods. Active intracellular metabolites of cidofovir have half-lives of 17–48 hours. This prolonged intracellular activity allows for an intermittent dosing schedule that is attractive compared with that for ganciclovir or foscarnet. About 90% of drug is excreted in the urine, primarily by renal tubular secretion. ,
Nephrotoxicity is the principal adverse event associated with systemic administration. , Cidofovir concentrates in renal cells in amounts 100 times greater than in other tissues, producing severe nephrotoxicity involving the proximal convoluted tubule if concomitant hydration and administration of probenecid are not employed. Renal toxicity manifests as proteinuria and glycosuria. To decrease the potential for nephrotoxicity, aggressive intravenous prehydration and coadministration of probenecid are required with each cidofovir dose. Within 48 hours before delivery of each dose of cidofovir, serum creatinine and urine protein concentrations must be determined, and the dose adjusted if indicated. Cidofovir should not be administered concomitantly with other nephrotoxic agents. Cidofovir is contraindicated in patients with a serum creatinine level >1.5 mg/dL, a calculated CrCl of 55 mL/min or lower, or a urine protein level of at least 100 mg/dL (i.e., ≥2+ proteinuria). The maintenance dose of cidofovir must be reduced from 5 to 3 mg/kg for an increase in serum creatinine of 0.3–0.4 mg/dL above baseline. Cidofovir therapy must be discontinued if the serum creatinine level increases by at least 0.5 mg/dL above baseline.
Because of poor oral bioavailability (2%–26%), cidofovir can be administered only intravenously or topically. Intravitreal administration has been associated with ocular hypotony.
Cidofovir has been evaluated for the treatment of CMV retinitis in patients with acquired immunodeficiency syndrome (AIDS). , Therapy delays progression of retinal disease. In one study, patients who had failed or were intolerant of traditional therapy (i.e., ganciclovir or foscarnet) were treated with a 2-week induction course of cidofovir (5 mg/kg administered weekly) followed by randomization to maintenance with a dose of 5 mg/kg or 3 mg/kg every other week. At the higher maintenance dose of cidofovir, disease progression was delayed for 115 days, compared with 49 days in the lower-dose cohort. Cidofovir also has been used successfully in the management of disease caused by acyclovir-resistant HSV isolates.
The safety and efficacy of cidofovir in children have not been studied. The use of cidofovir in children with AIDS warrants caution because of the risk of long-term carcinogenicity and reproductive toxicity.
Case reports and small case series suggest that cidofovir may be beneficial in the management of adenovirus infections and BK virus infections , in immunocompromised patients.
The recommended induction dose of cidofovir for patients with CMV disease with a serum creatinine level ≤1.5 mg/dL, a calculated CrCl >55 mL/min, and a urine protein level <100 mg/dL (i.e., equivalent to <2+ proteinuria) is 5 mg/kg administered once per week for 2 consecutive weeks. The recommended maintenance dose of cidofovir is 5 mg/kg administered once every 2 weeks. The drug should be administered with probenecid, and hydration should be ensured.
Famciclovir (Famvir) is the inactive diacetyl ester prodrug of penciclovir (Denavir), an acyclic nucleoside analogue with a spectrum of activity against herpesviruses similar to that of acyclovir. Its activation by viral and host cell enzymes and its mechanism of action are akin to those of acyclovir, except that penciclovir is neither an obligate DNA chain terminator nor an inactivator of DNA polymerase. However, laboratory studies suggest that penciclovir triphosphate retards the rate of subsequent nucleotide incorporation.
Penciclovir is approximately 100-fold less potent than acyclovir in inhibiting herpesvirus DNA polymerase activity. However, it remains an effective antiviral agent because of high intracellular concentrations and long half-life. Because penciclovir must be activated by the virus-encoded TK enzyme, TK-deficient viral strains are resistant to acyclovir and penciclovir. Penciclovir persists in high intracellular concentrations much longer than acyclovir. The antiviral effect is more sustained, and the drug can be administered less frequently than acyclovir.
After oral administration of famciclovir, the bioavailability of penciclovir is about 70%. Food delays absorption but does not affect the peak plasma drug concentration. Peak concentrations of drug after intravenous administration of 10 mg/kg are approximately sixfold higher than those attained after oral doses of 250 mg. Drug half-life is 2.5 hours. Almost 75% of administered drug is recovered unchanged in the urine, and dose reduction is recommended for those with compromised renal function. A 12-hour dosing interval is recommended for those with CrCl values between 30 and 50 mL/min/1.73 m 2 , and a 24-hour interval is recommended for those with CrCl values <30 mL/min/1.73 m 2 . Measurable penciclovir concentrations are not detectable in plasma or urine after topical administration of penciclovir cream.
Famciclovir pharmacokinetics have been assessed in a single-dose pediatric study of infants 1–12 months of age given famciclovir sprinkles orally. Infants <6 months had substantially lower systemic exposure compared with those 6- to 12–months of age. A small safety and pharmacokinetic study assessed a single, large dose of famciclovir in adolescents with recurrent herpes labialis.
Famciclovir is as well tolerated as acyclovir. Complaints of nausea, diarrhea, and headache occurred in clinical trials, although at frequencies similar to those reported by placebo recipients. No clinically significant drug interactions have been reported, but concentrations of famciclovir among volunteers increase by about 20% in the patients receiving concomitant cimetidine or theophylline administration.
The indications for and recommended dosages of famciclovir are presented in Table 295.9 . Dosing in infants and young children has been studied. Dosing recommendations for children 6 months to 12 years are based on pharmacokinetic modeling and have not been studied adequately to warrant dosing recommendations in this age group.
| Infection | Dosage | Duration |
|---|---|---|
| Zoster | 500 mg tid | 7 days |
| Recurrent genital HSV, immunocompetent host (episodic therapy) | 1000 mg bid | 1 day |
| Recurrent genital HSV, immunocompetent host (suppressive therapy) | 250 mg bid | Up to 1 year |
| Recurrent genital HSV, immunocompromised host (episodic therapy in compromised hosts) | 500 mg bid | 7 days |
| Recurrent orolabial HSV, immunocompromised host (episodic therapy) | 500 mg bid | 7 days |
| Recurrent orolabial HSV, immunocompetent host (episodic therapy) | 1500 mg | Administer as a single dose |
Topical penciclovir for the treatment of recurrent herpes labialis reduces the time to healing and the duration of pain by about one-half of a day. , Application of medicine should begin as early as possible, preferably during the prodromal phase, and should be continued every 2 hours during waking hours for 4 days.
Foscarnet (Foscavir), or phosphonoformic acid (PFA), is an inorganic pyrophosphate analogue that inhibits all known human herpesviruses, including most ganciclovir-resistant CMV isolates and acyclovir-resistant HSV and VZV strains ( Table 295.4 ). Foscarnet also is active against HIV. This antiviral agent directly inhibits DNA polymerase by blocking the pyrophosphate binding site and preventing cleavage of pyrophosphate from deoxynucleotide triphosphates. Foscarnet is a noncompetitive inhibitor of viral DNA polymerases or HIV reverse transcriptase and is not incorporated into the growing viral DNA chain.
Foscarnet is approximately 100-fold more active against viral enzymes than against host cellular enzymes. Resistance occurs as a result of DNA polymerase mutations. Strains of CMV, HSV, and VZV with threefold to fivefold reduced sensitivity to foscarnet have been reported. , These isolates may respond to therapy with acyclovir or cidofovir.
Foscarnet is absorbed poorly after oral administration, with a bioavailability of about 20%. This limits foscarnet administration to the intravenous route. Maximal serum concentration attained after a dose of 60 mg/kg is approximately 500 μmol/L. Data regarding tissue distribution are limited, but CSF concentrations are about two thirds of those in serum.
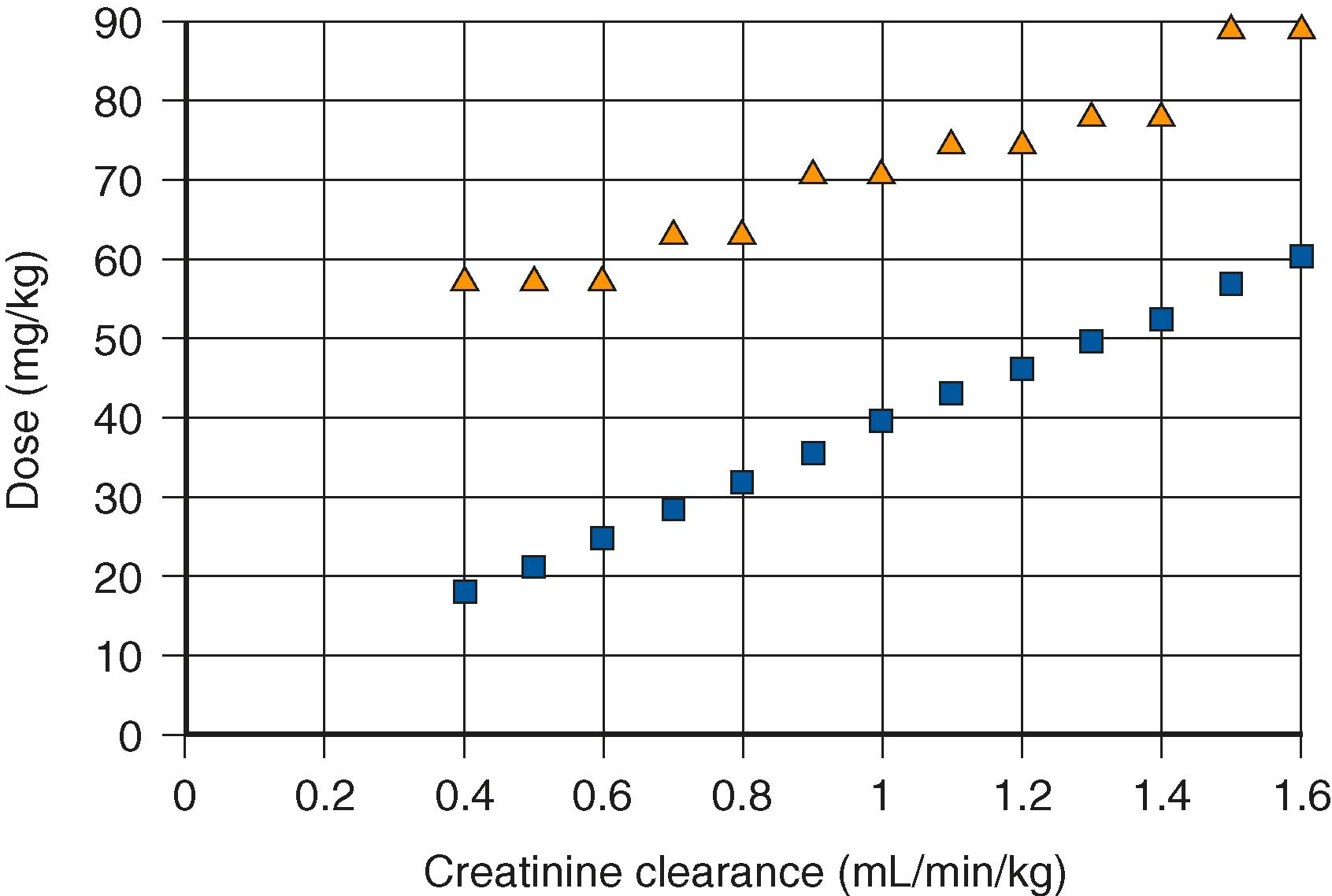
Eighty percent of an administered dose of foscarnet is eliminated unchanged in the urine. The half-life is 48 hours, and dosage adjustments are necessary even for minimal degrees of renal dysfunction ( Table 296.6 ). The degree of dose reduction is proportional to the degree of reduction in CrCl; when CrCl is 50% of normal, the dose should be reduced by about 50% ( Fig. 295.1 ). Hemodialysis efficiently eliminates foscarnet, and an extra dose of drug is recommended after dialysis for 3 or more hours. There are no pharmacokinetic data for foscarnet in neonates.
The most common and serious adverse effects are nephrotoxicity and metabolic derangements. Azotemia, proteinuria, acute tubular necrosis, crystalluria, and interstitial nephritis can occur, and serum creatinine concentrations increase, usually during the second week of therapy, in as many as 50% of patients. In most affected patients, renal function returns to normal within 2–4 weeks after discontinuing foscarnet. Risk factors for developing renal dysfunction include preexisting renal disease, concurrent use of other nephrotoxic drugs, dehydration, rapid injection of large doses, and continuous intravenous infusion of drug.
Drug-related electrolyte disturbances can occur, including symptomatic hypocalcemia, hypercalcemia, hypophosphatemia, and hyperphosphatemia. Hypocalcemia is caused by direct chelation of ionized calcium by the drug, and patients can have paresthesias, tetany, seizures, and arrhythmias. Metabolic disturbances can be minimized if foscarnet is administered by slow infusion, with rates not exceeding 1 mg/kg/min.
Common drug-associated CNS symptoms include headache, tremor, irritability, seizures, and hallucinations. Fever, nausea, vomiting, abnormal serum hepatic enzymes, anemia, granulocytopenia, and genital ulcerations are reported. Genital ulcerations appear to be associated with high urinary concentrations of drug. Concomitant use of amphotericin B, cyclosporine, gentamicin, and other nephrotoxic drugs increases the likelihood of renal dysfunction associated with foscarnet therapy. Hypocalcemia is more common with coadministration of pentamidine, and anemia and neutropenia are more common when patients are treated with foscarnet and zidovudine.
The most important indication for foscarnet is therapy for sight-threatening chorioretinitis caused by CMV in patients with AIDS. Several controlled trials have demonstrated that foscarnet is as effective as ganciclovir in managing this infection and may offer a survival advantage because of its inherent activity against HIV. In refractory cases of chorioretinitis, combination foscarnet and ganciclovir therapy have been given. Foscarnet also is effective in the treatment of ganciclovir-resistant strains of CMV infections. Limited data for patients with AIDS suggest a foscarnet benefit for gastrointestinal and pulmonary CMV infections. ,
The usual dosage regimen for CMV infection is 180 mg/kg/day in 3 divided doses for 14–21 days, followed by a daily maintenance dose of 90–120 mg/kg. About 90% of patients have stabilization of their retinitis, and the time to progression of infection is prolonged to about 3 months.
Infections caused by acyclovir-resistant strains of HSV and VZV have been successfully controlled with foscarnet. , The dosage of foscarnet used for these infections is 120 mg/kg/day in 3 divided doses. In patients with AIDS, foscarnet therapy should be initiated within 7–10 days of suspicion of infection caused by acyclovir-resistant HSV or VZV. Therapy should be continued until lesions have resolved.
Ganciclovir (Cytovene) is a nucleoside analogue that is different from acyclovir by having an extra hydroxymethyl group on the acyclic side chain. Its greatest in vitro activity is against CMV ( Table 295.4 ), although it is also as active as acyclovir against HSV-1 and HSV-2 and almost as active against VZV.
As with acyclovir and penciclovir, the first step in ganciclovir phosphorylation is carried out by a virus-encoded enzyme, and the final steps are achieved by cellular enzymes. CMV lacks the gene for TK, and the enzyme that catalyzes the initial phosphorylation of ganciclovir in CMV-infected cells is the phosphotransferase encoded by the UL97 gene. , Intracellular ganciclovir triphosphate concentrations are at least 10-fold higher in CMV-infected cells than in noninfected cells, and intracellular ganciclovir triphosphate has a half-life of 24 hours or longer.
Ganciclovir triphosphate is a competitive inhibitor of herpesvirus DNA polymerases, although it has some activity against cellular DNA polymerases. This potential for incorporation into host cellular DNA accounts for ganciclovir’s substantial toxicity. Incorporation of ganciclovir triphosphate into the growing viral DNA chain results in slowing and subsequent cessation of DNA chain elongation.
Valganciclovir (Valcyte) was approved by the FDA in 2001. Because valganciclovir is well absorbed after oral administration, it can be a favorable alternative to intravenously administered ganciclovir for the treatment and suppression of CMV infections in immunocompromised hosts. Valganciclovir is an l -valine ester prodrug of ganciclovir and has the same mechanism of action and antiviral spectrum as ganciclovir.
Ganciclovir resistance among CMV isolates is conferred by mutations in the UL97 gene or the CMV DNA polymerase gene. Of these two mechanisms of antiviral resistance, ganciclovir-resistant CMV isolates with mutations in the UL97 open reading frame are the predominant phenotype. Specific mutations in the UL97 region can be detected rapidly in plasma and serum by direct sequencing of polymerase chain reaction (PCR)–amplified CMV DNA.
Resistant strains of CMV should be suspected when progressive disease and continued recovery of virus occur despite ganciclovir therapy. In one study, 8% of 72 patients with AIDS had progressive infection associated with isolation of ganciclovir-resistant strains of CMV after 3 months of continuous ganciclovir therapy. Resistance may be more likely in patients treated with oral ganciclovir than in those treated with intravenous ganciclovir because of the selective pressure applied by the lower concentrations of drug achieved with oral administration. In contrast, CMV isolates from solid-organ transplant recipients who have been exposed to ganciclovir appear less likely to develop resistance. Foscarnet may be useful in the treatment of CMV infections caused by ganciclovir-resistant isolates.
Occasionally, strains of HSV that are resistant to acyclovir because of TK deficiency also are less sensitive to ganciclovir. Ganciclovir-resistant isolates of HSV due to mutations in DNA polymerase have been demonstrated in the laboratory but have not become a clinical problem.
The mechanism of resistance to ganciclovir and valganciclovir are the same. Small studies have indicated that rates of resistance among valganciclovir recipients are similar to those of intravenous ganciclovir recipients and are less than those of patients taking ganciclovir orally.
Peak serum concentrations of ganciclovir attained after 5 mg/kg administered intravenously range from 8 to 11 μg/mL. Concentrations sufficient to inhibit susceptible strains of CMV are attained in aqueous humor, subretinal fluid, CSF, and brain tissue. Most of an administered dose of ganciclovir is eliminated unchanged in the urine; the elimination half-life is 2–3 hours. Dose reduction, roughly proportional to the degree of reduction in CrCl, is necessary in patients with impaired renal function ( Table 295.6 ). , For CrCl values of 50 to <80 mL/min/1.73 m 2 , one-half of the usual dose should be given every 12 hours. The same dose should be given every 24 hours for CrCl values of 25 to <50 mL/min/1.73 m 2 , and 25% of the usual dose should be given every 24 hours for CrCl values of <25 mL/min/1.73 m 2 . Because ganciclovir is efficiently removed by hemodialysis, a supplemental dose is recommended after dialysis.
The pharmacokinetics of ganciclovir in the neonatal population are similar to those of adults. After intravenous administration of 6 mg/kg of ganciclovir, peak concentrations of 7 μg/mL are achieved. The mean elimination half-life is 2.4 hours. Pharmacokinetics of oral valganciclovir have been compared with those of parenteral ganciclovir in this population. Intravenous ganciclovir clearance almost doubled, and the AUC 12 was reduced by almost one-half over the first 6 weeks of life. In comparison, only a marginal decrease in AUC 12 was seen after administration of valganciclovir oral solution, possibly because bioavailability increased by 32% over the same period.
The concentration of ganciclovir in the CNS is 24%–70% of the plasma level, and the brain concentration is approximately 38% of plasma level.
Valganciclovir is rapidly converted to ganciclovir, with a mean plasma half-life of about 30 minutes. The absolute bioavailability of valganciclovir exceeds 60%, and it is enhanced by about 30% with concomitant administration of food. Oral valganciclovir produces exposures of ganciclovir exceeding those attained with oral ganciclovir and are similar to those reported after standard intravenous administration of ganciclovir. Patients with impaired renal function require dosage reduction roughly proportional to their reduction in CrCl.
The most important toxic effect of ganciclovir is myelosuppression. Dose-related neutropenia, defined as a >50% decrease in absolute neutrophil count from baseline or <1000/mm 3 , is the most consistent hematologic disturbance. The incidence of neutropenia during a 2-week course is about 40%. It is dose-limiting in about 15% of courses, and it is reversible on cessation of the drug. The likelihood of neutropenia occurring after oral administration of ganciclovir is lower, with 14%–24% of patients developing an absolute neutrophil count of <1000/mm 3 .
Hematopoietic growth factors may be useful in preventing or counteracting neutropenia. Thrombocytopenia (<50,000 platelets/mm 3 ) occurs in approximately 20% of treated patients. Anemia occurs in only about 2% of ganciclovir recipients.
Approximately 5% of ganciclovir recipients experience some combination of headache, confusion, altered mental status, hallucinations, nightmares, anxiety, ataxia, tremors, and seizures. About 2% of recipients develop fever, rash, and abnormal levels of serum hepatic enzymes. Intraocular injection of ganciclovir can cause transient increases in intraocular pressure with associated intense pain and amaurosis lasting up to 30 minutes.
In preclinical test systems, ganciclovir is mutagenic, carcinogenic, and teratogenic. It also causes irreversible reproductive toxicity in animals.
The most common side effects associated with valganciclovir therapy include diarrhea (41%), nausea (30%), neutropenia (27%), anemia (26%), and headache (22%). Up to 5 years of treatment with valganciclovir is well tolerated in patients with AIDS, and the type and incidence of adverse events experienced long term appear to be similar to those observed at 1 year.
Ganciclovir is indicated for the treatment and prevention of life- and sight-threatening CMV infections occurring in immunosuppressed patients. It is approved in the US for the treatment and suppression of retinitis caused by CMV in immunocompromised patients and for the prevention of CMV disease in transplant recipients. It is frequently used for other serious CMV infections.
Almost 90% of patients with CMV retinitis have improvement or stabilization of the ocular disease and clearing of the condition, demonstrated by a >100-fold reduction in viral titers, from urine, blood, the and throat after 1–2 weeks of ganciclovir therapy. Because relapse of retinitis is virtually inevitable in patients with AIDS, ganciclovir should be continued long term for CMV suppression after induction therapy.
Patients in whom ganciclovir therapy has failed may benefit from treatment with a combination of foscarnet and ganciclovir. Patients with AIDS who have CNS disease caused by CMV also have been treated successfully with a combination of ganciclovir and foscarnet. As many as 25% of ganciclovir recipients develop resistance within 9 months of the initiation of therapy. ,
Patients with AIDS and solid-organ transplant recipients who have gastrointestinal disease attributed to CMV appear to benefit from ganciclovir therapy. , Ganciclovir monotherapy does not appear to benefit bone marrow recipients with CMV gastrointestinal infections.
Limited and uncontrolled data suggest that ganciclovir therapy may be useful in patients with AIDS and CMV pneumonia. Bone marrow transplant recipients with CMV pneumonia fail to respond to ganciclovir therapy alone but may benefit from therapy with intravenous CMV hyperimmunoglobulin and ganciclovir given together. ,
Ganciclovir has been evaluated in the treatment of neonates congenitally infected with CMV. In a phase 3 randomized, controlled trial, ganciclovir therapy (6 mg/kg per dose administered every 12 hours intravenously for 6 weeks) protected infants from hearing deterioration beyond 1 year of life, and it may improve neurologic outcomes. Transient positive effects on growth also were seen, as was a decreased time to resolution of elevated serum hepatic transaminase levels. Approximately two-thirds of treated patients, however, developed neutropenia, and one-half required dose modification.
A phase 1/2 pharmacokinetic and pharmacodynamic study of oral valganciclovir established that a 16-mg/kg dose of oral valganciclovir produced similar systemic exposure to that of a 6-mg/kg dose of intravenous ganciclovir. This dose of valganciclovir was then evaluated in a randomized, double-blind, controlled trial enrolling infants with symptomatic congenital CMV disease. Durations of therapy for 6 weeks and 6 months were compared. The longer duration of therapy improved audiologic and neurodevelopmental outcomes more than that seen with 6 weeks of treatment. Neutropenia was less prevalent than with parenterally administered ganciclovir.
Ganciclovir is useful for the prevention of CMV infections in high-risk immunocompromised patients, including bone marrow and solid-organ transplant recipients ( Table 295.8 ). Strategies that have been used to reduce the frequency of posttransplantation disease include routine administration of ganciclovir to all recipients at risk and pre-emptive administration to those who have a positive culture for CMV after transplantation. Pre-emptive therapy has been demonstrated to reduce CMV disease in recipients of liver, lung, heart, and bone marrow transplants. CMV-seropositive recipients of heart, lung, or liver transplants who receive routine prophylaxis with ganciclovir for 1 month also have significant reductions in posttransplant CMV infection and disease. , Pre-emptive administration of ganciclovir improves survival of bone marrow transplant recipients, , whereas routine initiation of prophylactic ganciclovir before bone marrow transplantation in patients who are CMV seropositive does not impact survival. ,
The usual therapeutic and prophylactic dose of intravenous ganciclovir is 10 mg/kg/day divided every 12 hours for 2–3 weeks. For continued suppressive therapy to prevent relapse of infection (e.g., patients with AIDS) or long-term prophylaxis, either of the following may be used: 5 mg/kg as a single daily dose each day of the week or 6 mg/kg administered as a single dose 5 days per week. Prophylactic oral ganciclovir (1000 mg 3 times daily) significantly reduces the risk of CMV disease in patients with advanced AIDS.
Indications for valganciclovir are similar to those for ganciclovir. However, based on limited controlled trials, valganciclovir is approved only for induction and maintenance therapy for CMV retinitis. The recommended dose of valganciclovir for induction therapy is 900 mg twice daily for 2 weeks. The recommended dose for maintenance therapy is 900 mg once daily. Orally administered valganciclovir appears to be as effective as intravenous ganciclovir for induction treatment and is convenient and effective for the long-term management of CMV retinitis in patients with AIDS.
The greater systemic exposure to ganciclovir delivered by valganciclovir when used prophylactically is safe and is associated with delayed development of viremia compared with oral ganciclovir in solid-organ transplant recipients. Valganciclovir also is effective as pre-emptive therapy and as treatment for CMV disease in solid-organ transplant recipients.
Letermovir inhibits the CMV DNA terminase complex (pUL51, pUL56, and pUL89) utilized by CMV in the terminal phase of the replication life cycle of viral DNA processing and packaging. This enzyme complex cuts the CMV genome units, which are generated in tandem before being packaged into preformed virus capsids. Letermovir is highly specific to CMV and is approximately 1000-fold more potent than ganciclovir against CMV but has no significant antiviral activity against other herpesviruses or non-human CMV strains. In vitro studies have identified letermovir resistance mutations mapping primarily to the UL56 gene and more rarely to the UL89 and UL51 genes encoding the three subunits of the HCMV terminase complex. Clinical isolates with letermovir resistance have been reported. Emerging data suggest that letermovir may have a low viral genetic barrier to resistance, and, therefore, additional clinical experience with this new drug is needed before a complete understanding of resistance will be reached. Letermovir is fully active against viral populations with substitutions conferring resistance to CMV DNA polymerase inhibitors (cidofovir, foscarnet, and ganciclovir), and conversely these DNA polymerase inhibitors are fully active against viral populations with substitutions conferring resistance to letermovir.
Letermovir has a good oral bioavailability (35%). Maximum plasma concentrations (C max ) of letermovir are achieved approximately 1.5 hours after oral administration, and the half-life (t 1/2 ) is 10 hours. Letermovir is highly bound to protein, with the estimated percentages of drug bound to plasma proteins being 99% in humans. Letermovir is eliminated primarily unchanged via biliary excretion into feces, and excretion into urine is negligible.
Letermovir is an inhibitor of OATP1B1/3 transporters, and co-administration with drugs that are substrates of OATP1B1/3 transporters may result in a clinically relevant increase in plasma concentrations of co-administered OATP1B1/3 substrates. These drugs include amiodarone, warfarin, phenytoin, ergotamine, atorvastatin, pitavastatin, simvastatin, rosuvastatin, tacrolimus, omeprazole, and CYP3A substrates such as fentanyl, midazolam, and quinidine. No clinically significant interactions were observed in clinical drug-drug interaction studies of letermovir and acyclovir, digoxin, mycophenolate mofetil, posaconazole, ethinyl estradiol, and levonorgestrel.
Letermovir was licensed in the US in November 2017, in both oral and intravenous formulations, for the prophylaxis of CMV infection in adult CMV-seropositive recipients of an allogenic HCT. The adult dose of letermovir is 480 mg administered once daily orally or as an intravenous infusion over 1 hour, through 100 days post-transplant. If letermovir is co-administered with cyclosporine, the dosage of letermovir should be decreased to 240 mg once daily. In the phase 3 prophylaxis study evaluating the efficacy of oral and intravenous daily doses of letermovir in stem cell transplant recipients seropositive for CMV, the percentage of patients who developed clinically significant CMV infection was significantly lower in the letermovir group (38%) than in the placebo arm (61%) by week 24. All-cause mortality at week 24 posttransplantation was lower in the LMV groups (10%) compared with the placebo arm (16%). Letermovir currently is being evaluated in a phase 2 age-deescalating pediatric trial in children following hematopoietic stem cell transplantation being conducted by the manufacturer, and a National Institutes of Health-sponsored study of pharmacokinetics and safety in young infants with congenital CMV infection is in development.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here