Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Although endovascular intervention is an important part of the treatment of aneurysms, expertise and proficiency in open surgical management of cerebral aneurysms are essential requirements.
Each aneurysm location requires its own unique set of nuances and methods, which are essential for proper and safe clip ligation.
Cotton (in addition to patties) is an important adjunct to aneurysm repair and allows for retractorless surgery in many instances.
Becoming efficient and precise with Sylvian fissure dissection and opening will facilitate all aneurysm surgeries, and these are basic techniques that need to be mastered.
Intracranial saccular or berry aneurysms are common, occurring in approximately 1% to 3% of the population. Based on a review of 1450 unruptured intracranial aneurysms (UIAs) in 83 study populations, the overall prevalence is estimated to be 3.2%, with a mean age of 50 years and a higher prevalence in women, in patients with autosomal dominant polycystic kidney disease, and in those with a family history of a ruptured cerebral aneurysm. Other associated disorders with a higher incidence of intracranial aneurysms include multiple endocrine neoplasia type I, hereditary hemorrhagic telangiectasia, Ehlers-Danlos syndrome type IV, neurofibromatosis type I, and Marfan syndrome.
Anterior circulation aneurysms comprise roughly 85% of intracranial aneurysms and are classified based on their anatomic location, most commonly at the anterior communicating artery, middle cerebral artery bifurcation, ophthalmic artery, and the branching points of the internal carotid artery (ICA), including the ophthalmic, superior hypophyseal, posterior communicating, and anterior choroidal arteries and the ICA bifurcation ( Table 17.1 ). The cavernous segment of the ICA is also a common location for aneurysm formation. These aneurysms are generally extradural and have a different natural history and set of therapeutic options.
| Aneurysm Location | Frequency a |
|---|---|
| Cavernous part of carotid artery | 8.3% |
| Internal carotid artery | 29.9% |
| Anterior communicating or anterior cerebral artery | 12.3% |
| Middle cerebral artery | 29.0% |
| Posterior communicating artery | 8.5% |
| Vertebrobasilar system (other than basilar tip) | 4.9% |
| Tip of basilar artery | 7.0% |
a Reported frequencies result from the International Study of Unruptured Intracranial Aneurysms (ISUIA) trial of 4060 patients.
Intracranial aneurysms are increasingly found on cross-sectional imaging studies, including computed tomography (CT) and magnetic resonance imaging (MRI), which are performed for other clinical reasons; however, as many as 50% of aneurysms are found after an initial subarachnoid hemorrhage. Although screening studies of CT and MRI are useful, computed tomography angiography (CTA) and magnetic resonance angiography (MRA) can typically define the location and morphology of aneurysms with a greater degree of certainty. Aneurysms that are >3 to 4 mm are commonly identified on these noninvasive imaging studies, depending on the equipment, software, and clinical experience. For routine screening or following intracranial aneurysms, noninvasive imaging studies are preferred when possible, and although these can also be used for treatment planning in some situations, catheter angiography remains the gold standard for defining aneurysms and their morphology. The advent of three-dimensional (3D) rotational angiography has significantly improved the ability to resolve and model the angioarchitecture, associated perforators, and parent vessels.
A comprehensive understanding of the vascular anatomy along with a robust knowledge of aneurysm location and nuances are of supreme importance. There are general tenets that apply to all intracranial aneurysms, as well as location-specific nuances should be considered. Anatomy, both general vascular anatomy and that of aneurysms and other cerebral vascular disease, should be a fundamental foundation that can help with the study and mastery of cerebral aneurysms and their treatment.
The origin of the ICA in the neck is usually lateral to the external carotid artery at the bifurcation and courses cephalad toward the skull base. Carotid-basilar anastomoses are important to recognize, both diagnostically and in treatment planning. Most common is the primitive trigeminal artery (PTA), which connects the cavernous ICA with the embryonic dorsal longitudinal neural arteries. Beginning at the bifurcation, the carotid is divided into seven numbered and named segments, each identifying its anatomic location: C1, cervical segment; C2, petrous segment (vertical and horizontal segments); C3, lacerum segment; C4, cavernous segment; C5, clinoidal segment; C6, ophthalmic segment; and C7, the communicating segment ( Fig. 17.1 ).
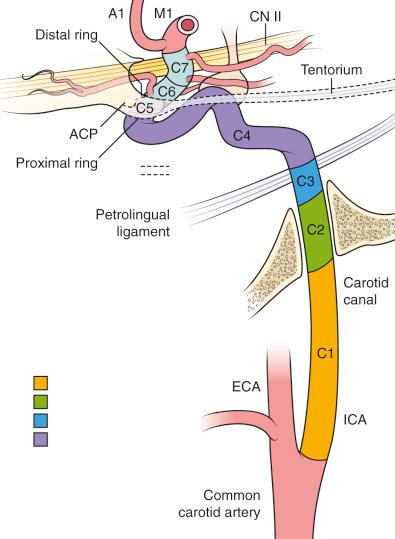
Anatomically, the cervical segment (C1) begins at the carotid bifurcation (at approximately the fourth cervical vertebra) and courses to the skull base. The petrous segment (C2) begins at the entrance of the carotid into the carotid canal at the base of the skull, where it courses vertically then horizontally (anteromedially). The ICA then exits the carotid canal, to begin the lacernal segment (C3), where the carotid continues over the foramen lacerum; this short segments ends at the petrolingual ligament. The cavernous segment (C4) passes through the cavernous sinus, from the lateral dural ring proximally, and exits at the distal dural ring. The clinoidal segment (C5) represents a very short segment that extends from the distal to the proximal dural rings, beneath the anterior clinoid process. The ophthalmic segment (C6) starts at the proximal dural ring, whereby the carotid enters the subarachnoid space, and ends at the posterior communicating artery. The absence of a reliable angiographic landmark at the clinoidal-ophthalmic (C5–C6) junction makes this difficult to identify on angiography; adjunctive studies such as MRI or CTA can be helpful. The C6 segment gives rise to the ophthalmic artery, and this has traditionally been used to help identify the beginning of this segment. In practice, cerebral aneurysms that are at, or distal to, the ophthalmic artery are considered to be at least partially intradural and within the subarachnoid space. The carotid siphon, also known as the anterior loop (in the Dolenc classification), contains the distal cavernous segment, the entire clinoidal segment, and the proximal ophthalmic segment. The communicating segment (C7) begins at the posterior communicating artery and concludes at the ICA bifurcation, where the ICA divides into the anterior and middle cerebral arteries.
Branches that are diagnostically or clinically relevant are important to understand and identify. The cervical segment (C1) has no regular branches. The petrous segment (C2) has the caroticotympanic artery and, inconsistently, the vidian artery. The cavernous segment (C4) has several branches including the meningohypophyseal artery, which divides into the inferior hypophyseal artery, the dorsal meningeal artery and the tentorial artery of Bernasconi and Cassinari, the inferolateral trunk, and the capsular arteries of McConnell. Although these represent important functional vascular components and can be involved in dural arteriovenous fistulas as well as blood supply to meningiomas, they do not regularly play a role in cerebral aneurysms. Additionally, cavernous segment aneurysms are not infrequently identified, but, with exceptions that are typically limited to giant aneurysms, they are extradural. Unfortunately, large or strategically placed aneurysms can occasionally become symptomatic from focal pressure on structures within the cavernous sinus and may require treatment—typically using endovascular techniques. Although the ophthalmic segment (C6) gives rise to the ophthalmic artery in 90% of anatomic dissections, in a small percentage of cases it may originate in the clinoidal segment (C5) or, rarely, more proximally. A branch of the ophthalmic artery, the lacrimal artery, gives rise to the recurrent meningeal artery, passes retrograde through the superior orbital fissure, and anastomoses with branches of the middle meningeal artery, which can have important clinical implications. Additionally, a number of extraorbital ophthalmic artery branches have extensive anastomoses with ethmoidal and facial branches of the external carotid artery and can be an important source of blood flow to the distal ICA in the event of a more proximal occlusion. The second important named artery of the C6 segment is the superior hypophyseal artery (SHA); this and the ophthalmic artery can be sites of aneurysm formation. The communicating segment (C7) has two branches. First, the posterior communicating artery branches inferolaterally and immediately wraps beneath, traveling posteriorly to merge with the posterior cerebral artery (PCA) at the P1–P2 junction. A fetal PCA is a normal variant of the circle of Willis, where the ipsilateral P1 is atretic or hypoplastic, and the dominant supply to the distal posterior cerebral artery is from the posterior communicating artery. As such, when a fetal PCA is identified, it must be preserved. The second branch is the anterior choroidal artery, which may be duplicative and is also a common site of aneurysm formation. The ICA terminus is the bifurcation of the A1 and M1, and it has distal branches that perforate the anterior perforating substance and supply the internal capsule.
The anterior cerebral artery (ACA) arises from the ICA above the optic nerve as A1 and travels ventromedially to the anterior communicating artery complex (ACOMM); during this course it has several (as many as 12) medial lenticulostriate arteries that branch both posteriorly, supplying the anterobasal brain, and inferiorly, supplying the optic chiasm and nerves ( Fig. 17.2 ). Additional perforators can come posteriorly from the ACOMM to supply the hypothalamus and the columns of the fornix as well as other structures, and as such, obliteration of the communicating artery in aneurysm management should be avoided whenever possible. The recurrent artery (of Heubner) is the largest and typically the longest ACA perforator, which branches from the proximal A2 or distal A1. The ACOMM can be hypoplastic or atretic in some patients, but the patency of the lenticulostriate arteries can be assumed to be present.
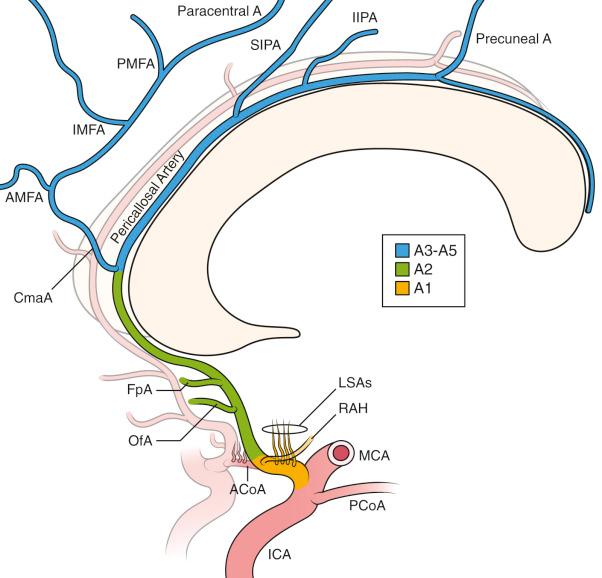
After branching to the contralateral A1 segment through the communicating artery, within the interhemispheric fissure, the A2 segment transverses the interhemispheric fissure, paired with its contralateral counterpart. Variability of the communicating segment is the rule, and a number of anatomic variants have been commonly described, with duplication, triplication, fenestrations, loops, and other anomalies often found. The A2 segment extends from the ACOMM of the corpus callosum. The orbitofrontal and frontopolar arteries branch consistently from this segment. At the rostrum of the corpus callosum, the A2 branches into the pericallosal artery, now A3, and its more superficial branch, the callosomarginal artery. This represents a common site of aneurysm formation in this segment, and anatomic variability typically is seen with the location of the branch site. The callosomarginal artery, which travels in the cingulate sulcus, branches into the medial frontal artery and the paracentral artery. A common anatomic variant is the absence of the callosomarginal artery, with terminal branches arising directly from the pericallosal artery. Distal A3 branches include prefrontal (often from the callosomarginal artery), paracentral, and parietal branches.
The second main trunk from the ICA bifurcation is the middle cerebral artery (MCA). The MCA begins at this major branch point and travels within the Sylvian fissure (lateral fissure) to yield multiple branches ( Fig. 17.3 ). The branching and origin of the MCA are relatively consistent anatomically, although variants exist including duplication or hypoplasia. The M1 segment travels deep within the Sylvian fissure, and its orientation and length are variable. The medial lenticulostriate arteries branch from the M1 segment are of great importance clinically, and their presence and preservation should be emphasized. Knowledge of these when dissecting along the M1, as well as preservation with manipulation of this larger segment, prevents damage. Larger named branches along this segment include the anterior temporal and polar temporal arteries, either of which can serve as bypass donor arteries if needed.
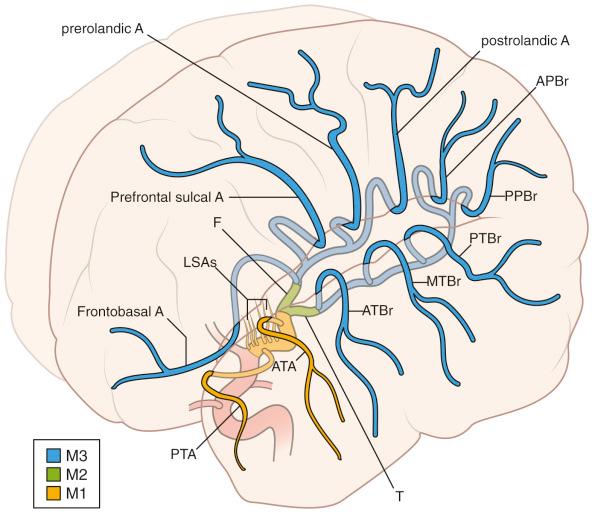
The MCA bifurcation, like the anterior communicating artery complex, is variable. Classically, the bifurcation branches into a superior and an inferior division, whereas in approximately 15% of patients a trifurcation is identified. Other patterns are also seen, including separating into multiple small branches. Generally, when a superior trunk is identified, it will supply branches in the lateral frontobasal artery, prefrontal sulcal artery, and precentral and central (rolandic) terminal branches. The inferior trunk forms inferior temporal branches (anterior, middle, and posterior), an angular branch, and often two parietal branches (anterior and posterior). Variability is the rule with terminal branches, and variances are expected. Identifying the vascular anatomy for a given patient is of particular importance in patients with MCA pathology, as small branches are often obscured from the surgeon's view.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here