Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Acute anorectal pathology constitutes a significant proportion of the general surgeon’s workload. The problems encountered range from the acute pain of thrombosed haemorrhoids and perianal sepsis to the management of anorectal bleeding, trauma and irreducible rectal prolapse. Effective management depends upon sound knowledge of anorectal anatomy, accurate classification of the pathology and timely intervention to ensure minimal disruption to normal function. Given its mode of presentation, clinical trials in the management of acute anorectal pathology are infrequent, yet where present are discussed here.
The anal canal is a 3–4 cm tube running downwards and backwards from the anorectal angle to the anus. It is divided in half by the dentate line, which demarcates hindgut-derived columnar epithelium above and stratified squamous epithelium below that merges at the anus with the perianal skin. Above the dentate line is the anal transitional zone, lined by cuboidal epithelium for 1–2 cm. The hindgut sensory innervation is supplied by autonomic hypogastric nerves, sensitive only to stretch. Anoderm below the dentate line is innervated from the somatic inferior rectal nerves, making it sensitive to pain, pressure and temperature. However, the dentate line is not a reliable boundary between somatic and autonomic sensation; pain can certainly be felt within the anal transitional zone, which explains why some patients experience severe and immediate pain after rubber band ligation of haemorrhoids despite the band being applied apparently well above the dentate line.
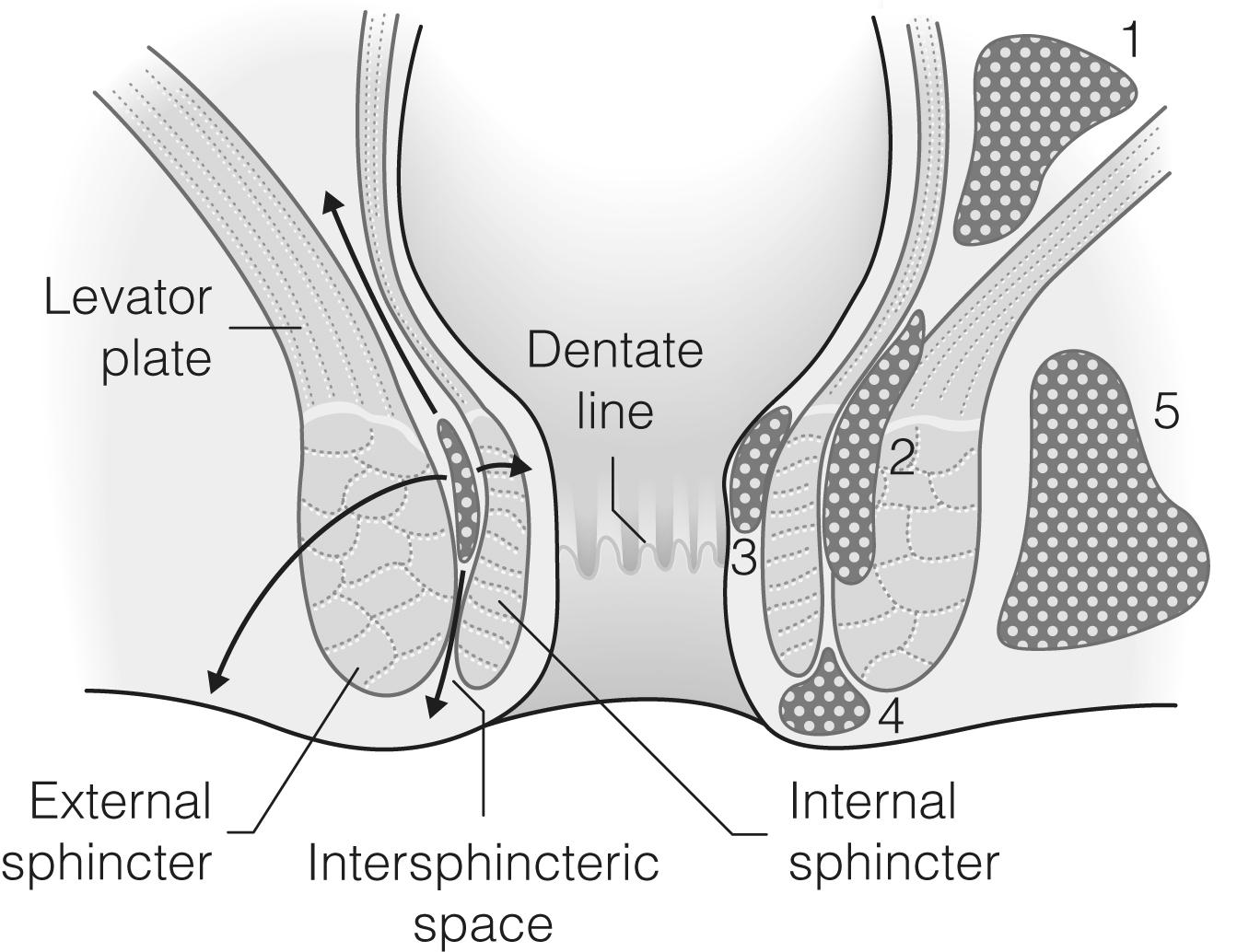
The anorectal sphincter complex can be thought of as a gut tube within a funnel of muscle essential for the maintenance of normal continence. The inner muscle layer is the involuntary internal sphincter, a thickened continuation of the circular muscle of the rectum. Outside this is the funnel of the voluntary external sphincter, formed from striated muscle, continuous at its superior edge with the levator plate. Between these two muscle layers is the intersphincteric space, which contains mucous-secreting anal glands. These glands drain through the internal sphincter via their respective crypts of Morgagni at the level of the dentate line.
Three submucosal anal cushions comprising fibroelastic tissue and arteriovenous anastomoses are found within the anal canal, usually at the 3, 7 and 11 o’clock positions. They normally appose to form a tight seal within the canal, which helps to maintain continence, but in some patients they may enlarge to form troublesome symptomatic haemorrhoids.
Anorectal abscesses, defined as sepsis within the soft tissues that surround the anus, are among the commonest surgical emergencies, with operative treatment being required in about 1 in 5000 of the population in the UK annually. They occur predominantly in adults, frequently in the third and fourth decades of life, with a male preponderance. An anorectal abscess is thought to originate within the intersphincteric space, due to obstruction and suppuration of the anal crypt glands (the cryptoglandular theory). Furthermore, 90% of anorectal abscesses arise as a result of this process and are termed primary anorectal abscesses. The resultant primary infection will produce an intersphincteric abscess which can spread, in a vertical, horizontal or circumferential direction, along several anatomical planes to collect in a number of potential anorectal spaces: perianal, intersphincteric, pelvi-rectal (ischiorectal, or post anal) or supralevator. Circumferential spread results in a horseshoe abscess ( Fig. 17.1 ). The relative frequency with which abscesses occur in the various anatomical locations is shown in Table 17.1 .
| Anatomical site | Number | Percentage |
|---|---|---|
| Perianal abscess | 437 | 42.7 |
| Ischiorectal | 233 | 22.8 |
| Intersphincteric | 219 | 21.4 |
| Supralevator | 75 | 7.3 |
| Submucosal | 59 | 5.8 |
Both aerobic and anaerobic bacteria are responsible for the abscess formation, with the predominant anaerobic bacteria being Bacteroides spp., Peptostreptococcus and Clostridium ; the most commonly isolated aerobic and facultative bacteria are Staphylococcus aureus , Streptococcus , Enterobacteriaceae and Enterococcus .
Suppurating skin infections, including carbuncles, furuncles and infected apocrine glands, can also cause primary abscesses. The responsible bacterium in these cases is almost invariably staphylococcus and as these abscesses do not communicate with the anal canal they are not associated with fistula formation.
Secondary abscesses are much less common, accounting for just 10% of presentations. They are a manifestation of distinct underlying disease processes, with Crohn’s disease, colorectal neoplasia, diabetes mellitus, AIDS and tuberculosis all being potential causes. They can also occur as a complication of anorectal surgery or as a consequence of trauma.
Anorectal abscesses present with signs and symptoms of acute inflammation, with pain being the most common symptom. On examination a perianal abscess can usually be seen as a red, tender, fluctuant swelling near the anal verge; however, ischiorectal abscesses often present as a less distinct brawny swelling on one side of the anus and intersphincteric abscesses usually cannot be seen externally at all. Although this last type of abscess can be felt through the anal wall as a smooth, tender collection, digital rectal examination is usually excruciatingly painful. This diagnosis should therefore be suspected in patients with severe anal pain and fever. Satisfactory examination is usually only possible under general anaesthesia. In a few patients (particularly those who are immune-compromised or with diabetes mellitus), the perianal sepsis can be associated with cellulitis, which can progress to life-threatening necrotising infection if not treated promptly.
All patients presenting with anorectal sepsis or pain should have an examination of the anorectum, including proctosigmoidoscopy. It is our opinion that this can usually only be performed satisfactorily under general anaesthesia. As the diagnosis is usually obvious, few people would routinely recommend preoperative imaging (although some advocate its use, arguing that by identifying cavities and fistulas there is a reduction in the incidence of recurrence ).
Diagnostic radiology is seldom required to image uncomplicated anorectal sepsis. Its role in the management of acute anorectal sepsis is for investigating complex fistula disease in Crohn’s disease or patients with supralevator sepsis to differentiate an intrapelvic source from extension of an intersphincteric or ischiorectal abscess. Imaging is also useful if the diagnosis is unclear, if there is non-resolution of symptoms after operative intervention or in cases of recurrent sepsis. Magnetic resonance imaging (MRI) is a sensitive and specific modality for identifying anorectal abscesses and associated fistula tracts and is the technique of choice in UK practice and beyond. ,
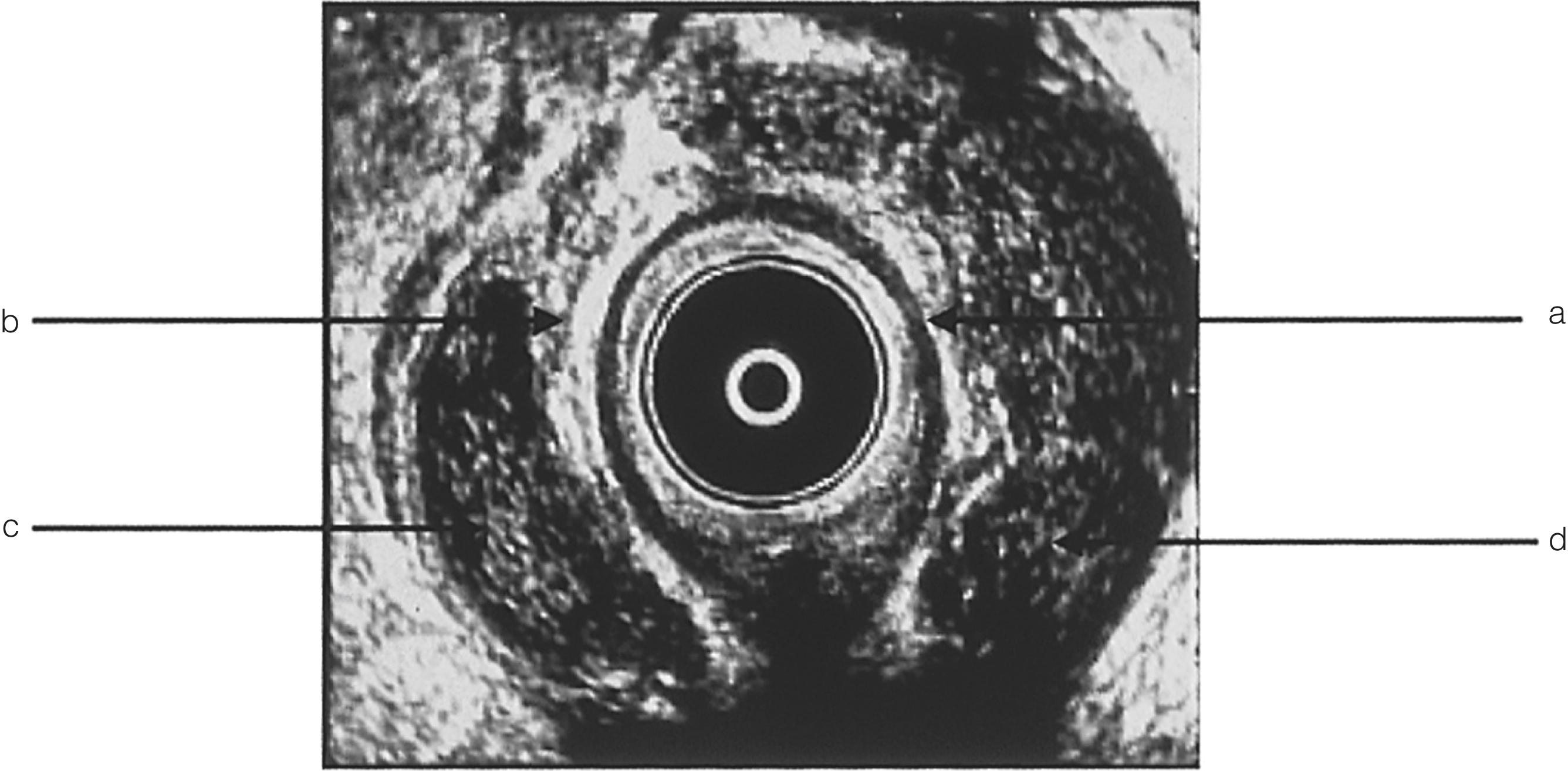
Endoanal ultrasound is an alternative method of determining the origin of the sepsis and delineating fistula anatomy ( Fig. 17.2 ). In experienced hands, it is as accurate as MRI. ,
Undrained pus is remarkably destructive and hence sepsis must be drained promptly whilst minimising iatrogenic damage to the sphincter complex and preventing recurrence. In most patients, this involves external drainage via a curvilinear (not radial) incision at the lateral edge of the abscess cavity. This should not be placed too laterally as it will make subsequent fistula management more challenging. Needle aspiration alone is not recommended due to risk of inadequate drainage and abscess recurrence. Adequate drainage should result in prompt resolution of the patient’s symptoms; if not, a repeat examination under anaesthetic (EUA) or MRI is recommended to ensure that all sepsis has been completely drained.
Historically, it was suggested that thoroughly ‘deroofing’ the abscess cavity with a wide cruciate incision was beneficial, but this does little other than giving the patient a larger wound that will take longer to heal. Primary wound closure after incision has been abandoned as it offers little immediate benefit in terms of time to wound healing and probably increases the chance of recurrent sepsis.
If the abscess cavity is very large, an alternative to making a huge incision is to insert a de Pezzer or Malecot catheter via a smaller skin incision. One study showed that using such treatment reduced hospital stay (1.4 vs 4.5 days) and duration of wound management, with no disadvantages seen at long-term follow-up.
Antibiotics are not indicated routinely and should be used only as an adjunct to surgical treatment in immune-compromised patients, or when there is evidence of florid cellulitis or suspicion of necrotising infection.
It is the opinion of the authors that all anorectal abscesses should be drained under general or spinal anaesthetic, although in some centres perianal abscess drainage is commonly performed under local anaesthesia. Drainage of perianal abscesses under general or spinal anaesthetic allows a comprehensive examination to be undertaken and prevents a significant proportion of re-operations. A large retrospective case study of 500 patients treated for perianal abscess at the Mayo Clinic was reported in 2001 and revealed a 7.6% re-operation rate. The reasons for re-operation included incomplete drainage of the abscess cavity at the first operation, missed abscesses (most often posterior collections) and postoperative bleeding. There was no association reported between patient variables (such as age, immune suppression or diabetes), emphasising the need for a thorough primary examination.
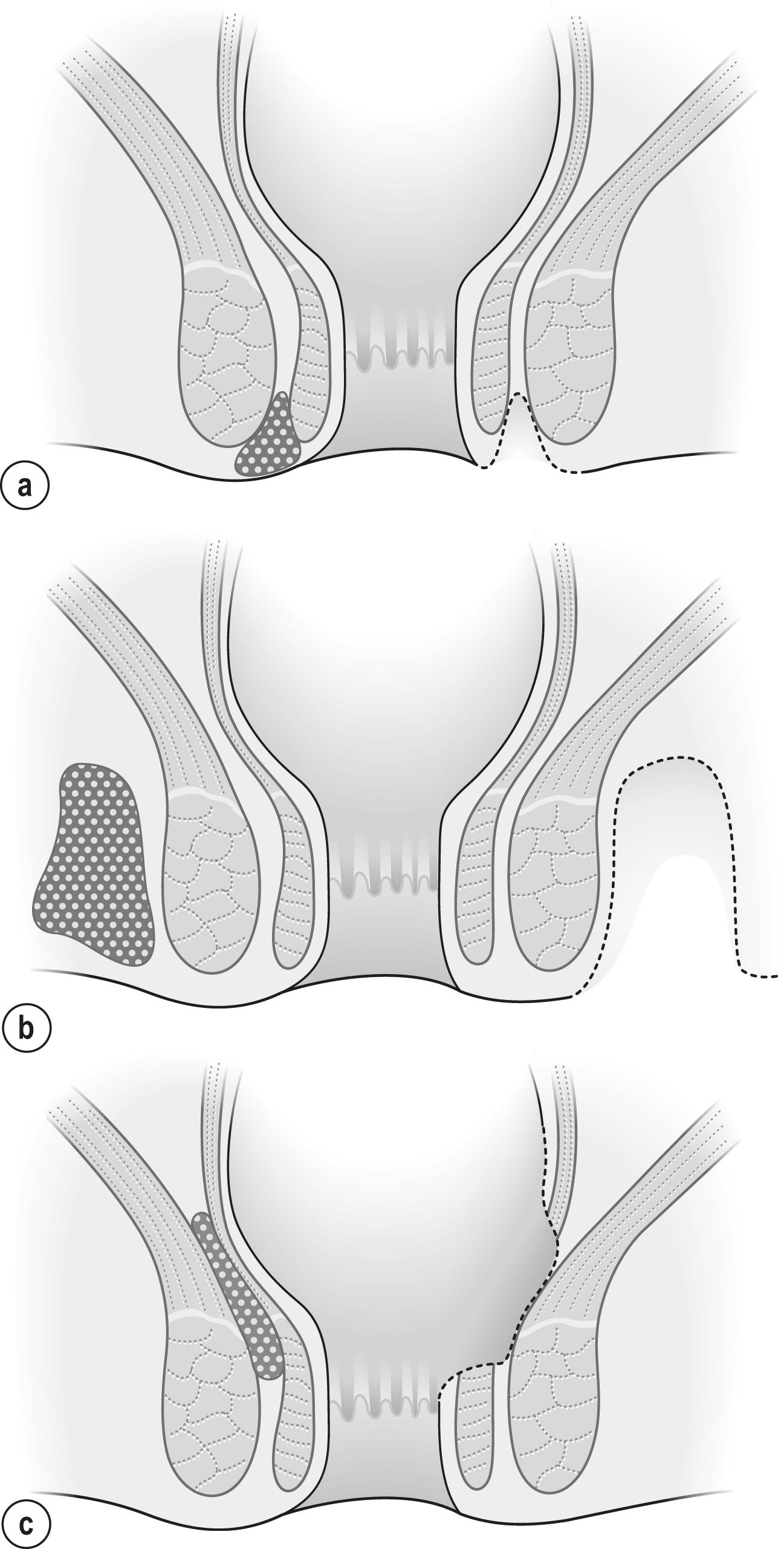
The management of specific abscesses is shown diagrammatically in Fig. 17.3 . Simple perianal abscesses should be drained externally and the cavity gently curetted ( Fig. 17.3a ). With ischiorectal abscesses, the cavity is often large ( Fig. 17.3b ) and should be incised as near to the anal verge as possible in order to minimise the distance to the external opening of any subsequent fistula. A large horseshoe abscess in the ischiorectal space is better drained through multiple short incisions than one large circumferential incision. As mentioned earlier, the size of the drainage wound can be minimised if a drainage catheter is used. Intersphincteric abscesses are more challenging and require drainage into the anorectum, which requires excision of part of the internal sphincter ( Fig. 17.3c ). Submucosal abscesses, although rare, are drained into the anal canal. Supralevator abscesses pose a unique challenge compared with the drainage of other anorectal abscesses. They develop in a space above levator ani, lateral to the rectum. Drainage transrectally is associated with high recurrence rates, whilst concomitant drainage through the ischiorectal fossa results in high fistula-in-ano, which can be difficult to manage. It is therefore recommended that such abscesses be approached radiologically or via an open transabdominal approach. In true pelvic abscesses unrelated to spread from the anal glands, drainage can be achieved into the rectum or vagina. If the abscess is related to pelvic pathology, the primary disease process will need to be resected along with drainage of the pus to prevent recurrence.
Routine bacteriological swabs at the time of abscess drainage do not predict fistula or recurrence rates, and are therefore unnecessary. , In a recent meta-analysis, an empiric course of postoperative antibiotics following drainage was associated with a reduction in fistula formation in otherwise healthy patients, although quality of the evidence is low.
Internal packing of anorectal abscess cavities has historically been commonly practised within the UK. However, a 2016 Cochrane review concluded that there is a paucity of high-quality evidence to confirm whether packing influences time to healing, recurrence or fistula rates. A recent UK multicentre observational study (2016) demonstrated significant levels of pain, resource utilisation and expense associated with internal postoperative packing and as a result, an ongoing multicentre, phase III randomised control trials was designed to confirm this (PPAC2, currently recruiting). In our opinion, postoperative packing should be omitted as it serves only to cause discomfort, inconvenience and cost, with no clear clinical advantage.
![]() Perianal abscesses are best incised under general anaesthesia with the cavity left open.
Perianal abscesses are best incised under general anaesthesia with the cavity left open.
![]() Routine bacteriology swabs do not alter management and are unnecessary. ,
Routine bacteriology swabs do not alter management and are unnecessary. ,
![]() There is no evidence that packing the abscess cavity is clinically beneficial.
There is no evidence that packing the abscess cavity is clinically beneficial.
![]() Needle aspiration rather than incision and drainage risks abscess recurrence.
Needle aspiration rather than incision and drainage risks abscess recurrence.
Perianal abscesses are associated with anal fistulas in about 60% of patients from the outset , ; only 27–37% of these persist after the acute inflammation has resolved. Fistula-in-ano increases the likelihood of recurrent anorectal sepsis, a non-healing wound or persistent discharge, and thus the requirement for repeat interventions. For these reasons, many surgeons consider fistula treatment at the primary drainage procedure. However, identifying a fistula when acute inflammation is present can be difficult, especially for the less experienced surgeon. The probing of fistula tracts within friable, oedematous tissue can also lead to the creation of false fistula tracts which can be difficult to manage, and may cause disproportionate damage to the anal sphincter complex.
![]() The majority of anorectal abscesses are adequately treated with incision and drainage alone and if a fistula tract is not obvious, it should not be sought.
The majority of anorectal abscesses are adequately treated with incision and drainage alone and if a fistula tract is not obvious, it should not be sought.
A Cochrane review of six randomised control studies from five different centres from 1987 to 2003 looked at concomitant fistula surgery at the time of abscess drainage in terms of recurrence, need for further surgery and postoperative incontinence. , The eligibility criteria and treatments offered varied considerably between the trials, making definitive recommendations difficult. Three studies also reported fistula rates of 83–90%, which is higher than would be expected, raising questions about whether iatrogenic tracks may have been created.
The majority of the studies included in the review dealt only with the surgical treatment of low fistulas. In one study, fistulotomy was performed as a second procedure on day 3 of the acute admission. Most studies excluded those with recurrent anorectal sepsis, previous surgery and inflammatory bowel disease (IBD).
All the studies showed that recurrence was less likely after concomitant fistula surgery (risk ratio 0.07–0.24), although follow-up times varied. The overall conclusions of the Cochrane meta-analysis were that synchronous fistulotomy is appropriate for low, uncomplicated fistula tracts. It should not be performed for high fistulas or anterior fistulas in women. It should also be avoided for groups where the risk of incontinence is high (e.g. those who have undergone previous anorectal surgery or have IBD).
![]() Synchronous fistulotomy can be performed at the time of abscess drainage for low, uncomplicated fistula tracts to reduce the risk of abscess recurrence and further operations without compromising continence.
Synchronous fistulotomy can be performed at the time of abscess drainage for low, uncomplicated fistula tracts to reduce the risk of abscess recurrence and further operations without compromising continence.
These abscesses should be drained like any other anorectal abscess, but definitive treatment will require appropriate management of the malignant lesion. The presence of anorectal sepsis in this context, however, indicates advanced disease either from direct tumour infiltration or perforation, which has implications for further treatment. Consideration should be given to a defunctioning stoma at the time of sepsis drainage in this subset of patients.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here