Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Disorders of the immune system are diverse and range from mild to severe in their manifestations and impact on normal function. In this chapter, we review the physical findings and characteristic symptoms of children with hypersensitivity reactions and immune deficiencies, as well as diagnostic techniques and radiographic findings. Topics have been chosen on the basis of their prevalence and importance in the pediatric population and their association with characteristic physical findings.
Hypersensitivity disorders of the human immune system have been traditionally classified by Gell and Coombs into four broad groups ( Table 4.1 ). This provides a simplified framework for understanding these reactions. Type I reactions occur quickly after the sensitized individual is exposed to an antigen and are mediated by specific immunoglobulin E (IgE) antibody. Cross-linking of IgE on the surface of mast cells and basophils leads to release of histamine and other inflammatory mediators. This mechanism is responsible for the common disorders of immediate hypersensitivity, such as allergic rhinitis and urticaria. “Anaphylactoid” reactions are clinically similar and are caused by the degranulation of mast cells and basophils in the absence of specific IgE. Type II reactions involve antibodies directed against antigenic components of peripheral blood or tissue cells or foreign antigens, resulting in cell destruction. Examples of this type include autoimmune hemolytic anemia, pemphigus vulgaris, and Goodpasture syndrome. In type III reactions, antigen/antibody complexes form and are deposited in the lining of blood vessels, stimulating tissue inflammation mediated by complement and activated white blood cells. Examples of this type of reaction are serum sickness and the immune complex–mediated renal diseases. Type IV reactions involve T cell–mediated tissue inflammation and typically occur more than 48 hours after exposure. Examples of this type are tuberculin (purified protein derivative [PPD]) reactions and contact dermatitis (see Chapter 8 ).
| Hypersensitivity Type | Interval Between Antigen Exposure and Reaction | Antibody or Cell | Antigenic Target | Mechanism of Disease/Tissue Injury | Disorder(s) |
|---|---|---|---|---|---|
| Type I: Immediate, IgE-mediated | Within minutes to 1–2 h | Mast cell and basophil derived histamines, prostaglandins, leukotrienes | |||
| IgE-mediated | IgE | Foods, drugs, insect venoms, pollens | Anaphylaxis, Urticaria Rhinitis Bronchospasm |
||
| Anaphylactoid | Direct mast cell activation | Radiocontrast media, drugs (e.g., Vancomycin) | |||
| Type II: Antibody-mediated | Variable (days to weeks) | IgG, IgM | Cell surface proteins Extracellular matrix proteins |
Complement, phagocytes Leukocyte-derived inflammatory mediators |
Immune thrombocytopenia and hemolytic anemia Goodpasture syndrome |
| Type III: Immune complex-mediated | Variable (days to weeks) a | IgG complexed with circulating antigen | Nucleoproteins, other various endogenous or exogenous proteins | Complex deposition in vascular endothelium, complement activation, leukocyte-derived inflammation | Systemic lupus erythematosus Serum sickness |
| Streptococcal cell wall antigens | Poststreptococcal glomerulonephritis | ||||
| Type IV: Delayed, T cell–mediated | |||||
|
>48–72 h | Th1 cells | Contactants e.g., Nickel, Uroshiol (poison ivy) Mycobacterium tuberculosis |
Interferon-gamma, TNF-alpha, macrophages | Contact dermatitis (with IVc) Tuberculin skin test reactions |
|
1–6 weeks | Th2 cells | Antigen presented by cells or direct T cell stimulation | IL-4, IL-5, IL-13, eosinophils, inflammatory mediators | DRESS, chronic asthma, chronic allergic rhinitis |
|
4–28 days | Cytotoxic T cells | Cell-associated antigen or direct T cell stimulation | Perforin, granzyme B, FasL | Fixed drug eruptions, SJS/TEN, Bullous exanthems |
|
>72 h | T cell–mediated neutrophilic inflammation | Antigen presented by cells or direct T cell stimulation | Neutrophils, IL-8, cytokines, inflammatory mediators | Behcet’s, AGEP |
a More rapid reactions can occur if prior antigenic exposure/sensitization.
The development of type I hypersensitivity depends on hereditary predisposition, sensitization by exposure to an antigen, and subsequent reexposure to the antigen leading to an allergic reaction. Antigens that stimulate allergic reactions are known as allergens, and the mechanism of allergen-induced mediator release in type I hypersensitivity reactions is shown in Fig. 4.1 . IgE antibodies directed toward specific allergens are bound to the high-affinity IgE receptor on mast cells and basophils. When allergen causes cross-linking of IgE antibodies on the cell surface, the cell becomes activated, leading to the release of preformed mediators and the generation of the early and late mediators of anaphylaxis. The preformed mediators include histamine, tryptase, chymase, heparin, and other proteases that drive the earliest symptoms of anaphylaxis. A serum tryptase level is currently the best biologic marker of anaphylaxis, but it is still a relatively insensitive test. Serum tryptase levels should be obtained close to the onset of anaphylaxis as levels peaks 1 to 2 hours after the onset of symptoms and may return to baseline in 6 to 8 hours. The early and late mediators generated by mast cell activation include prostaglandins, leukotrienes, and cytokines. These mediators, which are generated over minutes to hours, continue to drive the clinical symptoms of the allergic reaction and initiate an inflammatory cascade that leads to the recruitment of eosinophils, basophils, and lymphocytes.
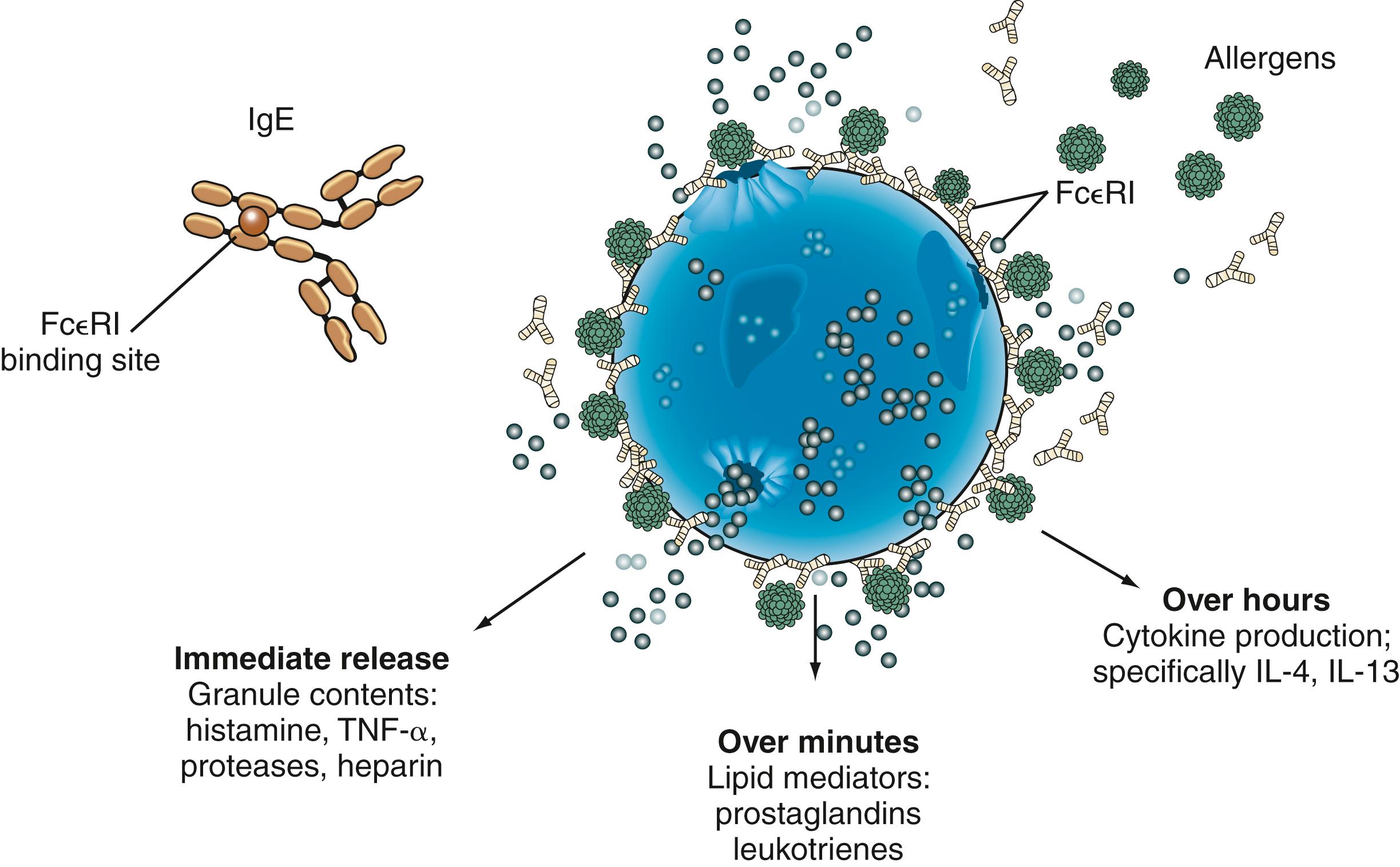
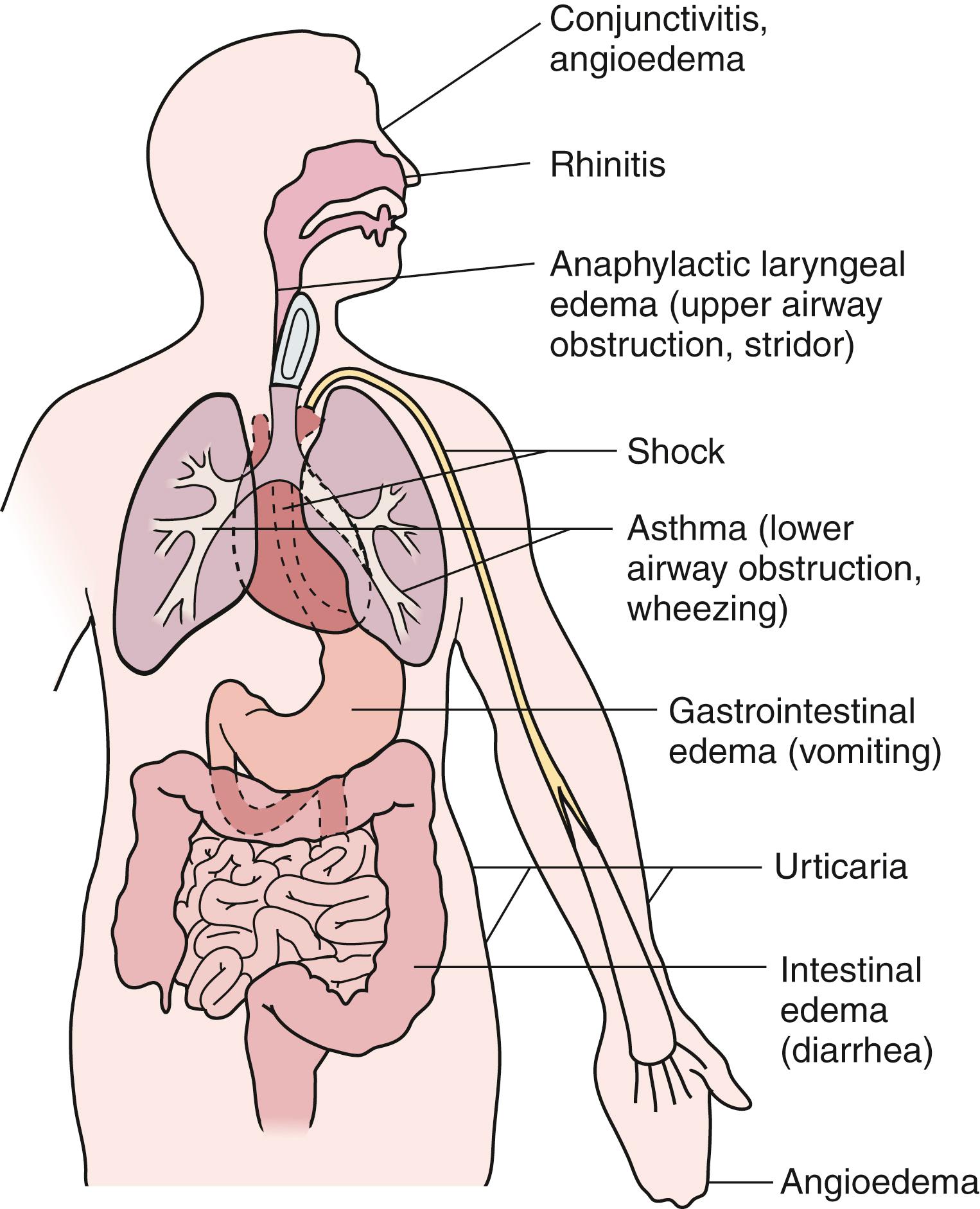
Type I reactions may occur in one or more target organs including the upper and lower respiratory tracts, cardiovascular system, skin, conjunctivae, and gastrointestinal (GI) tract. Manifestations depend on the systems involved, as shown in Fig. 4.2 . The most common manifestation of type I reaction is seasonal allergic rhinitis with a prevalence of at least 25%. The most serious manifestation of type I hypersensitivity is anaphylaxis, which can simultaneously involve all of the systems mentioned earlier.
Type I hypersensitivity has been diagnosed by skin testing for almost 150 years. The percutaneous skin test, also known as either a scratch or prick test, is an in vivo method to detect the presence of IgE antibody to specific allergens. The test is typically performed with a plastic lancet on either the forearm or upper back and involves a superficial disruption of the epidermis that is almost always painless ( Fig. 4.3 ). The prick test is interpreted 15 minutes after by measuring the maximal diameter of both the wheal and the flare. Skin test results are compared with a negative control, which is usually glycerol-saline, and a positive control, which is typically histamine. Intradermal allergy testing involves the injection of a small amount (0.02 to 0.03 mL) of a nonirritating concentration of allergen underneath the skin. Intradermal tests are considered to be more sensitive than prick tests but less specific. They are beneficial in the evaluation of drug allergies and insect venom hypersensitivity and are often performed as the second step after skin prick testing. Skin prick testing, however, is considered safer than intradermal testing and has better correlation with environmental and food allergy symptoms.
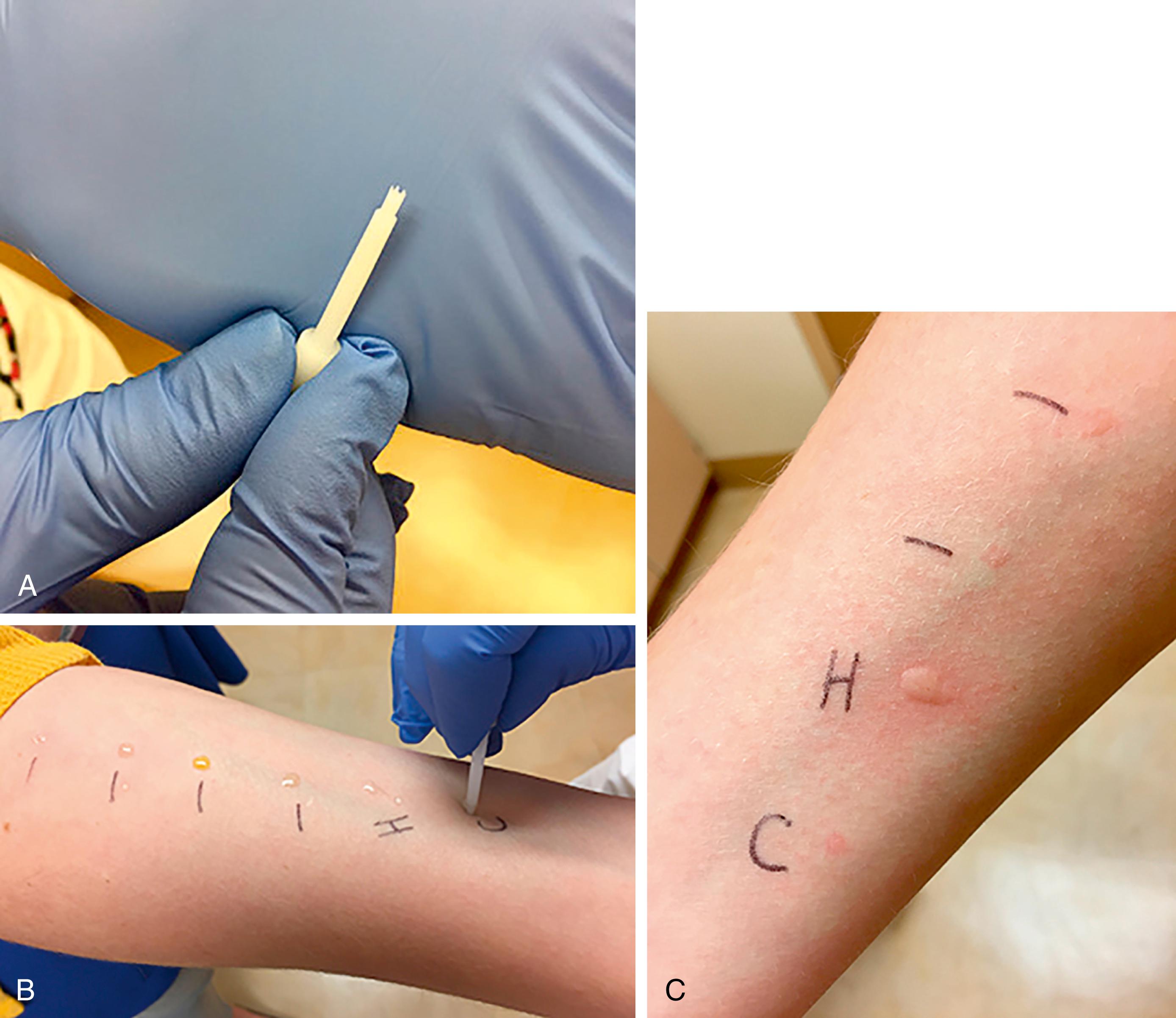
Allergy skin testing is not recommended in four clinical situations: (1) when antihistamines have been used in the recent past—this will suppress true positive test results evidenced by a suboptimal histamine control; (2) when skin disease limits the area available for testing; (3) during either an asthma exacerbation or episode of anaphylaxis; and (4) when a patient is taking a beta-blocking medicine or an angiotensin-converting enzyme (ACE) inhibitor (considered a “relative” contraindication), because these can interfere with epinephrine treatment in rare cases of test-induced anaphylaxis. When skin testing is not possible, in vitro testing is a good alternative. The in vitro tests can be accomplished with just a few milliliters of serum and also may be advantageous for some patients who have a difficult time sitting through allergy tests. In vitro allergy tests are more expensive and sometimes less sensitive than allergy prick tests ( Table 4.2 ). In vitro tests are especially helpful in the evaluation of patients with possible food allergies, which is further discussed later.
| Variable | Skin Test | In Vitro |
|---|---|---|
| Risk of allergic reaction | Rare | No |
| Sensitivity | Very good | Good a |
| Affected by antihistamines | Yes | No |
| Affected by corticosteroids | Not usually | No |
| Affected by extensive dermatitis or dermatographism | Yes | No |
| Broad selection of antigens | Yes | Yes |
| Immediate results | Yes | No |
| Discomfort | Mild | Moderate |
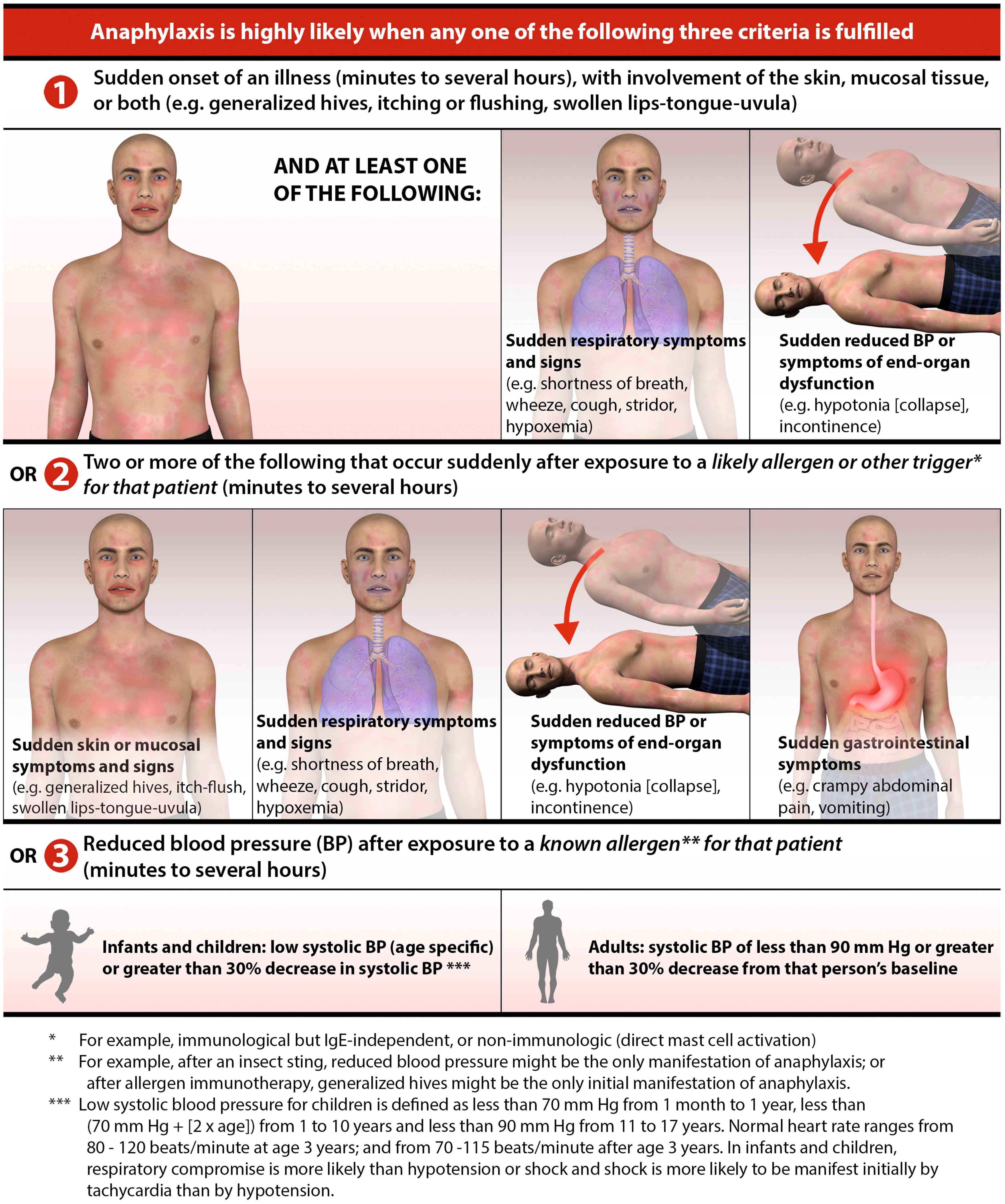
Anaphylaxis results from widespread degranulation of mast cells after cross-linking of antigen-specific IgE on the mast cell surface. Anaphylactoid reactions are clinically similar, however, mast cells degranulate without receiving signals through the cross-linking of IgE. While anaphylaxis is a clinical diagnosis, a report issued jointly by the National Institute of Allergy and Infectious Diseases and the Food Allergy and Anaphylaxis Network established criteria to define likely anaphylactic episodes ( Fig 4.4 ). The onset of anaphylaxis is typically rapid within minutes to 1 to 2 hours after exposure to the offending antigen. The most common triggers of anaphylaxis in children are foods (such as milk, egg, peanut, and treenuts) followed by insect stings. The pattern of system involvement can vary based on the antigen, dose, route of exposure and concomitant factors such as exercise, nonsteroidal antiinflammatory drugs (NSAIDs), and/or alcohol use. Symptoms can range from isolated urticaria to cardiovascular collapse ( Table 4.3 ). Airway obstruction and hypotension are the most severe manifestations of anaphylaxis. The upper or lower airway or both can be affected. Upper airway obstruction is due to laryngeal edema, whereas lower airway involvement can be due to edema and/or bronchospasm. Hypotension results from vasodilation and may be complicated by loss of intravascular volume. Vascular collapse may be aggravated by decreased myocardial function. In addition to the airway and cardiovascular system, other organ systems are also involved in anaphylaxis. The skin is the most commonly involved organ with urticaria and angioedema often present. GI involvement can manifest as vomiting, diarrhea, and abdominal pain due to cramping or edema.
| Signs and Symptoms | Percentage |
|---|---|
|
62–90 |
|
62–90 |
|
45–55 |
|
2–5 |
|
|
|
45–50 |
|
50–60 |
|
15–20 |
|
30–35 |
|
|
|
25–30 |
|
|
|
5–8 |
|
4–5 |
|
1–2 |
For treatment of anaphylactic reactions, intramuscular epinephrine is the initial therapy of choice which is superior in efficacy as compared to the subcutaneous route. Patients at risk should carry self-injectable epinephrine and be trained in its use. There are currently 5 different automated injectable epinephrine devices: EpiPen, Adrenaclick, Auvi-Q, Symjepi and several generic epinephrine autoinjectors ( Fig. 4.5 ). All exist in two doses: either 0.15 or 0.3 mg, while the Auvi-Q device is also available in a 0.1 mg dose. Operating technique varies somewhat among the devices, so it is important for families to become familiar with their specific device. Epinephrine is most effective when it is used as rapidly as possible with the onset of anaphylaxis. Other medications should not delay prompt delivery of epinephrine, which is the only medication that has been proven to be effective in treating anaphylaxis. In cases of hypotension, large-volume fluid resuscitation is essential. In severe cases of anaphylactic shock pressor support may be required. It is commonly recommended to call to 911 after administration of epinephrine followed by evaluation and observation in an emergency department. Albuterol inhalation should be considered for any lower airway symptoms and especially in patients with asthma. Systemic steroids are often administered, however their effectiveness in the treatment of anaphylaxis and in the prevention of biphasic anaphylaxis has never been proven. Antihistamines can be used for reactions confined to the skin and as second-line therapy to epinephrine in more severe reactions.

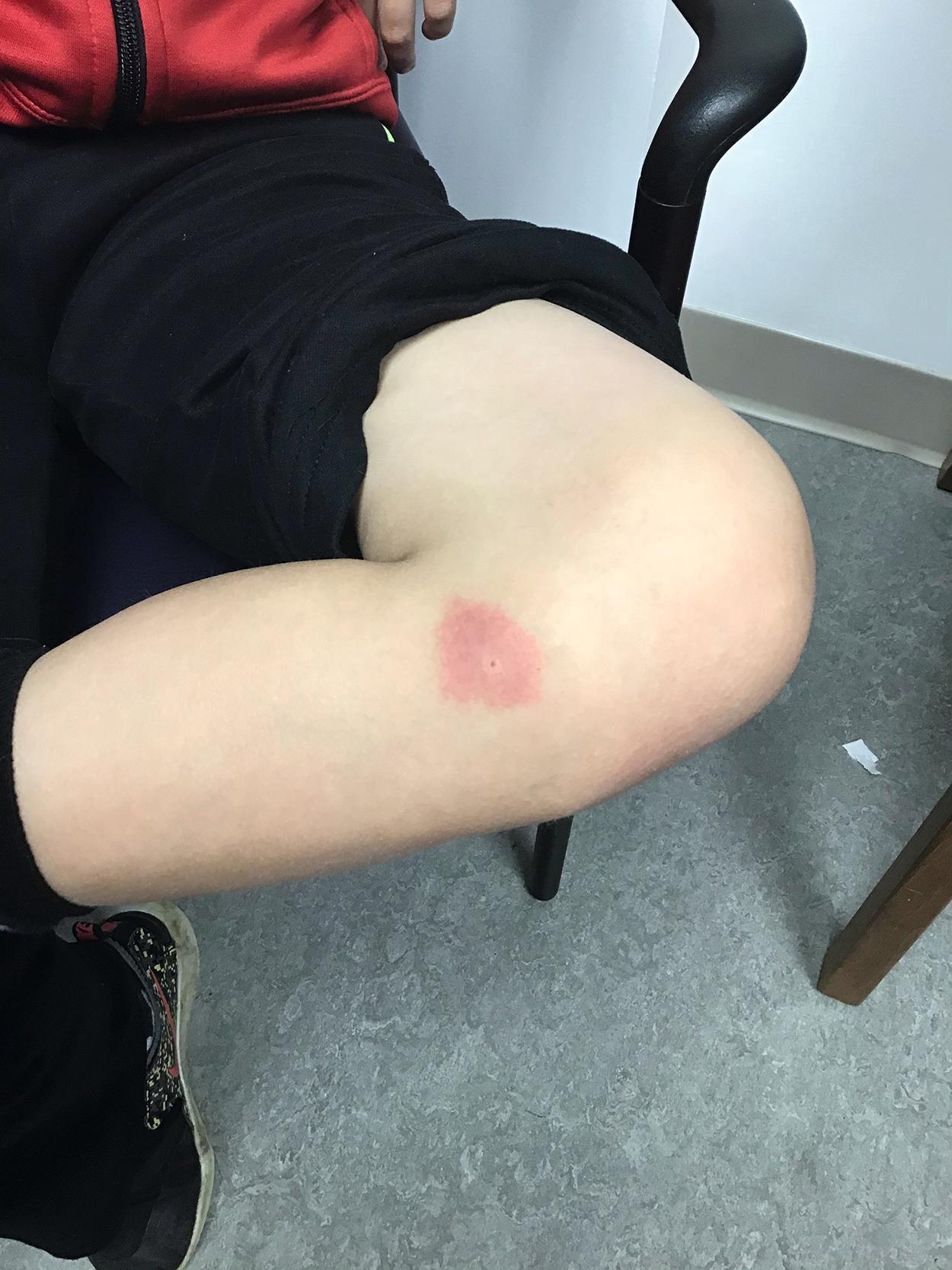
Life-threatening reactions to Hymenoptera are rare in childhood. A typical local reaction from a bee sting involves erythema, induration, pain, and/or itching secondary to local effects of the insect venom. ( Fig 4.6 ) Hypersensitivity reactions may manifest as large local reactions (LLRs), generalized urticaria or anaphylaxis. LLRs involve extreme swelling (>10 cm) contiguous to the sting and may involve lymphangitic streaking towards nodes in the groin or axilla. They typically progress in size 12 to 24 hours after the sting and may take 5 to 10 days to resolve. While these reactions may look like an infection, LLRs occur more rapidly than cellulitis and respond to therapy such as cool compresses antihistamines and NSAIDs. Corticosteroids may rarely be required. Patients with LLRs have an excellent long-term prognosis. While LLRs may occur again, the risk for anaphylaxis with future stings is only 4% to 10%. Allergy testing does not predict risk for anaphylaxis with future stings and is therefore not recommended in the evaluation of patients with LLRs.
Children and adults who have a history of generalized urticaria without respiratory, cardiovascular, or GI symptoms after an insect sting also have a good long-term prognosis. The risk for severe anaphylaxis after experiencing a mild cutaneous-only systemic reaction is less than 3% which is not greater than that of the general population. Allergy testing or desensitization with immunotherapy is generally not recommended unless other risk factors are present such as frequent unavoidable exposure or underlying medical conditions such as mastocytosis. The decision about whether to prescribe an epinephrine autoinjector to children with a history of generalized reactions confined to the skin or LLRs from stinging insects is left to physician discretion.
Children with a history of systemic anaphylaxis involving extracutaneous systems after a hymenoptera sting have a 30% to 40% risk of a similar reaction with future stings and should undergo allergy testing for consideration of immunotherapy. Testing should be performed no sooner than 4 weeks after the reaction to decrease the potential for false-negative test results. Allergen immunotherapy for Hymenoptera sensitivity is the most effective form of allergen immunotherapy available. Venom immunotherapy for wasps, yellow jackets, and hornets reduces the risk for anaphylaxis by greater than 95%, whereas it is slightly less effective for honeybees, providing complete protection in 80% to 85% of patients on therapy. Most venom immunotherapy protocols start with weekly injections for about 10 to 16 weeks followed by maintenance injections every 1 to 2 months for a duration of 3 to 5 years in most cases.
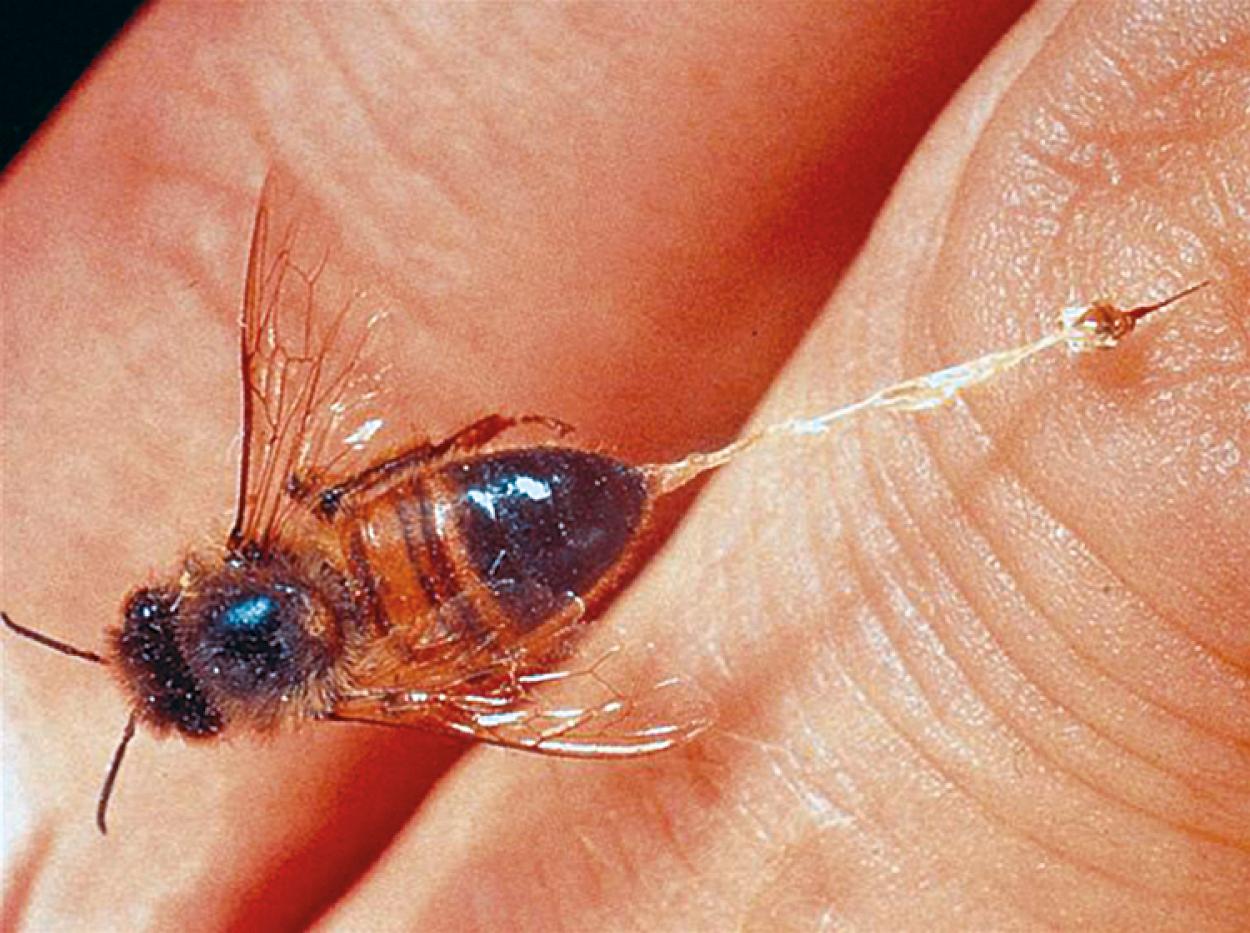
The Hymenoptera that have been associated with anaphylaxis come from three subfamilies: Apidae (honeybees); Vespidae (yellow jackets, wasps, and white- and yellow-faced hornets); and Formicidae (fire ants). In the United States, yellow jackets are the most common cause of Hymenoptera-induced anaphylaxis. Yellow jackets typically nest in the ground, are scavengers for food, and, consequently, are frequently encountered at picnics and around garbage cans. The yellow jacket is small with tight yellow and black bands. The honeybee is the least aggressive of the Hymenoptera family. Stings from honeybees occur most commonly in beekeepers and after accidental contact. The honeybee’s stinger is barbed and is retained in the skin after stings ( Fig. 4.7 ). If the stinger is visible, it should be gently flicked away from the skin with a fingernail. The stinger and venom sac should not be removed by pinching between the thumb and forefinger, because this process can express additional venom. Wasps and hornets are very territorial and will sting to protect their nests. The fire ant is an increasingly important cause of Hymenoptera-induced anaphylaxis. Fire ants are found in the south and southeastern United States, but their natural habitat appears to be expanding.
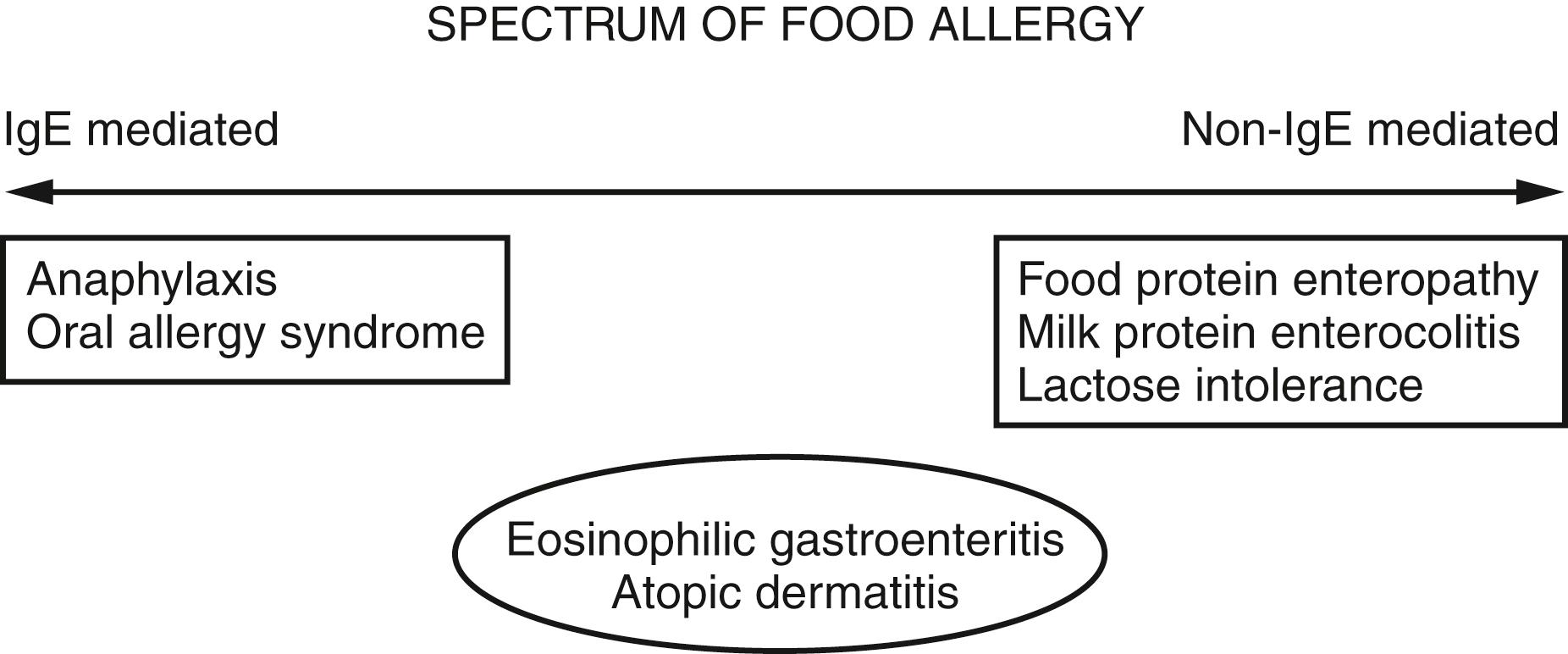
If tolerance, or lack of an immune or other reaction, to a food represents a normal host response to ingestion, an adverse food reaction can be broadly defined as any abnormal reaction. These adverse food reactions can be further subdivided into those that are immune-mediated and not. Within the immune-mediated reactions are IgE-mediated (those most commonly referred to as food allergy), non-IgE mediated, mixed IgE and non-IgE, and cell-mediated ( Fig. 4.8 ). Nonimmune-mediated reactions are commonly referred to as intolerances (e.g., lactose intolerance), and although these reactions can lead to significant symptoms, allergy testing is not helpful or indicated in the evaluation of these disorders. IgE-mediated food allergy reactions are highly reproducible (i.e., occur with every exposure to the food), and often are triggered by ingestions of very small quantities. Anaphylaxis is the best described and understood type I hypersensitivity to food. Although contact reactions (erythema, pruritus, hives) are common if an individual touches his or her food allergen, the most severe allergic reactions occur with ingestion. Milk, egg, wheat, soy, and peanut account for more than 90% of pediatric food allergic reactions. The onset of type I hypersensitivity to milk and egg is almost always in the first year of life. Fortunately, sensitivity to milk and egg is eventually outgrown in the vast majority of patients, frequently by school age.
Most allergic reactions to foods occur with the first few ingestions of a food. In contrast to type I hypersensitivity to milk and egg, less than 25% of patients will outgrow their peanut or tree nut sensitivity. Peanuts and tree nuts cause the majority of life-threatening reactions to foods. Other risk factors for life-threatening reactions from food-induced anaphylaxis include asthma, adolescence, and the delayed administration of epinephrine. A past history of mild reactions does not rule out the possibility of a future life-threatening episode of food-induced anaphylaxis. Some patients with type I hypersensitivity to peanut will develop type I hypersensitivity to one or more tree nuts. Nuts are also frequently processed together, increasing the risk of cross-contact exposures with nuts. A simplified approach includes avoiding all nuts if a patient is allergic to either peanuts or tree nuts, however recommendations should be individualized as many tree nut–allergic children can ingest peanut butter and many peanut-allergic children can ingest tree nuts as long as they avoid foods that may contain traces of the nut to which they are allergic.
Another common form of type I–mediated hypersensitivity to food is oral allergy syndrome (also known as pollen-food allergy syndrome ). Patients with this syndrome typically have underlying seasonal allergic rhinitis and develop pruritus and mild angioedema of the oropharynx when ingesting fresh fruits and vegetables. The reaction is due to cross-reactivity between heat-labile proteins in some fruits and vegetables and outdoor seasonal pollens. The reaction is often eliminated by heating the vegetable or fruit. The reaction does not generally extend beyond the oropharynx, and patients typically do not need to carry epinephrine. In a minority of cases, reactions may be severe, so taking a thorough history is essential before recommendations are made for avoidance and the need for epinephrine.
Type I hypersensitivity to foods can be an unrecognized trigger in up to one-third of younger children with severe atopic dermatitis although it is an uncommon trigger in older children and those with mild atopic dermatitis. It has rarely been described in adults with atopic dermatitis. The evaluation of type I hypersensitivity to foods in children with atopic dermatitis should be reserved for patients whose disease cannot be managed with good skin care and intermittent use of low- to moderate-potency topical antiinflammatory medications. When food hypersensitivity plays a role in poorly controlled atopic dermatitis, six foods account for the vast majority of reactions: milk, egg, wheat, soy, and peanut.
One of the challenges with diagnosing type I hypersensitivity to foods is the poor specificity of both the skin prick and in vitro tests. These tests have a 20% to 50% false positive rate. Therefore, the “gold standard” for the diagnosis of food allergy is a double-blind, placebo-controlled food challenge (DBPCFC). A distinction must be drawn between patients who are “sensitized,” based on skin or serum testing, and those who are clinically allergic, based on a history of reactions to ingestions. IgE-mediated reactions occur quickly and reproducibly with ingestion of the triggering food. Over the years, research using in vitro allergy tests has identified levels of specific IgE against certain foods that predict a reaction during a DBPCFC with 95% likelihood. In vitro food-specific IgE levels are most useful clinically when used in association with a patient’s ingestion history and (if indicated) skin test results. In a nationally representative survey study, approximately 17% of the overall population in the United States displayed serum sensitization to either cow’s milk, egg, peanut, or shrimp, and this prevalence rate was even higher (28%) among children from 1 to 5 years old. Yet, on the basis of these serum levels in the same study, only approximately 2.5% of the United States population was predicted to be clinically allergic to one of these foods. The data concerning positive predictive levels depend on the age of the patient and are not available for all foods ( Table 4.4 ). The in vitro allergy test may also be repeated over time to try to help with the identification of patients who may have outgrown their sensitivity. The sensitivity of both the skin prick and in vitro tests is excellent but not 100%. If there is a strong clinical history of a type I reaction to a food, it may be necessary to perform an oral challenge in a medically supervised setting before food hypersensitivity can be fully ruled out.
| Allergen | Decision Point (kU/L) | Sensitivity (%) | Specificity (%) | Positive Predictive Value (%) | Negative Predictive Value (%) |
|---|---|---|---|---|---|
| Egg | 7 | 61 | 95 | 98 | 38 |
|
2 | 95 | |||
| Milk | 15 | 57 | 94 | 95 | 53 |
|
5 | 95 | |||
| Peanut | 14 | 57 | 100 | 100 | 36 |
| Fish | 20 | 25 | 100 | 100 | 89 |
| Soybean | 30 | 44 | 94 | 73 | 82 |
| Wheat | 26 | 61 | 92 | 74 | 87 |
| Tree nuts | ≈15 | — | — | ≈95 | — |
a Boyano-Martinez T, Garcia-Ara C, Diaz-Pena GM, et al. Validity of specific IgE in children with egg allergy. Clin Exp Allergy . 2001;31:1464–1469.
b Garcia-Ara C, Boyano-Martinez T, Diaz-Pena GM, et al. Specific IgE levels in the diagnosis of immediate hypersensitivity to cow’s milk protein in infants. J Allergy Clin Immunol . 2001;107:185–190.
Current treatment for IgE-mediated food allergic reactions consists of avoidance of the trigger food, as well as tools to manage an accidental ingestion reaction. Many patients with this severe food allergy will require access to an epinephrine autoinjector, and patients/families should be instructed in its use. These patients and their families should be provided with an anaphylaxis action plan outlining appropriate symptom-based treatment. Although avoidance is the current standard of care, there are currently multiple potential “active” therapies being investigated for IgE-mediated food allergy. These interventions aim to raise the threshold of the amount of allergen that can lead to an allergic reaction if there is an accidental ingestion of that particular food. To date, the most well studied of these approaches involve exposing the allergic individual to very small amounts of the food allergen in question, either orally (oral immunotherapy [OIT]), sublingually (sublingual immunotherapy [SLIT]), or through the skin (epicutaneous immunotherapy [EPIT]). As of this writing, the only U.S. Food and Drug Administration (FDA)-approved immunotherapy treatment for food allergy is Palforzia, which involves the graded administration of pharmaceutical grade peanut powder for children 4 to 17 years. Phase III studies for EPIT for peanut have been completed and are under consideration for approval by the FDA.
Triggers of eosinophilic gastrointestinal disorders (EGIDs) are more challenging to evaluate as eosinophilic infiltration of the gut is thought to be caused by a mixed reaction involving type I (IgE-mediated) hypersensitivity and other immune pathways. Patients with EGIDS often have type I hypersensitivity to certain foods and sometimes inhaled aeroallergens that are inadvertently swallowed. The pathogenesis of EGIDs is thought, however, to also involve non–IgE-mediated pathways such that a negative skin prick or in vitro test does not eliminate the possibility that food may be causing or contributing to the disease. Similarly, a positive allergy test to a food does not definitely establish causation. Patch testing has also been studied to determine potential food triggers of EGIDs revealing equally concerning limitations in sensitivity and specificity. Patients with eosinophilic inflammation of the upper GI tract tend to have problems with dysphagia, food impaction, abdominal pain, and vomiting. Patients with eosinophilic inflammation of the lower GI tract tend to have diarrhea, failure to thrive, and abdominal pain. Treatments for EGIDs include elemental diets, selected dietary elimination, proton pump inhibitors, and swallowed steroids while biologic therapies targeting eosinophilic pathways and other mediators involved in the process are currently under evaluation. The long-term prognosis is not completely understood and the disease often follows a relapsing and remitting course.
The term food allergy also encompasses the food reactions elicited through non–IgE-mediated immune pathways. The classic example of a non–IgE-mediated hypersensitivity to food is seen in milk protein proctocolitis of infancy (also known as food-induced eosinophilic proctitis ). These children usually present with bloody diarrhea in the first few months of life and this improves within days of removing milk proteins from the diet. Many of these children are breastfed, and they react to either cow’s milk or another protein in the maternal diet. Approximately half of children with milk protein enterocolitis will also experience symptoms with soy protein–based formulas. Since milk protein proctocolitis is not IgE-mediated, allergy testing is not recommended in its evaluation. Almost all infants will outgrow this condition by 1 to 2 years old.
Food protein–induced enterocolitis syndrome (FPIES) is another non–IgE-mediated reaction with a more severe presentation. Infants typically present 1 to 4 hours after an ingestion of the triggering food, with profuse vomiting sometimes to the point of dehydration hypotension and lethargy. Fecal leukocytes, a rise in the peripheral absolute neutrophil count, acidosis, and methemoglobinemia may also be seen, whereas skin lesions are not present. Cow’s milk, soy, rice, and oat are the most common triggers of FPIES, although it has been described with a variety of other foods. Infants frequently are diagnosed with recurrent sepsis or infectious gastroenteritis before the condition of FPIES is realized. Since FPIES is not IgE-mediated, the gold standard for diagnosis is a food challenge. While skin prick testing and in vitro IgE testing are not helpful in determining causality or resolution, a positive test may have prognostic value in predicting a longer course to resolution and/or the risk for IgE-mediated potentially anaphylactic symptoms on reintroduction of the food. Many cases, however, resolve by 2 to 6 years and the triggering foods are tolerated without issue.
Drug hypersensitivity reactions (DHRs) are defined as unexpected reproducible objective signs and symptoms caused by a specific drug at a normally tolerated dose. DHRs can be divided into allergic drug hypersensitivity reactions which are immunologically mediated and nonallergic drug hypersensitivity. Allergic DHRs are mediated by drug-specific antibodies, T cells, or both and can be further defined using the classification by Gell and Coombs (see Table 4.1 ). Type I hypersensitivity reactions to drugs are mediated by drug-specific IgE bound to mast cells and basophils. IgE is cross-linked by the drug, leading to mast cell and basophil activation and release of mediators that drive the clinical reaction. Type I hypersensitivity can range from mild reactions involving only the skin (generalized urticaria and/or angioedema) to anaphylaxis with laryngeal edema, bronchospasm, hypotension, and cardiovascular collapse. These reactions occur within minutes of exposure and require prior sensitization. IgE-mediated reactions occur early in the treatment course, usually after the first or second dose of a drug. A non-urticarial rash that develops after more than a few days of therapy is seldom due to IgE-mediated mechanisms.
Penicillin is the only drug that has been widely studied as a cause of type I hypersensitivity, and consequently, it is the only drug for which well-standardized allergy testing is available. Once penicillin enters the body, it undergoes spontaneous conversion to a reactive intermediate that then binds to nearby proteins, forming penicilloyl amides. Penicilloyl amides account for 95% of tissue-bound penicillin and are known as the major antigenic determinant. A small percentage of penicillin can be found in the body as the native drug, penicillin, or other metabolites called minor antigenic determinants (penicilloate and penilloate). Skin testing done with the major antigenic determinant, penicillin, and the minor determinants has a negative predictive value approaching 98% to 100%. At present, in the United States only the major determinant and the drug itself, penicillin G, are available for skin testing. Patients who have had either a severe or recent urticarial reaction to a penicillin-type antibiotic should be evaluated cautiously with penicillin skin testing and if negative, an oral challenge to the triggering drug, which in many cases in children was amoxicillin.
The use of cephalosporins in patients with a history of penicillin allergy is controversial. Before 1980, a reported 10% to 20% of penicillin-allergic patients reacted to cephalosporins; but since 1980, that number has dropped to 2%. Much of the concern about cross-reactivity between these two classes of drugs was based on their common beta-lactam ring but also likely involved occasional contamination of early cephalosporins with trace amounts of penicillin. Although clinical cross-reactivity between these two drugs has lessened over the years, there are clear reports of anaphylactic reactions after the use of cephalosporins in penicillin-allergic patients. This cross-reactivity is thought in many cases to be due to a similar or identical side chain shared between the two specific beta-lactam antibiotics. The combination of all these factors has contributed to the persistent concern about the safety of cephalosporins in patients with penicillin allergy. Unfortunately, skin testing for cephalosporins has not been standardized, and the negative predictive value of these tests is not known. In patients with a remote clinical history and mild reactions, it may be reasonable to treat with second- or third-generation cephalosporins without testing. In patients with a recent or severe reaction to a cephalosporin, recommendations for use of future beta-lactam antibiotics are determined based on the history, potentially penicillin testing and review of the side chains involved in the original reaction.
Anaphylaxis from drugs other than penicillin must be diagnosed on the basis of clinical presentation. Patients with type I hypersensitivity to drugs can still potentially receive the drug, if there is not acceptable alternative, through a drug desensitization protocol. This procedure attempts to induce drug tolerance by slow delivery of increasing drug doses over a period of 6 to 12 hours. The mechanism by which tolerance is achieved is not well understood. Patients maintain the desensitized state for only as long as they are receiving the medication. Once the drug is discontinued, the tolerant state is lost and future treatment would require repeating the protocol.
Nonallergic DHRs manifest as anaphylactoid (also called pseudoallergic ) reactions. These are due to generalized mast cell mediator release that does not involve specific IgE. The exact mechanism of anaphylactoid reactions is not known, therefore, diagnostic testing is not available. Several drug-induced causes of anaphylactoid reactions are radiocontrast media, NSAIDs vancomycin and fluoroquinolones.
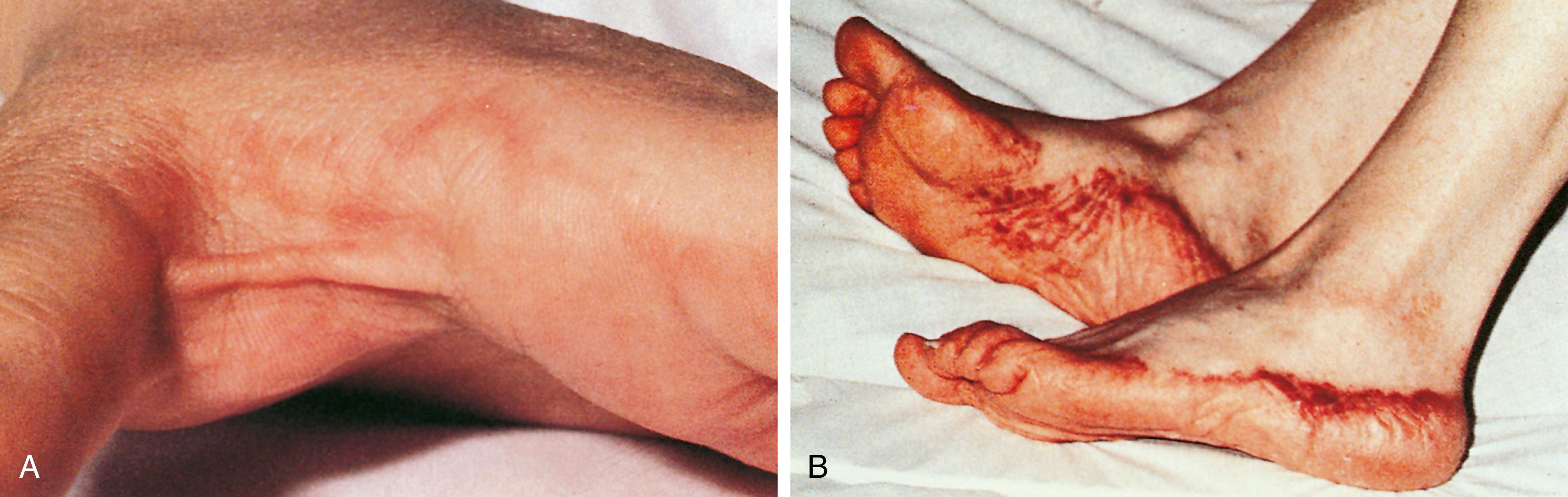
DHRs can also manifest through type II, III, and IV Gell and Coombs reactions. The classic Gell and Coombs type II reaction is due to immunoglobulin G (IgG) antibodies directed against drug bound to cell surfaces, causing either hemolytic anemia or thrombocytopenia. Serum sickness represents a Gell and Coombs type III reaction. Serum sickness usually begins 1 to 3 weeks after drug exposure and involves various symptoms, including fever, malaise, arthralgias, arthritis, urticaria, and lymphadenopathy. Many patients with serum sickness will develop a characteristic serpiginous, erythematous, or purpuric eruption at the junction of the palmar or plantar and dorsolateral aspects of the hands and feet, respectively ( Fig. 4.9 ). Patients with serum sickness may have reduced levels of complement components C3 and C4 along with the presence of circulating immune complexes. Serum sickness can be treated with antihistamines; and if the patient fails to improve, systemic steroids can be used. Symptoms usually resolve within a few weeks. Future avoidance of the drug inciting the reaction is recommended.
Delayed type IV DHRs are considered cell-mediated and commonly occur days to weeks after drug initiation. Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN) involve cytotoxic CD8 + T cells and natural killer (NK) cells that cause widespread injury to keratinocytes in epidermis. These reactions affect the skin mucous membranes and eyes. The lesions on the skin may begin as dusky erythematous macules or papules that evolve into targetoid lesions vesicles and bullae followed by sloughing of the skin. SJS involves less than 10% of the body surface area (BSA) while TEN involves greater than 30% of the BSA. The process is considered SJS/TEN overlap when 10% to 30% of the BSA is affected. TEN is almost always drug induced, whereas SJS may be induced by either drugs or infection with Mycoplasma or herpes simplex virus (HSV). When drug exposure is the inciting cause, the exposure precedes the onset of skin findings by 1 to 3 weeks. Other than discontinuation of the culprit drug and supportive care, there are no universally recommended therapies for SJS/TEN. While not clearly established, use of several immunomodulating modalities such as corticosteroids, cyclosporin, anti-tumor necrosis factor antibodies and intravenous immune globulin (IVIG) have shown varied benefits.
Another delayed immune-mediated DHR is the syndrome known as drug rash with eosinophilia and systemic symptoms (DRESS) . DRESS was initially described with anticonvulsant therapies but is now known to be caused by a number of different drugs. DRESS reactions usually begin 2 to 8 weeks into drug therapy. Clinical characteristics include a generalized rash, fever, facial edema, lymphadenopathy, and organ dysfunction. The liver is most commonly involved but any internal organ can be affected. Laboratory findings include eosinophilia, atypical lymphocytosis, elevated transaminases, and occasionally increased creatinine with kidney involvement. The exact pathogenesis is not yet fully understood but is thought to involve both effector and regulatory T cells. Treatment involves discontinuation of the inciting drug and supportive therapy. High potency topical steroids are recommended for skin symptoms while systemic glucocorticoids are reserved for cases of severe organ involvement. Cyclosporin has shown benefit as second-line therapy in a limited number of cases.
Allergic rhinitis, characterized by inflammation, edema, and weeping of the nasal mucosa, is the most common allergic disorder and occurs in up to 25% of the population. Diagnosis is based on characteristic history, physical findings, and testing for antigen-specific IgE. Common presenting symptoms include nasal congestion and pruritus, clear rhinorrhea, and paroxysms of sneezing. Whereas older children may blow their noses frequently, younger children do not. Instead, they sniff, snort, and repetitively clear their throats. Nasal pruritus stimulates grimacing and twitching ( Fig. 4.10 ) and rubbing of the nose (“allergic salute”). Frequent irritation of the nasal mucosa can lead to epistaxis. In the case of allergy to seasonal pollens, the symptoms may be acute, have a sudden onset, and be confined to the period during which the particular airborne pollen is detectable. Seasonal allergic rhinitis tends to be triggered by tree pollens in the spring, grasses in the early summer and ragweed or other pollens in the late summer and early fall.

Interestingly, seasonal allergic rhinitis is often called hay fever, although fever is not a symptom. Manifestations sometimes can be severe enough to induce flulike symptoms of fatigue and malaise. In contrast, symptoms may be chronic and more indolent in the case of allergy to perennial allergens, including molds, house dust mites, and animal dander.
Symptoms of allergic rhinitis can overlap with those of infectious rhinitis and include (1) disturbed sleep and snoring; (2) morning dryness and irritation of the throat as a result of mouth breathing; (3) lassitude, fatigue, and irritability from sleep interruption; (4) early nighttime cough; and (5) if the maxillary, frontal, and ethmoidal sinuses are affected, a sensation of pressure over the cheeks, forehead, and bridge of the nose. The presence of naso-ocular watering and pruritus with pruritus of the throat or ears that persists for longer than the normal duration of a viral upper respiratory infection should prompt consideration of an underlying IgE-mediated disorder. The extent to which allergic rhinitis affects a patient’s quality of life is often underappreciated.
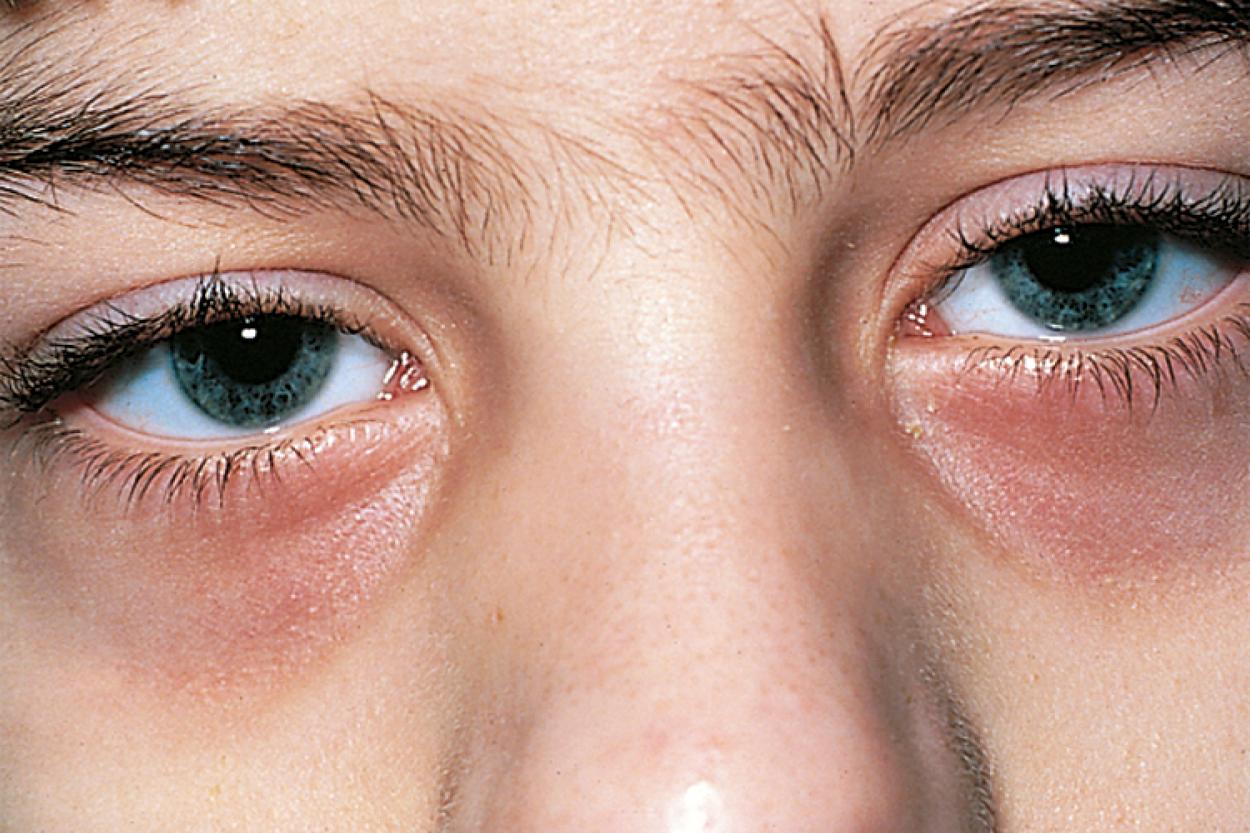
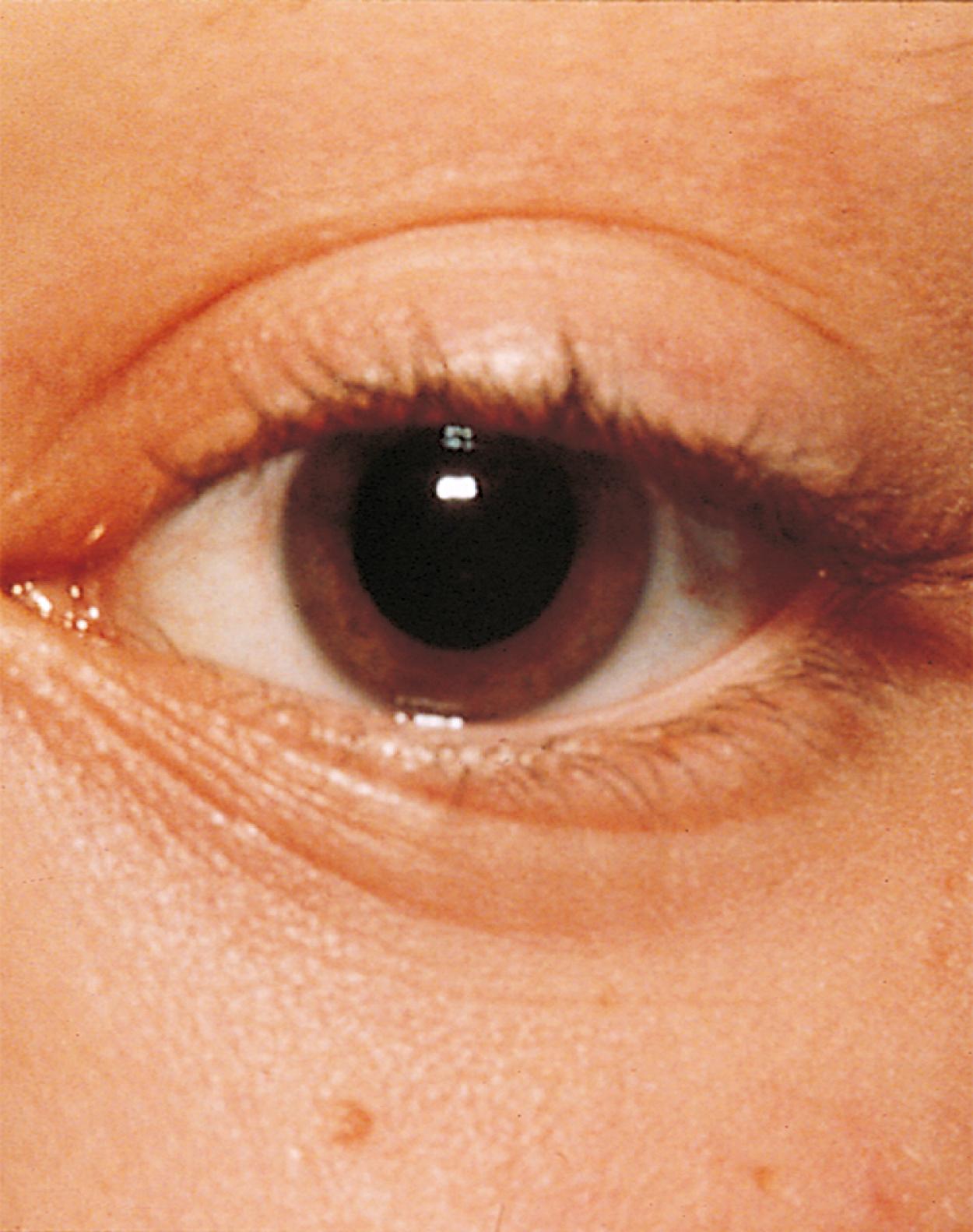
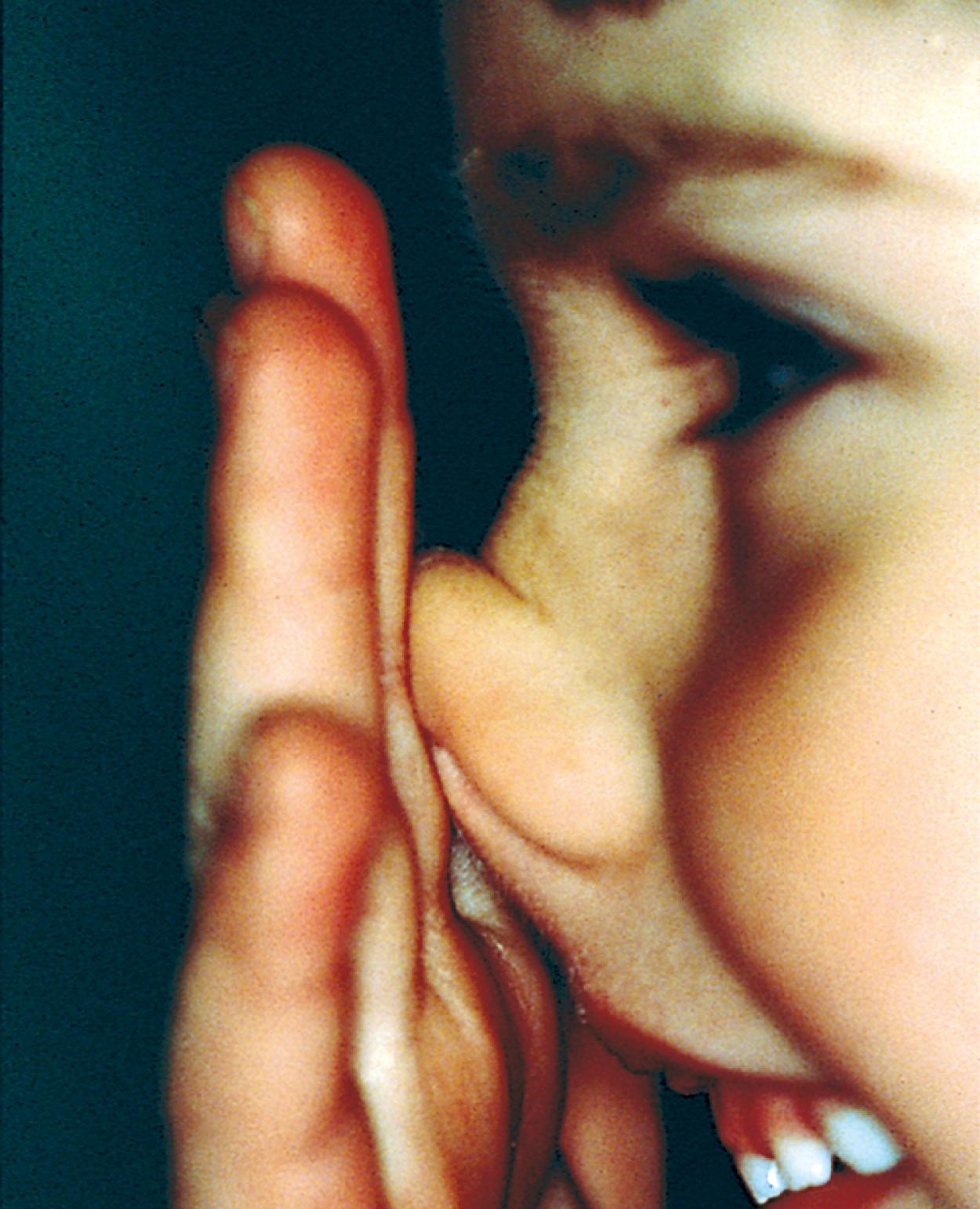
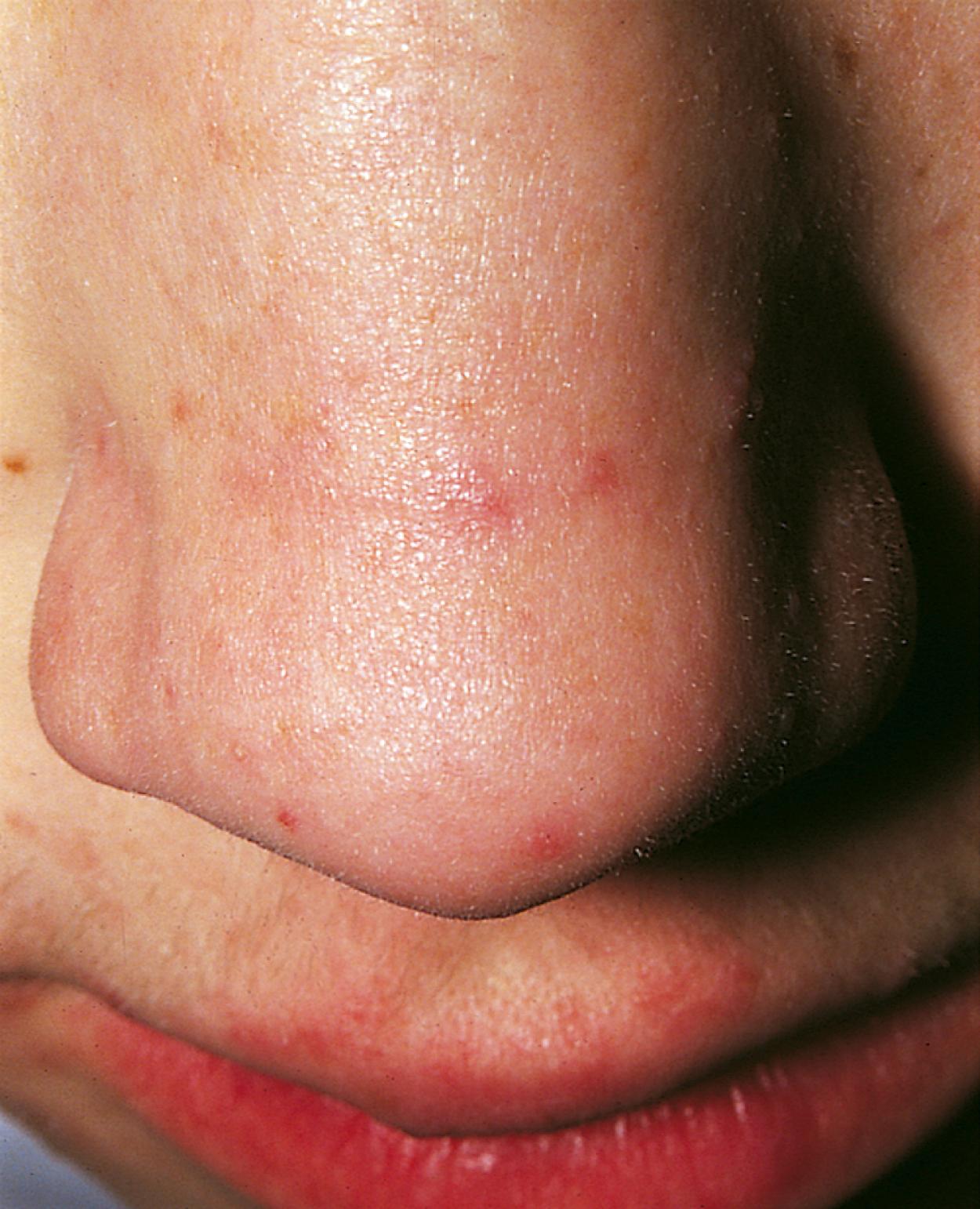
Many children with long-standing allergic rhinitis can be recognized by their facial characteristics. Ocular manifestations of the allergic disposition include cobblestoning of the conjunctivae (see Fig. 4.33 ), the allergic shiner, and Dennie sign. Allergic shiners—bluish discolorations or dark circles beneath the eyes—are commonly observed in patients with allergic rhinitis ( Fig. 4.11 ). This finding represents chronic venous congestion secondary to inflammation and can also be seen in the setting of infections and in those with a genetic predisposition. Dennie sign refers to prominent folds or creases on the lower eyelid ( Fig. 4.12 ) running parallel to the lower lid margin. Although these lines were originally thought to indicate a predisposition to allergy, they can be present in any condition associated with periocular pruritus and scratching or chronic nasal congestion. Frequent upward rubbing of the nose with the palm of the hand (the allergic salute; Fig. 4.13 ) promotes development of a transverse nasal crease across the lower third of the nose ( Fig. 4.14 ). Chronic obstruction produced by nasal mucosal edema may result in mouth breathing and typical open-mouthed, adenoid-type facies ( Fig. 4.15 ).
On nasal examination, attention should be focused on the position of the nasal septum; nasal patency; mucosal appearance; and presence and character of secretions, polyps, or foreign bodies (see Chapter 24 ). The typical physical examination findings in allergic rhinitis include a marked decrease in nasal patency resulting from swollen inferior turbinates, which appear pale and bluish gray ( Fig. 4.16 ). The mucosa appears edematous and secretions are clear and watery to mucoid in character.
Depending on the specific allergens, allergic rhinitis may be acute, recurrent, or chronic and must be distinguished from a number of nonallergic conditions. This necessitates a thorough medical and family history and careful examination. In some instances, response to a trial of medication and/or observations over time may be necessary to confirm the diagnosis. When symptoms are seasonal or regularly associated with exposure to specific allergens, the distinction is generally clear. In evaluating patients with perennial or recurrent but nonseasonal symptoms, allergy, recurrent infection, nonallergic rhinitis with eosinophilia syndrome (NARES) and vasomotor rhinitis must be considered. Unilateral rhinorrhea should prompt further investigation. Foul smelling mucoid discharge suggests a foreign body, whereas unilateral clear rhinorrhea could be due to a cerebral spinal fluid leak.
Children with frequent upper respiratory infections and/or persistent nasal congestion can present a diagnostic challenge. In some cases, the phenomenon is due to recurrent viral infections, particularly in children in their first year of daycare or nursery school. In other patients, tonsillar and adenoidal hypertrophy provides favorable conditions for recurrent infections (see Chapter 24 ). Atopic (allergic) children may have increased risk of infection because of impaired flow of secretions due to mucosal edema. Recurrent viral sinus infections must also be considered as they typically begin with thin clear nasal secretions overlapping with that of allergic rhinitis before evolving to produce a thicker often opaque discharge which is often difficult to distinguish from a bacterial sinus infection based on appearance alone.
Other forms of rhinitis that must be distinguished from allergic rhinitis are enumerated in Table 4.5 . Although characterized by eosinophilia, NARES does not produce nasal pruritus, and patients lack specific IgE antibodies as measured by skin testing or in vitro serum testing. Patients with vasomotor rhinitis do not complain of pruritus, have a clear discharge without eosinophils, and also lack specific IgE antibodies. Vasomotor rhinitis is thus considered a form of noninflammatory rhinitis and is often triggered by nonspecific stimuli, such as cold air exposure or smoke. The condition is diagnosed most frequently in adults but may occur in children. Congestion or rhinorrhea may predominate in this disorder. Rhinitis medicamentosa is a condition seen in patients who have been using alpha-adrenergic vasoconstrictor nose drops (phenylephrine or oxymetazoline) as decongestants for prolonged treatment periods. The disorder is characterized by rebound vasodilation that produces an erythematous, edematous mucosa in association with a profuse clear nasal discharge.
| Allergic | Nonallergic | ||
|---|---|---|---|
| NARES | Vasomotor | ||
| Usual onset | Childhood | Childhood | Adulthood |
| Family history of allergy | Usual | Coincidental | Coincidental |
| Collateral allergy symptoms | Common | Unusual | Unusual |
|
Frequent | Occasional | Occasional |
|
Common | Unusual | Unusual |
|
Profuse | Profuse | Profuse |
|
Moderate to marked | Moderate to marked | Moderate to marked |
| Physical Examination | |||
| Edema | Moderate to marked | Moderate | Moderate |
| Secretions | Watery | Watery | Mucoid to watery |
| Nasal eosinophilia | Common | Common | Occasional |
| Allergic Evaluation | |||
| Skin tests | Positive | Coincidental | Coincidental |
| IgE antibodies | Positive | Coincidental | Coincidental |
| Therapeutic Response | |||
| Antihistamines | Fair to good | Fair | Poor to fair |
| Decongestants | Fair | Fair | Poor to fair |
| Corticosteroids | Good | Good | Poor |
| Cromolyn | Fair | Unknown | Poor |
| Immunotherapy | Good | None | None |
Some children with perennial allergic rhinitis have congestion so constant and severe that it produces signs of chronic nasal obstruction. This must be distinguished from other acquired and congenital causes (see Chapter 24 ). The history, physical findings, and results of allergy tests for specific IgE and nasal smears, along with therapeutic trials of antihistamines and intranasal corticosteroids, will all help lead to a diagnosis.
Many patients with allergic rhinitis have mild symptoms that are adequately controlled by intermittent antihistamine administration and/or environmental controls. It is not uncommon, however, for acute naso-ocular symptoms to be attributed to allergies inappropriately. Allergy testing with confirmation of specific IgE can guide ideal timing of medical therapy and investment into appropriate environmental controls as well as eliminate the use of unnecessary medications in children whose symptoms may not be due to allergies. Additional treatment options for those with poorly controlled allergy symptoms include topical antiinflammatory agents and aeroallergen immunotherapy. Subcutaneous immunotherapy injections (allergy shots) and SLIT tablets have been shown to be effective alternative treatment options for patients who are not well controlled with medications or who desire to reduce the amount of medications required to control their symptoms. The typical effective course of immunotherapy is 3 to 5 years.
Respiratory distress in children (tachypnea with or without grunting, flaring, retractions, and cyanosis) should be promptly evaluated and treated. The first step in approaching respiratory distress is to differentiate upper from lower airway disorders. At times, various degrees of upper and lower airway obstruction may coexist, as in laryngotracheobronchitis.
Upper airway obstruction causes difficulty moving air into the chest, whereas lower airway obstruction causes difficulty moving air out of the chest. This difference results in characteristic physical findings. In general, lower airway obstruction produces prolongation of the expiratory phase of respiration and typical expiratory wheezing, whereas upper airway obstruction prolongs the inspiratory phase. Wheezing is defined as musical or whistling auscultatory sounds heard more often on expiration than on inspiration, although in severe obstruction both inspiratory and expiratory wheezing are often present. Inspiratory stridor, seen with upper airway obstruction, can mimic wheezing. Both can be detected concomitantly. Stridor is defined as a crowing sound usually heard during the inspiratory phase of respiration. It tends to be loud when the obstruction is subglottic and quiet when obstruction is supraglottic. Mild to moderate increases in respiratory and heart rates are common in upper airway obstruction, whereas lower airway disorders such as pneumonitis and asthma often lead to markedly increased respiratory and heart rates. Retractions are often generalized (suprasternal, intracostal, and subcostal) in severe airway obstruction of any etiology.
Asthma is the most common chronic respiratory condition affecting children. The Centers for Disease Control and Prevention (CDC) reported more than 6 million children (1 in 12) have asthma (CDC National Center for Environmental Health, 2018). In 2016, more than half of children with asthma had one or more attacks, occurring more frequently in children younger than the age of 5. Each year, 1 in 6 children with asthma go to the emergency room and 1 in 20 children with asthma are hospitalized because of their disease. Of note, asthma hospitalizations of children did decline by 50% from 10% in 2003 to 5% in 2013. Data analyzed from the 2001 to 2016 National Health Interview Survey for children 0 to 17 demonstrated that there was a higher prevalence in boys, non-Hispanic black children and Puerto Rican children and children from low-income households. This survey also noted a decrease in asthma prevalence and asthma attacks ( Fig. 4.17 ).
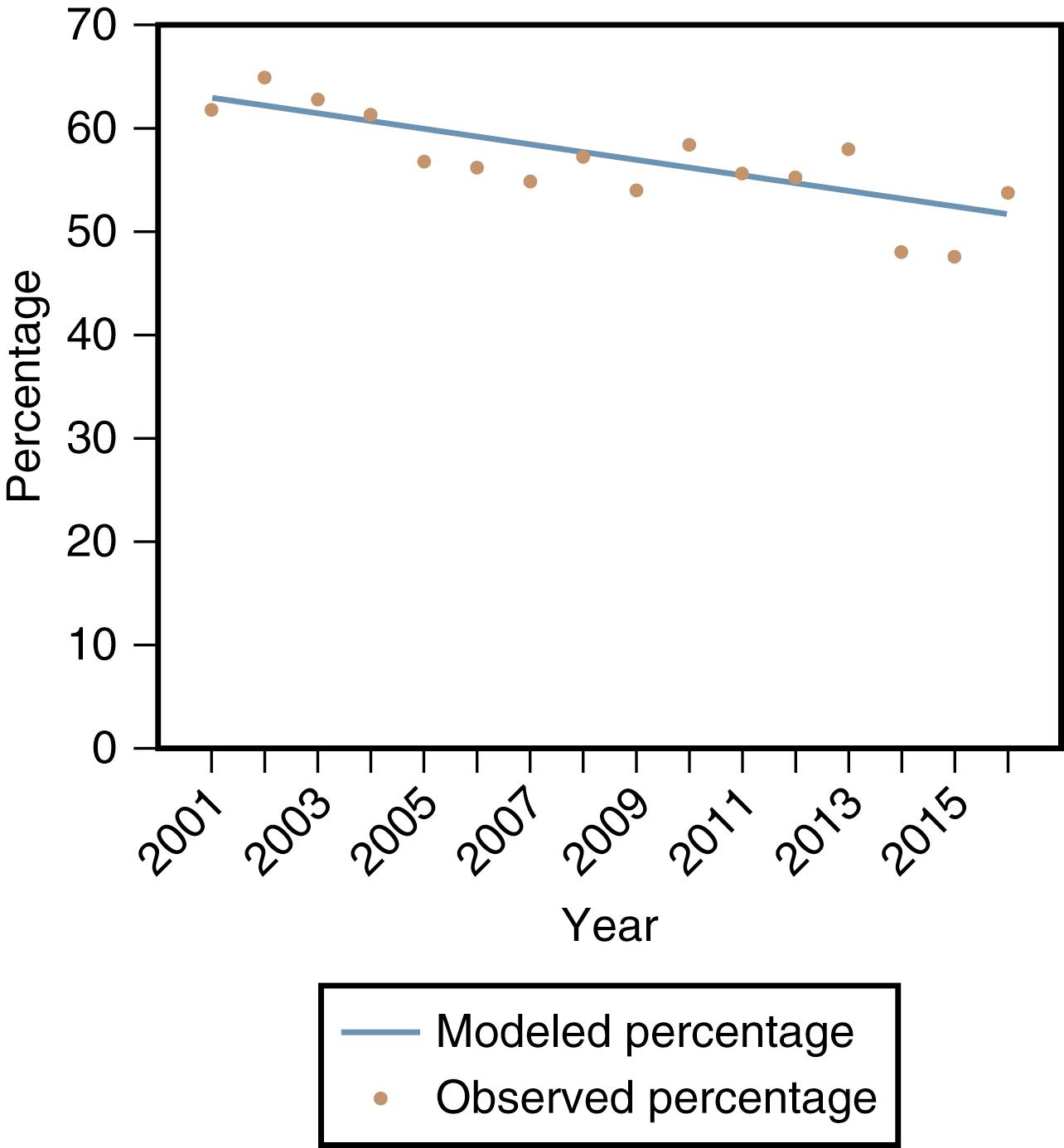
Defining characteristics of asthma, as elucidated by the National Heart, Lung, and Blood Institute (NHLBI), include the following: (1) lower airway obstruction that is partially or fully reversible either spontaneously or with bronchodilator or antiinflammatory treatments, (2) the presence of lower airway inflammation, and (3) increased lower airway responsiveness (bronchial hyperreactivity) (NHLBI National Asthma Education and Prevention Program [NAEPP], 2016). The last is characterized by inherent hyperreactivity of the airways to stimuli, including allergens, infection, exercise, chemical agents (such as methacholine), cold or dry air, emotions, and weather changes ( Fig. 4.18 ).
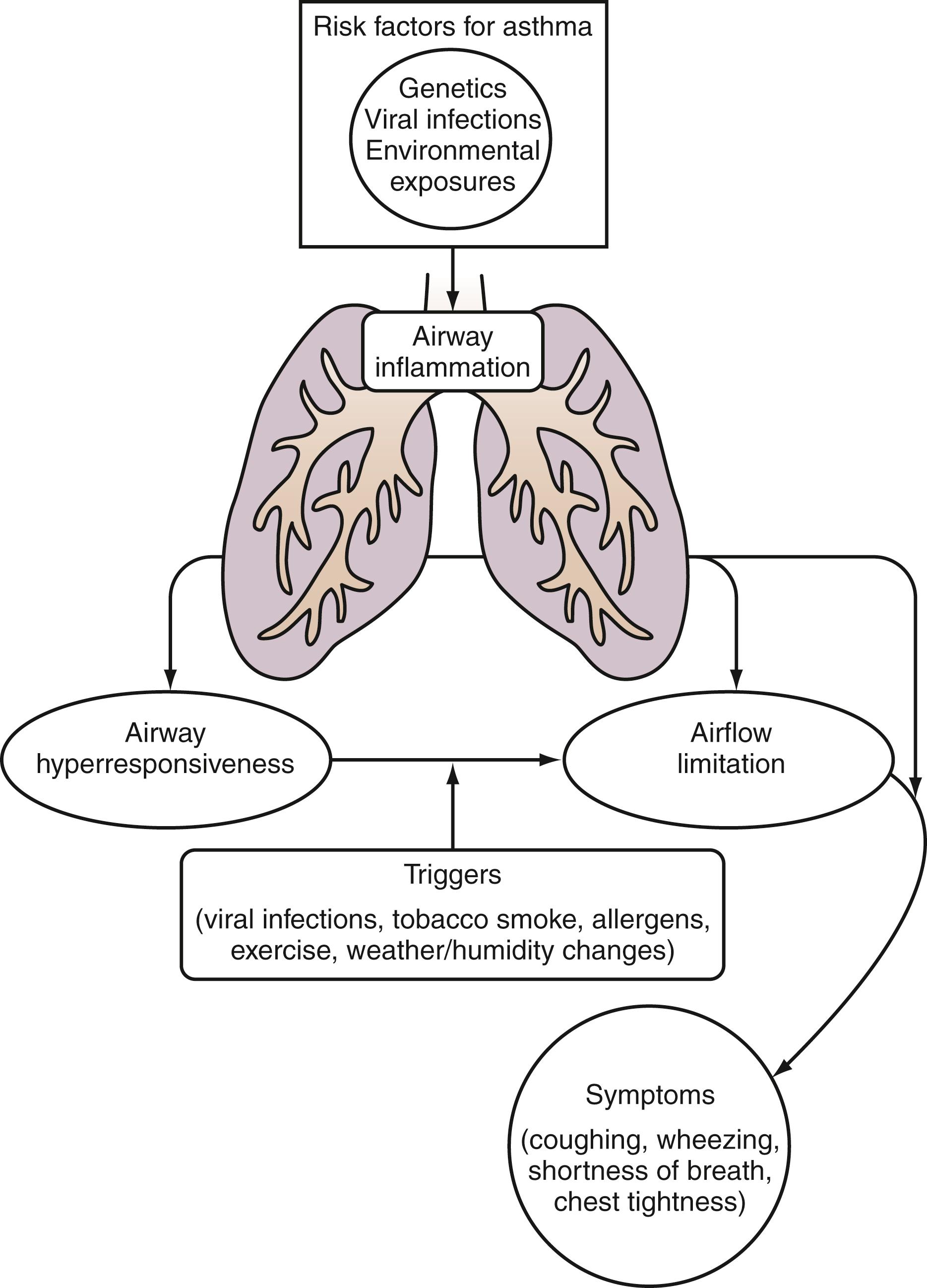
Many cases of asthma, particularly in children, have an atopic basis. Specific allergens implicated in atopic patients are pollen, mold spores, house dust mites, and animal dander, whereas drugs, food, and insect venoms typically cause similar symptoms of wheezing and respiratory distress as part of anaphylaxis (see earlier discussion). On exposure, these allergens, via cross-linking specific IgE, produce the characteristic features of asthma: mucosal edema, increased mucus production, and smooth muscle contraction that result in airway inflammation, airway hyperreactivity, and bronchoconstriction. These responses combine to produce a state of reversible obstruction of the large and small airways that is the hallmark of asthma.
Patients with asthma should be evaluated to determine the important triggers for their disease. Affected individuals are often aware of the specific trigger for exacerbations of their asthma. Viruses are the most common precipitants of acute asthma in children, especially rhinoviruses, respiratory syncytial virus, and parainfluenza viruses. These infections usually affect the upper and lower airways, producing rhinorrhea, nasal congestion, and wheezing, which tends to develop insidiously. In contrast, allergen-triggered episodes typically lack fever and have a more abrupt onset of wheezing.
The diagnosis of asthma, especially in younger children who cannot do pulmonary function tests, is frequently based on historical findings alone, indicating the importance of taking a thorough history. Updated NHLBI and American Academy of Pediatrics (AAP) guidelines have stressed the importance of complementing good history taking with objective measurements of lung function. Important parts of the medical history include description and pattern of the symptoms, precipitating factors, development of disease and treatments tried, family history, social history, history of exacerbations, impact on the patient and family, and the patient’s perception of the disease ( Fig. 4.19 ). In-office pulmonary function testing can be attempted in children of 5 years old and older, but it is important to understand that normal results in an asymptomatic patient do not exclude asthma. Peak flow monitoring is not recommended for use in diagnosis but can sometimes be used for monitoring. The greatest shortcomings of peak flow monitoring are that it is effort dependent, requires compliance, and does not measure small-airway function. In addition, there is a wide variability in types of peak flow meters, along with different reference ranges. When peak flow monitoring is used, patients should be given a written plan with instructions on what to do as their peak flow falls along with increase in symptoms.

As with many common diseases, asthma incidence is influenced by both genes and environment (see Fig. 4.23 ). Family history often reveals affected siblings, parents, or other relatives. An environmental survey can determine possible provocative factors, especially allergens, infections, occupational exposures, smoking, exercise, stress, climate, and medication use (NSAIDs, beta-blockers). The history should emphasize the frequency, duration, and intensity of suspected episodes ( Box 4.1 ). Individuals with asthma commonly present with recurrent episodes of wheezing that, depending on the severity, may require emergency treatment. Episodes may be infrequent and/or seasonal or may occur daily. The spectrum of presenting complaints, however, is broad, and affected individuals may complain only of mild, occasional wheezing or shortness of breath with exercise and/or colds or a persistent dry, hacking cough (particularly at night or in the morning). Breathlessness and chest tightness are also commonly reported.
a No single indicator is diagnostic in itself, but the presence of several increases the probability of asthma. Objective measures are essential to establish the diagnosis of asthma .
History of recurrent:
Coughing
Wheezing
Shortness of breath or rapid breathing
Chest tightness
Symptoms made worse by:
Viral infection
Tobacco smoke, wood smoke, and other irritants (e.g., strong odors or fumes)
Exercise
Allergens (e.g., house dust mites, pollens, cockroaches, molds, animal dander)
Changes in weather/humidity
Crying, laughing
Symptoms occur/worsen at night, waking the child and/or parent
Reversible airflow limitation by spirometry in children older than 4 years old and diurnal variation in peak flow
Wheezing (high-pitched whistling sounds when exhaling) may or may not be present
The frequency and severity of acute asthma episodes and the level of symptoms between episodes can be used to grade asthma severity and ultimately assess and monitor asthma control as shown in Table 4.5 and see Table 4.6 . Stepwise therapy recommendations were updated in the 2020 Focused Updates to the Asthma Management Guidelines by the Expert panel Working Group of the National Heart, Lung and Blood Institute as noted in Tables 4.6–4.10 .
| Components of Severity | Intermittent | Persistent | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mild | Moderate | Severe | |||||||||||
| Ages 0–4 Years | Ages 5–11 Years | Ages ≥12 Years | Ages 0–4 Years | Ages 5–11 Years | Ages ≥12 Years | Ages 0–4 Years | Ages 5–11 Years | Ages ≥12 Years | Ages 0–4 Years | Ages 5–11 Years | Ages ≥12 Years | ||
| Impairment | Symptoms | ≤2 days/week | ≥2 days/week but not daily | Daily | Throughout the day | ||||||||
| Nighttime awakenings | 0 | ≤2×/month | 1–2×/month | 3–4×/month | 3–4×/month | >1×/week but not nightly | >1×/week | Often 7×/week | |||||
| SABA a use for symptom control (not to prevent EIB a ) | ≤2× days/week | >2 days/week but not daily | >2 days/week but not daily and not more than once on any day | Daily | Several times per day | ||||||||
| Interference with normal activity | None | Minor limitation | Some limitation | Extremely limited | |||||||||
| Lung Function | Not applicable | Normal FEV 1 between exacerbations | Normal FEV 1 between exacerbations | Not applicable | Not applicable | Not applicable | |||||||
| → FEV 1 a (% predicted) | >80% | >80% | >80% | >80% | 60–80% | 60–80% | <60% | <60% | |||||
| → FEV 1 /FVC a | >85% | Normal b | >80% | Normal b | 75–80% | Reduced 5% b | <75% | Reduced 5% b | |||||
| Generally, more frequent and intense events indicate greater severity . | |||||||||||||
| Risk | Asthma exacerbations requiring oral systemic corticosteroids c | 0–1/year | ≥2 exacerb. In 6 months, or wheezing ≥4× per year lasting >1 day AND risk factors for persistent asthma | ≥2/year | Generally, more frequent and intense events indicate greater severity. | ||||||||
| Consider severity and interval since last asthma exacerbation. Frequency and severity may fluctuate over time for patients in any severity category. Relative annual risk of exacerbations may be related to FEV 1 . a |
|||||||||||||
a EIB, Exercise-induced bronchospasm; FEV 1 , forced expiratory volume in 1 second; FVC, forced vital capacity; ICS, inhaled corticosteroid; SABA, short-acting beta 2 -agonist.
b Normal FEV 1 /FVC by age: 8–19 years, 85%; 20–39 years, 80%; 40–59 years, 75%; 60–80 years, 70%.
c Data are insufficient to link frequencies of exacerbations with different levels of asthma severity. Generally, more frequent and intense exacerbations (e.g., requiring urgent care, hospital or intensive care admission, and/or oral corticosteroids) indicate greater underlying disease severity. For treatment purposes, patients with ≥2 exacerbations may be considered to have persistent asthma, even in the absence of impairment levels consistent with persistent asthma.
| Components of Control | Well Controlled | Not Well Controlled | Very Poorly Controlled | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Ages 0–4 Years | Ages 5–11 Years | Ages ≥12 Years | Ages 0–4 Years | Ages 5–11 Years | Ages ≥12 Years | Ages 0–4 Years | Ages 5–11 Years | Ages ≥12 Years | ||
| Impairment | Symptoms | ≤2 days/week | ≤2 days/week but not more than once on each day | ≤2 days/week | >2 days/week | >2 days/week or multiple times on ≤2 days/week | >2 days/week | Throughout the day | ||
| Nighttime awakenings | ≤1×/month | ≤2×/month | >1×/month | ≥2×/month | 1–3×/week | >1×/week | ≥2×/week | ≥4×/week | ||
| Interference with normal activity | None | Some limitation | Extremely limited | |||||||
| SABA a use for symptom control (not to prevent EIB a ) | ≤2 days/week | >2 days/week | Several times per day | |||||||
| Lung Function | Not applicable | Not applicable | Not applicable | |||||||
| → FEV 1 a (% predicted) or peak flow (% personal best) | >80% | >80% | 60–80% | 60–80% | <60% | <60% | ||||
| → FEV 1 /FVC a | >80% | Not applicable | 75–80% | Not Applicable | <75% | Not applicable | ||||
| Validated questionnaires b | ||||||||||
| → ATAQ a | Not applicable | Not applicable | 0 | Not applicable | Not applicable | 1–2 | Not applicable | Not applicable | 3–4 | |
| → ACQ a | ≤0.75 c | ≥1.5 | Not applicable | |||||||
| → ACT a | ≥20 | 16–19 | ≤15 | |||||||
| Risk | Asthma exacerbations requiring oral systemic corticosteroids | 0–1/year | 2–3/year | ≥2/year | >3/year | ≥2/year | ||||
| Consider severity and interval since last asthma exacerbation. | ||||||||||
| Reduction in lung growth/Progressive loss of lung function | Not applicable | Evaluation requires long-term follow-up care. | Not applicable | Evaluation requires long-term follow-up care. | Not applicable | Evaluation requires long-term follow-up care. | ||||
| Treatment-related adverse effects | Medication side effects can vary in intensity from none to very troublesome and worrisome. The level of intensity does not correlate to specific levels of control but should be considered in the overall assessment of risk. |
|||||||||
a ACQ, Asthma Control Questionnaire©; ACT, Asthma Control TestTM; ATAQ, Asthma Therapy Assessment Questionnaire©.
b Minimal important difference: 1.0 for the ATAQ; 0.5 for the ACQ; not determined for the ACT.
c ACQ values of 0.76–1.4 are indeterminate regarding well-controlled asthma.
| Intermittent Asthma | Management of Persistent Asthma in Individuals Ages 0–4 Years | |||||
|---|---|---|---|---|---|---|
| Treatment | Step 1 | Step 2 | Step 3 | Step 4 | Step 5 | Step 6 |
| Preferred | PRN SABA and At the start of RTI: Add short course daily ICS a |
Daily low-dose ICS and PRN SABA | Daily low-dose ICS-LABA and PRN SABA a or daily low-dose ICS + montelukast, b or daily medium-dose ICS, and PRN SABA | Daily medium-dose ICS-LABA and PRN SABA | Daily high-dose ICS-LABA and PRN SABA | Daily high-dose ICS-LABA + oral systemic corticosteroid and PRN SABA |
| Alternative | Daily montelukast b or Cromolyn, b and PRN SABA | Daily medium-dose ICS + montelukast b and PRN SABA | Daily high-dose ICS + montelukast b and PRN SABA | Daily high-dose ICS + montelukast b + oral systemic corticosteroid and PRN SABA | ||
| For children age 4 years only, see Step 3 and Step 4 on Management of Persistent Asthma in Individuals Ages 5–11 Years diagram | ||||||
| Assess Control | ||||||
Consult with asthma specialist if Step 3 or higher is required. Consider consultation at Step 2. |
||||||
a Updated based on the 2020 guidelines.
b Cromolyn and montelukast were not considered for this update and/or have limited availability for use in the United States. The FDA issued a Boxed Warning for montelukast in March 2020.
| Intermittent Asthma | Management of Persistent Asthma in Individuals Ages 5–11 Years | |||||
|---|---|---|---|---|---|---|
| Treatment | Step 1 | Step 2 | Step 3 | Step 4 | Step 5 | Step 6 |
| Preferred | PRN SABA | Daily low-dose ICS and PRN SABA | Daily and PRN combination low-dose ICS-formoterol a | Daily and PRN combination medium-dose ICS-formoterol a | Daily high-dose ICS-LABA and PRN SABA | Daily high-dose ICS-LABA + oral systemic corticosteroid and PRN SABA |
| Alternate | Daily LTRA, b or Cromolyn, b or Nedocromil, b or Theophylline, b and PRN SABA | Daily medium-dose ICS and PRN SABA or daily low-dose ICS-LABA, or daily low-dose ICS + LTRA, b or daily low-dose ICS + Theophylline, b and PRN SABA | Daily medium-dose ICS-LABA and PRN SABA or daily medium-dose ICS + LTRA b or daily medium-dose ICS + Theophylline, b and PRN SABA | Daily high-dose ICS + LTRA b or daily high-dose ICS + Theophylline, b and PRN SABA | Daily high-dose ICS + LTRA b + oral systemic corticosteroid or daily high-dose ICS + Theophylline b + oral systemic corticosteroid, and PRN SABA | |
| Steps 2–4: Conditionally recommend the use of subcutaneous immunotherapy as an adjunct treatment to standard pharmacotherapy in individuals ≥5 years of age whose asthma is controlled at the initiation, build up, and maintenance phases of immunotherapy a | Consider Omalizumab a , c | |||||
| Assess Control | ||||||
Consult with asthma specialist if Step 4 or higher is required. Consider consultation at Step 3. |
||||||
a Updated based on the 2020 guidelines.
b Cromolyn, Nedocromil, LTRAs including montelukast, and Theophylline were not considered in this update and/or have limited availability for use in the United States, and/or have an increased risk of adverse consequences and need for monitoring that make their use less desirable. The FDA Issued a Boxed Warning for montelukast in March 2020.
c Omalizumab is the only asthma biologic currently FDA-approved for this age range.
| Intermittent Asthma | Management of Persistent Asthma in Individuals Ages 12+ Years | |||||
|---|---|---|---|---|---|---|
| Treatment | Step 1 | Step 2 | Step 3 | Step 4 | Step 5 | Step 6 |
| Preferred | PRN SABA | Daily low-dose ICS and PRN SABA or PRN concomitant ICS and SABA a | Daily and PRN combination low-dose ICS-formoterol a | Daily and PRN combination medium-dose ICS-formoterol a | Daily medium-high dose ICS-LABA + LAMA and PRN SABA a | Daily high-dose ICS-LABA + oral systemic corticosteroids + PRN SABA |
| Alternate | Daily LTRA b and PRN SABA or Cromolyn, b or Nedocromil, b or Zileuton, b or Theophylline, b and PRN SABA | Daily medium-dose ICS and PRN SABA or daily low-dose ICS-LABA, or daily low-dose ICS + LAMA, a or daily low-dose ICS + LTRA, b and PRN SABA or daily low-dose ICS + Theophylline b or Zileuton, b and PRN SABA | Daily medium-dose ICS-LABA or daily medium-dose ICS + LAMA, and PRN SABA a or daily medium-dose ICS + LTRA, b or daily medium-dose ICS + Theophylline, b or daily medium-dose ICS + Zileuton, b and PRN SABA | Daily medium-high dose ICS-LABA or daily high-dose ICS + LTRA, b and PRN SABA | ||
| Consider adding Asthma Biologics (e.g., anti-IgE, anti-IL5, anti-IL5R, anti-IL4/IL13) c | ||||||
| Steps 2–4: Conditionally recommend the use of subcutaneous immunotherapy as an adjunct treatment to standard pharmacotherapy in individuals ≥5 years of age whose asthma is controlled at the initiation, build up, and maintenance phases of immunotherapy a | ||||||
| Assess Control | ||||||
Consult with asthma specialist if Step 4 or higher is required. Consider consultation at Step 3. |
||||||
a Updated based on the 2020 guidelines.
b Cromolyn, Nedocromil, LTRAs including Zileuton and montelukast, and Theophylline were not considered for this update, and/or have limited availability for use in the United States, and/or have an increased risk of adverse consequences and need for monitoring that make their use less desirable. The FDA issued a Boxed Warning for montelukast in March 2020.
c The AHRQ systematic reviews that informed this report did not include studies that examined the role of asthma biologics (e.g., anti-IgE, anti-IL5, anti-IL5R, anti-IL4/IL13). Thus, this report does not contain specific recommendations for the use of biologics in asthma in Steps 5 and 6.
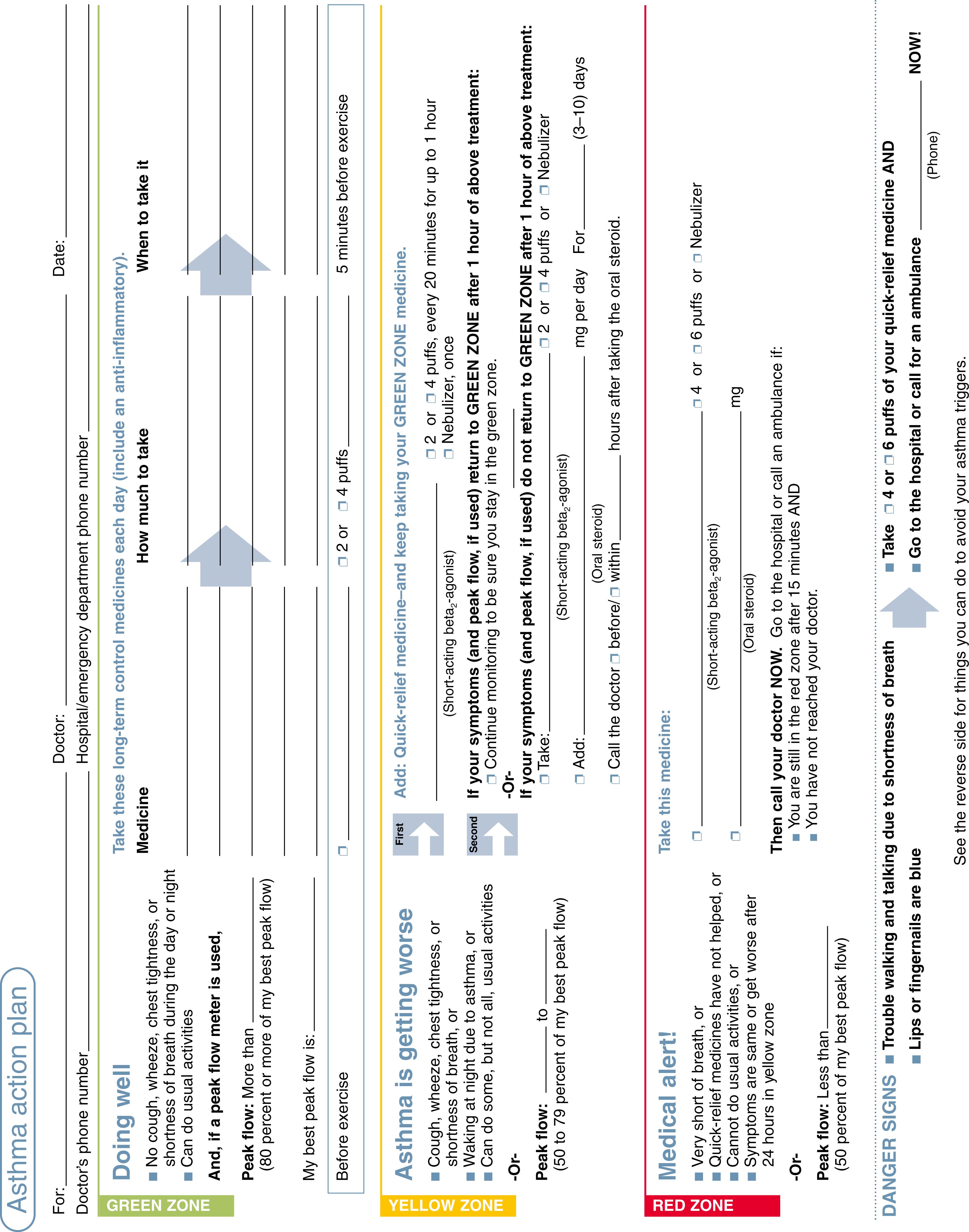
The early stages of an asthma exacerbation in children are characterized by the onset of cough, chest tightness, and chest retractions or audible wheezing. The parents should be educated to observe their child for the warning signs and identify the onset of asthmatic exacerbation at home. A written asthma action plan that details use of rescue medicines (such as albuterol) can minimize emergency department visits for asthma ( Fig. 4.20 ).
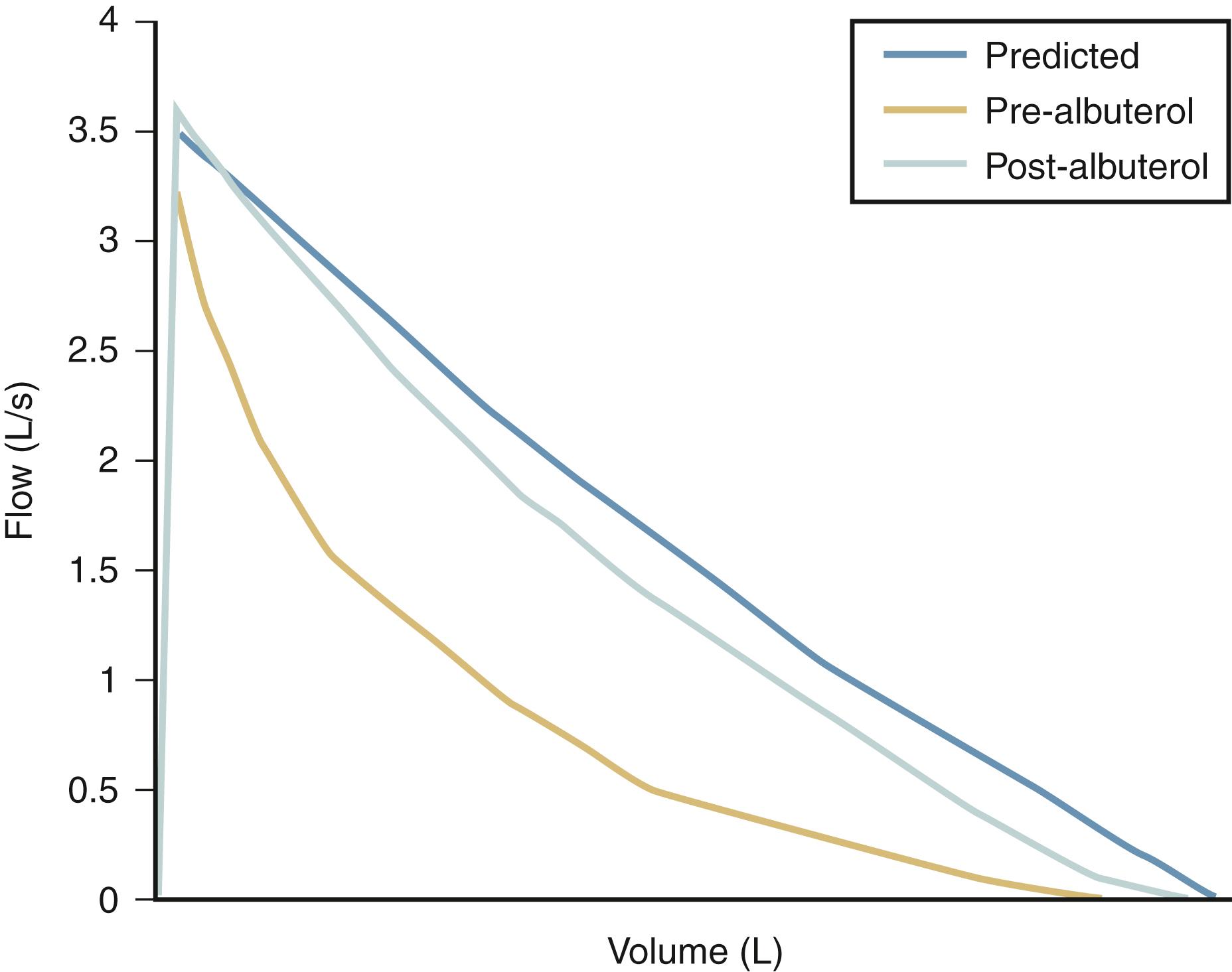
Asthma should be considered part of the differential diagnosis in any child with recurrent or chronic lower respiratory symptoms or signs. Even though a high index of suspicion must be maintained, excessive or erroneous diagnoses may result if they are made hastily without appropriate supportive evidence; normal children or those with potentially more severe disorders may be mistakenly diagnosed with asthma and inappropriately treated. Box 4.2 provides a differential diagnosis for asthma. Parents must be instructed that physician assessment is essential during suspected episodes of asthma so that wheezing or other signs of lower airway obstruction and reversibility may be documented. Pulmonary function tests in asthmatic children older than 5 years may show airway obstruction at baseline or after appropriate challenge with methacholine, exercise, or cold air and document reversibility after administration of an aerosolized bronchodilator ( Fig. 4.21 ). For children younger than 5 years old or for those in whom testing is unreliable, the diagnosis must be made on the basis of historical and physical findings and clinical response to bronchodilator or antiinflammatory medications. Lack of an immediate response to a bronchodilator does not eliminate asthma as a consideration, however. In the instance of poor response to prescribed medications, appropriate inhaler technique should be addressed, as well as the importance of using a spacer device with any metered dose inhaler to ensure adequate medication delivery to the distal airways ( Fig. 4.22 ).
| Diagnosis | Age | Findings that Differentiate from Asthma | Diagnostic Tests |
|---|---|---|---|
| Bronchiolitis | <24 months | No atopy, typically not responsive to SABA, context of fever and viral respiratory tract infection | Clinical diagnosis |
| Viral-Induced wheeze | Any age (<4 years more common) | Wheezing only with viral respiratory tract infections, less atopy; usually resolves by age 4 years | PFT if frequent or severe |
| Bronchopulmonary dysplasia | <24 months | Symptoms since birth in premature infant | Clinical diagnosis |
| Chronic aspiration/GFRD | Any | Chronic cough especially after meals, gastrointestinal symptoms | Gastroenterology referral, endoscopy, pH probe |
| Tracheomalacia/bronchomalacia | <24 months | Symptoms since birth, constant (as opposed to intermittent or triggered), stridor present with tracheomalacia | Laryngoscopy |
| Bronchiectasis | Any | Symptoms since birth, wet chronic cough, recurrent wheeze with focal lung findings, recurrent pneumonia in same location on CXR, sometimes with hemoptysis | Chest CT, bronchoscopy |
| Food allergy | Any | First-time wheeze in context of new food exposure and other signs of food allergy flip swelling, vomiting, hives, hypotension) | Allergy testing, PFT |
| Anxiety/panic attack | School age or older | No wheezing, no improvement with SABA | Clinical diagnosis, PFT results normal |
| Vocal cord dysfunction | Adolescent + | Acute onset of symptoms within minutes of exercise or exposure to irritant symptoms quickly resolve and do not respond to SABA, sensation of airway closing, throat tightness, no symptoms during sleep, occasionally will hear inspiratory stridor on examination, hoarse voice | PFT shows classic pattern of dynamic airflow obstruction, laryngoscopy |
| Heart failure | Any | Fever (viral myocarditis) missed congenital heart disease, failure to thrive, symptoms worsen with feeding in young infant or with laying down, murmur, poor central pulses, hepatomegaly, crackles | Echocardiography, electrocardiography, CXR |
| Foreign body aspiration | <4 years (could be any age) | Acute onset of symptoms, unilateral findings, history of choking spell | Anteroposterior/posteroanterior or bilateral decubitus CXR, bronchoscopy |
| Mass effect (from vascular anomaly or tumor) | Any | Unilateral wheeze or stridor (may be positional, if due to a mass, ring, or sling around upper airways), symptoms worse with laying down, weight loss and other systemic signs it oncologic | Chest CT, bronchoscopy |
| Underlying immunodeficiency | Any | Recurrent bacterial pneumonia | CXR, immunology evaluation |
| Allergic bronchopulmonary aspergillosis | Any | Difficult-to-control asthma symptoms, chronic cough and intermittent fevers | Sputum, Aspergillus IgE and IgG, CXR |
| Cystic fibrosis | Any | Failure to thrive (may still have a normal newborn screen) | Sweat chloride testing, genetic testing |

A thorough physical examination provides valuable information regarding the diagnosis of asthma and its severity and chronicity. The physical findings in asthma vary with the state of activity of the disease process at the time of examination. The findings of acute asthma are markedly different from those of chronic and latent or quiescent asthma. Between episodes, the examination is usually entirely normal, although prolongation of the expiratory phase is sometimes noted. Clubbing as a sign of chronic asthma is rare and suggests another chronic pulmonary disease.
During acute asthma, the following historical features should be noted: time of onset, possible triggers, present medications, comparison with previous episodes, and presence of complicating factors (e.g., vomiting, fever, or chest pain). Examination should document accessory muscle use, retractions, and color. Auscultation should be done to assess air exchange, wheezing, and inspiratory-to-expiratory ratio. The ability to speak (words, phrases, or complete sentences) is a useful measure of dyspnea. Lethargy, decreased air exchange, and increased work of breathing are the most worrisome signs indicating acute deterioration.
In addition, rales are often heard, and pulse, respiratory rate, and blood pressure are frequently elevated. Pulsus paradoxus, an exaggerated decrease in systolic blood pressure during inspiration ( Table 4.11 ), correlates highly with degree of airway obstruction, although it is rarely measured clinically. This phenomenon may result from physical forces on the pericardium that impede venous return and reduce cardiac output during forced inspiration. Normally, the inspiratory decrease in systolic blood pressure is less than 10 mm Hg and is not discernible during routine sphygmomanometry. In acute asthma, it is usually greater than 10 mm Hg (up to 30 and 40 mm Hg) and is easily detectable.
| Blood Pressure in Relation to Time and Respiratory Phase (mm Hg) | |||||
|---|---|---|---|---|---|
| Expiration | Inspiration | Expiration | Inspiration | Expiration | |
| Normal (no airway obstruction) | 125/70 | 120/70 | 125/70 | 120/70 | 125/70 |
| Asthma (airway obstruction) | 125/70 | 100/70 | 125/70 | 100/70 | 125/70 |
| Method | |||||
|
|||||
Individuals with asthma may be distinguished by their characteristic symptoms and signs during acute episodes, which typically change with the degree of airway obstruction. Symptoms usually consist of progressively increasing shortness of breath and difficulty breathing with or without rhinorrhea, low-grade fever, and vomiting. On examination, expiratory wheezing or a prolonged expiratory phase may be the only manifestation of mild asthma. However, as the obstructive process progresses, the expiratory phase becomes longer and the wheezing becomes louder and occurs on both inspiration and expiration. Eventually, airways collapse and signs of hyperinflation develop (low diaphragms, decreased lateral excursions of the chest wall with breathing, and hyperresonance to percussion). Visible sternocleidomastoid contractions; increased anteroposterior chest diameter; circumoral cyanosis; and suprasternal, intercostal, and substernal retractions occur. Subjectively, the patient experiences chest tightness and anxiety and works harder to breathe. Accessory muscle use and retractions can develop with or without marked wheezing on auscultation. To maximize air exchange, the child assumes a characteristic sitting posture, bending slightly forward. Frequent examinations are warranted, and any change in sensorium requires prompt evaluation. As respiratory muscles tire, the patient becomes lethargic and cyanotic, even with supplemental oxygen. A decrease in wheezing with increased air entry represents response to therapy. Decreased wheezing with decreased air entry is an ominous sign. With extreme fatigue, respiratory muscles fail, retractions decrease, and respiratory failure is imminent unless appropriate therapy is promptly initiated. After initial examination, serial assessment of the degree of respiratory distress, using the parameters outlined in Table 4.12 , assists determination of the response to therapy.
| Mild | Moderate | Severe | Life-Threatening | |
|---|---|---|---|---|
| Symptoms | ||||
| Breathlessness a | While walking | While at rest (infant—softer, shorter cry, difficulty feeding) | While at rest (infant—stops feeding) | |
| Talks in: | Sentences | Phrases | Words | |
| Alertness | May be agitated | Usually agitated | Usually agitated | Drowsy or confused |
| Signs | ||||
| Respiratory rate b | Increased | Increased | Significantly increased | Often normal or decreased |
| Accessory muscle use | Unusual | Common | Common | Paradoxic abdominal movements |
| Wheeze | Moderate, often only end-expiratory | Loud, throughout exhalation | Usually loud, in both inspiration and expiration | Absence of wheeze and air movement |
| Pulse | Normal to tachycardic | Tachycardic | Tachycardic | Bradycardic |
| Pulsus paradoxus c | <10 mm Hg | 10–20 mm Hg | 20–40 mm Hg | Absence suggests respiratory muscle fatigue |
| Functional Assessment | ||||
| PEF (percent predicted or personal best) | >70% | 40%–69% | <40% | <25% |
| PaO 2 (room air) | Normal | >60 mm Hg | <60 mm Hg; possible cyanosis | Cyanosis |
| PaCO 2 | <42 mm Hg | <42 mm Hg | >42 mm Hg | >42 mm Hg |
| SaO 2 percent (room air) | >95% | 90%–95% | <90% | <90% |
a Parents’ or physician’s impression of degree of child’s breathlessness.
b See Table 4.9 for normal respiratory rates by age.
c Pulsus paradoxus does not correlate with phase of respiration in small children.
A particularly useful aspect of the physical examination is the respiratory rate, which increases as the degree of airway obstruction progresses. Respiratory rates of normal children are shown in Table 4.13 .
| Age | Sleeping | Awake | ||||
|---|---|---|---|---|---|---|
| No. | Mean | Range | No. | Mean | Range | |
| 6–12 months old | 6 | 27 | 22–31 | 3 | 64 | 58–75 |
| 1–2 years old | 6 | 19 | 17–23 | 4 | 35 | 30–40 |
| 2–4 years old | 16 | 19 | 16–25 | 15 | 31 | 23–42 |
| 4–6 years old | 23 | 18 | 14–23 | 22 | 26 | 19–36 |
| 6–8 years old | 27 | 17 | 13–23 | 28 | 23 | 15–30 |
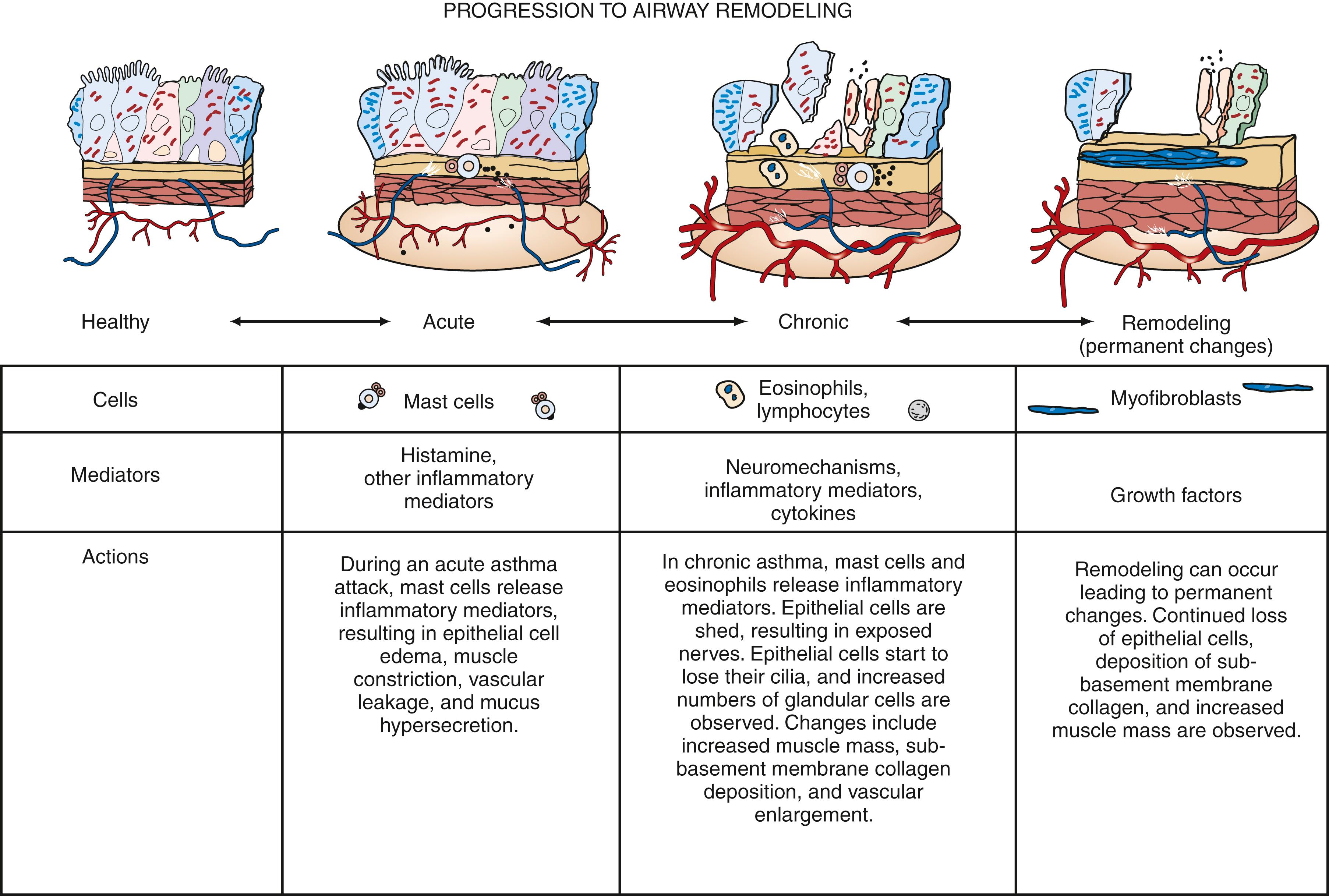
The underlying pathology of asthma is lung inflammation. Histopathologic features of acute asthma include airway infiltration with inflammatory cells, increased intraluminal mucus with plugging of small airways, edema, bronchoconstriction, and smooth muscle hypertrophy. Because asthma has bronchoconstrictive and inflammatory components, the ideal therapeutic regimen should incorporate a combination of bronchodilator and antiinflammatory agents. Characteristic features of asthmatic inflammation include mast cell activation, inflammatory cell infiltration, edema, denudation and disruption of the bronchial epithelium, collagen deposition beneath the basement membrane, goblet cell hyperplasia, and smooth muscle thickening. These morphologic changes may not be completely reversible and may contribute to airway remodeling ( Fig. 4.23 ).
Education is a key component of asthma therapy, incorporating information about disease pathogenesis; avoidance of environmental triggers (including second-hand tobacco smoke exposure); benefits and risks of medications; and a written, individualized therapeutic plan for chronic and acute management that outlines the goals of asthma therapy, as well as indications and correct use for prescribed medications.
The radiographic features of hyperinflation, peribronchial cuffing, and atelectasis, which are characteristic of uncomplicated acute asthma, are illustrated in Fig. 4.24 . Complications are diagnosed radiographically but may be suggested by symptoms and signs. Pneumothorax ( Fig. 4.25 ) should be suspected in any patient with asthma who develops chest pain associated with dyspnea, cyanosis, tachypnea, and occasionally cough. Examination reveals respiratory distress, marked hyperinflation and decreased chest wall excursion, and decreased or absent breath sounds on the affected side. With tension pneumothorax, the trachea, mediastinum, and cardiac landmarks may be shifted to the opposite side. Pneumomediastinum and subcutaneous emphysema ( Fig. 4.26 ), usually involving the neck and supraclavicular areas, are more common than pneumothorax. When mild, they may be asymptomatic and may be detected incidentally on radiographs. With more extensive air dissection, the patient may complain of neck and chest pain, and the subcutaneous emphysema may be visibly evident as a soft tissue swelling of the neck and chest that is crepitant on palpation. Pneumothorax and pneumomediastinum can produce characteristic auscultatory findings, including a crackling “mediastinal crunch” at the base of the heart and a systolic crunch or knock. The latter sound has been referred to as noisy pneumothorax and is frequently audible to the patient and physician without the aid of a stethoscope.
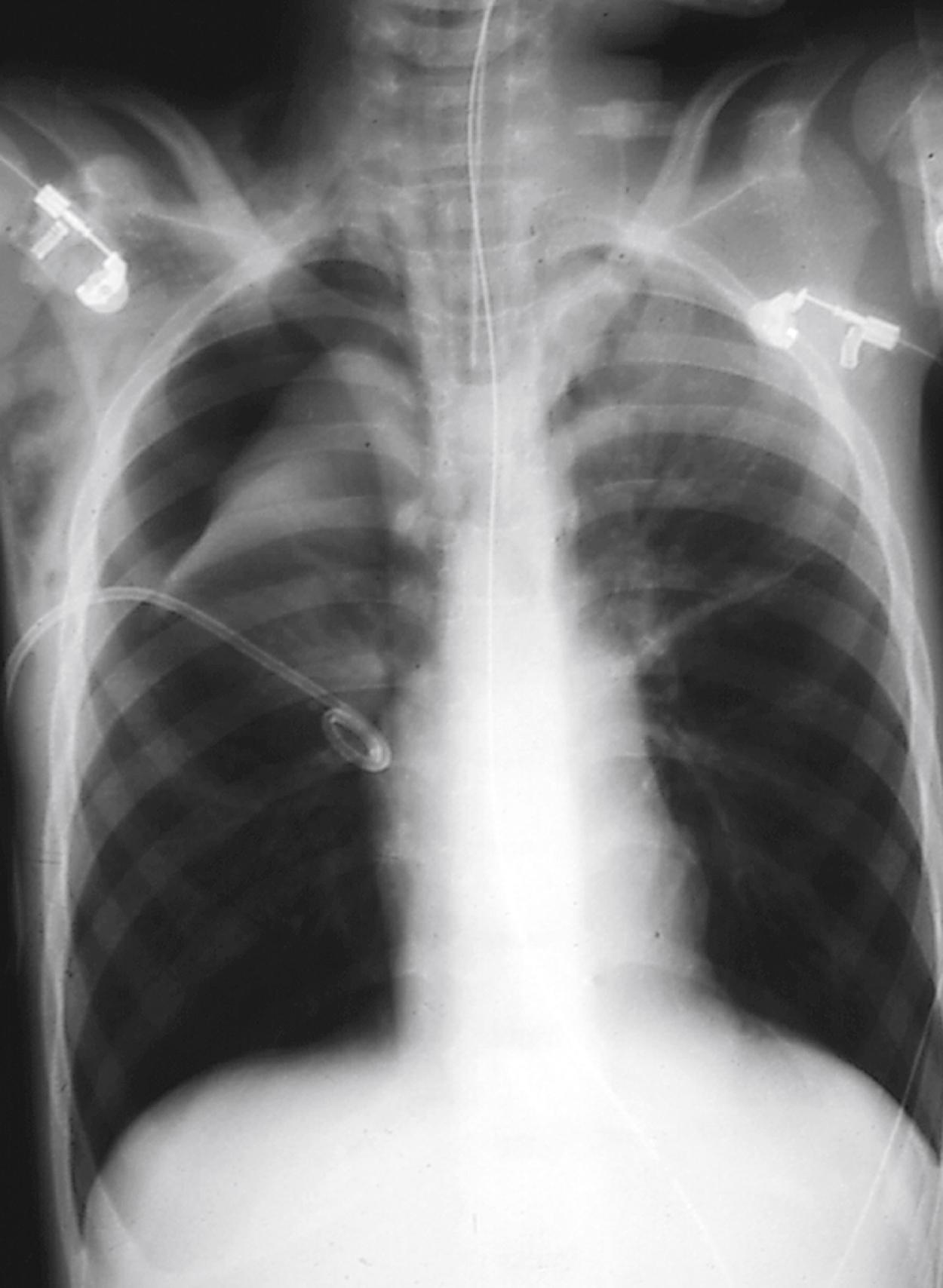
Other complications that may be observed on physical examination include those induced by chronic systemic steroid use, such as weight gain, “moon-type” facies, hirsutism, polycythemia (red, ruddy complexion) ( Fig. 4.27 ), and short stature (see Chapter 9 ). The introduction of effective antiinflammatory therapy, particularly inhaled corticosteroids, has made such sequelae rare.
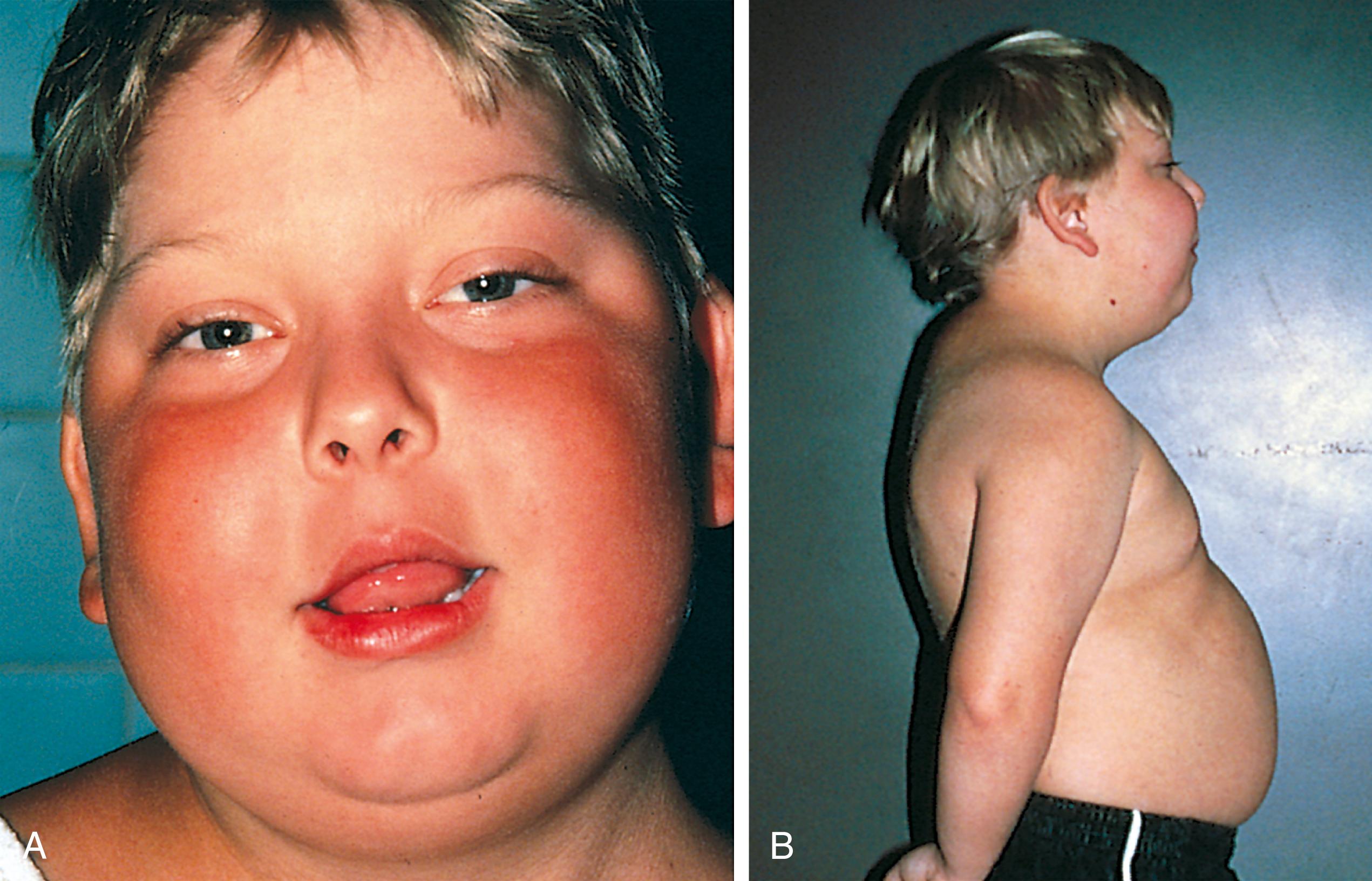
In children with recent onset of wheezing, asthma must be differentiated from other disorders associated with wheezing. In infants, this differentiation includes bronchiolitis, the features of which are listed in Table 4.14 (see Chapter 17 ). This differentiation may be difficult, because up to 50% of children with recurrent bronchiolitis are later diagnosed with asthma, and asthma exacerbations are often triggered by viral infection. However, the distinction remains a clinically useful one for the following reasons: (1) the children with bronchiolitis who do not develop asthma may be inappropriately labeled as asthmatic; and (2) children younger than 2 years old frequently do not respond to inhaled bronchodilators. In infants and young children who wheeze with viral upper respiratory infections, different patterns of illness may emerge over time.
| Asthma | Bronchiolitis | |
|---|---|---|
| Primary etiologies | Viruses, allergens, exercise, and so on | Respiratory syncytial virus, other viruses |
| Age at onset | 50% by 2 years old | <24 months old |
| 80% by 5 years old | ||
| Recurrent wheezing | Yes (characteristic) | 70% (≤2 episodes) |
| 30% progress to asthma (≥3 episodes) | ||
| Onset of wheezing | Acute if allergic or exercise induced | Insidious |
| Concomitant symptoms of upper respiratory infection | Yes, if infectious | Yes |
| Family history of allergy and asthma | Frequent | Infrequent in children with ≤2 episodes |
| Nasal eosinophilia | With allergic rhinitis | Absent |
| Chest auscultation | If viral, as in bronchiolitis | Fine, sibilant rales, and coarse inspiratory and expiratory wheezes |
| Nonviral: High-pitched expiratory wheezes | ||
| Concomitant allergic manifestations | Yes, if allergic asthma | Usually absent |
| Immunoglobulin E (IgE) level | Elevated (if allergic) | Normal |
| Response to bronchodilator | Yes (characteristic) | Unresponsive or partially responsive |
The Tucson Children’s Respiratory Study, a large, longitudinal assessment of respiratory illnesses in children, identified different patterns of wheezing illness in children. As summarized in Fig. 4.28 , 51% of children enrolled in this study never wheezed, 20% had transient infant wheezing (defined as wheezing during the first 3 years of life but no wheezing at 6 years old), 14% had persistent wheezing (defined as wheezing during the first 3 years of life with wheezing present at 6 years old), and 15% had late-onset wheezing (defined as no wheezing during the first 3 years of life but wheezing present at 6 years old). Children with transient wheezing had significantly lower lung function in infancy (before any wheezing illness) than all the other groups ( Table 4.15 ). At 6 years old, children with early transient wheezing had significantly lower lung function than those who had never wheezed, and children with persistent wheezing had the lowest levels of lung function of all the groups.
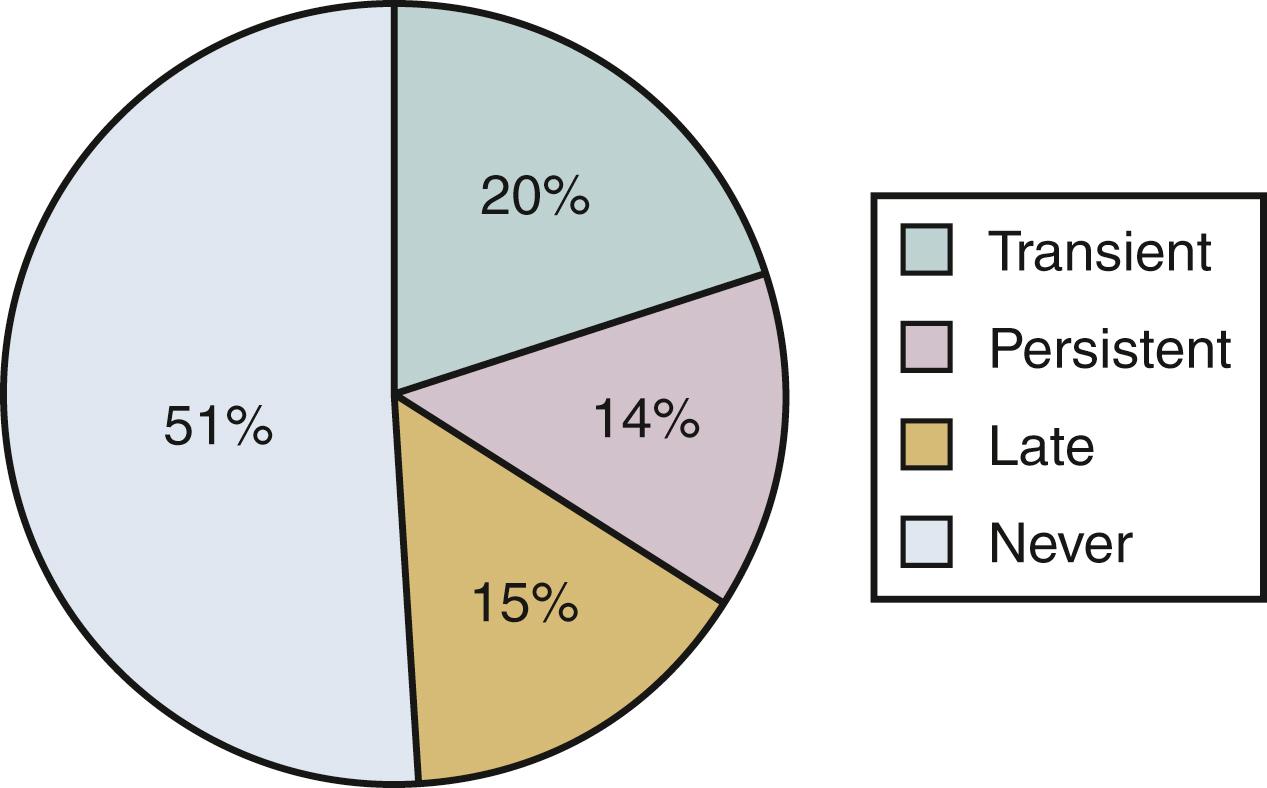
| Group | Lung Function ( FRC) |
||
|---|---|---|---|
| n (%) | Infancy | 6 Years Old | |
| No wheezing | 425 (51.5%) | Normal | Normal |
| Transient wheezing | 164 (19.9%) | Diminished | Diminished |
| Persistent wheezing | 113 (13.7%) | Normal | Diminished |
| Late-onset wheezing | 124 (15%) | Normal | Normal |
It is hypothesized that children with early transient wheezing have congenitally smaller airways, which predispose to wheezing with viral illnesses. As their airways grow in size with age, these children outgrow wheezing with viral infections. In contrast, children with persistent wheezing are born with normal airway function, which deteriorates over time, reflecting the effects of the chronic asthmatic disease process. Distinguishing transient versus persistent wheezing during infancy and early childhood remains problematic. Infant pulmonary function testing is still experimental and not widely available, there are currently no reliable genetic markers, and the use of any single biochemical marker is controversial. However, a combination of clinical and biochemical markers may be useful in predicting persistent wheezing ( Box 4.3 ). These include increased serum IgE level, atopic dermatitis and rhinitis (unrelated to upper respiratory infection) during the first year of life, severe lower respiratory infections requiring hospitalization, and diminished airway function as measured by spirometry at 6 years old. Other factors associated with persistent wheezing include a family history of asthma and/or allergy and perinatal exposure to passive tobacco smoke. A clinical index has been developed to predict which children with recurrent wheezing are at risk for persistent asthma ( Table 4.16 ).
Family history of asthma (maternal > paternal)
Atopy
↑ IgE or positive allergy skin tests
Eczema
Rhinitis
Allergen exposure (dust mites, animals)
RSV bronchiolitis
Gender (males > females)
Environmental smoke exposure
Severity in childhood
| Major Criteria | Minor Criteria |
|---|---|
|
|
a The loose index for prediction of asthma consists of early wheezing plus at least one of two major criteria or two of three minor criteria. The stringent index for prediction of asthma consists of early frequent wheezing plus at least one of two major criteria or two of three minor criteria.
Although children with pneumonia (particularly of viral origin) may wheeze, they are more likely to have rales or normal findings on auscultation, with the diagnosis suggested by tachypnea in association with retractions, nasal flaring, or expiratory grunting. Other causes of wheezing are listed in Table 4.17 . Airway compression by anomalous vessels (see Chapter 5 ) or mass lesions is often distinguishable from bronchiolitis by the absence of signs of infection and from asthma by failure to respond to bronchodilators. The history and presence of infiltrates help in the diagnosis of aspiration, which can mimic asthma closely, often responding to bronchodilator therapy. Radiographic studies (such as barium swallow with fluoroscopy) can be helpful in distinguishing among these entities ( Fig. 4.29 ). pH probe testing may be required to identify gastroesophageal reflux (see Chapter 17 ), although a trial of antireflux medicines is often attempted.
| Symptoms and Signs | Diseases Associated With Wheezing | |
|---|---|---|
| In Infants | In Older Children | |
| Positional changes | Anomalies of the great vessels, gastroesophageal reflux | Gastroesophageal reflux |
| Failure to thrive | Cystic fibrosis, tracheoesophageal fistula, bronchopulmonary dysplasia | Cystic fibrosis, chronic hypersensitivity pneumonitis, α 1 -antitrypsin deficiency, bronchiectasis |
| Factors associated with feeding | Tracheoesophageal fistula, gastroesophageal reflux | Gastroesophageal reflux |
| Environmental triggers | Allergic asthma | Allergic asthma, allergic bronchopulmonary aspergillosis (ABPA), acute hypersensitivity pneumonitis |
| Sudden onset | Allergic asthma, croup | Allergic asthma, foreign body aspiration, croup, acute hypersensitivity pneumonitis |
| Fever | Bronchiolitis, pneumonitis | Infectious asthma, acute hypersensitivity pneumonitis, croup |
| Rhinorrhea | Bronchiolitis, pneumonitis | Infectious or allergic asthma, croup |
| Concomitant stridor | Tracheal or bronchial stenosis, anomalies of the great vessels, croup | Foreign body aspiration, croup |
| Clubbing | — | Cystic fibrosis, bronchiectasis, bronchopulmonary dysplasia |
In older children who have sudden onset of wheezing and respiratory distress, the differential diagnosis includes respiratory infections, left ventricular failure, aspiration, and vocal cord dysfunction. Respiratory infections (such as croup) may be distinguished by their characteristic histories and tendencies to involve the upper airways (see Chapter 17 ). Lower respiratory infections (pneumonia) generally produce fever and more localized findings of rales, a decrease in the number of and change in the quality of breath sounds, and egophony. Left ventricular failure, especially with pulmonary edema, may present with acute respiratory distress and wheezing. A history of cardiac disease, diffuse crackles or basilar rales, and a third heart sound on auscultation help distinguish this condition from asthma. Aspiration of a foreign body that lodges in a mainstem bronchus may produce wheezing ( Fig. 4.30 ). A history of a choking episode and physical findings of unilateral wheezing and hyperresonance aid in distinguishing aspiration from asthma but do not confirm the diagnosis, because wheezing resulting from foreign body aspiration may respond at least in part to bronchodilator therapy. Vocal cord dysfunction is becoming more recognized in children and adolescents and is often confused with asthma by families, school nurses, as well as primary care and emergency room physicians. Symptoms include acute-onset dyspnea and often stridor, which can be mistaken for wheezing to the untrained observer, secondary to partial or complete adduction of the vocal cords during inspiration. Symptoms rarely awaken individuals from sleep, and there are many triggers that are similar to asthma, including exercise, inhalational irritants, psychosocial stressors, and rhinitis. Bronchodilators are not effective and objective measurements of pulmonary function, including pulse oximetry and chest radiographs, are unremarkable during acute episodes. Some episodes of vocal cord dysfunction can become severe enough to result in syncope from hyperventilation, which often successfully resolves the acute episode as the vocal cords relax.
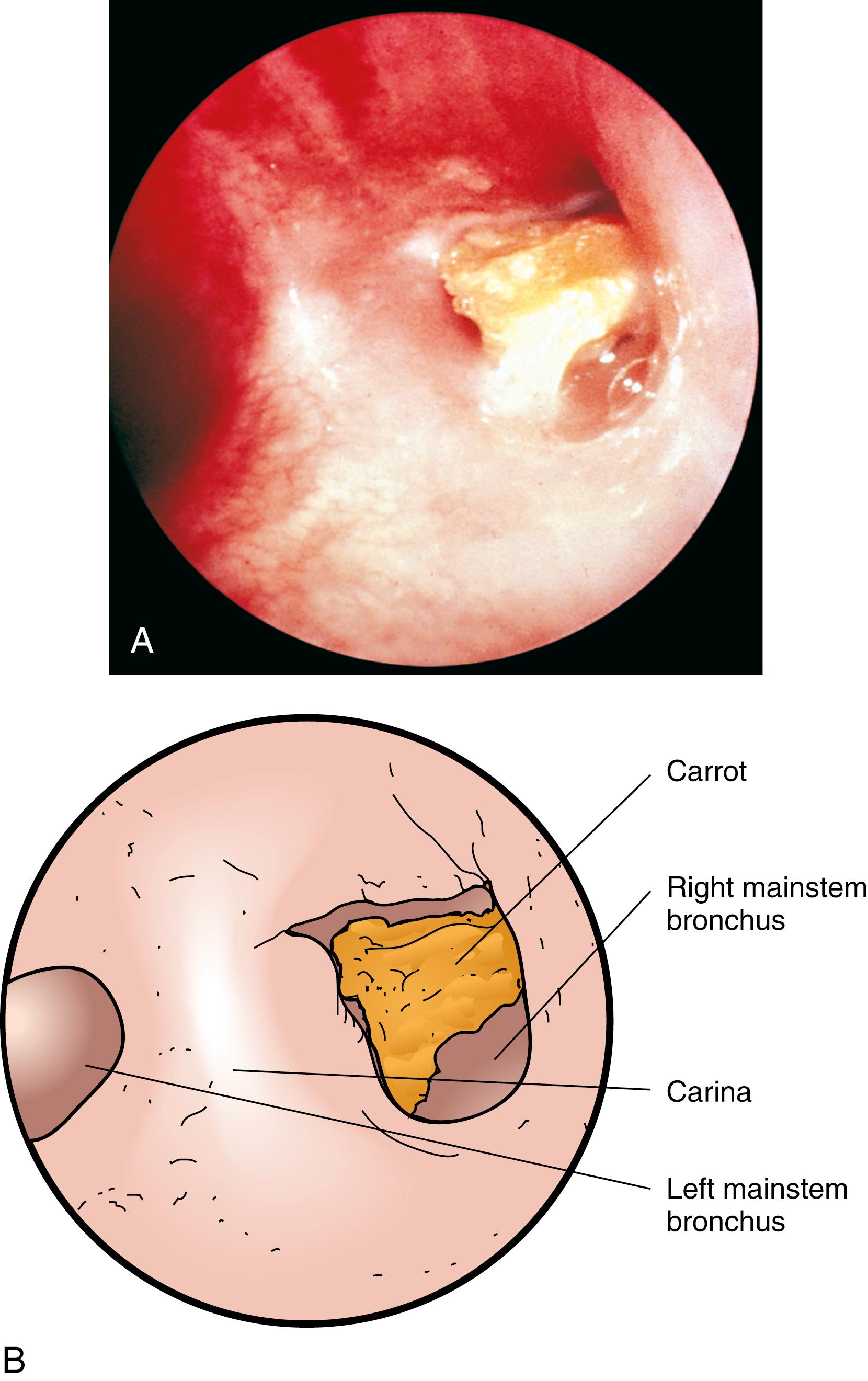
In the older child with mild, infrequent episodes of wheezing that respond to bronchodilator therapy, asthma is readily diagnosed. However, with daily wheezing, frequent exacerbations, lack of response to bronchodilators, or poor growth, other diagnoses must be considered, including chronic obstructive pulmonary disease, cystic fibrosis, α 1 -antitrypsin deficiency, and immunodeficiency. Chronic obstructive pulmonary diseases, which include chronic bronchitis, emphysema, bronchiectasis, and bronchopulmonary dysplasia, are distinguished by their lack of significant reversibility with bronchodilator therapy. Cystic fibrosis may present with chronic cough, wheezing, and recurrent infections. In addition, malabsorption with bulky, foul-smelling stools; failure to thrive; and clubbing of the nail beds are common. An inherited autosomal recessive disorder, α 1 -antitrypsin deficiency, is characterized by the onset of progressive emphysema in a young adult and is one cause of neonatal hepatitis.
Although IgE-mediated allergic respiratory diseases (allergic rhinitis, asthma) are the most common manifestations of inhalant sensitivities in humans, other immunologic respiratory diseases, involving non-IgE immune mechanisms, may result from the inhalation of antigens from the susceptible individual’s environment. A wide variety of inhaled biologic dusts may induce an inflammatory lung disease involving the interstitium, alveoli, and airways. The most common form of hypersensitivity pneumonitis is caused by inhalation of thermophilic actinomycetes ( Saccharopolyspora rectivirgula, formerly Micropolyspora faeni ), antigens present in moldy vegetable compost, and is termed farmer’s lung. Other forms (and their causative dusts and antigens) include malt-worker’s lung (moldy malt, Aspergillus species) and bird-breeder’s lung (avian dust, avian proteins). The disorder is termed hypersensitivity pneumonitis or extrinsic allergic alveolitis and appears to be mediated by a complex immune response involving both immune complexes (type III) and T cells (type IV).
Hypersensitivity pneumonitis is a syndrome with a broad spectrum of presenting symptoms and signs that have been subdivided into acute, subacute, and chronic. The clinical features depend on the following factors: (1) the nature of the inhaled dust, (2) the intensity and frequency of inhalation exposure, and (3) the immunologic responsiveness of the exposed individual. A concomitant upper respiratory infection or another pulmonary insult may be an important factor in induction. Development of sensitization to the inhaled organic dust requires several months to years, although organic dust toxic syndrome can present similarly after a single exposure to a large dose of antigen. The acute form of hypersensitivity pneumonitis is usually readily differentiated from asthma by the lack of wheezing, prominence of rales, and presence of severe systemic symptoms, such as high fever and myalgia. Subacute and chronic forms generally present with insidious development of respiratory symptoms.
Aspergillus species, in addition to being one cause of hypersensitivity pneumonitis and allergic asthma, cause a disorder termed allergic bronchopulmonary aspergillosis (ABPA). This entity is characterized by migrating pulmonary infiltrates and peripheral blood and sputum eosinophilia. Type I and type III hypersensitivity are thought to be involved in the pathogenesis. Affected individuals are usually atopic and have a history of asthma and/or cystic fibrosis. They often present with systemic symptoms and difficulty weaning off systemic corticosteroids. Sputum production is prominent. Physical findings include the general signs of lower airway obstruction (see the earlier section, Asthma).
Laboratory studies that assist in diagnosis include the following:
Peripheral blood examination reveals eosinophilia (generally >1000/mm 3 ).
The serum IgE level is markedly elevated, often greater than 1000 ng/mL, and specific IgE to Aspergillus fumigatus is elevated.
Skin testing for A. fumigatus is positive. Although mold- sensitive patients with asthma without ABPA may have a positive reaction, a negative reaction rules out ABPA.
Serum precipitating antibody (IgG) to A. fumigatus is found in most patients, but the titer has a poor correlation with disease activity.
Chest radiography commonly shows infiltrates in active ABPA. The upper lobes are commonly involved, and infiltrates characteristically shift rapidly from one site to the other. Remarkably, radiographic findings do not correlate well with clinical severity, and patients with extensive consolidation may be asymptomatic ( Fig. 4.31 ).
High-resolution computed tomography (CT) scanning may show bronchiectasis, especially late in the course of the disease.
Ocular allergic reactions may involve the eyelid and/or conjunctiva. The eyelids have a rich blood supply and loose connective tissue assisting edema collection from either allergic inflammation or trauma. Immediate hypersensitivity reactions that produce eyelid angioedema may be triggered by a number of stimuli, including pollens, dusts, insect stings or bites, foods, or drugs. The most common cause of mild reactions is topical exposure to environmental allergens. These reactions are characterized by periorbital edema, pruritus, and erythema after exposure to an allergen ( Fig. 4.32 ). Severe acute episodes can be distinguished from cellulitis by lack of induration, absence of tenderness and fever, and the fact that involvement is usually bilateral (see Chapter 24 ).

Allergic conjunctivitis may be acute or chronic, and seasonal or perennial, depending on the sensitizing allergens. Commonly implicated allergens include weed, tree, and grass pollens; molds; dust; and animal dander. In the acute seasonal form, onset may be rapid and may coincide with the appearance of pollen. This condition frequently accompanies seasonal allergic rhinitis and is commonly due to ragweed, grass, and tree pollens. Itching and excessive tearing are the most prominent symptoms. Pruritus often interferes with sleep, and vision may be impaired by excessive discharge.
Physical findings depend on the degree of chronicity. In the acute form, these findings consist of diffuse bilateral conjunctival edema and hyperemia. Photophobia, profuse tearing, and mild lid swelling are commonly associated. In the chronic form, the conjunctivae appear pale, with mild edema and hyperplasia of the papillae. This may result in a fine, granular appearance of the conjunctivae, which is termed allergic cobblestoning ( Fig. 4.33 ). The clinical diagnosis may be confirmed by finding eosinophilia on a smear of conjunctival secretions and skin testing for the suspected allergens.
The differential diagnosis of allergic conjunctivitis includes atopic keratoconjunctivitis and vernal conjunctivitis. Atopic keratoconjunctivitis occurs mostly in adult patients with atopic dermatitis and is characterized by erythema and thickening of the conjunctivae ( Fig. 4.34 ). This may progress to scarring and vascularization of the cornea in severe cases, and ocular disease activity parallels that of cutaneous disease.
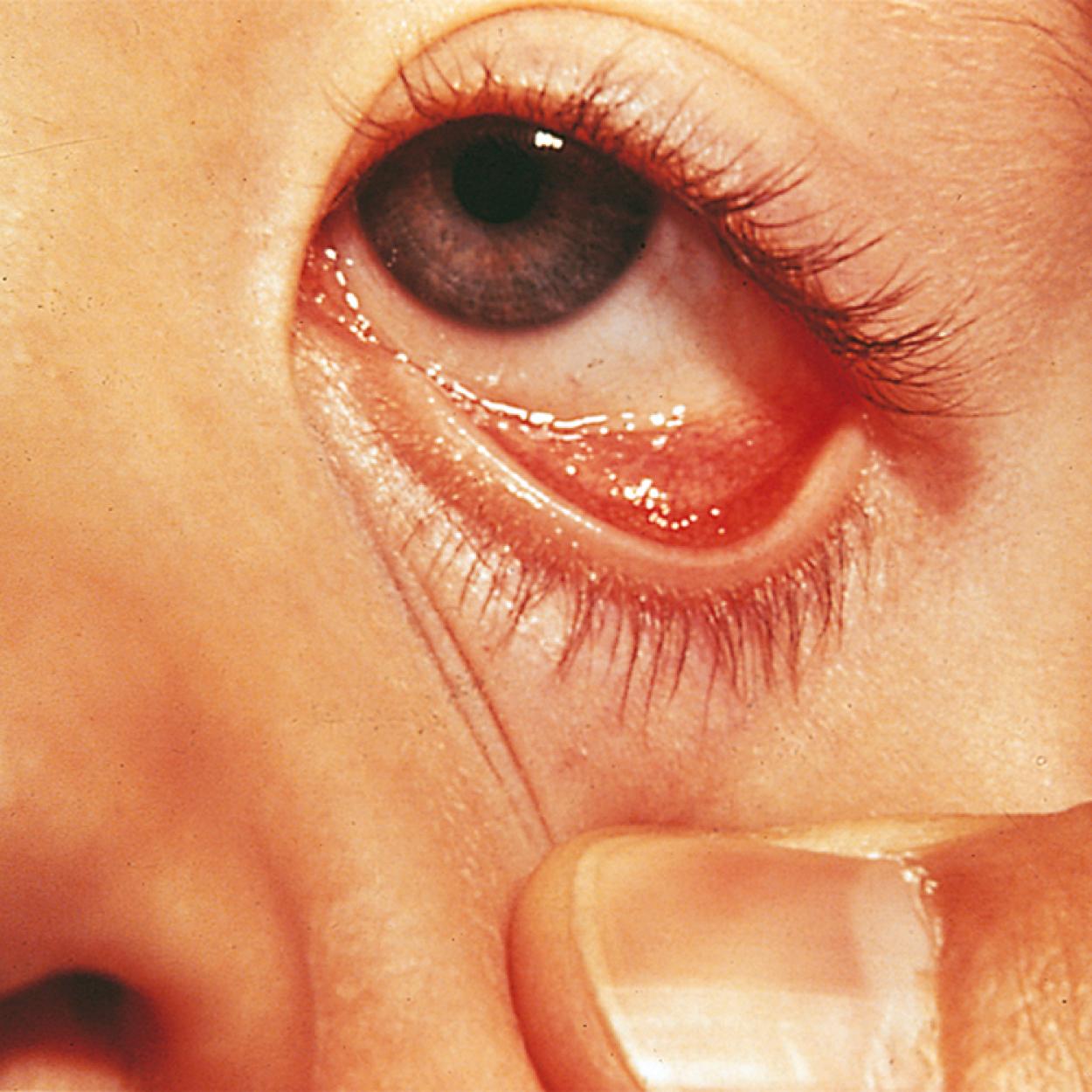
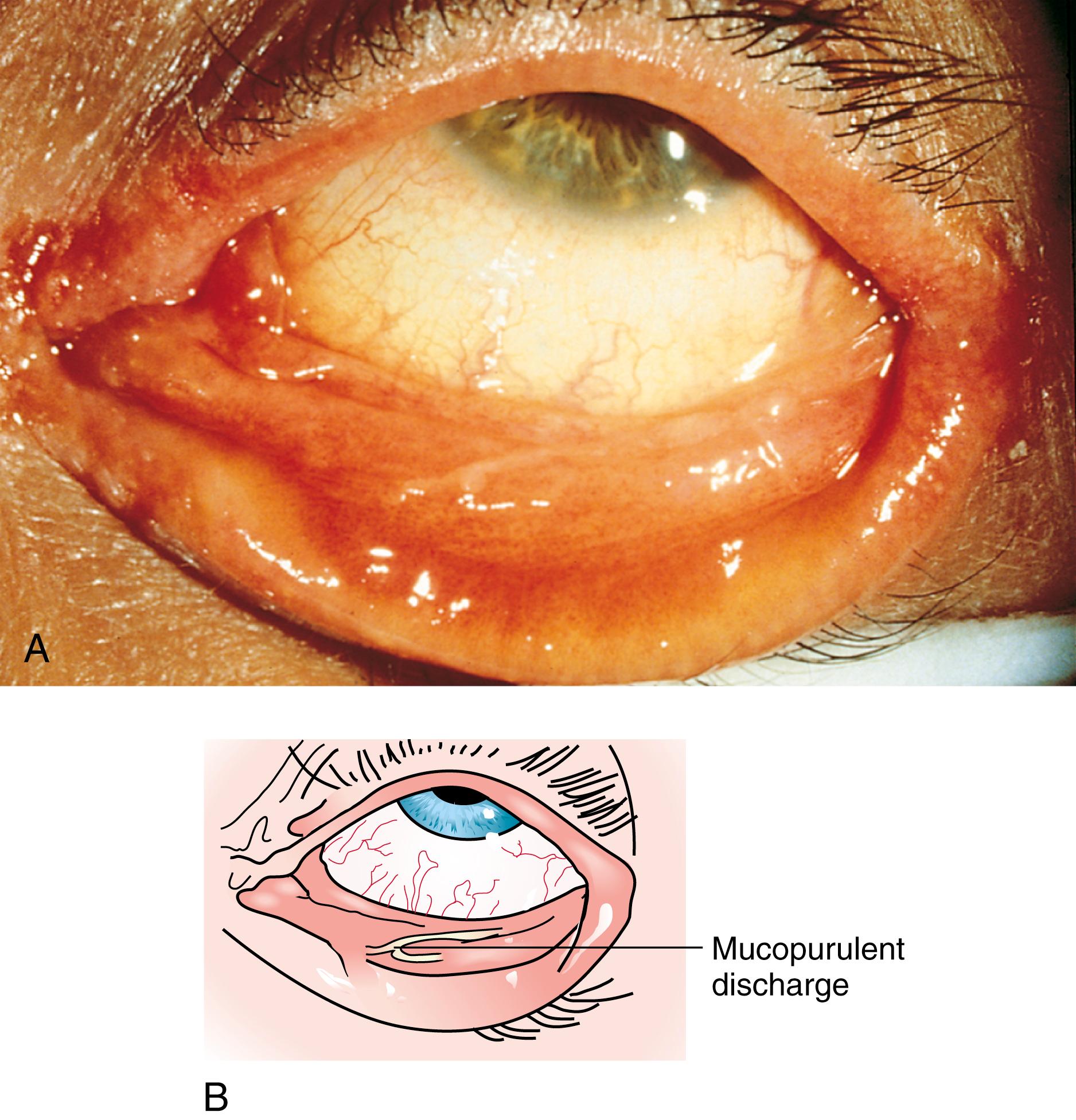
Vernal conjunctivitis is uncommon and chronic in nature. Young, atopic boys are affected most frequently. Symptoms include severe itching, photophobia, blurring of vision, and lacrimation. Physical examination reveals white, ropy secretions containing many eosinophils. A palpebral form manifests hypertrophic nodular papillae that resemble cobblestones on the upper eyelids ( Fig. 4.35 ). The papillae consist of dense fibrous tissue with eosinophilic infiltrates. In the bulbar form, nodules appear as gelatinous masses called Trantas’ dots, which are usually found at the corneoscleral junction ( Fig. 4.36 ). The symptoms are generally severe and frequently require referral to ophthalmology for topical steroids. This disease usually remits with maturity and is rarely seen in adults. Giant papillary conjunctivitis, which appears clinically and histologically to be a mild form of vernal conjunctivitis, is associated with the use of hard and soft contact lenses ( Fig. 4.37 ). The stimulus is believed to be foreign material that accumulates on the surface of the contact lenses. Whether this material is antigenic and the condition an immune-mediated disease is not known.
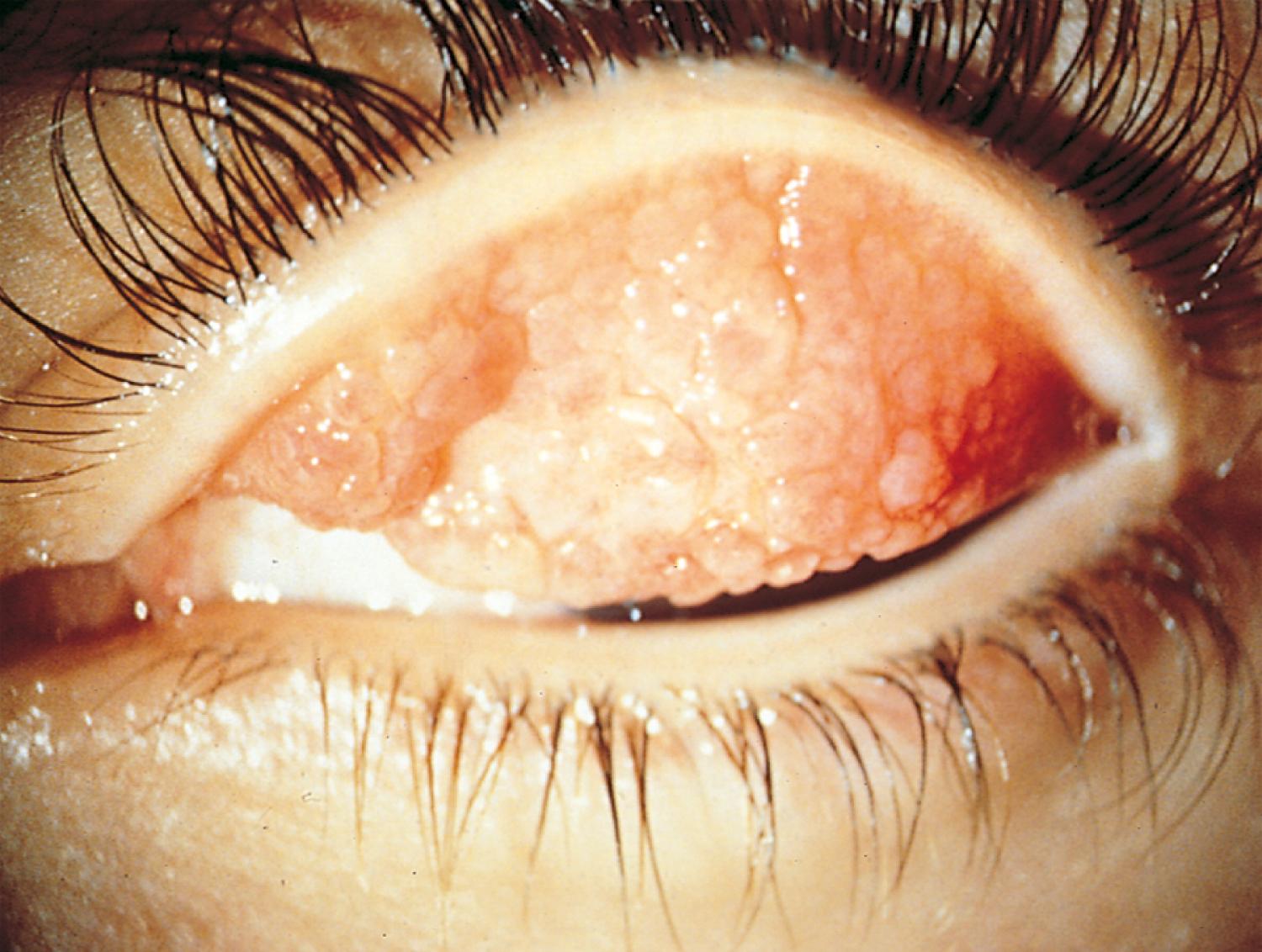
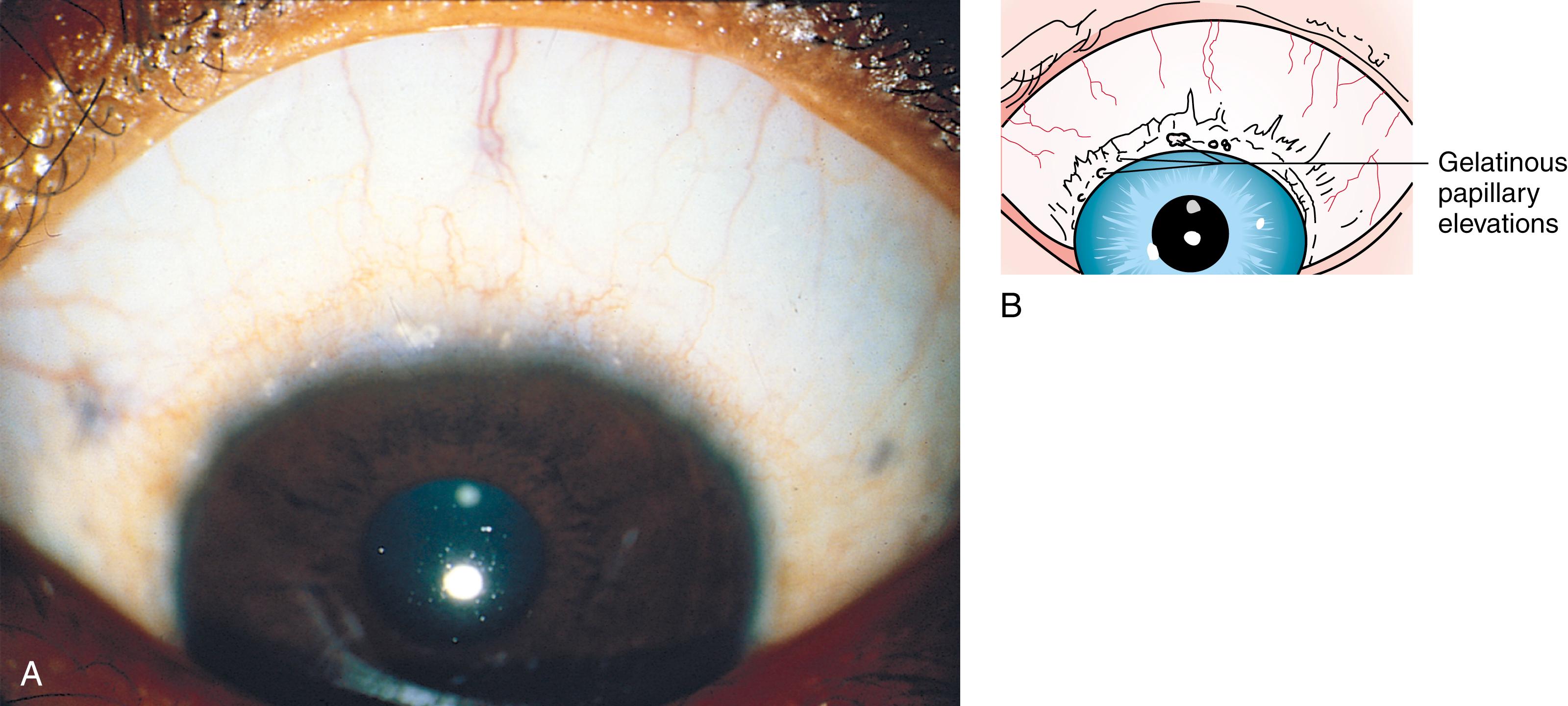
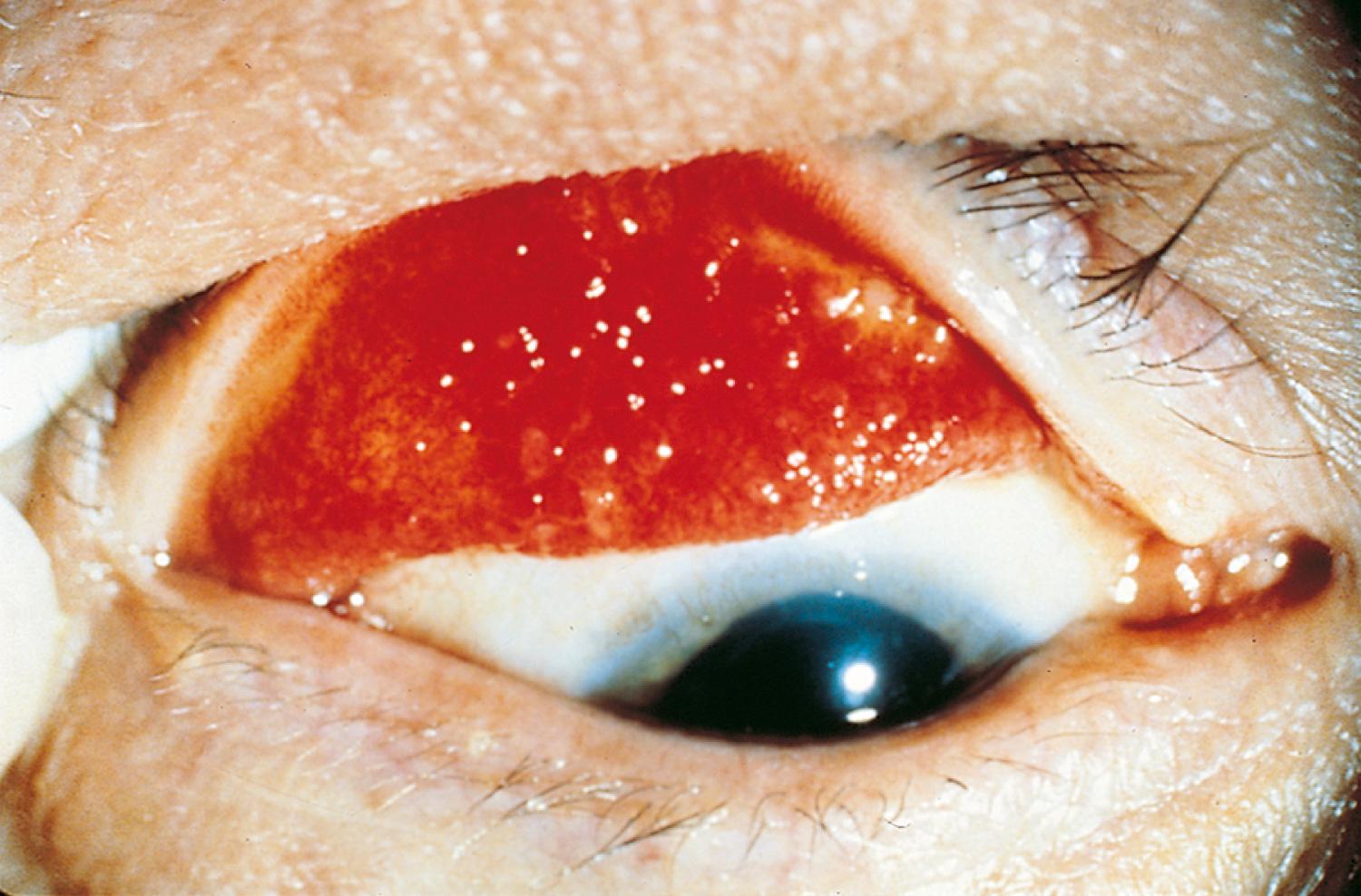
Episodes of urticaria occur in 20% of the population at some point in their lifetime. Angioedema is less common. Both processes involve degranulation of mast cells and basophils and can be due to any one of multiple triggers. Acute episodes of urticaria and/or angioedema due to type I IgE-mediated hypersensitivity reactions to drugs foods or insect stings occur within minutes to 1 to 2 hours of exposure to the allergen. Non-IgE-mediated mechanisms can also cause degranulation of mast cells through activation of other pathways such as complement and immune complex formation or direct mast cell degranulation. The end result for these various processes is vasodilation with increased vascular permeability followed by edema. When this occurs in the dermis it results in characteristic urticaria while occurrence in the deeper subcutaneous tissue results in angioedema.
Urticarial lesions are well circumscribed, raised, palpable wheals that blanch with applied pressure ( Fig. 4.38 ). They are usually erythematous but may be pale or white with red halos. Typically, the lesions are intensely pruritic; however, in some instances the pruritus is mild. Classic urticaria should not last in any one location for greater than 24 hours and is not associated with residual skin changes such as blistering bruising or changes in skin pigmentation. Angioedema is characterized by diffuse subcutaneous tissue swelling. The overlying skin may be erythematous or normal in coloration. Angioedema may be more painful than pruritic and typically involves loose tissues such as the face, hands, feet and perineum ( Fig. 4.39 ). Angioedema, unlike other types of edema, is not gravity dependent or symmetric. Unlike hives, angioedema may take hours to days to completely resolve.
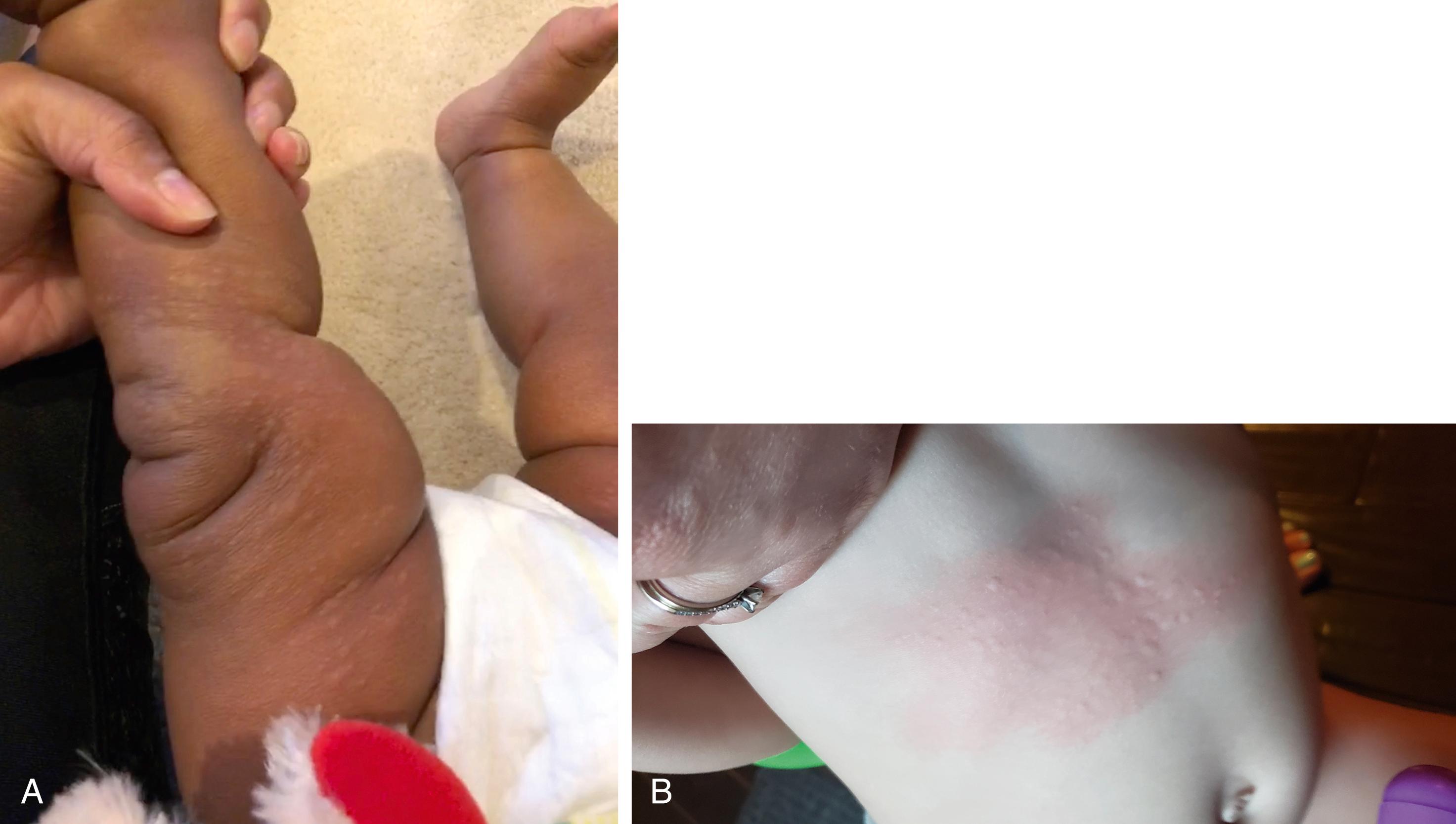
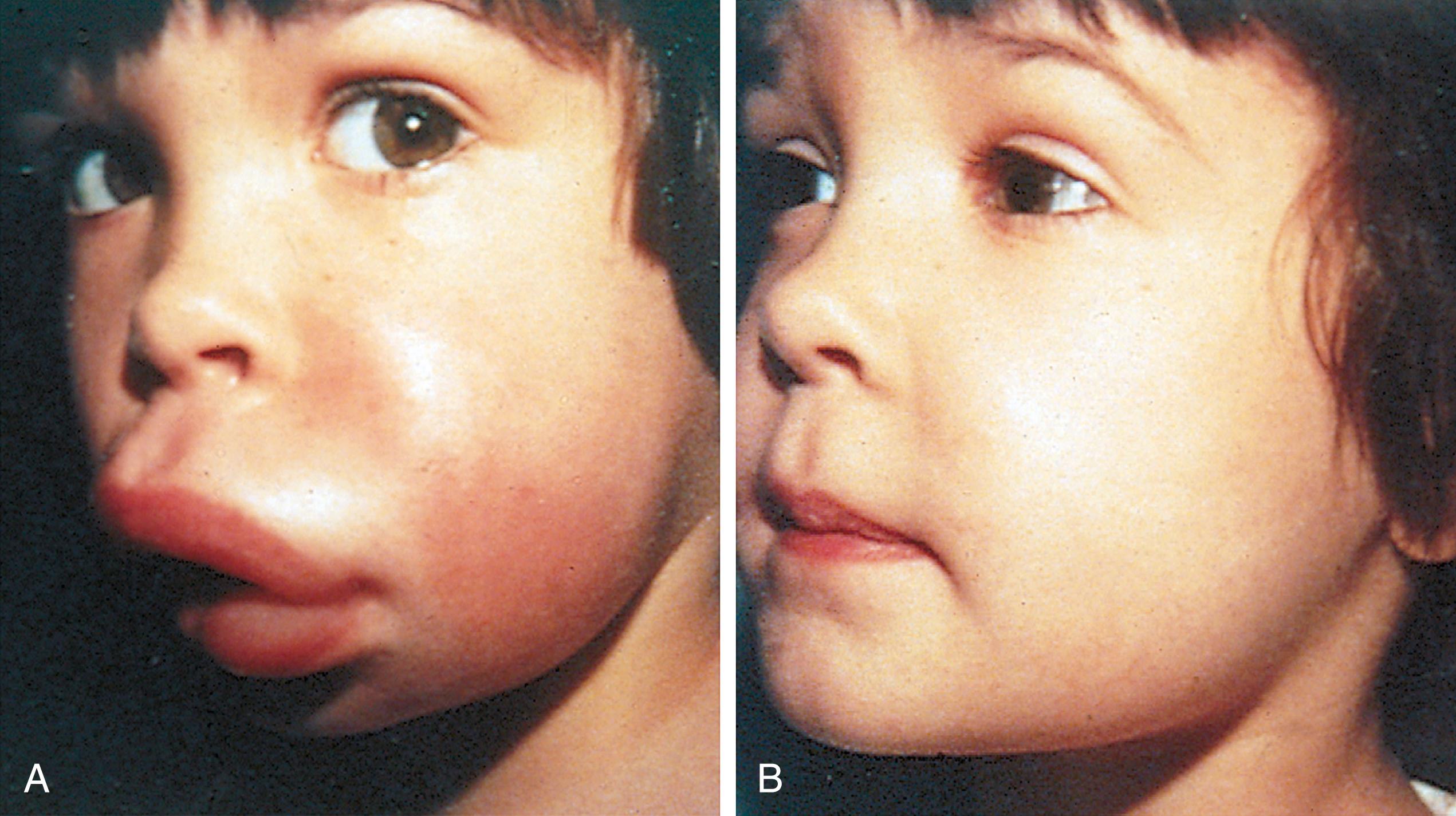
When evaluating causes of urticaria and/or angioedema, a thorough history looking for inciting exposures can identify a cause. In cases of allergen triggers, the symptoms should occur only on exposure and with every exposure. Physical triggers of urticaria include heat scratching pressure vibration and occasionally cold exposure. The term idiopathic is used when there is no clear definable reproducible trigger of urticaria and angioedema. Many of these cases in children are triggered by the immune system’s response in fighting off a viral infection. Most of these cases resolve in 6 weeks or less. When the duration exceeds 6 weeks, the condition is categorized as chronic urticaria and/or angioedema. Research suggests that many of these cases are autoimmune in nature with research showing evidence for autoantibodies to the IgE receptor on mast cells and basophils. Other less common causes of urticaria include aquagenic, solar urticaria, cholinergic and exercise-induced urticaria. The history and distribution of lesions often help in identifying the source, which can then be confirmed by challenge ( Fig. 4.40 ). Dermographism, translated literally as the “ability to write on the skin” is a form of trauma-induced pressure urticaria. It can be elicited by stroking the skin with a fingernail or tongue blade. The initial white line secondary to reflex vasoconstriction is supplanted by pruritic, erythematous linear swelling, as seen in a classic wheal and flare reaction ( Fig. 4.41 ).
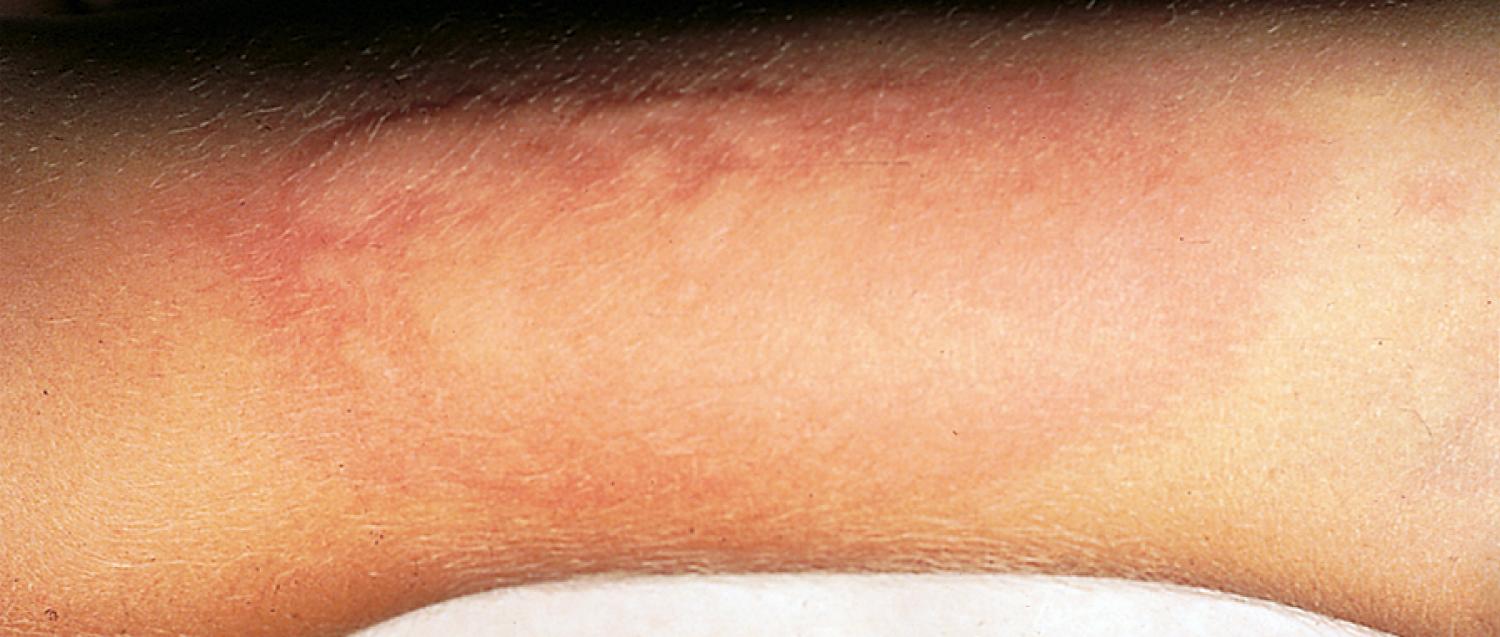
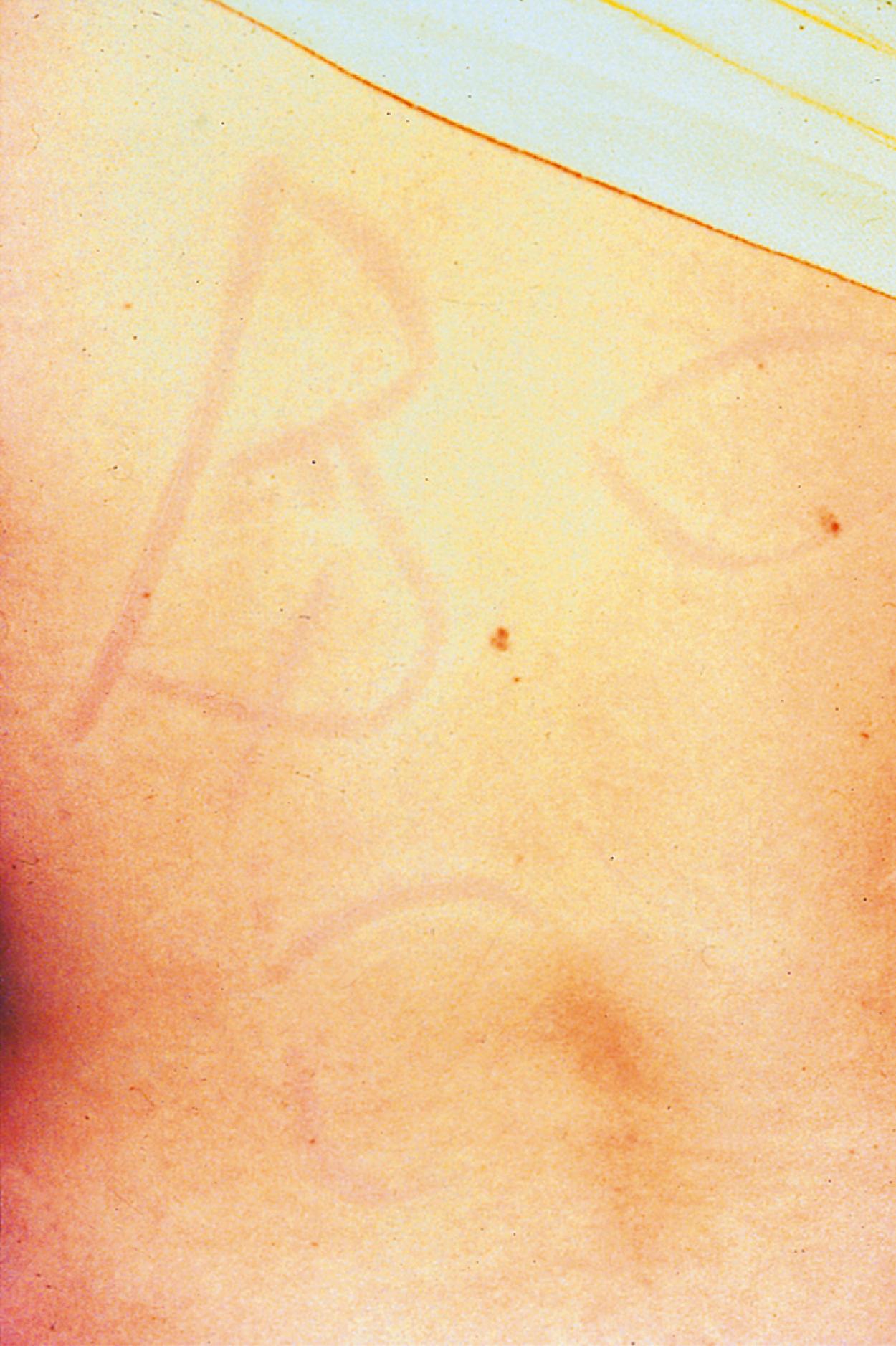
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here