Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
In addition to airway assessment, neurologic examination and communication with the surgeon is invaluable before the induction of anesthesia for neurosurgical procedures.
Patients with an unstable cervical spine may be unable to cooperate with an awake flexible scope intubation (FSI) because of intoxication, hypoxia, or head injury. The need for a cervical spine injury (CSI) patient’s airway to be secured is often urgent because of the CSI or associated head or facial injury.
A rigid cervical collar may make airway management difficult, impeding mouth opening and application of cricoid pressure. Therefore with manual in-line immobilization in place, the front part of the collar should be removed or opened before attempted intubation.
Patients with acromegaly frequently have obstructive sleep apnea and should be induced and ventilated with caution. Direct laryngoscopy and video-assisted laryngoscopy (VAL) are the most effective intubation techniques.
Elective patients who demonstrate neurologic symptoms of the extremities with neck flexion or extension should have awake, topicalized tracheal intubation.
Become familiar with alternative airway devices and practice in normal airways before treating patients with difficult airways.
Be attentive and inspect the degree of neck flexion in patients positioned prone, lateral, or in any head fixation device.
A cuff leak test may be helpful before extubation after prolonged surgery in the prone position; always have reintubation strategies and plans. Consider trial extubation over an airway exchange catheter.
Airway management in the neurosurgical patient can be a challenging endeavor. In achieving and maintaining a patent airway it is important to consider its impact on the central nervous system (CNS) and the well-being of the patient. The variety of neurosurgical procedures, anesthetic techniques, and airway devices has increased dramatically over the past decade. The evolution of neurosurgical practice and the growth of complex spine surgery provide a myriad of clinical challenges.
The goal of this chapter is to address issues specific to the dedicated neuroanesthesiologist, as well as those related to the neurosurgical patient that might be encountered by the generalist anesthesia provider. This discussion reflects the ever-changing considerations in the airway management of neurosurgical patients and offers solutions to common clinical problems that may occur.
The American Academy of Neurological Surgeons (AANS) estimates that almost 1 million neurosurgical procedures are performed annually in the United States. Spine procedures are performed at three times the rate of cranial surgeries. When considering the range of potential neurosurgical procedures, the variety of patient pathophysiology is substantial. A patient presenting for neurosurgery may appear to be completely normal or can present with clinical symptoms of intracranial hypertension. The airway might be assessed as “normal,” but the patient’s head is fixed in a frame. Also, the patient may present with acromegaly for pituitary surgery or may have a previous history of difficult intubation (DI). Additionally, the unanticipated difficult airway becomes an even greater challenge in patients at risk for cerebral aneurysm rupture. Other challenges include the spine surgery patient in the prone position and considerations for extubation after prolonged surgery. Patients with CNS disease can be sensitive to the effects of hypnotic agents, rendering them susceptible to apnea when premedication is given. These are just a few examples of challenges that will be further addressed in this chapter.
Although neurosurgical procedures comprise only 7% of cases in the American Society of Anesthesiologists (ASA) closed-claims database, they are associated with settlements that are 1.6 to 4 times more than general surgical procedures. Understanding the patient’s physiologic requirements, in addition to the surgeon’s plan, is extremely important in these patients. It is wise to have a number of techniques for achieving, maintaining, and rescuing the neurosurgical airway.
Intracranial pressure (ICP) is the pressure within the rigid skull. Airway management in the face of intracranial hypertension is a frequent challenge for the neuroanesthesiologist, neurointensivist, and emergency physician. The patient who does not require immediate airway control may benefit from the simple maneuver of elevating the head. The head-up position may have beneficial effects on ICP through changes in airway pressure, central venous pressure, and cerebrospinal fluid displacement. Cerebral perfusion pressure (CPP) is the effective perfusion pressure driving blood through the brain, defined as the difference between mean arterial pressure (MAP) and ICPs (CPP = MAP − ICP). A frequent consideration in the neurosurgical patient is the need to balance and maintain intracranial dynamics, avoiding increases in ICP, yet maintaining cerebral perfusion.
Laryngoscopy and intubation, if performed with difficulty or improperly, can severely compromise intracranial dynamics and increase morbidity. Both the sympathetic and the parasympathetic nervous systems mediate cardiovascular responses to tracheal intubation. Acute increases in ICP and MAP during laryngoscopy and tracheal intubation have been well documented. In 1975 Burney and Winn measured ICP in 12 patients undergoing craniotomy and 2 patients for carotid arteriography. ICP did not change in response to the injection of contrast medium but increased significantly and dramatically in response to laryngoscopy and intubation. The increase appeared related to the initial ICP of these patients, possibly representing exhaustion of compensatory mechanisms. Special attention must be given to this factor during manipulation of the larynx in neurosurgical patients with initially increased ICP or space-occupying intracranial lesions.
Techniques to blunt this sympathetic response include (1) an additional dose of opioids or propofol; (2) the use of beta-blockers or other antihypertensive agents; and (3) the use of intravenous (IV) lidocaine. Esmolol or lidocaine as an IV bolus of 1.5 mg/kg before laryngoscopy and intubation does not completely prevent the increase in MAP and ICP. , Etomidate has been shown to cause an early “burst suppression” pattern on the electroencephalogram (EEG), minimal changes in CPP, and a marked reduction in ICP. This decrease in ICP is maintained during the first 30 seconds and the following 60 seconds after intubation, as MAP and heart rate remain unchanged. Although not practical, this approach demonstrates the extent of efforts often made to obtund this response. Numerous methods have been advocated to prevent undesirable cardiovascular disturbances at intubation. Whereas the cardiovascular response can be dramatic and substantial, the ICP response may lag and persist longer. Once the patient is intubated, ventilation parameters may be adjusted to the clinical situation.
Airway assessment of the neurosurgical patient requires similar considerations, as described in other chapters of this textbook. The patient who has undergone previous surgery and has a history of DI warrants particular attention. A review of the anesthetic record should reveal which techniques produced success or failure. Difficult mask ventilation is of particular concern because of the potential for causing hypercarbia and the detrimental changes previously described.
Patients who are neurologically intact may demonstrate no evidence of intracranial pathology or alteration. In addition to the history and physical examination, preoperative computed tomography (CT) or magnetic resonance imaging (MRI) scans of the head may give valuable information, because lesions associated with greater than 10 mm in midline shift or cerebral edema usually indicate intracranial hypertension. These patients should be appropriately managed to avoid undue increases in ICP and cerebral blood flow (CBF). Such measures include proper head positioning, preoxygenation, and appropriate dosing of induction agents and relaxants to achieve a smooth intubation. The primary challenge in anesthetizing a patient with a supratentorial mass lesion is to avoid further increases in ICP when one has limited intracranial compliance. There is no “ideal anesthetic” for this group of patients, and the perioperative management should be individualized. However, the practitioner should be aware of the effects of anesthetic agents on intracranial dynamics.
The preoperative use of midazolam for anxiety in these patients should not cause harm if they are carefully observed. A 1- to 2-mg dose of IV midazolam in adult patients may facilitate the induction of anesthesia without altering intracranial dynamics. Opioids, on the other hand, should be restricted to very small amounts and given preoperatively under constant supervision because of possible hypercarbia and resultant effects. The efficacy was recognized early on as a technique for avoiding intracranial hypertension. In the 1980s deep inhalation anesthesia was replaced by a combination of IV induction agents, notably thiopental, in combination with fentanyl. Barbiturates, methohexital and pentobarbital, produce a dose-dependent reduction in CBF and cerebral metabolic rate of oxygen (CMRo 2 ) consumption. ICP is reduced by barbiturates, likely because of the reduction in CBF and cerebral blood volume (CBV). Propofol has replaced thiopental (no longer manufactured in the United States) as the induction agent of choice for neuroanesthesia. Despite initial concerns about decreasing MAP and CPP, propofol provides a smooth transition to unconsciousness without an increase in heart rate, as observed with thiopental. This often produces less hypertension with laryngoscopy and intubation.
Clinical doses of most opioids have minimal to modest depressive effects on CBF and CMRo 2 . Early studies demonstrate that ICP is either not elevated or slightly decreased with fentanyl alone or in combination with droperidol. Reported ICP increases in patients with space-occupying lesions have been attributed to hypercapnia. The variability in response to opioids appears to be caused by the background anesthetic. When vasodilating drugs are used as part of the anesthetic management, the effect of the opioid is consistently that of a vasoconstrictor. Sufentanil was thought to produce an increase in ICP in patients with intracranial mass effect, but this was later attributed to a decrease in MAP. Alfentanil produces little change or a slight decrease in CBF. The beneficial effect of synthetic opioids is their ability to blunt the hemodynamic response to laryngoscopy and intubation without affecting intracranial dynamics. Remifentanil produces the most profound and consistent response, with a lack of hypertension, tachycardia, or an increase in ICP. A continuous infusion throughout induction may provide the most effective hemodynamic control, while adequate ventilation is maintained.
The volatile agents, including nitrous oxide, can be considered dose-dependent cerebral vasodilators. As a component of neuroanesthesia, volatile agents are typically used in moderate doses, in combination. Their effects on cerebral circulation and metabolism of sevoflurane and desflurane are largely comparable to isoflurane. Both induce a direct vasodilation of the cerebral vessels, resulting in a less pronounced increase in CBF, compared with the decrease in cerebral metabolism.
Induction may be followed by ventilation with a volatile agent to deepen the anesthetic, decrease CMRo 2 (and CBF), and provide bronchodilation in patients with asthma or chronic obstructive pulmonary disease (COPD). Sevoflurane is useful in both pediatric and adult patients by allowing inhalation induction without the adverse effects of coughing or breath-holding. A frequently employed technique in the cooperative patient is the use of active hyperventilation before induction to initiate hypocapnia and decrease CBF as the patient loses consciousness. The use of topical anesthesia applied to the larynx and trachea can also prevent further response to laryngoscopy and intubation. The large number of techniques recommended to suppress cardiovascular responses indicates that no single method has gained widespread acceptance ( Table 39.1 ).
| Technique | Precaution(s) |
|---|---|
| Avoid hypercapnia |
|
| Avoid hypoxia |
|
| Avoid marked hypertension |
|
| Avoid severe neck rotation |
|
| Avoid compression of jugular veins |
|
| Elevate head |
|
| Decrease blood viscosity and intracerebral vascular volume |
|
| Avoid sustained increases in intrathoracic pressure |
|
| Avoid cerebral venodilators |
|
The obtunded patient with symptoms of intracranial hypertension requires additional attention to detail, avoiding premedication and maneuvers that increase coughing. If a rapid sequence induction is not indicated and the patient’s airway anatomy is adequate for laryngoscopy, anesthetic induction may proceed with voluntary hyperventilation with 100% oxygen by mask, if possible. After loss of consciousness, manual hyperventilation should occur both before and after the administration of muscle relaxant. Opioid administration may begin at this time to prevent the sympathetic response to laryngoscopy. IV lidocaine (1 mg/kg) may be administered to blunt the hemodynamic and ICP response to laryngoscopy. Alternatively, a beta-blocker or an additional dose of propofol may be given. Esmolol or lidocaine, 1.5 mg/kg as an IV bolus before laryngoscopy and intubation, does not completely prevent the increase in MAP and ICP. Complete neuromuscular blockade should be verified before laryngoscopy to prevent cough and associated increases in ICP. Proper airway management is essential to avoid the dual threat of hypoxia and hypercarbia. An obstructed airway may also lead to a rise in intrathoracic pressure. This may produce an elevated venous pressure, increased intracranial blood volume, and elevated ICP. If the patient can be mask ventilated but intubation is difficult, one may choose to proceed with video-assisted laryngoscopy (VAL) or another alternative device to facilitate intubation.
The intubating laryngeal mask airway (ILMA) is particularly useful in the failed intubation sequence, and the ability to ventilate is extremely important in neurosurgical patients. The success of the ILMA as a ventilatory device has been well established, as demonstrated in several of the early evaluation studies. , While it is used less often with the availability of VAL, it remains very useful in the setting of a failed flexible scope intubation (FSI).
The patient for aneurysm surgery who presents with a difficult airway is particularly problematic. If the airway is anticipated or known to be difficult, FSI is often the method of choice. This is assuming that one is skillful in using the flexible intubation scope (FIS) and is prepared to perform this technique in the awake, cooperative patient (see Chapter 25 ). IV fentanyl and midazolam may be carefully administered if the patient does not exhibit signs of intracranial hypertension. An arterial line is sometimes placed before induction for careful monitoring. Additional techniques include remifentanil infusion (0.05 µg/kg/min) or dexmedetomidine infusion. , Both techniques require careful patient monitoring and may be useful. Once the glottis is viewed, a dose of lidocaine may be given via the FIS to prevent coughing and “bucking” with intubation.
Alternative techniques for failed sedation or topicalization include awake placement of the ILMA or other techniques that do not produce excessive hemodynamic responses. The concomitant administration of beta-blockers or vasodilators may be necessary for blood pressure control. Essentially, the ASA Difficult Airway Management Algorithm should be followed with close monitoring of blood pressure and heart rate at all times until the airway is secured.
Traumatic brain injury (TBI) remains a prevalent problem in the United States and the world. The incidence of TBI is 175 to 300 per 100,000 population and accounts for 56,000 deaths per year in the United States. With the increased use of seatbelts, motor vehicle accidents are now secondary to gunshot wounds as the leading cause of TBI. Early intubation of the head-injured patient is critical and is often established in the field if providers are so trained. It is essential for the optimal management of the patient, providing for efficient ventilation and oxygenation, helping to prevent aspiration of gastric contents, and allowing for suction of the lungs and pulmonary toilet. However, patients who are unconscious and breathing adequately may be transported with oxygen by mask throughout their initial assessment. This is intuitive in the apneic and unresponsive patient with a Glasgow Coma Scale (GCS) score of 8 or less.
The provider caring for the patient with TBI must understand that although primary mechanisms of injury (primary insults) are a large determinant of patient outcome, attention to secondary insults, such as hypoxia, hypotension, intracranial hypertension, and decreased CPP, can impact morbidity, mortality, and quality of life of the TBI patient. Evidence supports the impact of secondary harm, with mortality from TBI nationally decreasing over the decades. Hypoxia in TBI patients is a frequent occurrence, particularly in the prehospital setting. Interestingly, hypoxia was identified in 44% of patients with TBI on arrival in the emergency department. Similarly, Jeremitsky and colleagues report that hypoxia is one of three predictors of mortality in adult brain-injured patients (with hypothermia and hypoperfusion). Hypoxia dramatically impacts morbidity and mortality in TBI, and hypercapnia further increases mortality.
Hypotension is the secondary insult that has been most frequently cited as contributing to poor outcome after TBI. Hypotension is independently related to mortality in multivariate analysis. Information from the Traumatic Coma Data Bank demonstrates that a systolic blood pressure of less than 80 mm Hg was one of five factors that worsened patient outcome at 6 months. Hypotension during any phase in the brain trauma patient’s hospital course is associated with a greater likelihood of severe disability and vegetative state. However, early in the course of brain trauma, especially when combined with hypoxia, hypotension is devastating. When hypotension and hypoxia occur together, mortality is 75%.
Techniques minimizing head movement should be used in TBI patients and by the most skilled clinicians. However, concern about a cervical fracture should never take precedence over relieving hypoxemia. It is of critical importance to ensure that appropriate monitoring is present throughout airway maneuvers. Nasal intubation should be avoided in head-injured patients, particularly in those with known or suspected basilar skull fractures and sinus injuries. Alternative airway devices, such as video laryngoscopes, any of the indirect rigid laryngoscopes, ILMA, or optical stylets, may be useful when the head must remain immobilized. Most emergency patients are assumed to have a full stomach, so it is important to weigh the risk of aspiration, which is a potential problem during laryngoscopy and intubation. If the situation warrants, surgeons should be prepared to perform a rapid cricothyrotomy, if intubation attempts fail and ventilation becomes impossible.
Spinal injuries occur in approximately 13% to 30% of polytrauma patients, and cervical spine injury (CSI) represents about 0.9% to 3% of all polytrauma patients. , The relative risk of CSI is increased in the presence of severe head injury by a factor greater than 8. In the United States cervical trauma has an incidence of approximately 5 per 10,000 population annually, making up 4% of all blunt trauma. In trauma victims with a GCS score of 13 to 15 the incidence of CSI is 1.4%, but this rises dramatically to 10.2% if the GCS score is less than 8. It is of vital importance to capture all injuries in the unconscious polytrauma patient within an emergent time frame. If a CSI is missed or its detection delayed, the incidence of secondary neurologic deficit increases from 1.4% to 10.5%. For this reason, the Advanced Trauma Life Support (ATLS) protocol was created and is updated and broadly followed in most trauma centers. When a diagnosis of CSI is delayed, almost one-third of patients may develop permanent neurologic deficit. One of the areas of controversy is how to best “clear” the cervical spine in the trauma patient. The detection of CSI requires a variety of modalities that vary in sensitivity, including clinical evaluation, plain radiography, CT, MRI, and dynamic fluoroscopy.
To clear the cervical spine clinically, the following criteria must be met:
GCS score of 15, with the patient alert and oriented
Absence of injuries that may draw attention away from a CSI
Absence of drugs or intoxicants that may interfere with the patient’s sensorium
Absence of signs or symptoms on examining the neck, specifically:
No midline pain or tenderness
Full range of active movement
No neurologic deficit attributable to the cervical spine
Clearly, there will be only a small number of trauma patients who fulfill these criteria.
The cross-table lateral view alone, even if technically adequate and interpreted by an expert, will still miss 15% of cervical injuries. Of cross-table lateral films taken in emergency rooms, approximately a quarter of the films are anatomically inadequate, necessitating further imaging modalities for evaluation, usually of the cervicothoracic junction. A three-view cervical series includes the cross-table lateral view, open-mouth odontoid view, and anteroposterior (AP) view ( Figs. 39.1 – 39.3 ). Using these views, the sensitivity increases to detect 90% of those with an actual injury. Again, anywhere from 25% to 50% of these series may be inadequate anatomically. In low-risk patients plain radiography is an efficient diagnostic examination with a specificity of 100%. In high-risk patients plain radiography is a good adjunctive screening test in conjunction with a CT scan, with a sensitivity of 93.3% and specificity of 95%.
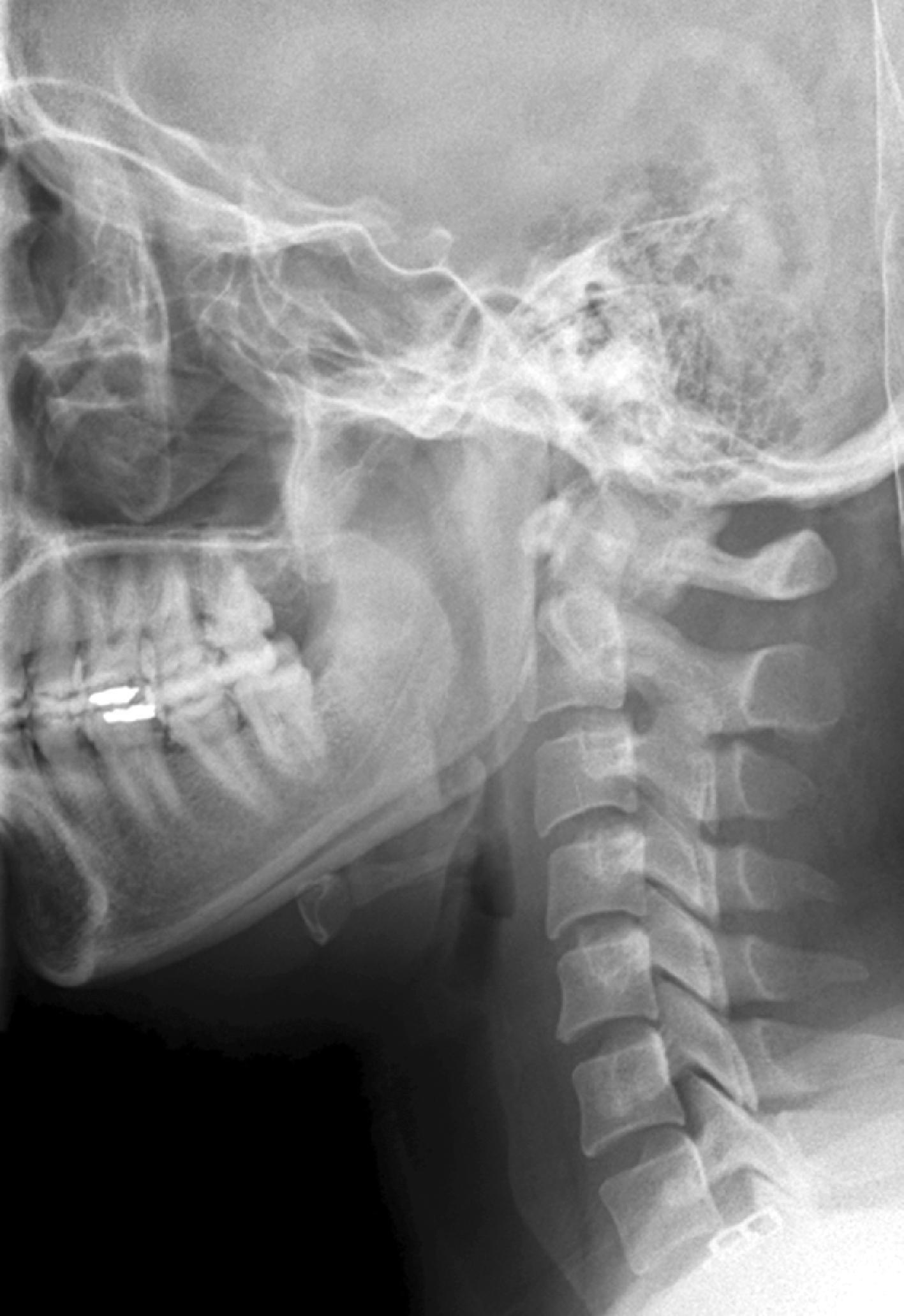
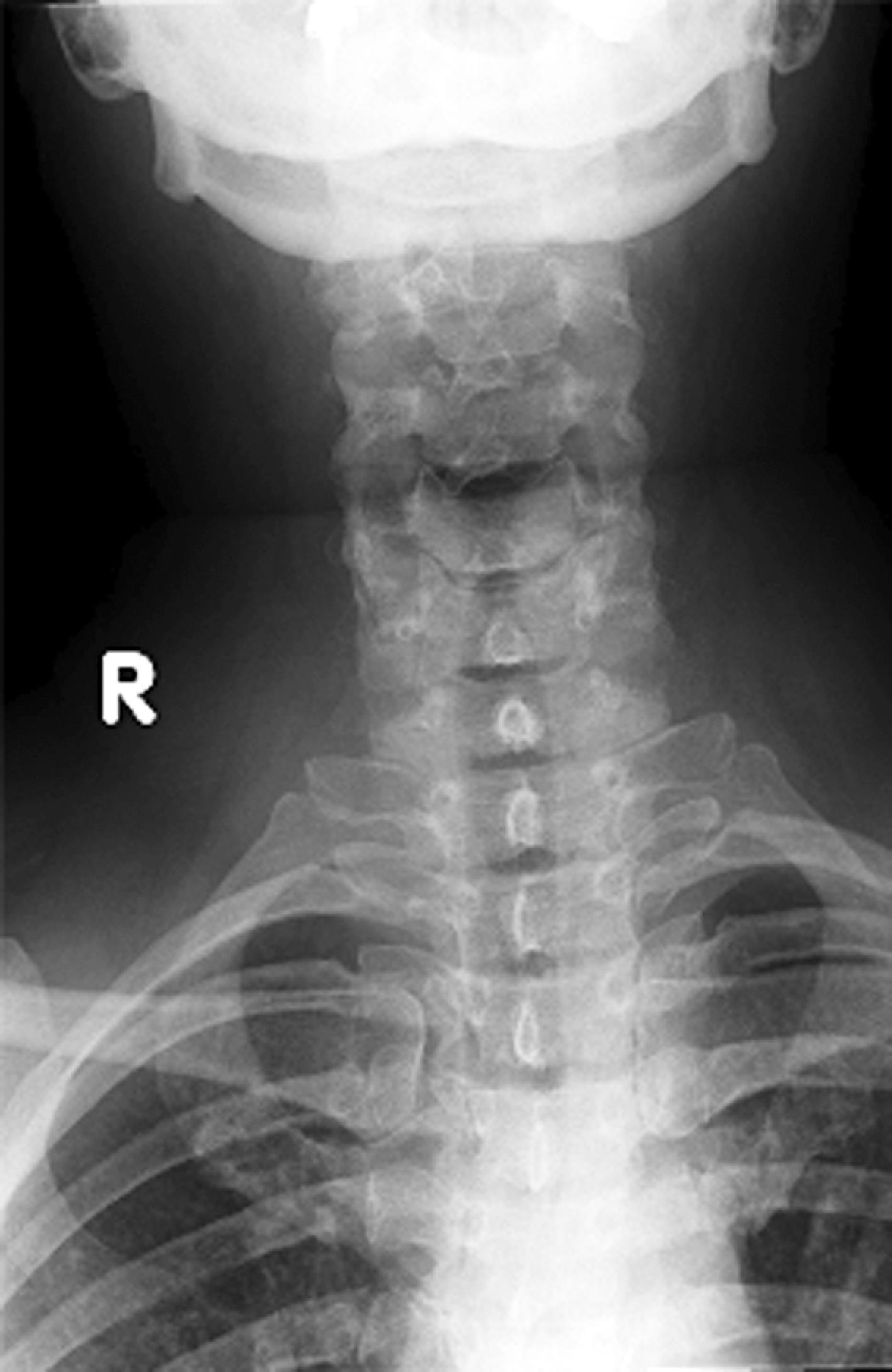
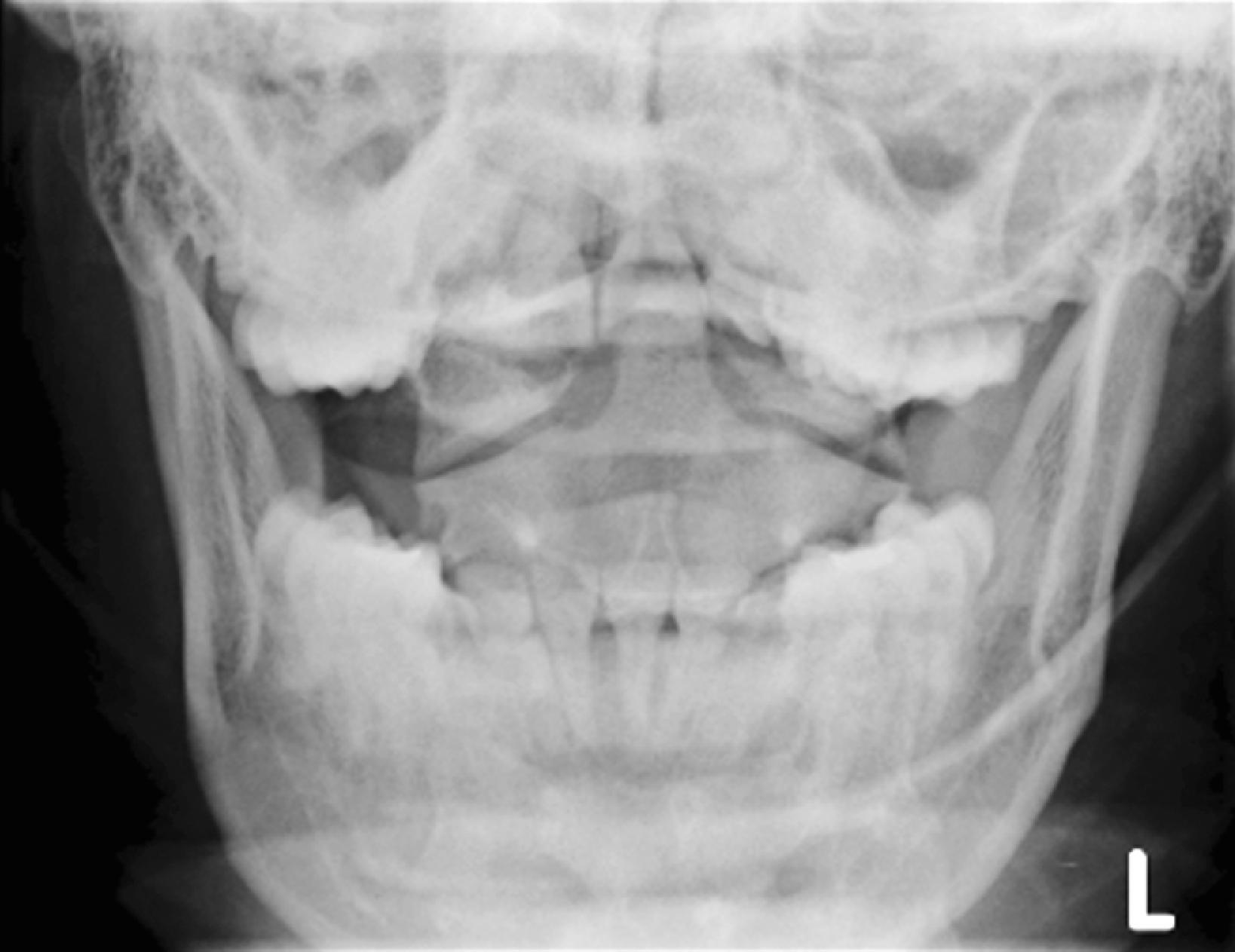
CT scanning, either of the entire cervical spine or directed at areas missed by plain radiographs, provides a complementary approach when used in addition to the three-view cervical series, reducing the risk of missing a CSI to less than 1%. In the evaluation of the cervical spine a helical CT scan has higher sensitivity and specificity than plain radiographs in the moderate-risk and high-risk trauma population, but it is more costly. In fact, a helical CT scan is the preferred initial screening test for the detection of cervical spine fractures among moderate-risk to high-risk patients seen in urban trauma centers, reducing the incidence of paralysis resulting from false-negative imaging studies and institutional costs when settlement costs are taken into account.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here