Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Living donor liver transplantation (LDLT) has emerged to relieve the global shortage of deceased donor liver grafts. After the first success in a child was reported by Strong et al of Brisbane in 1990, significant advancements in various fields have been achieved that allow dramatic growth and a wider application of this lifesaving operation. Among these, the advent of adult right liver LDLT, which overcomes the restriction of donor-recipient size matching and evades small-for-size syndrome, has had the most significant impact.
Incidentally, the first right liver LDLT was anecdotally reported to have been performed in Kyoto not for an adult but for a 9-year-old recipient avoid an anatomical anomaly of the donor’s left hepatic arteries. The first adult right liver LDLT was performed in Hong Kong on May 9, 1996, in an adult with fulminant Wilson’s disease who had a body weight of 90 kg. The donor was his elder brother, who weighed 74 kg. When the first series of seven patients from Hong Kong was reported in 1997, there was a cautionary note as to the future role of this operation. Indeed, in the initial phase, ethical controversy regarding the safety of the donor posed a major obstacle toward a wider application of the procedure. Nonetheless, improved understanding of the anatomy of the liver and advances in surgical techniques have dramatically improved the outcome of both donors and recipients. Recipients now can have excellent long-term survival while the risks borne by the donors are shown to be acceptable. The application of this procedure has expanded tremendously, and it has become the main workload in centers active in adult LDLT. LDLT accounted for nearly 90% of the liver transplants performed in Asia excluding mainland China, and nearly 90% of the adult LDLTs were right liver LDLT.
Adult right liver LDLT is one of the most complicated and technically demanding procedures, and there are numerous technical variations that have been described in both the donor and the recipient operations. In this chapter the standard techniques as adopted in our center will be presented, focusing on the tips and tricks and with reference to other important variations.
A description of adult right liver LDLT is not complete without a few brief remarks on the evaluation of a right liver donor and recipient.
The potential recipient evaluation for LDLT should not be different from that for deceased donor liver transplantation. The indications and contraindications for transplantation are generally the same. Candidates for LDLT should also be candidates for deceased donor liver transplant. Some centers, however, have adopted extended criteria for LDLT, especially in patients with hepatocellular carcinoma because a graft from a living donor is a dedicated gift and is not subject to any allocation system that mandates a strict set of criteria to justify organ use. Transplant centers need to balance the donor risks versus the recipient benefit and decide on a limit beyond which a transplant becomes futile. The recipient’s coping ability and attitude toward LDLT as a treatment option is carefully assessed by the surgeon and then by the clinical psychologist.
Retransplantation, Budd-Chiari syndrome, and portal vein thrombosis, especially with extension into the superior mesenteric vein, had been regarded as contraindications for LDLT because of the more difficult challenge in vascular reconstruction for right liver LDLT. With better technique and experience, however, these technical hurdles can be overcome and hence are not considered as absolute contraindications.
Although LDLT is not the preferred procedure for a high-urgency situation in Europe and the United States, it was the impetus for development of adult right liver LDLT in Hong Kong and other parts of Asia. All seven recipients in our first reported series were high-urgency patients in the intensive care unit. Right liver LDLT not only shortens the waiting time but allows excellent timing for the transplantation of these critically ill patients. The transplant rate and survival rate are both improved. A high-urgency patient is a prime indication for right liver LDLT in Asia.
The evaluation of a potential living liver donor should proceed in a stepwise fashion to ensure that the volunteer remains psychologically and physically healthy in the long-term after the organ donation. Only healthy individuals 18 to 60 years of age are eligible. ABO incompatibility is no longer an absolute contraindication for LDLT. After careful planning and workup, donor interchange can also convert two pairs of ABO-incompatible LDLTs into two compatible ones.
Donation of the right liver represents removing two thirds of the liver in contrast to keeping two thirds as in a donor left hepatectomy. The former has an estimated donor mortality of 0.5% and the latter 0.1%. Hence a right liver graft is generally chosen only if the left liver volume as estimated by volumetry using computed tomography is too small for the needs of the recipient. Previous studies have indicated that graft survival will be compromised if the graft volume–to–standard liver volume ratio is less than 40% or the graft weight–to–recipient body weight ratio is less than 0.8%. The mode of failure for small-for-size grafts is often manifested as small-for-size syndrome and is characterized by coagulopathy, cholestasis, ascites, sepsis, encephalopathy, and eventually graft failure. With maturation of surgical techniques, the minimum graft volume–to–standard liver volume ratio can be lowered from 40% to 35%, and perhaps even lower, without compromising outcome. Nonetheless, the graft size requirement may become higher in sicker patients with more severe portal hypertension and for liver grafts from older donors. For the safety of the donor, a remnant left liver of 30% or more of the total liver volume is required. The presence of fatty change has to be factored into the liver graft and remnant size estimation. In general, a fatty change of over 20% as verified by liver biopsy would prohibit a safe right liver donation.
The anatomy of the liver and its vasculature must be studied in detail using three-phase contrast-enhanced computed tomography or magnetic resonance imaging. Anatomical variants in the hepatic artery and portal vein need to be identified. Appreciation of the hepatic venous drainage, especially that of segment IVb/V/VIII and sizable (>5 mm) inferior right hepatic veins (RHVs), is of utmost importance. At our center, all right liver grafts include the middle hepatic vein (MHV), which allows optimal venous drainage. Preoperative biliary imaging is not performed.
Access is gained through a short right subcostal incision of only the right rectus muscle with upper midline extension. The two curved blades of the Bookwalter retractor pull the rib cage laterally and anteriorly to open up the aperture made by the costal margins. The ligamentum teres is ligated and divided, and the falciform ligament is taken down. Following careful laparotomy for unexpected intra-abdominal pathological conditions, intraoperative ultrasonography is performed to study the anatomy, especially that of the junction of the MHV and left hepatic vein with the inferior vena cava (IVC). The relation of the segment IVb vein to the MHV, already known from computed tomography, is ascertained. This also registers the flow characteristics of the hepatic arteries, portal veins, and hepatic veins bilaterally for reference throughout the operation.
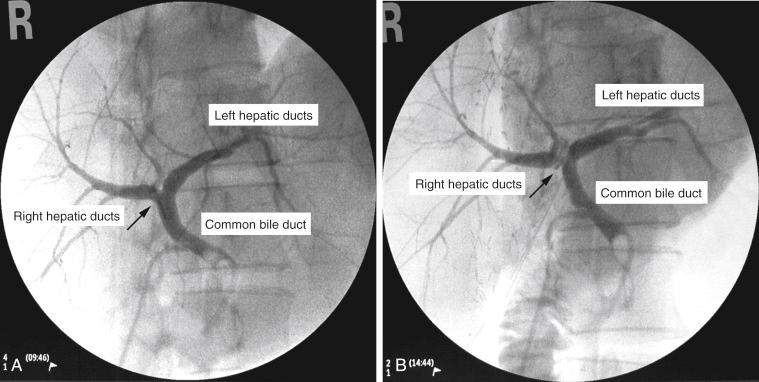
A good-quality operative cholangiogram using a C-arm fluoroscope is mandatory. The Calot triangle is dissected to isolate the cystic duct and cystic artery. After cannulation of the cystic duct with a balloon catheter, the gallbladder is excised. Minimal sharp dissection of the hilar plate just enough to identify the junction of the right hepatic duct with the common hepatic duct is done. The planned site of right hepatic duct division is then marked with a large metal clip externally to correlate with the findings of an operative cholangiogram. The balloon of the catheter is inflated, and the biliary tract is outlined by real-time operative cholangiogram under fluoroscopy. Swinging the C-arm of the fluoroscope laterally to the right (a right anterior-oblique view) allows a true “anterior-posterior” display of the right and left duct, and the parallax technique clarifies the location of the right anterior and posterior sectoral ducts ( Fig. 50-1 , A ). The liver capsule just superior of the planned site of right hepatic duct transection is marked by diathermy.
Hilar dissection is then continued to isolate the right hepatic artery and the right portal vein. The right hepatic artery dissection should be limited to the right side of the common hepatic duct, and the branch to segment IV should be preserved. If any branches to segment IV need to be sacrificed, the artery is temporarily clamped and a Doppler study is performed to ascertain that there are good collaterals from the left hepatic artery. The right hepatic artery and hilar plate should be completely separated, but the space between the right hepatic artery and the right hepatic duct should not be disrupted in order to preserve the blood supply of the latter. To isolate adequate length of the right portal vein, a number of branches to the caudate lobe need to be ligated and then divided.
The right triangular and coronary ligaments are then taken down to mobilize the right liver. Excessive rotation and compression of the right liver results in ischemic liver injury and should be avoided. Intermittent rotation may be required. The right liver is freed from the right adrenal, which may be adherent, and bleeding is controlled by the argon beam coagulator and plication with sutures. Short hepatic veins on the right side of the midline of the IVC are individually isolated and divided between ligatures. Inferior RHV(s) larger than 5 mm are preserved for reconstruction in the recipient. These are particularly crucial when the right liver graft is relatively small. The IVC ligament contains vessels and should be suture ligated after division. The RHV is then encircled with a vessel loop. A nylon tape is placed posterior to the caudate lobe, passing between the roots of the RHV and MHV, for the hanging maneuver to facilitate the liver transection. Temporary right liver inflow control with atraumatic vascular clamps is performed, and the line of transection, the Cantlie line, is marked with electrocautery. The line of demarcation is usually poorly defined between segments VIII and IVb at the cephalic end and is guided by extrahepatic identification of the RHV and MHV. The transection line should be directed toward the root of the MHV. The line on the inferior surface of the liver is just to the left of the gallbladder fossa and is extended to join the planned point of division of the right hepatic duct as marked earlier.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here