Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The adrenal medulla occupies the central portion of the adrenal gland. Adrenomedullary cells are called chromaffin cells because they stain brown with chromium salts. Chromaffin cells differentiate in the center of the adrenal gland in response to cortisol; some chromaffin cells also migrate to form paraganglia. The largest cluster of chromaffin cells outside the adrenal medulla is near the level of the inferior mesenteric artery and is referred to as the organ of Zuckerkandl.
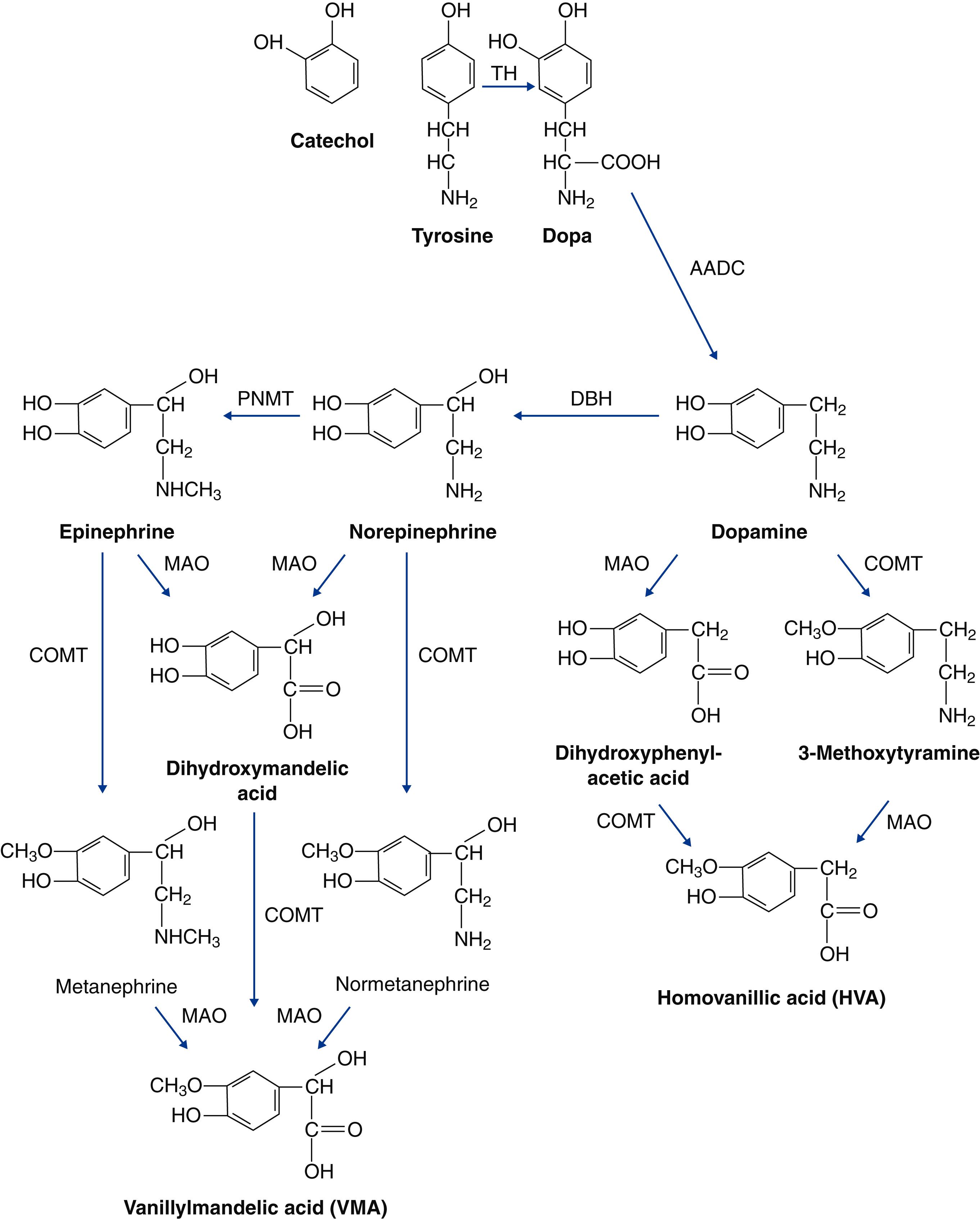
Catecholamines are substances that contain catechol ( o -dihydroxybenzene) and a side chain with an amino group—the catechol nucleus ( E-Fig. 209-1 ). Epinephrine, which is synthesized and stored in the adrenal medulla, is released into the systemic circulation. Norepinephrine is synthesized and stored not only in the adrenal medulla but also in the peripheral sympathetic nerves. Dopamine, the precursor of norepinephrine, is found in the adrenal medulla and peripheral sympathetic nerves.
Catecholamines have many cardiovascular and metabolic actions, including increasing the heart rate, blood pressure, myocardial contractility, and velocity of cardiac conduction. Three types of specific adrenergic receptors mediate their biologic actions: α, β, and DA. Their receptor subtypes are α 1 , α 2 , β 1 , β 2 , β 3 , DA 1 , and DA 2 . The α 1 subtype is a postsynaptic receptor that mediates vascular and smooth muscle contraction; stimulation causes vasoconstriction and increased blood pressure. The α 2 -receptors are located on presynaptic sympathetic nerve endings and, when activated, inhibit the release of norepinephrine; stimulation causes suppression in central sympathetic outflow and decreased blood pressure. Stimulation of the β 1 -receptor causes positive inotropic and chronotropic effects on the heart, increased renin secretion in the kidney, and lipolysis in adipocytes, as well as bronchodilation, vasodilation in skeletal muscle, glycogenolysis, and increased release of norepinephrine from sympathetic nerve terminals. The β 3 -receptor regulates energy expenditure and lipolysis. DA 1 receptors are localized to the cerebral, renal, mesenteric, and coronary vasculature; stimulation causes vasodilation in these vascular beds. DA 2 receptors are presynaptic and localized to sympathetic nerve endings, sympathetic ganglia, and brain; their stimulation inhibits the release of norepinephrine, inhibits ganglionic transmission, and inhibits the release of prolactin.
Catecholamines are synthesized from tyrosine by a process of hydroxylation and decarboxylation (see E-Fig. 209-1 ). Tyrosine, which is derived from the diet or synthesized from phenylalanine in the liver, enters neurons and chromaffin cells by active transport. The conversion of tyrosine to 3,4-dihydroxyphenylalanine (dopa) by tyrosine hydroxylase is the rate-limiting step in the synthesis of catecholamines. α-Methyl- p -tyrosine (metyrosine) is a tyrosine hydroxylase inhibitor that may be used therapeutically in patients with catecholamine-secreting tumors. Aromatic l -amino acid decarboxylase catalyzes the decarboxylation of dopa to dopamine. Dopamine is actively transported into granulated vesicles where it is hydroxylated to norepinephrine by the dopamine β-hydroxylase. These reactions occur in the synaptic vesicle of adrenergic neurons and the chromaffin cells of the adrenal medulla. In the adrenal medulla, norepinephrine is released from the granule into the cytoplasm, where phenylethanolamine N -methyltransferase converts it to epinephrine. Expression of phenylethanolamine N -methyltransferase is positively regulated by glucocorticoids. Catecholamine-secreting tumors that secrete primarily epinephrine are localized to the adrenal medulla. In normal adrenal medullary tissue, approximately 80% of the catecholamine released is epinephrine.
The biologic half-life of circulating catecholamines is between 10 and 100 seconds, so plasma concentrations of catecholamines can fluctuate widely. Catecholamines are removed from the circulation by either reuptake by sympathetic nerve terminals or metabolism through two enzyme pathways (see E-Fig. 209-1 ), followed by sulfate conjugation and renal excretion. Almost 90% of catecholamines released at sympathetic synapses are taken up locally by the nerve endings (i.e., uptake 1). Uptake 1 can be blocked by cocaine, tricyclic antidepressants, and phenothiazine. Extraneuronal tissues also take up catecholamines (i.e., uptake 2). Most of these catecholamines are metabolized by catechol- O -methyltransferase. Metanephrine and normetanephrine are oxidized by monoamine oxidase to vanillylmandelic acid by oxidative deamination. Monoamine oxidase may also oxidize epinephrine and norepinephrine to 3,4-dihydroxymandelic acid, which is then converted by catechol- O -methyltransferase to vanillylmandelic acid. In the storage vesicle, norepinephrine is protected from metabolism by monoamine oxidase. Monoamine oxidase and catechol- O -methyltransferase metabolize dopamine to homovanillic acid (see E-Fig. 209-1 ).
Catecholamine-secreting tumors that arise from the chromaffin cells of the adrenal medulla and the sympathetic ganglia are referred to as pheochromocytomas and extra-adrenal catecholamine-secreting paragangliomas, respectively. Because the tumors have similar clinical presentations and are treated with similar approaches, many clinicians use the term pheochromocytoma to refer to both entities. However, the distinction between pheochromocytoma and paraganglioma is important because of differing risks for malignant transformation and different approaches to genetic testing for associated neoplasms.
Catecholamine-secreting tumors are rare, with an annual incidence of two to eight cases per million people. Over the past 3 decades, the diagnosed incidence of these tumors increased about five-fold because of smaller incidental tumors (adrenal incidentalomas; Chapter 208 ) detected on radiographic scans in patients with few or no symptoms. Catecholamine-secreting tumors occur with equal frequency in men and women, primarily in the third, fourth, and fifth decades of life. These tumors are rare in children; when discovered, they may be multifocal, extra-adrenal, metastatic, and associated with a hereditary syndrome.
Approximately 40% of patients with catecholamine-secreting tumors have germline pathogenic variants (inherited mutations present in all cells of the body) in genes associated with the genetic disease. Hereditary catecholamine-secreting tumors typically are manifested at a younger age than sporadic neoplasms. Sporadic pheochromocytoma is typically diagnosed on the basis of symptoms or incidental discovery on computed imaging, whereas syndromic pheochromocytoma and paraganglioma are frequently diagnosed earlier in the course of disease because of biochemical surveillance or genetic testing.
Multiple endocrine neoplasia (MEN) type 2A is an autosomal dominant disorder ( Chapter 212 ). The phenotype includes adrenal pheochromocytoma in 50% (usually bilateral and sometimes asynchronous), medullary carcinoma of the thyroid in 100%, hyperparathyroidism in 20 to 30%, and cutaneous lichen amyloidosis in 5%. Medullary carcinoma of the thyroid is usually detected before the pheochromocytoma. Numerous activating pathogenic variants in the RET proto-oncogene have been documented in persons with MEN type 2A.
MEN type 2B, which is also an autosomal dominant disorder, represents approximately 5% of all MEN type 2 cases. The phenotype includes pheochromocytoma in 50% (usually bilateral), aggressive medullary carcinoma of the thyroid in 100%, mucosal neuromas (typically involving the tongue, lips, and eyelids) in most patients, thickened corneal nerves, intestinal ganglioneuromatosis, and marfanoid body habitus. MEN 2B–associated tumors are caused by pathogenic variants in the RET protein’s intracellular domain, (see Chapter 212 ).
Von Hippel-Lindau (VHL) disease ( Chapter 385 ) is an autosomal dominant disorder in which nearly 100% of patients have an identifiable pathogenic variant in the VHL tumor suppressor gene. Certain missense mutations appear to be associated with a “pheochromocytoma only” presentation. The disease is characterized by pheochromocytoma (frequently bilateral), paraganglioma (skull base, mediastinal, abdominal, pelvic), hemangioblastoma (involving the cerebellum, spinal cord, or brain stem), retinal angioma, clear cell renal cell carcinoma, pancreatic neuroendocrine tumor, endolymphatic sac tumor of the middle ear, serous cystadenoma of the pancreas, and papillary cystadenoma of the epididymis and broad ligament. Pheochromocytoma occurs in about 10 to 20% of patients with the disease.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here