Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
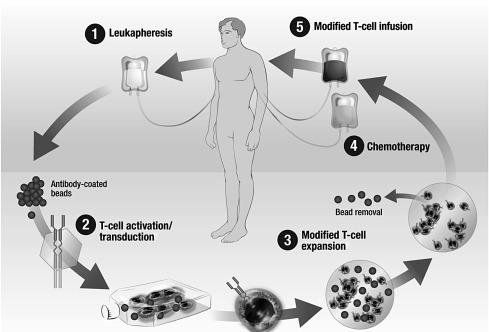
The fields of vaccinology and adoptive cell transfer (ACT) converge in the shaping of the acquired cellular and humoral responses of the B- and T cells of the immune system toward desired antigens on pathogens or self-targets. However, the approaches differ markedly, with vaccines often requiring immunizations and boosting over a period of months, whereas ex vivo culture of T cells and gene transfer can generate sufficient T cells in less than a week ( Fig. 30.1 ). The process of ACT is more complex but the power of the resultant T cells may justify the expense and effort in some areas such as cancer and chronic infection, where vaccines often fail.
A scientific and technological revolution in the war against cancer has taken place in the past century. Paul Ehrlich was probably the first to envision targeted therapies with “magic bullets,” an idea that was actualized by the invention of monoclonal antibodies and has inspired generations of scientists to devise powerful molecular cancer therapeutics. In this review the potential uses of chimeric T cells, modified to express antibody fragments is discussed.
The idea that passive transfer of primed immune cells can generate immunity in the recipient of this transfer is a relatively old idea in the history of immunology. As first proposed by Billingham, Brent, and Medawar in 1954, who coined the term “adoptive immunity,” numerous animal studies have demonstrated the effectiveness of adoptive transfer of immunity toward cancer and infectious disease. Immunity has remarkable specificity toward its targets, and specificity can be controlled through strategies such as in vivo and ex vivo priming and genetic engineering to install receptors of defined specificities. Moreover, it has the potential to induce longstanding effects via the establishment of immunologic memory.
In early studies, the transfer of pig and nonhuman primate lymphocytes were tested and found inadequate for ACT due to transient engraftment. However, with the advent of efficient tools for genetic editing, it is not inconceivable that the use of xenogeneic cells for ACT might be reconsidered. Currently, human T cells with alpha beta T-cell receptors (TCRs) are currently the cell of choice for most clinical trials, reviewed in Ref. . Gene transfer of MHC class I-restricted TCRs can “convert” a population of polyclonal CD8+ T cells to cytotoxic T-lymphocytes (CTLs) of monoclonal TCR specificity. The adoptive transfer of engineered CD4+ T cells has promise for adoptive therapy of cancer and HIV, reviewed in Refs. .
Recent advances in the understanding of the biology of γδ T cells suggest that these cells have promise for ACT. For example, human γδ T cells can prime αβ T cells with an efficiency similar to that of dendritic cells. Human γδ T cells can be engineered to express chimeric antigen receptors (CARs) and αβ TCRs. The use of γδ T cells may provide a significant safety feature over the use of TCR-engineered αβ T cells, taking advantage of the finding that α and β TCR chains cannot pair with γ and δ TCR chains.
Natural killer T (NKT) cells are functionally related to γδ T cells since they also bridge innate and adaptive immune responses and can enhance or suppress immunity. The best characterized human NKT cell subpopulation, referred to as invariant NKT (iNKT) cells, expresses CD161 and an invariant Vα24Jα18 TCR that recognizes α-galactosylceramide presented by the MHC class I-like molecule CD1d. After activation, iNKT cells have MHC-independent cytotoxic activity against various tumors and secrete high levels of interferon-γ, although this function becomes impaired in patients with cancer. Compared with mouse NKT cells, human NKT cells are rare and comprise <1% of total lymphocytes. However, human NKT cells, unlike mouse NKT cells, can undergo substantial expansion in vitro. The first ACT studies of iNKT cells have been conducted and show safety and some evidence of antitumor activity.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here