Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Gastrointestinal bleeding (GIB) is a term that describes the loss of blood from along the alimentary canal. GIB is classified by its anatomic location relative to the ligament of Treitz. Upper GIB (UGIB) is defined as being proximal to the ligament of Treitz. Upper intestinal hemorrhage is the most common presentation of GIB and is commonly from peptic ulcer disease (PUD) or esophageal varices. Pancreatic, liver, and other biliary origins of blood loss also are encompassed by this term.
Lower GIB (LGIB) accounts for 30% to 40% of all bleeds and is defined as distal to the ligament of Treitz. This most often originates from the colon from diverticular disease or angiodysplasias.
The term massive GIB refers to intestinal blood loss leading to hemodynamic instability or transfusion requirement, whereas occult GIB refers to anemia that persists or recurs after negative endoscopic evaluation and imaging workup.
Overall, bleeding is the most common cause of hospitalization from gastrointestinal (GI) disease in the United States, accounting for over 507,000 admissions annually with a total cost of over $5.8 billion. The reported median length of hospital stay for GI hemorrhage is 3 to 6 days, with median cost ranging from $6700 to $20,370. The peak incidence has steadily decreased by 1% annually since the mid-1990s due to the advent of proton pump inhibitors (PPIs), increased treatment of Helicobacter pylori , and avoidance of nonsteroidal antiinflammatory drugs (NSAIDs). Following this trend, mortality has dramatically decreased from the historical rates of 6% to 12% for UGIB to more contemporary rates of less than 2%. , While these advancements have steadily decreased hospital admissions for UGIB, admissions related to LGIB have increased.
GIB may present in a subtle way as a diagnosis of unexplained microcytic anemia or the finding of a positive hemoccult stool study. In contrast, it may also present as massive life-threatening exsanguination. Depending on the manifestation, treatment may take place in the outpatient setting or be emergently managed. Regardless, a multidisciplinary approach is required to fully tend to the condition and to determine the location and best therapeutic approach for the blood loss.
Rapid triage of hemorrhaging patients while localizing the areas of blood loss is essential for resuscitation and prompt intervention.
Assessment should adhere to the ABCDEs: airway, breathing, circulation, disability, and exposure. Patency of the airway and adequacy of breathing are the priority. Severe hematemesis or decreased mental status from shock or hepatic encephalopathy may compromise oxygenation, ventilation, and airway protective reflexes. If an airway is needed, it should be secured with attention to the patient’s hemodynamic status. Intravenous (IV) access with two large-bore (14- or 16- gauge) catheters should be obtained. Occasionally, in the massively hemorrhaging patient, central access with resuscitative lines is needed to maintain hemodynamic support. Invasive hemodynamic monitoring should also be considered in such patients. A urinary catheter should be placed to follow adequacy of resuscitation and preservation of renal function. The severity of hemorrhage can generally be determined quickly using simple clinical parameters. Tachypnea, tachycardia, hypotension, agitation, and mental status changes are all indicators of a severe degree of hemorrhage.
Hypotension is a harbinger of death. Systolic blood pressures under 90 mm Hg do not typically manifest until a patient has experienced a 30% to 40% blood loss. In well-compensated hosts, many of these signs may be absent or subtle, manifesting only as anxiety, tachypnea, or cool skin. The clinical response may be further blunted in patients taking beta blockers or those patients who are at the extremes of age.
Additional management priorities include obtaining a type and crossmatch, complete blood count, metabolic panel, coagulation profile, and liver function tests. Serum lactate can be utilized as an endpoint of resuscitation when elevated. It is important to note that in acute severe blood loss, the serum hematocrit is not a reliable marker of the amount of blood loss as it may take hours to dilute as fluid shifts from the interstitium and the patient receives volume resuscitation.
The strategy for fluid resuscitation should be guided by the severity of hemorrhage. With large volume blood loss, the utilization of massive transfusion protocols can make universal donor blood products rapidly available in prespecified ratios with proven survival benefit in bleeding patients. The ideal ratio of these products for GIB has not been well studied, although evidence suggests that a ratio of blood constituents (plasma and platelets) that approximates the whole blood (one unit fresh-frozen plasma per two units of packed red blood cells [RBCs] administered) lost during hemorrhage may be beneficial. , Furthermore, rapid blood loss can be complicated by preexisting or hemorrhage-related coagulopathy with deficiencies in both pro- and antithrombotic factors. Normalization of prothrombin time, partial thromboplastin time, fibrinogen levels, and platelet count are important adjuncts in management A role for viscoelastic tests such as thromboelastography or thromboelastometry has not been well studied in GIB but has shown benefit in other hemorrhage-related clinical scenarios.
Use of a rapid infuser can be utilized to keep up with rapid blood loss and may help warm products to minimize hypothermia. These patients often require admission to the intensive care unit and urgent intervention to localize and control hemorrhage. Clinical response to volume to support mentation, radial pulse pressure, and at least a systolic blood pressure of 90 mm Hg, while following endpoints of resuscitation, such as lactate clearance and urine output, guides adequacy of resuscitation.
It is important to distinguish massively hemorrhaging patients from stable patients with GIB in whom a restrictive transfusion strategy should be employed. A landmark study by Villanueva compared a restrictive transfusion threshold of a hemoglobin level of 7 to 9 g/dL to a liberal threshold of 9 to 11 g/dL. This randomized controlled trial found lower rates of mortality, rebleeding, and other adverse outcomes with the restrictive strategy of 7 g/dL. Importantly, this trial specifically excluded patients who were massively exsanguinating. In light of this study, such massively hemorrhaging patients should be considered a different patient population in terms of management strategy.
Running parallel with the active resuscitation of the patients is the equally important search to localize the site of blood loss ( Fig. 47.1 ).
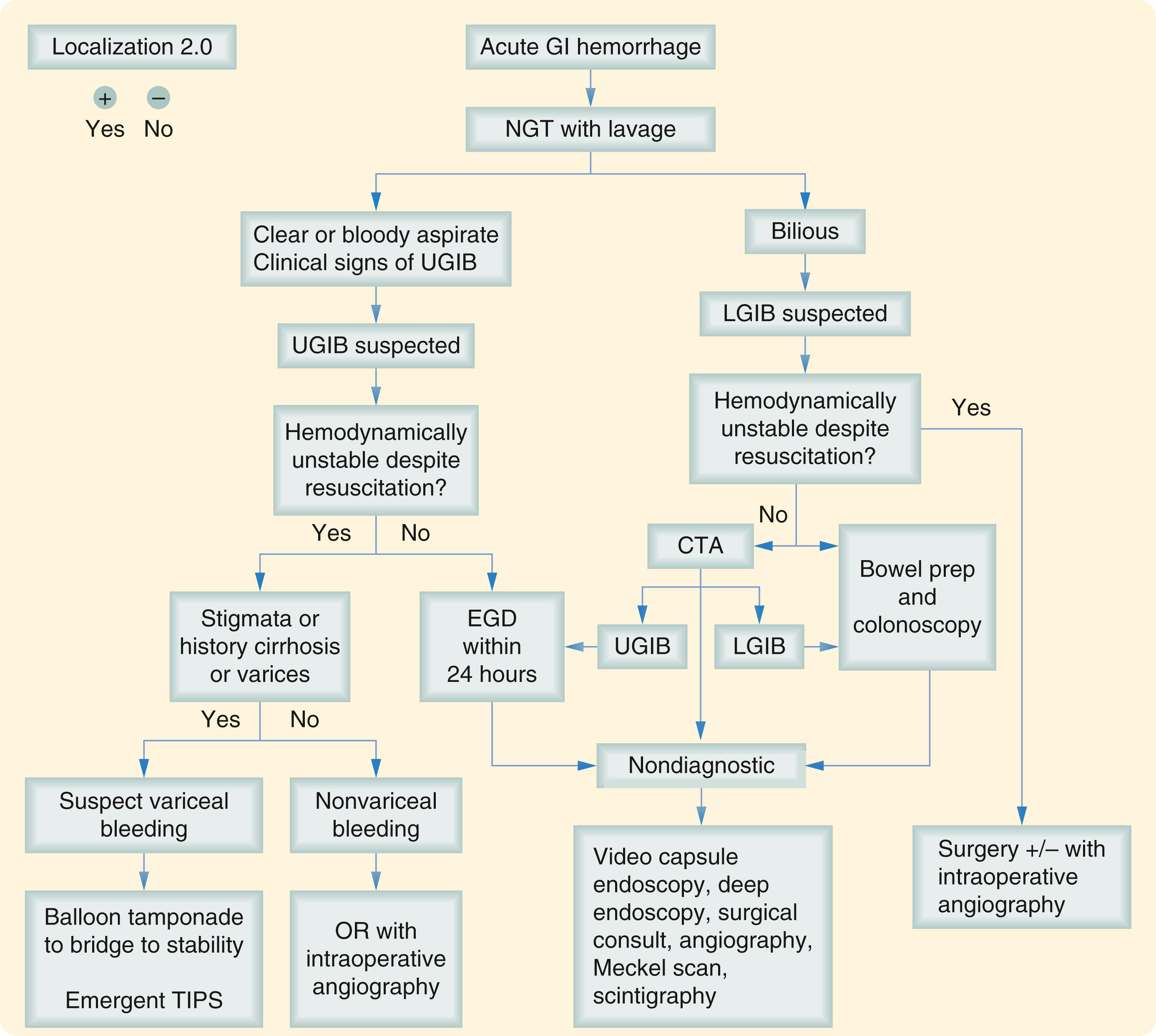
History and physical exam guide the assessment and may direct the examiner to a presumptive diagnosis. This information, combined with the characteristic of the blood loss, is helpful for determining an upper versus lower source.
Hematemesis is the emesis of blood or coffee ground gastric secretions, most commonly from a UGIB. Bleeding from the nasal or oropharyngeal space also may be swallowed or pool dependently in the stomach and thus lead to a non-GI source of hematemesis.
Melena, a malodorous, black stool with a tar-like quality, is also indicative of a proximal source of bleeding. Melena is the byproduct of hemoglobin degradation by digestive enzymes and intestinal bacterial flora. Hematin is produced from the degradation of hemoglobin, leading to a brownish blue coloration. Over 90% of melena arises from a UGIB, though it can originate from anywhere in the GI tract if intestinal transit is slow. Hematochezia, the passage of bright red blood from the anus, is most commonly from hemorrhoidal disease. However, brisk UGIB with swift transit through the intestinal tract may also manifest as bright red blood per rectum and is an important etiology to rule out upon evaluation.
The first steps in differentiating a UGIB from a LGIB is aided by a nasogastric tube (NGT) lavage ( Fig. 47.1 ). It can detect the presence of blood above the ligament of Treitz while also irrigating the stomach in preparation for an endoscopy. Aspiration of bile is required to assure sampling of postpyloric secretions. An aspiration, which is bilious and nonbloody, can effectively rule out an active UGIB. However, a nonactive upper GI (UGI) source is not definitively ruled out by a negative lavage if hemostasis occurred several hours prior.
The next localization steps after NGT lavage is then based on clinical suspicion, patient stability, and bleeding rate.
Given enough clinical history or suspicion, one may target treatment to the most likely diagnosis, for instance, performing endoscopy for suspected ulcer disease or other therapeutic interventions without further localization. In an actively bleeding patient in whom further localization is needed, the balance between patient stability and timely evaluation is essential. The goal is to efficiently localize the area of bleeding prior to hemodynamic instability to avoid the rare but difficult circumstance of a forced procedure without source identification. An exsanguinating or moribund patient with unlocalized bleed should proceed to a hybrid room for visceral angiography and/or operative intervention.
In patients whose hemodynamics can support further workup, the multidetector computed tomography angiogram (CTA) is emerging as the first-line study for localization of GIB. CTA can detect bleeding rates as low as 0.3 mL/min when performed under specific protocols that include three-phase IV contrast, multidetector scanners (64 slice) with no enteral contrast. , The sensitivity and specificity of CTA approaches 100% and over 90%, respectively, and may reduce overall radiation and IV contrast of a visceral angiogram. In contrast to the CTA, technetium-99-m-labeled RBC scintigraphy can detect bleeding as low as 0.04 mL/min. A second advantage of tagged RBC scans is that repeat images can be acquired up to 24 hours after the initial labeling of RBCs. However, use of this modality sacrifices the precision of hemorrhage localization since it generally can only suggest the quadrant of the abdomen from where the bleeding is occurring. Regardless, in a stable patient with a slow intermittent bleed, RBC scintigraphy can sometimes provide a valuable estimation of bleeding site that can guide endoscopic or angiographic interventions. , ,
Patients with bleeding localized to the UGI tract should proceed to upper endoscopy within 24 hours of presentation, if not sooner, to diagnose and treat the source. If bleeding cannot be localized with imaging or angiography and UGIB has been ruled out, a colonoscopy should be performed if the patient is stable and can tolerate a full bowel preparation. A mechanical bowel prep assures a high-quality colonoscopy, as any stool or retained blood will often obscure the bleeding site. Diverticular bleeding is statistically the most common source of LGIB and visualization of bleeding within diverticula is severely impaired by the presence of stool and old blood. The exception to this is if a brisk bleed from the descending colon or rectum is suspected. In this case, a careful LGI endoscopy may be undertaken, as any colonoscopy without prep increases the risk of perforation.
To aid in the preparation of the colon to improve diagnostic accuracy, the patient should receive a “rapid prep,” which is a minimum of 4 L of polyethylene glycol solution given within a period of approximately 4 hours, followed by colonoscopy within 1 to 2 hours. This large volume of fluid is often difficult to tolerate, so administration through an NGT may be necessary. Administering prokinetics to improve gastric emptying can also reduce nausea and discomfort associated with the high volume of prep solution.
The colonoscopy should always include intubation of the terminal ileum to rule out a more proximal source of bleeding. A large working channel is highly recommended to facilitate suctioning of stool and clots. Water-jet irrigation should also be used to flush debris from the mucosa and improve visualization.
If UGI and colonic sources of bleeding have been excluded, the next most common location for bleeding is the small bowel, sometimes referred to as “middle GIB.” This accounts for 5% of acute GIBs. CTA remains the first test of choice. In a stable patient, there are additional options for further localization of bleeding if the CTA is nondiagnostic. These include video capsule endoscopy and push, device-assisted (balloon or spiral enteroscopy), or intraoperative enteroscopy.
Video capsule endoscopy is noninvasive and is designed for imaging of the small bowel. It is the diagnostic modality of choice for overt GIB in a stable patient when upper and lower sources have been ruled out. , Bleeding is the most common indication for capsule endoscopy. The diagnostic rates are reported to be between about 35% to 67% and highest in acute GIB rather than obscure blood loss. Other factors that are associated with a positive capsule study are male sex, age over 60 years, and hospitalization at the time of test performance. A full bowel preparation maximizes visualization of the mucosa. If there is recurrent bleeding, the capsule endoscopy can be repeated. The results should be used to guide further endoscopic or surgical therapy. If deep enteroscopy or device-assisted enteroscopy is performed, the transit time of the capsule can aid in determining the starting point of the endoscopy. The main risk of capsule endoscopy is capsule retention that can occur in up to 1.5% of patients when used for overt GIB.
Patients at high risk for capsule retention are those with heavy NSAID use, tumors, Crohn disease, prior small bowel radiation, or surgery. A dissolvable “test” capsule is sometimes used first to ensure that the video capsule will not get stuck. These higher-risk patients may be better served by the alternative diagnostic strategies of computed tomography (CT) enterography or magnetic resonance (MR) enterography.
Multiphase CT enterography can be superior to capsule endoscopy in detecting bleeding from tumors. Typically, it is performed with a neutral or low-density oral contrast to distend the small bowel and includes IV contrast. Scans are performed in the arterial, enteral, and delayed phase. If a patient is stable and can tolerate capsule endoscopy and CT enterography, these studies are complementary to each other in discovering a small bowel source of bleeding.
A positive study with capsule endoscopy or CT/MR enterography should be followed with a push or device-assisted enteroscopy as these have therapeutic capability.
Push enteroscopy using a small-caliber colonoscope can reach about 50 to 70 cm past the ligament of Treitz and leads to successful diagnosis in 40% of patients with obscure small bowel bleeding.
Double-balloon endoscopy is quickly gaining favor in the diagnosis and treatment of small bowel lesions. The procedure uses the peristaltic “push/pull” motions from inflation and deflation of two balloons to course the small intestine along its length. This technique is capable of examining the entire small bowel, with successful identification of 77% to 85% of occult bleeding sources. Although technically challenging, it has a diagnostic efficacy greater than that of video capsule endoscopy.
Patients with altered anatomy, for example, Roux-en-Y bypasses, should undergo device-assisted endoscopy to evaluate the excluded portions of bowel. Intraoperative endoscopy, during laparotomy or laparoscopy, is a last resort when other modalities have failed to localize a source of small bowel bleeding and the patient continues to require transfusions or repeat hospitalizations. Surgery may also be necessary if a device-assisted enteroscopy cannot be performed without lysis of adhesions. This typically uses a sterile, small caliber colonoscope that is passed bidirectionally with the surgeon assisting to pass the bowel over the endoscope. Any suspicious areas are marked for possible resection or are dealt with endoscopically if feasible.
The cause of obscure-overt bleeding is often a common lesion that is missed on initial evaluation. Repeated upper endoscopy and lower endoscopy should be performed and may identify lesions in up to 35% of patients. Tagged RBC scan and angiography can be helpful next steps but require ongoing hemorrhage. Small bowel enteroclysis, which uses barium, methylcellulose, and air to assist in image resolution, has been replaced largely in practice by CT enterography. CT enterography can identify gross lesions such as small bowel tumors and inflammatory conditions such as Crohn disease but cannot visualize angiodysplasias, the main cause of obscure small bowel hemorrhage.
Provocative angiographic testing, which involves administration of anticoagulants, fibrinolytics, or vasodilators to increase hemorrhage during angiography, has been employed in small series with favorable results, but reluctance to induce uncontrolled hemorrhage has limited its use. Surgical backup and the ability to salvage the patient in an operating room is essential to the planning of such a procedure.
Due to the divergence in diagnostic and therapeutic maneuvers, UGIB is often further subdivided into either nonvariceal or variceal bleeding.
PUD is responsible for up to two-thirds of UGIBs and can develop in the stomach or duodenum. Approximately 10% to 15% of patients with PUD will develop bleeding as part of their disease course. PUD results from an imbalance between mucosal barriers and other aggravating factors. The major etiologic factors in PUD are H. pylori and NSAIDs, and in some patients, these can act synergistically in the development of ulcers through their additive insults to the gastroduodenal mucosa. Worldwide, it is estimated that up to 77% of duodenal ulcers are associated with H. pylori infection. The bacterium causes an inflammatory reaction within the mucosa that impairs mucosal defense and allows ulcer formation. NSAIDs also disrupt the mucosal barrier but via a different mechanism. NSAIDs inhibit cyclooxygenases (i.e., COX-1 and COX-2), thus impairing prostaglandin synthesis, enhancing neutrophil adherence and subsequent mucosal injury. NSAIDs additionally inhibit the release of nitric oxide (NO) and hydrogen sulfide (H 2 S), further inhibiting protective mechanisms of the mucosa. In those patients with regular NSAID use, PUD has a prevalence of 15% to 20%. Erosion of the mucosal surface leads to injury, ulceration, and chronic blood loss that can be further exacerbated by antiplatelet agents, anticoagulants, and selective serotonin reuptake inhibitors. Significant bleeding does not occur until the erosion reaches an artery of the submucosa or an even larger vessel in the case of a penetrating ulcer. The most significant hemorrhage occurs when duodenal or gastric ulcers penetrate branches of the gastroduodenal or left gastric arteries, respectively.
Treatment of PUD begins with effective prophylaxis. Aggressive treatment of H. pylori , reduction of NSAIDs, and the use of alternative NSAIDS preparations such as COX-2 inhibitors for chronic therapy have further decreased the incidence of PUD. However, it has been the landmark discovery of PPIs that has most drastically impacted the treatment of PUD. Since their introduction in 1989, PPIs have become a mainstay in the treatment of acid-related disorders. Multiple randomized controlled trials have proven their efficacy in healing ulcers compared to placebo as well their superiority over H 2 -inhibitors. PPIs inhibit the final common pathway of acid secretion by targeting the H + /K + -ATPase of parietal cells. Acid is suppressed until replacement pumps are synthesized (up to 36 hours), well beyond the required 18 to 20 hours of a pH greater than 3 required for effective ulcer healing.
See localization algorithm for an outline of approach ( Fig. 47.2 ). After initial resuscitation, patients should undergo esophagogastroduodenoscopy (EGD). Those with high clinical risk may benefit from EGD as soon as possible, with evidence supporting intervention within 12 hours of presentation, rather than the more liberal time frame of 24 hours. While awaiting EGD, patients should be treated with a PPI. Prokinetics should be considered, as a metaanalysis has supported erythromycin prior to endoscopy to reduce the need for second endoscopy, amount of blood transfusion, and hospital length of stay. An NGT can be helpful for diagnosis, but it is unlikely to clear enough clot to improve endoscopic visualization of the gastric mucosa.
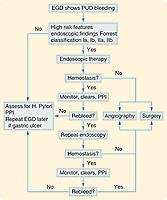
The next steps after index endoscopy depend on the findings. The Forrest classification was developed to assess the risk of rebleeding based on endoscopic findings and groups patients into high, intermediate, and low risk of rebleeding ( Table 47.1 ). Endoscopic therapy is recommended for ulcers with active bleeding as well as those with a visible ulcer (Forrest I–IIa). In cases with an adherent clot (Forrest IIb), the clot is removed and the ulcer evaluated. Ulcers with a clean base or black spot secondary to hematin deposition (Forrest IIc–III) do not require endoscopic treatment and are managed medically. Approximately 25% of patients undergoing EGD for UGIB will require an endoscopic intervention. If the endoscopy is unable to achieve hemostasis, angiography should be performed. Surgery is the next step if angiography fails or is not available.
| Classification | Rebleeding Risk | |
|---|---|---|
| Grade Ia | Active, pulsatile bleeding | High |
| Grade Ib | Active, nonpulsatile bleeding | High |
| Grade IIa | Nonbleeding visible vessel | High |
| Grade IIb | Adherent clot | Intermediate |
| Grade IIc | Ulcer with black spot | Low |
| Grade III | Clean, nonbleeding ulcer bed | Low |
All patients with confirmed peptic ulcer bleed should receive PPI therapy. Preendoscopic high-dose IV PPI therapy has been associated with decreased frequency of high-risk findings on endoscopy (Forrest Ia–IIa), leading to less need for endoscopic intervention. If PPI has not been started prior to endoscopy, a bolus of 80 mg should be given, followed by an infusion at 8 mg an hour for 72 hours. This approach has been shown to reduce risk of further bleeding, need for surgery, and mortality. Continuing PPI therapy postendoscopy also has been associated with reduced risk of further bleeding, need for surgery, and mortality. Interestingly, no difference in these outcomes was established between oral and IV preparations in a recent meta-analysis. However, heterogeneity in the study design, PPI dosing, and endoscopic findings limit absolute conclusions in the absence of a well-designed noninferiority trial.
Eradication of H. pylori infection, if present, has been shown in multiple studies and metaanalysis to result in less rebleeding. Confirmation of successful therapy should be confirmed by breath or stool test or by biopsy on repeat endoscopy. Once H. pylori has been eradicated, there is no need for long-term acid suppression as it does not decrease the rebleed risk of 1.3%.
All ulcerogenic medications such as NSAIDs or selective serotonin reuptake inhibitors should be discontinued in favor of alternatives. NSAIDs should be avoided as much as possible, although if they must be resumed, a combination of COX-2 selective NSAID and PPI should be used. Rebleeding rates with COX-2 inhibitors alone versus traditional NSAIDs plus a PPI are similar at 4% to 9%. A double-blind trial showed that COX-2 inhibitors plus PPI has a much lower rebleed risk as compared to COX-2 selective NSAIDs alone (0% vs. 8.9%). H. pylori eradication further improves the risk profile of NSAIDs.
The benefits of low-dose aspirin (ASA) for cardiovascular risk reduction are outweighed in some patients by the risk for GIB. The decision to continue ASA is based on its clinical indication for primary or secondary risk reduction. When used for primary risk reduction, the risk of recurrent GIB outweighs the benefit of ASA prophylaxis. A recent randomized controlled trial studied the benefits of ASA in primary prevention of cardiovascular events. An analysis of secondary endpoints showed that ASA did not reduce cardiovascular events when compared to placebo but did increase the risk of significant hemorrhage in multiple areas of the body, including upper and lower GI. However, ASA for secondary risk should be restarted in combination with a PPI within 1 to 7 days after bleeding cessation. This recommendation is based on a randomized trial that compared patients who had high-risk features on endoscopy and resumed ASA immediately with those on placebo for 8 weeks. All patients received PPI infusion followed by oral therapy. At 30 days, the group receiving ASA did have a nonsignificant increased risk of bleeding. This risk was far outweighed by the notable finding that patients receiving ASA, compared to placebo, had a lower mortality at 30 days (1.3% vs. 9%) and 8 weeks (1.3% vs. 12.9%).
Multiple endoscopic therapies have been used to treat PUD, including injection, thermal coagulation, plasma argon coagulation, mechanical clips, and fibrin glue. The recommended approach is to use thermal coagulation or clips, with or without epinephrine injection. This is based on meta-analysis showing that epinephrine monotherapy has a higher risk of rebleed compared to clipping or thermal coagulation. Injection therapy should be combined with other modalities such as heat or clipping. Epinephrine is diluted 1:10,000 or 1:20,000 and is injected into all four quadrants of a bleeding lesion. The rate of initial hemostasis with epinephrine monotherapy is as high as 100%, although the risk of rebleeding remains high and thus mandates use of another modality. The ideal volume of injection is unknown, although generally, 0.2 to 2 mL is injected in each quadrant. Large volume injection (>13 mL) is associated with improved hemostasis, suggesting that part of the mechanism is by tamponade and compression of the bleeding vessel. In addition to injection, heat or mechanical therapy is usually added and together can achieve initial hemostasis in up to 90% of bleeding ulcers. Heat can be in the form of monopolar or bipolar cautery, heater probe, or argon plasma coagulation. All heat modalities have similar efficacy and are effective in achieving initial hemostasis, reducing recurrent bleeding, surgery, and mortality.
Hemoclips are less effective than thermal therapy, although they may be advantageous in dealing with a spurting vessel for which they can provide immediate control of hemorrhage. Limitations of hemoclips are the difficulty in applying to fibrotic lesions and prolonged procedure time as only one clip can be applied at a time. It is important to properly place the first clip as improperly placed clips can impede placement of subsequent clips. Clips can be useful if subsequent angiographic intervention is done, as they can assist in localizing the bleeding.
A less commonly used hemostatic therapy is sclerosant injection, such as absolute alcohol. This modality is effective but risks tissue damage, thus making it less attractive.
Rebleeding of an ulcer is associated with a significant increase in mortality. Patients at high risk of rebleeding should be identified early using previously described criteria and observed under higher levels of care such as in an intensive care unit. With rebleeding, a second attempt at endoscopic control is recommended and is successful in 75% of patients.
Angiography is diagnostic as well as therapeutic and should be considered if a patient has failed endoscopic management or if the bleeding has not been localized. Access is obtained through the common femoral artery. The vessel interrogated first is based on clinical suspicion for location of the bleed. For suspected UGIB, the celiac artery and its branches are interrogated first as most UGIB is from gastric or duodenal ulcers supplied by branches of the celiac artery. The superior mesenteric artery and inferior mesenteric artery can also be evaluated if no bleeding is identified. The presence of clips or prior imaging can help guide further subselective catheterization.
In patients with repetitive, nondiagnostic workups, provocative maneuvers with systemic anticoagulation must be weighed against the risk of uncontrolled hemorrhage.
Transcatheter arterial embolization is effective at hemorrhage control when a bleeding source is found. Superselective embolization allows control of bleeding while maintaining adequate collateral flow to prevent bowel infarct. Examples include selective embolization of the left gastric artery or gastroduodenal artery for bleeding ulcers or the vasa recta or terminal branches of the inferior mesenteric artery for LGIB. There are various embolic agents such as coils, polyvinyl alcohol particles, Gelfoam, glue, and plugs. Coils and polyvinyl alcohol particles are most commonly used. Gelfoam is unique in that it is a temporary agent made of porcine adipose tissue and will recanalize over weeks to months, although the actual time course is often unpredictable. Success rates for UGIB embolization are cited at 44% to 100% and 88% to 93% for LGIB.
Vasopressin infusion is less frequently used now that improved methods of transcatheter embolization are available. The mechanism of action is that vasopressin constricts arteries to reduce blood flow to the site of hemorrhage. Downsides to its use are cardiac side effects and high rates of rebleeding after cessation of infusion and the need to maintain vascular access in situ for 24 to 48 hours to continue the infusion. The cardiac effects can be mitigated to some degree with a nitroglycerin infusion to maintain coronary perfusion. Vasopressin infusion can be useful if there is diffuse bleeding or as a bridge to surgical intervention if superselective cannulation cannot be achieved.
Despite significant advances in endoscopic therapy, approximately 10% of patients with bleeding ulcers still require surgical intervention for effective hemostasis. To assist in this decision making, several clinical and endoscopic parameters have been proposed that are thought to identify patients at high risk for failed endoscopic therapy. The Forrest classification is the best predictor of rebleeding (see Table 47.1 ). Other endoscopic factors associated with increased risk of rebleeding are active bleeding at time of endoscopy, large ulcer size (i.e., >2 cm), posterior duodenal wall ulcer, and lesser gastric curve ulcer. Patients with these characteristics need closer monitoring and possibly earlier surgical intervention. Clearly, clinical judgment and local expertise must play a critical role in this decision.
Indications for surgery have traditionally been based on the blood transfusion requirements, success of endoscopic therapy, and recurrent bleeding after repeat endoscopy. Increased blood transfusions have been clearly associated with increased mortality. Although a less definitive criterion than it was in the past, most surgeons still consider an ongoing blood transfusion requirement more than 6 units as an indication for surgical intervention, particularly in the elderly, although an 8- to 10-unit loss may be more acceptable for the younger population. Current indications for surgery for peptic ulcer hemorrhage are summarized in Box 47.1 . Secondary or relative indications include a rare blood type or difficult crossmatch, refusal of transfusion, shock on presentation, advanced age, severe comorbid disease, and a bleeding chronic gastric ulcer for which malignancy is a concern.
Hemodynamic instability despite vigorous resuscitation (>6-unit transfusion)
Failure of endoscopic techniques to arrest hemorrhage
Recurrent hemorrhage after initial stabilization (with up to two attempts at obtaining endoscopic hemostasis)
Shock associated with recurrent hemorrhage
Continued slow bleeding with a transfusion requirement exceeding 3 units/day
The first step in the operative management for a duodenal ulcer is exposure of the bleeding site. Most of these lesions are in the duodenal bulb; stay sutures are placed on either side of a longitudinal duodenotomy or duodenopyloromyotomy. Hemorrhage typically can be controlled initially with pressure and then direct suture ligation with nonabsorbable suture. Anterior ulcers can be directly ligated. More commonly, a posterior ulcer erodes into the pancreaticoduodenal or gastroduodenal artery. Suture ligature of the vessel proximal and distally, typically in a superior and inferior orientation, as placement of medial-stitch to control the pancreatic branches typically arrests the bleed. The duodenotomy is closed transversely with an NGT above and a nasojejunal tube placed beyond the repair for distal enteral access. Omental buttressing of the suture line may assist in healing. A surgical drain can be left in place if there is significant concern for a potential leak.
Traditionally, a definitive acid-reducing operation was considered if the patient was hemodynamically stable. This practice has largely been abandoned in the era of H. pylori eradication and PPI therapy, such that there has been a dramatic reduction in the rates of definitive ulcer therapy (gastrectomy or vagotomy). The choice between various acid-reduction operations was guided by the hemodynamic condition of the patient and the presence or absence of long-standing history of refractory ulcer disease. The various operations for PUD are discussed in greater detail in Chapter 49 . A pyloroplasty combined with truncal vagotomy is the most frequently performed acid-reduction surgery used in the setting of a bleeding duodenal ulcer. There is some evidence to suggest that a parietal cell vagotomy may represent a better therapy for a bleeding duodenal ulcer in the stable patient, although some of this benefit may be abrogated if the pylorus has been divided. In a patient who has a known history of refractory duodenal ulcer disease or who has failed to respond to more conservative surgery, antrectomy with truncal vagotomy may be more appropriate. However, this procedure is more complex and should generally not be done in a hemodynamically unstable patient.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here