Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
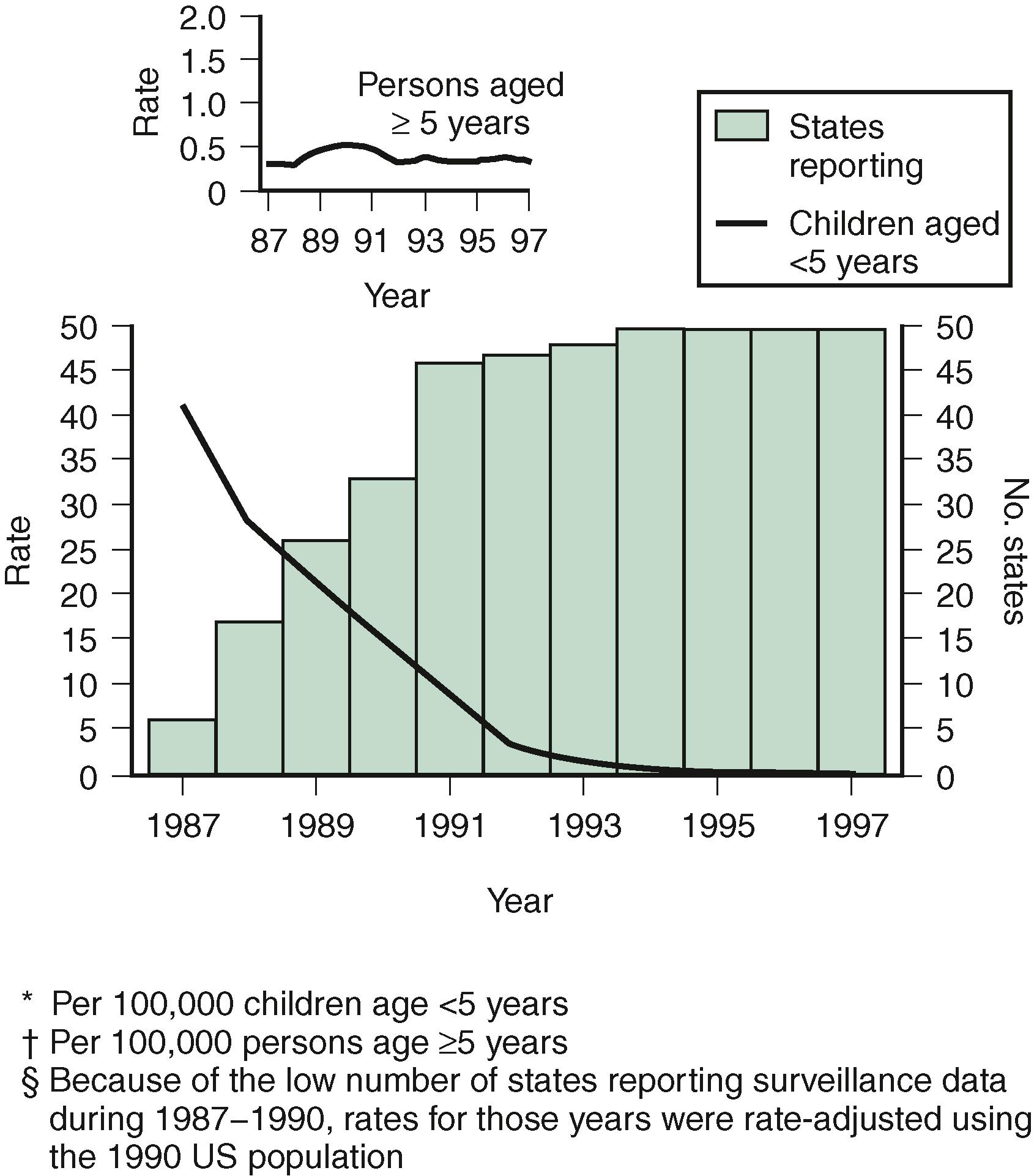
In otherwise healthy children, the three most common organisms causing hematogenously acquired acute bacterial meningitis worldwide are Streptococcus pneumoniae, Neisseria meningitidis, and Haemophilus influenzae type b (Hib). Previously, Hib accounted for up to 48% of all bacterial meningitis cases in the US ; however, the introduction of the Hib conjugate vaccination program led to a dramatic reduction in the incidence of Hib meningitis. In surveillance studies conducted by the Centers for Disease Control and Prevention (CDC), the incidence of Hib disease declined by 95% from 1987 to 1993 in children younger than 5 years (41 cases per 100,000 in 1987 to 2 per 100,000 in 1993) ( Fig. 40.1 ). Similar data from 1997 indicated a further decline to 1.3 cases per 100,000, and there was a further decline to 0.11 cases per 100,000 in 2008. The incidence also has decreased markedly in other areas of the world where Hib conjugate vaccines have been implemented for universal use in infants. , Currently, Hib accounts for only 7% of meningitis cases in the US, predominantly in unvaccinated adults and in children who have not completed the primary immunization series. ,
Pneumococcal and meningococcal (and previously Haemophilus ) meningitis typically occur more commonly during the winter. In 1994−1999, the annualized incidence of S. pneumoniae meningitis in children aged <2 years in the US was 7.7 per 100,000, 8 and 7 (4, 6B, 9, 14, 18F, 19F, and 23F) of >90 pneumococcal serotypes accounted for >80% of invasive disease in children in the US and other developed countries. Serotypes 5 and 1 also were prevalent in developing countries. The incidence of invasive disease caused by vaccine serotypes of S. pneumoniae fell by >95% after the implementation of universal 7-valent pneumococcal conjugate vaccine (PCV7) in US infants in 2000, from 80 cases per 100,000 population to <1 case per 100,000 by 2007. The rate of hospitalizations for pneumococcal meningitis in infants decreased by 66%. Between 2000 and 2010, the incidence of disease caused by nonvaccine serotype 19A increased, with many isolates resistant to β-lactam antibiotics, including penicillin and third-generation cephalosporins. , Universal implementation of PCV13 (including additional serotypes 1, 3, 5, 6B, 7F, and 19a) has led to a decrease in vaccine serotype invasive disease and only a modest further decrease in meningitis cases.
Pneumococcal meningitis can occur despite immunization with PCV, usually due to nonvaccine serotypes. After the introduction of PCV7, there was an increase in the proportion of cases of pneumococcal meningitis caused by serotypes not contained in the vaccine, particularly among children <2 years of age. , In population-based surveillance (1998–2007), serotypes 19A, 7F, 10A, and 22F were the most common non-PCV7 serotypes isolated from children with pneumococcal meningitis; approximately 60% of cases were caused by serotypes contained in the 13-valent pneumococcal conjugate vaccine (PCV13), which then replaced PCV7 in the US in 2010.
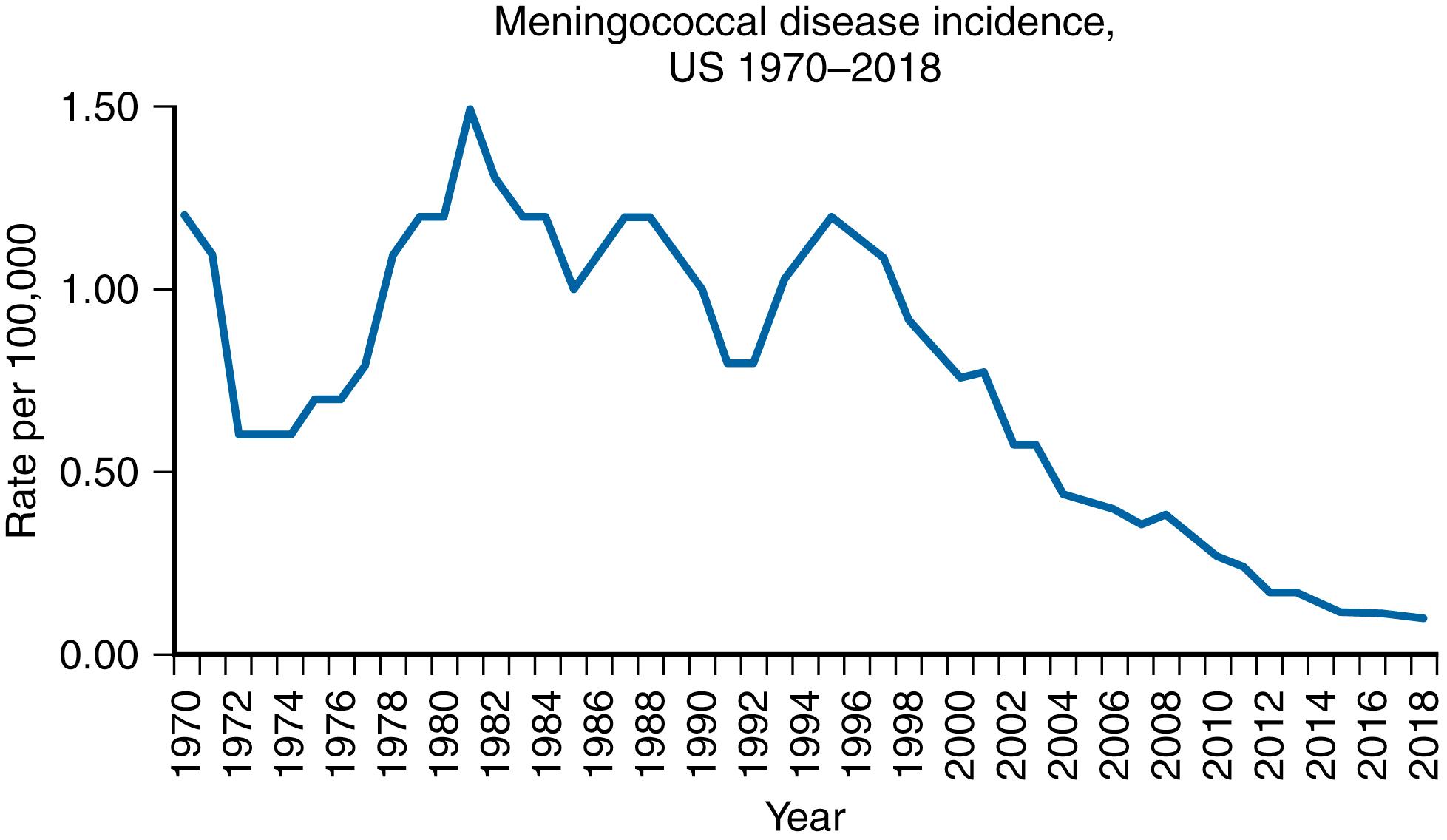
Six serogroups of N. meningitidis are responsible for severe disease and meningitis: serogroups A, B, C, X, Y, and W-135. Infection with N. meningitidis can be either epidemic or endemic, and although most cases in the US are endemic/sporadic, N. meningitidis is the only bacterium that can cause meningitis outbreaks in the US. Throughout America and Europe, serogroups B, C, W, and Y account for most cases of meningococcal meningitis, and serogroup B is the leading cause of endemic meningitis in resource-rich countries. , , The overall incidence of meningococcal disease was on the decline in 2005 when quadrivalent (A/C/Y/W-135) polysaccharide-protein meningococcal conjugate vaccine (MCV4) was licensed and recommended universally in the US for adolescents beginning at 11–12 years of age and for persons 2 through 54 years of age who have an increased risk for invasive meningococcal disease (IMD). , Since 2005, declines have continued among all age groups and for all vaccine-associated serogroups as well as for nonMCV4 serogroup B ( Fig 40.2 ). The overall case-fatality rates, however, remain at 10%–15%, and 11%–19% of survivors have long-term sequelae.
Neisseria meningitidis is an obligate human commensal that resides in the nasopharynx, with an estimated carriage rate between 0.6% and 34%. Rate may be higher in adolescents, young adults, and individuals living in overcrowded or confined spaces. , Approximately half a million cases of IMD occur worldwide annually with a mortality rate of ∼10%. The true incidence of IMD worldwide is difficult to ascertain because of variations in bacteriological surveillance in different countries and inaccurate and under-reporting in many resource-poor countries.
More than 90% of IMD is caused by 6 serogroups A, B, C, W, X, and Y. The distribution of serogroups causing disease varies with age group and geographical location.
Serogroup B is an important cause of endemic disease in much of the resource-rich world, including North America, Canada, Western Europe, Australasia, and South America. Since the successful introduction of vaccines in many parts of the world that are effective against group C, group B is now the cause of ∼60% of IMD in those countries. Nearly one-half of the burden of IMD occurs in children < 2 years of age.
In recent years, there has been a dramatic increase in some countries in the number of cases of IMD caused by serogroup W. In 2016/2017, there were 225 cases of group W disease of all ages in the UK, accounting for approximately one-third of reports. Group W also may contain the hypervirulent ST11 complex and has been associated with an atypical presentation of gastrointestinal symptoms and shock without a rash. Misdiagnosis is therefore common, and partly because of this, group W is associated with a high case-fatality rate. Serogroup Y is becoming an increasingly important cause of meningococcal disease in the US and more recently is increasingly reported in the UK. The changes in the epidemiology of carriage and invasive disease have led to alterations in vaccine schedules in the UK, including the introduction of the MenACWY vaccine in adolescents.
The annual incidence of IMD in the US varies in multiyear cycles. During 2005−2011, the annual incidence was 0.3 cases per 100,000, falling to 0.18 cases per 100,000 population in 2013, and to 0.11 in 2017. In 2018, there were 330 total cases of meningococcal disease reported. Incidence of meningococcal disease by year from 1970 through 2018, by age and serogroup are depicted in Figs. 40.2 and 40.3 and 125.3 and 125.4. The rates of disease are highest in children <1 year of age, followed by a second peak in adolescence. Among adolescents and young adults, those 16 through 23 years old have the highest rates of meningococcal disease.
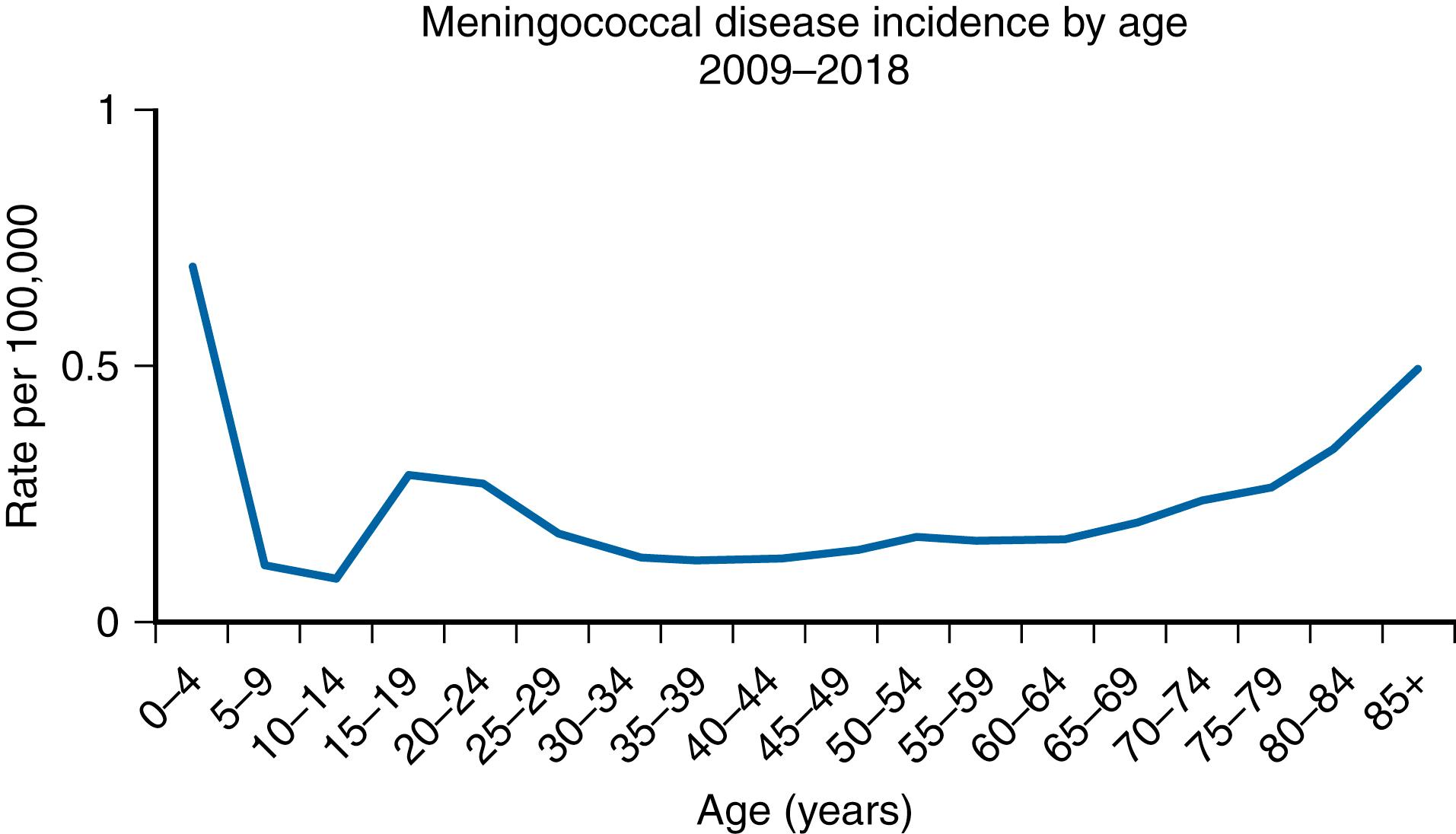
Serogroup B caused approximately 65% of IMD cases among children <5 years of age in 2009–2018 ( Fig. 125.4 ). Serogroups C, Y, or W (MCV4 serogroups) caused approximately two-thirds of cases of IMD among persons >11 years of age during this time period. However, in 2018, serogroups C, Y, or W caused approximately one-half of cases in persons >11 years of age. The emergence of infections due to serogroup W ST-11 has been the cause of some concern. The age distribution of these infections is somewhat different in that the infection has been observed primarily among individuals >18 years. A similar increase in the incidence of serogroup W ST-11 strains has been observed in Eastern Europe and Great Britain.
In the US, currently, the highest case-fatality ratios occur with serogroups W (21%) and C (14%) infection. The highest incidence and number of cases (25%) is in infants 2–3 months of age, and the highest mortality rate (approximately 28%) occurs in patients >85 years of age. The incidence is slightly higher among males and is higher among African Americans compared with other racial groups (0.27 vs. 0.20 cases per 100,000, respectively).
Underlying conditions are associated with increased risk for invasive infections in general, for meningitis, or for infection due to certain pathogens ( Table 40.1 ). Isolation from the cerebrospinal fluid (CSF) of an organism other than S. pneumoniae, N. meningitis , or Hib or isolation of an unusual serotype or a vaccine-preventable organism in a vaccinated child >2 months of age requires an explanation, with evaluation for unusual host susceptibilities such as an anatomic abnormality or immunologic disorder. Children with recurrent pneumococcal or meningococcal infections also should undergo thorough investigation, including neuroimaging and immunologic evaluation, with measurement of complement function. Patients with cochlear implants, especially using a positioner device, have a >30-fold increased incidence of pneumococcal meningitis and an increased risk of Hib meningitis. Certain host immune defects and events predispose to bacterial meningitis, with variable frequency ( Table 40.2 ). , Recent events can be important risk factors as well, such as exposure to a person with meningococcal or Hib infection, viral or other (especially respiratory or otic) infection, or travel to an area (sub-Saharan Africa) with endemic meningococcal disease.
| Condition | Organism |
|---|---|
| Cerebrospinal fluid leak (otorrhea, rhinorrhea) | Streptococcus pneumoniae, Haemophilus influenzae |
| Dermal sinus tracts meningomyelocele | Staphylococci, gram-negative enteric bacilli, intestinal bacteria (related to the site of the defect) |
| Persistent complement deficiency | Neisseria meningitidis |
| Asplenia (anatomic or functional) | S. pneumoniae, N. meningitidis, Salmonella spp. |
| Renal transplantation, T-lymphocyte deficiency | Listeria monocytogenes |
| Otic fistula (stapes footplate, oval window; cochlear implant) | S. pneumoniae |
| Ventriculoperitoneal shunt | Staphylococci (coagulase-negative and Staphylococcus aureus ), S. pneumoniae, H. influenzae, N. meningitidis (hematogenous), diphtheroids (e.g., contaminated shunt) |
| Antibody deficiency state (including HIV infection) | S. pneumoniae, N. meningitidis, H. influenzae type b |
| Penetrating trauma | Varies with nature and site of trauma (e.g., Pasteurella multocida after dog or cat bite, skin organisms after skull trauma, nasopharyngeal organisms after orbital or sinus trauma) |
| Surgery | Skin organisms, nosocomial pathogens |
| Host Problem | Organism(s) Favored | Frequency With Which Defect Leads to Meningitis |
|---|---|---|
| Absence of opsonizing antibody | Streptococcus pneumoniae | Common in all age groups |
| Haemophilus influenzae | Common in very young children | |
| Asplenia: surgical/functional | S. pneumoniae | Rare |
| Neisseria meningitidis | Very rare | |
| Complement deficiency | N. meningitidis | Very rare |
| Glucocorticoid excess | Listeria monocytogenes | Rare |
| HIV infection | S. pneumoniae | Common/presenting illness |
| L. monocytogenes | Rare | |
| Bacteremia/endocarditis | Staphylococcus aureus | Rare |
| Basilar skull fracture | S. pneumoniae | Very rare |
The propensity to cause meningitis and the mechanism of entry into the central nervous system (CNS) varies by pathogen ( Table 40.3 ). Bacterial meningitis usually follows stepwise progression: bacterial colonization of the nasopharynx (or skin), mucosal invasion (or a breach in skin barrier) leading to bloodstream infection, intravascular multiplication and passage through the blood-brain barrier, generation of inflammation within the subarachnoid space, and induction of neuronal and auditory cell damage.
| Organism | Site of Entry | Age Range | Predisposing Factors |
|---|---|---|---|
| Neisseria meningitidis | Nasopharynx | All ages | Usually none, rarely complement deficiency |
| Streptococcus pneumoniae | Nasopharynx, direct extension across skull fracture, or from contiguous or distant foci of infection | All ages | All conditions that predispose to pneumococcal bacteremia, or fracture of the cribriform plate, cochlear implant, cerebrospinal fluid otorrhea from basilar skull fracture, defects of the ear ossicle (Mondini defect) |
| Coagulase-negative staphylococci | Foreign body | All ages | Surgery and foreign body, especially ventricular devices |
| Staphylococcus aureus | Bacteremia, foreign body, skin | All ages | Endocarditis, surgery and foreign body, especially ventricular devices; cellulitis, decubitus ulcer |
| Haemophilus influenzae | Nasopharynx, contiguous spread from local infection | Infants and children who are unvaccinated | Diminished humoral immunity |
The 3 most common meningeal pathogens colonize the nasopharyngeal mucosa in 5%–40% of children or adolescents at any given time; for Hib and N. meningitidis, fimbriae mediate adherence to epithelial cells; for S. pneumoniae, specialized surface components, such as surface adhesion proteins, may be important. These encapsulated organisms are able to evade local host defenses and either invade between epithelial cells (Hib) or pass through epithelial cells ( N. meningitidis ) to reach the subepithelial tissues, where the organisms can invade the vascular endothelium. A viral upper respiratory tract infection can facilitate invasion by altering local tissue defenses. Increased hospitalization for invasive pneumococcal and meningococcal disease has been correlated with the viral respiratory season. , Intravascular replication leads to bacteremia and, with sufficient density of organisms, egress through the endothelium of the choroid plexus and cerebral capillaries into the ventricular fluid. A complex interplay between endothelial cells and microbial gene products appears to orchestrate the traversal of bacteria across the blood-brain barrier—transcellularly, paracellularly, or through infected phagocytes. For organisms that invade transcellularly, including E. coli, group B Streptococcus , and S. pneumoniae, the process is mediated by interactions with host receptors in the brain’s microvascular endothelial cells. For N. meningitidis, invasion is mediated by the outer membrane protein opcinvasin that binds to human fibronectin to attach and invade brain endothelial cells. Organisms multiply quickly and spread throughout the subarachnoid space because of the lack of intrinsic host defenses within the CSF. The host inflammatory response to bacteria leads to many of the alterations in CNS function and in the CSF that are characteristic of bacterial meningitis.
Communications between mucosal surfaces or skin and CSF that result from trauma, congenital malformations, or surgery can lead to direct invasion of the CNS by bacteria (Tables 40.1–40.3). Defects should be sought in patients with recurrent bacterial meningitis or meningitis due to unusual organisms. Bacterial meningitis also can be a complication of neurosurgery, spinal anesthesia, or placement of a ventricular shunt or external ventriculostomy device.
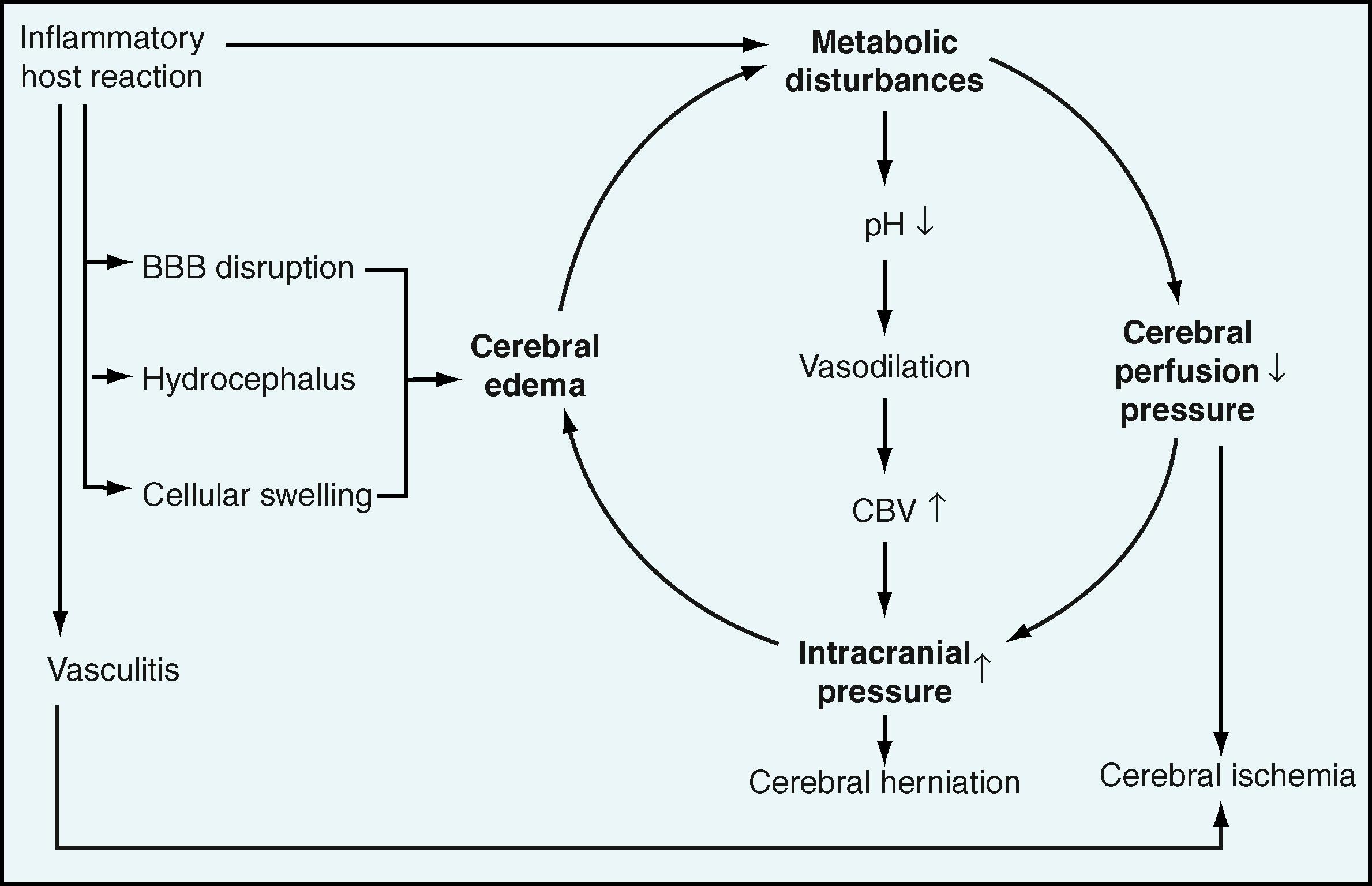
The pathologic features of bacterial meningitis have been described in detail. , Increased brain weight and flattened convolutions are evidence of cerebral edema. Temporal lobe or cerebellar herniation can occur. The brain and spinal cord are covered with a purulent subarachnoid exudate that consists primarily of neutrophils, which also infiltrate the perivascular spaces of blood vessels in the outer layers of the cortex of the brain and connective tissue sheaths of cranial and spinal nerves. Endothelial cells of small subarachnoid arteries and veins are swollen, leading to narrowing of the lumen and microscopic changes within brain parenchyma consistent with ischemia. The interstitial tissue of the choroid plexus is infiltrated with neutrophils. Phlebitis, venous thrombosis, arteritis, and brain necrosis are pathologic changes noted at necropsy in children who had untreated bacterial meningitis for <3 weeks. The foramina of Magendie and Luschka can be obstructed by exudate, resulting in obstructive hydrocephalus; however, communicating hydrocephalus is more common. This exudate, rich in bacterial products and proinflammatory substances, also can traverse the cochlear duct to involve the auditory tissue. Complex molecular events under study likely contribute to brain damage. It is currently believed that neuronal death is caused mainly by apoptosis through caspase-dependent and independent pathways. High levels of tumor necrosis factor-α have been observed in the CSF of patients with meningococcal meningitis when the sample was obtained within 48 hours of symptom onset. Index of the release of cytokines and chemokines per cell was significantly higher in pneumococcal meningitis. Levels of interferon γ alone were significantly higher in patients with meningitis due to S. pneumoniae compared with N. meningitidis . Changes in these mediators and in interleukin-1β (IL-1β) and IL-6 are the best-studied inflammatory responses in bacterial meningitis.
The molecular mechanisms responsible for oxidant-induced neuronal injury in experimental pneumococcal meningitis are summarized in Figs. 40.4 and 40.5 .
![Figure 40.4, Mechanisms of brain damage in experimental pneumococcal meningitis. NF-κB, a transcriptional activator of many genes involved in the pathogenesis of bacterial meningitis, encodes host factors including proinflammatory cytokines, chemokines (e.g., interleukin [IL]-8), and adhesion molecules. The proinflammatory cytokines IL-1β and tumor necrosis factor (TNF)- α are synthesized as inactive precursors that are processed to mature active forms by proteases (caspase 1 [Casp1], also known as IL-1β–converting enzyme, and TNF-α–converting enzyme [TACE]). IL-1β and TNF-α are potent activators of NF-κB. This process can lead to the uncontrolled expression of proinflammatory mediators and the increased expression of adhesion molecules both on the endothelium (e.g., intercellular adhesion molecule [ICAM]-1) and on neutrophils, leading to a subsequent massive influx of leukocytes into the subarachnoid space. Once present, activated leukocytes release a complex variety of potentially cytotoxic agents including oxidants and proteolytic enzymes (e.g., matrix metalloproteinase [MMP]), which may contribute to tissue destruction. Also, peroxynitrite may cause brain damage through a variety of independent mechanisms. The best studied are the attack of polyunsaturated fatty acids, leading to lipid peroxidation, and an alternative pathway that involves oxidant-induced DNA strand breakage and subsequent poly(adenosine diphosphate ribose) polymerase (PARP) activation, initiating an energy-consuming intracellular cycle that ultimately results in cellular energy depletion and cell death. Both mechanisms likely contribute to cell injury during pneumococcal meningitis. ATP, adenosine triphosphate; ECM, extracellular matrix; KC, keratinocyte-derived chemokine; MIP, macrophage inflammatory protein; NAD, nicotinamide adenine dinucleotide. Figure 40.4, Mechanisms of brain damage in experimental pneumococcal meningitis. NF-κB, a transcriptional activator of many genes involved in the pathogenesis of bacterial meningitis, encodes host factors including proinflammatory cytokines, chemokines (e.g., interleukin [IL]-8), and adhesion molecules. The proinflammatory cytokines IL-1β and tumor necrosis factor (TNF)- α are synthesized as inactive precursors that are processed to mature active forms by proteases (caspase 1 [Casp1], also known as IL-1β–converting enzyme, and TNF-α–converting enzyme [TACE]). IL-1β and TNF-α are potent activators of NF-κB. This process can lead to the uncontrolled expression of proinflammatory mediators and the increased expression of adhesion molecules both on the endothelium (e.g., intercellular adhesion molecule [ICAM]-1) and on neutrophils, leading to a subsequent massive influx of leukocytes into the subarachnoid space. Once present, activated leukocytes release a complex variety of potentially cytotoxic agents including oxidants and proteolytic enzymes (e.g., matrix metalloproteinase [MMP]), which may contribute to tissue destruction. Also, peroxynitrite may cause brain damage through a variety of independent mechanisms. The best studied are the attack of polyunsaturated fatty acids, leading to lipid peroxidation, and an alternative pathway that involves oxidant-induced DNA strand breakage and subsequent poly(adenosine diphosphate ribose) polymerase (PARP) activation, initiating an energy-consuming intracellular cycle that ultimately results in cellular energy depletion and cell death. Both mechanisms likely contribute to cell injury during pneumococcal meningitis. ATP, adenosine triphosphate; ECM, extracellular matrix; KC, keratinocyte-derived chemokine; MIP, macrophage inflammatory protein; NAD, nicotinamide adenine dinucleotide.](https://storage.googleapis.com/dl.dentistrykey.com/clinical/AcuteBacterialMeningitisBeyondtheNeonatalPeriod/3_3s20B9780323756082000409.jpg)
Children with bacterial meningitis usually have 1 of 3 patterns of illness: (1) nonspecific symptoms progressive over 2–5 days; (2) rapidly progressive symptoms and signs over 1–2 days; and (3) fulminant , rapid deterioration and shock early in the course of illness. The rapidly progressive form is frequently associated with severe cerebral edema. Suspected bacterial meningitis is a medical emergency, and immediate diagnostic steps must be taken to establish the specific cause. A careful history, physical examination, blood tests, and lumbar puncture (LP) should be performed before the initiation of therapy for meningitis. Contraindications to performing LP are uncommon. Examples are shown in Table 40.4 . In fulminant cases with hypotension and end-organ failure, rapid intervention is particularly necessary; administration of antibiotics may precede complete history, examination, and LP. In such cases, blood culture should be obtained before administration of antibiotics, and LP performed as soon as is feasible.
| Signs suggesting raised intracranial pressure |
|
| Shock | Defer until stabilization |
| Extensive or spreading purpura | Defer until shock and coagulopathy controlled |
| After convulsions | Defer until airway and breathing are stabilized |
| Coagulation abnormalities | Defer if results of coagulation studies are outside the normal range, platelet count is <100,000/μL, or patient is receiving anticoagulant therapy |
| Local superficial infection at the lumbar puncture site | Defer |
| Respiratory insufficiency, severe respiratory distress (nonintubated) | Contraindicated because of high risk for precipitating respiratory failure |
Recognizing the uncommon infant or child with meningitis or other invasive bacterial infection among the large numbers of children evaluated for febrile illnesses requires expertise and a high index of suspicion, careful elicitation of history, targeted observation, and examination.
Important aspects of the history in the child with suspected bacterial meningitis:
The course of illness.
The presence of symptoms consistent with meningeal inflammation.
The presence of seizures. Focal seizures have important prognostic implications.
The presence of predisposing factors ( Tables 40.1–40.3 ).
History of immunization (particularly Hib conjugate vaccine, PCV, and MCV). Receipt of a full series of any/all of these vaccines does not alter the need for CSF examination or initial empiric therapy, but depending upon age, may affect the need for chemoprophylaxis or evaluation of the immune system.
History of drug allergies, particularly anaphylactic reactions to antibiotics, which, if present, may affect the choice of antimicrobial therapy.
Recent receipt of antibiotics, which may affect the yield of blood and/or CSF culture.
The symptoms and signs depend, to some extent, upon the duration of illness, the host response to infection, and the age of the patient. The triad of fever, neck stiffness, and mental status changes is present in only 44% of adults with bacterial meningitis and in even fewer children. Nonspecific findings include abnormal vital signs such as tachycardia and fever, poor feeding, irritability, lethargy, and vomiting. An infant may become progressively more irritable and lethargic, refuse feeds, and manifest increasingly less interaction with caregivers. The fontanel of an infant may be bulging, but this sign is neither highly sensitive nor specific for meningitis; the finding requires urgent evaluation. The infant who demonstrates diminished activity or little interest in the environment requires urgent evaluation and intervention and may show little interest in the environment. An older child may complain of headache, vomiting, back pain, myalgia, and photophobia; may be confused or disoriented; and may verbalize specifically that the neck is stiff or sore.
Seizures occur in up to 30% of patients before or soon after hospital admission and tend to occur more frequently in pneumococcal or Hib meningitis than in meningococcal disease. Complex seizures at the time of presentation increase the likelihood of meningitis more than twofold. Focal neurologic signs or a reduced level of consciousness may be present. Coma on presentation, compared with irritability or lethargy alone, portends a worse prognosis. Kernig and Brudzinski signs and neck stiffness are more specific physical findings of meningeal inflammation and occur more commonly in children older than 12–18 months. The presence of meningeal signs increases the likelihood of meningitis, and absence decreases the likelihood. No classical symptoms and signs, however, distinguish accurately children with or without meningitis. 1848
Cranial nerve VI palsy and, less commonly, cranial nerve III or VII dysfunction suggest increased intracranial pressure (ICP). Papilledema is uncommon in a child with uncomplicated meningitis and, if present, suggests another diagnosis (brain abscess, epidural or subdural empyema, another cause of increased ICP) or a complication of meningitis (venous sinus thrombosis). Unilateral weakness (hemiparesis) often is a result of ischemia or infarction, usually associated with vasculitis or cerebral artery spasm. Occasionally in an older child, ataxia is the major neurologic sign.
Although meningeal signs are present at the time of admission in most patients, they are not invariably present. In one review of 1064 cases of acute bacterial meningitis in children older than 1 month, 16 (1.5%) had no meningeal signs during their entire period of hospitalization. Nuchal rigidity may not be elicitable in comatose patients or those with focal or diffuse neurologic deficits. In addition, nuchal rigidity can occur late in the course, particularly in young children. Generally, in children with bacterial meningitis, signs of meningeal irritation are expected in 60%–80% at the time of presentation and in approximately 25% of children with normal CSF findings. ,
Nuchal rigidity is manifest by the inability to place the chin on the chest, limitation of passive neck flexion, and Kernig and Brudzinski signs. Kernig sign is present if the patient, in the supine position with the hip and knee flexed at 90°, cannot extend the knee more than 135° and/or there is flexion of the opposite knee. Brudzinski sign is present if the patient, while in the supine position, flexes the lower extremities during attempted passive flexion of the neck.
A petechial or purpuric rash and shock classically are associated with meningococcal septicemia (frequently coexisting with meningitis) but also can occur occasionally in infection due to Hib or S. pneumoniae . A maculopapular rash, which is difficult to distinguish from a common enteroviral exanthem, can occur in up to 15% of children with meningococcemia. Focal infections such as pneumonia, pyogenic arthritis, buccal cellulitis, pericarditis, and endophthalmitis can be present concurrently with bacterial meningitis; their presence should not discourage evaluation of CSF unless the severity of the condition warrants deferral of the LP.
The differential diagnosis in children with fever and alteration in CNS function includes other infections, such as viral and fungal meningitis, viral and protozoal encephalitis, focal brain or parameningeal abscess, Lyme disease, and rickettsial infection. Noninfectious conditions also are considered, such as Kawasaki disease, autoimmune and collagen vascular disease, other vasculitides, acute disseminated encephalomyelitis, hypersensitivity to drugs (e.g., trimethoprim-sulfamethoxazole, immune globulin intravenous, antithymocyte globulin), and Reye syndrome. Differentiation of these disorders from bacterial meningitis frequently is complex, requiring specialized testing and neuroimaging.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here