Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Dengue fever is a benign syndrome caused by several arthropod-borne viruses and is characterized by biphasic fever, myalgia or arthralgia, rash, leukopenia, and lymphadenopathy. Dengue hemorrhagic fever (Philippine, Thai, or Singapore hemorrhagic fever; hemorrhagic dengue; acute infectious thrombocytopenic purpura) is a severe, often fatal, febrile disease caused by one of four dengue viruses. It is characterized by capillary permeability, abnormalities of hemostasis, and, in severe cases, a protein-losing shock syndrome (dengue shock syndrome), which is thought to have an immunopathologic basis.
A revised case definition adopted by the World Health Organization (WHO) in 2009 includes as severe dengue those cases accompanied by fluid loss leading to shock, fluid loss with respiratory distress, liver damage evidenced by elevations of ALT or AST to > 1000 U/L, severe bleeding, and altered consciousness or significant heart abnormalities.
There are at least four distinct antigenic types of dengue virus (dengue 1, 2, 3, and 4), members of the family Flaviviridae. In addition, three other arthropod-borne viruses (arboviruses) cause similar dengue fever syndromes with rash ( Table 295.1 ; see also Chapter 294 ).
| VIRUS | GEOGRAPHIC GENUS AND DISEASE | VECTOR | DISTRIBUTION |
|---|---|---|---|
| Togavirus | Chikungunya | Aedes aegypti | Africa, India, Southeast Asia, Latin America, United States |
| Aedes africanus | |||
| Aedes albopictus | |||
| Togavirus | O'nyong-nyong | Anopheles funestus | East Africa |
| Flavivirus | West Nile fever | Culex molestus | Europe, Africa, Middle East, India |
| Culex univittatus |
Dengue viruses are transmitted by mosquitoes of the Stegomyia family. Aedes aegypti, a daytime biting mosquito, is the principal vector, and all four virus types have been recovered from it. Transmission occurs from viremic humans by bite of the vector mosquito where virus multiplies during an extrinsic incubation period and then by bite is passed on to a susceptible human in what is called the urban transmission cycle. In most tropical areas, A. aegypti is highly urbanized, breeding in water stored for drinking or bathing and in rainwater collected in any container. Dengue viruses have also been recovered from Aedes albopictus, as in the 2001 and 2015 Hawaiian epidemics, whereas outbreaks in the Pacific area have been attributed to several other Aedes species. These species breed in water trapped in vegetation. In Southeast Asia and West Africa, dengue virus may be maintained in a cycle involving canopy-feeding jungle monkeys and Aedes species, which feed on monkeys.
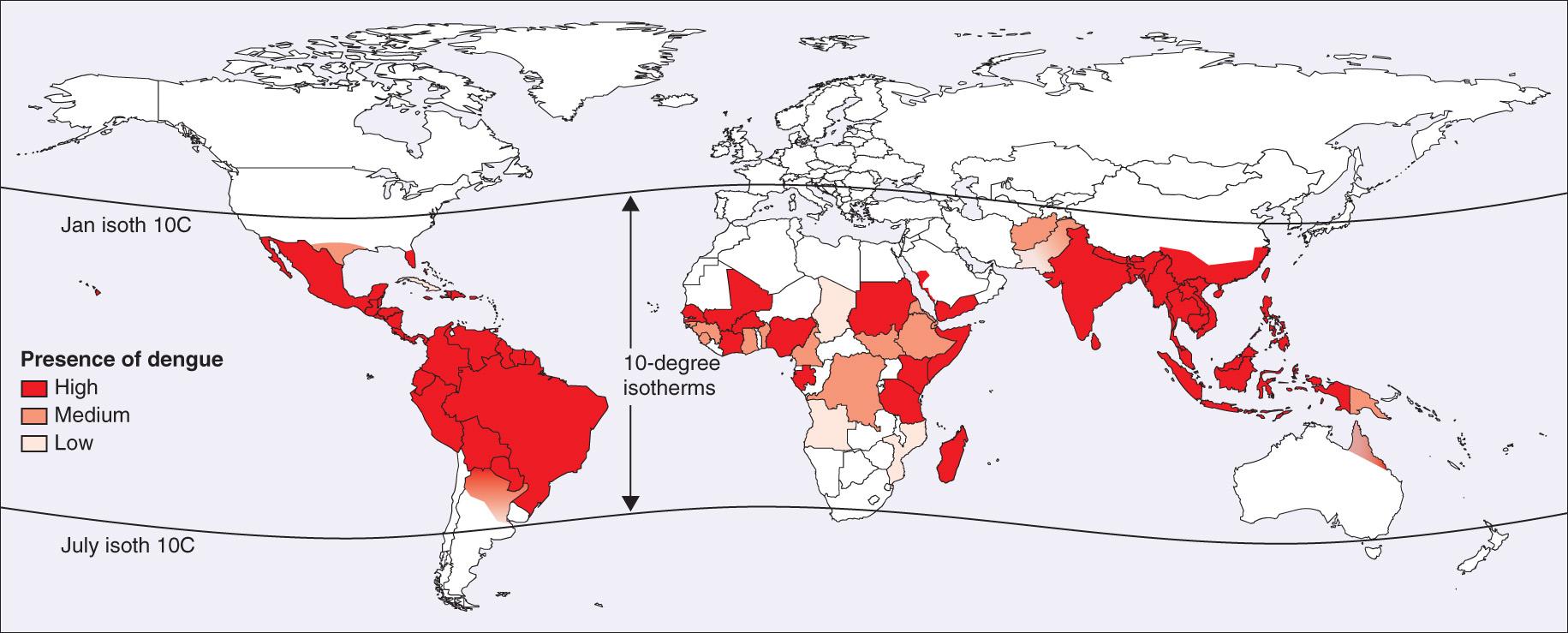
In the 19th and 20th centuries, epidemics were common in temperate areas of the Americas, Europe, Australia, and Asia. Dengue fever and dengue-like disease are now endemic in tropical Asia, the South Pacific Islands, northern Australia, tropical Africa, the Arabian Peninsula, the Caribbean, and Central and South America ( Fig. 295.1 ). Dengue fever occurs frequently among travelers to these areas. Locally acquired disease has been reported in Florida, Arizona, and Texas, and imported cases in the United States occur in travelers to endemic areas. More than 390 million dengue infections occur annually; approximately 96 million have clinical disease.
Dengue outbreaks in urban areas infested with A. aegypti may be explosive; in virgin soil epidemics, up to 70–80% of the population may be involved. Most overt disease occurs in older children and adults. Because A. aegypti has a limited flight range, spread of an epidemic occurs mainly through viremic human beings and follows the main lines of transportation. Sentinel cases may infect household mosquitoes; a large number of nearly simultaneous secondary infections give the appearance of a contagious disease. Where dengue is highly endemic, children and susceptible foreigners may be the only persons to acquire overt disease, because adults have become immune.
Dengue-like diseases may occur in epidemics. Epidemiologic features depend on the vectors and their geographic distribution (see Chapter 294 ). Chikungunya virus is enzootic in subhuman primates throughout much of West, Central, and South Africa. Periodic introductions of virus into the urban transmission cycle have led to pandemics, resulting in widespread endemicity in the most populous areas of Asia. In Asia, A. aegypti is the principal vector; in Africa, other Stegomyia species may be important vectors. In Southeast Asia, dengue and chikungunya outbreaks occur concurrently in the urban cycle. Outbreaks of o'nyong-nyong fever usually involve villages or small towns, in contrast to the urban outbreaks of dengue and chikungunya. West Nile virus is enzootic in Africa. Chikungunya is now endemic in urban cycles in tropical countries throughout the world. Intense transmission in Caribbean and Central and South American countries beginning in 2013 results in the emergence of limited chikungunya transmission in the United States.
Dengue hemorrhagic fever occurs where multiple types of dengue virus are simultaneously or sequentially transmitted. It is endemic in tropical America, Asia, the Pacific Islands, and parts of Africa, where warm temperatures and the practices of water storage in homes plus outdoor breeding sites result in large, permanent populations of A. aegypti. Under these conditions, infections with dengue viruses of all types are common. A first infection, referred to as a primary infection, may be followed by infection with a different dengue virus, referred to as a secondary infection. In areas of high endemicity, secondary infections are frequent.
Secondary dengue infections are relatively mild in the majority of instances, ranging from an inapparent infection through an undifferentiated upper respiratory tract or dengue-like disease, but may also progress to dengue hemorrhagic fever. Nonimmune foreigners, both adults and children, who are exposed to dengue virus during outbreaks of hemorrhagic fever have classic dengue fever or even milder disease. The differences in clinical manifestations of dengue infections between natives and foreigners in Southeast Asia are related to immunologic status. Dengue hemorrhagic fever can occur during primary dengue infections, most frequently in infants whose mothers are immune to dengue. Dengue hemorrhagic fever or severe dengue occurs rarely in individuals of African ancestry because of an as yet incompletely described resistance gene that is consistent with the low incidence of severe dengue throughout much of Africa and among African populations in the American tropics despite high rates of dengue infection.
The pathogenesis of dengue hemorrhagic fever is incompletely understood, but epidemiologic studies usually associate this syndrome with second heterotypic infections with dengue types 1-4 or in infants born to mothers who have had two or more lifetime dengue infections. Retrospective studies of sera from human mothers whose infants acquired dengue hemorrhagic fever and prospective studies in children acquiring sequential dengue infections have shown that the circulation of infection-enhancing antibodies at the time of infection is the strongest risk factor for development of severe disease. The absence of cross-reactive neutralizing antibodies and presence of enhancing antibodies from passive transfer or active production are the best correlates of risk for dengue hemorrhagic fever. Monkeys that are infected sequentially or are receiving small quantities of enhancing antibodies have enhanced viremias. In humans studied early during the course of secondary dengue infections, viremia levels directly predicted disease severity. When dengue virus immune complexes attach to monocyte/macrophage Fc receptors, a signal is sent that suppresses innate immunity, resulting in enhanced viral production. In the Americas, dengue hemorrhagic fever and dengue shock syndrome have been associated with dengue types 1-4 strains of recent Southeast Asian origin. Outbreaks of dengue hemorrhagic fever in all areas of the world are correlated with secondary dengue infections while recent outbreaks in India, Pakistan, and Bangladesh are related to imported dengue strains.
Early in the acute stage of secondary dengue infections, there is rapid activation of the complement system. Shortly before or during shock, blood levels of soluble tumor necrosis factor receptor, interferon-γ, and interleukin-2 are elevated. C1q, C3, C4, C5-C8, and C3 proactivators are depressed, and C3 catabolic rates are elevated. Circulating viral nonstructural protein 1 (NS1) is a viral toxin that activates myeloid cells to release cytokines by attaching to toll receptor 4. It also contributes to increased vascular permeability by activating complement, interacting with and damaging endothelial cells, and interacting with blood clotting factors and platelets. The mechanism of bleeding in dengue hemorrhagic fever is not known, but a mild degree of disseminated intravascular coagulopathy, liver damage, and thrombocytopenia may operate synergistically. Capillary damage allows fluid, electrolytes, small proteins, and, in some instances, red blood cells to leak into extravascular spaces. This internal redistribution of fluid, together with deficits caused by fasting, thirsting, and vomiting, results in hemoconcentration, hypovolemia, increased cardiac work, tissue hypoxia, metabolic acidosis, and hyponatremia.
Usually no pathologic lesions are found to account for death. In rare instances, death may be a result of gastrointestinal or intracranial hemorrhages. Minimal to moderate hemorrhages are seen in the upper gastrointestinal tract, and petechial hemorrhages are common in the interventricular septum of the heart, on the pericardium, and on the subserosal surfaces of major viscera. Focal hemorrhages are occasionally seen in the lungs, liver, adrenals, and subarachnoid space. The liver is usually enlarged, often with fatty changes. Yellow, watery, and at times blood-tinged effusions are present in serous cavities in approximately 75% of patients at autopsy.
Dengue virus is frequently absent in tissues at the time of death; viral antigens or RNA have been localized to hepatocytes and macrophages in the liver, spleen, lung, and lymphatic tissues.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here