Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The arthropod-borne viral infections are a group of mosquito- or tick-transmitted pathogens of several taxa manifested clinically mostly as neurologic infections, influenza-like illnesses, or acute viral exanthems. In temperate countries, arboviruses are transmitted during warmer weather; however, in tropical and subtropical countries, arboviruses may be transmitted year around either in an urban cycle (human to mosquito to human) or by arthropods that feed on other vertebrate species and then feed on humans.
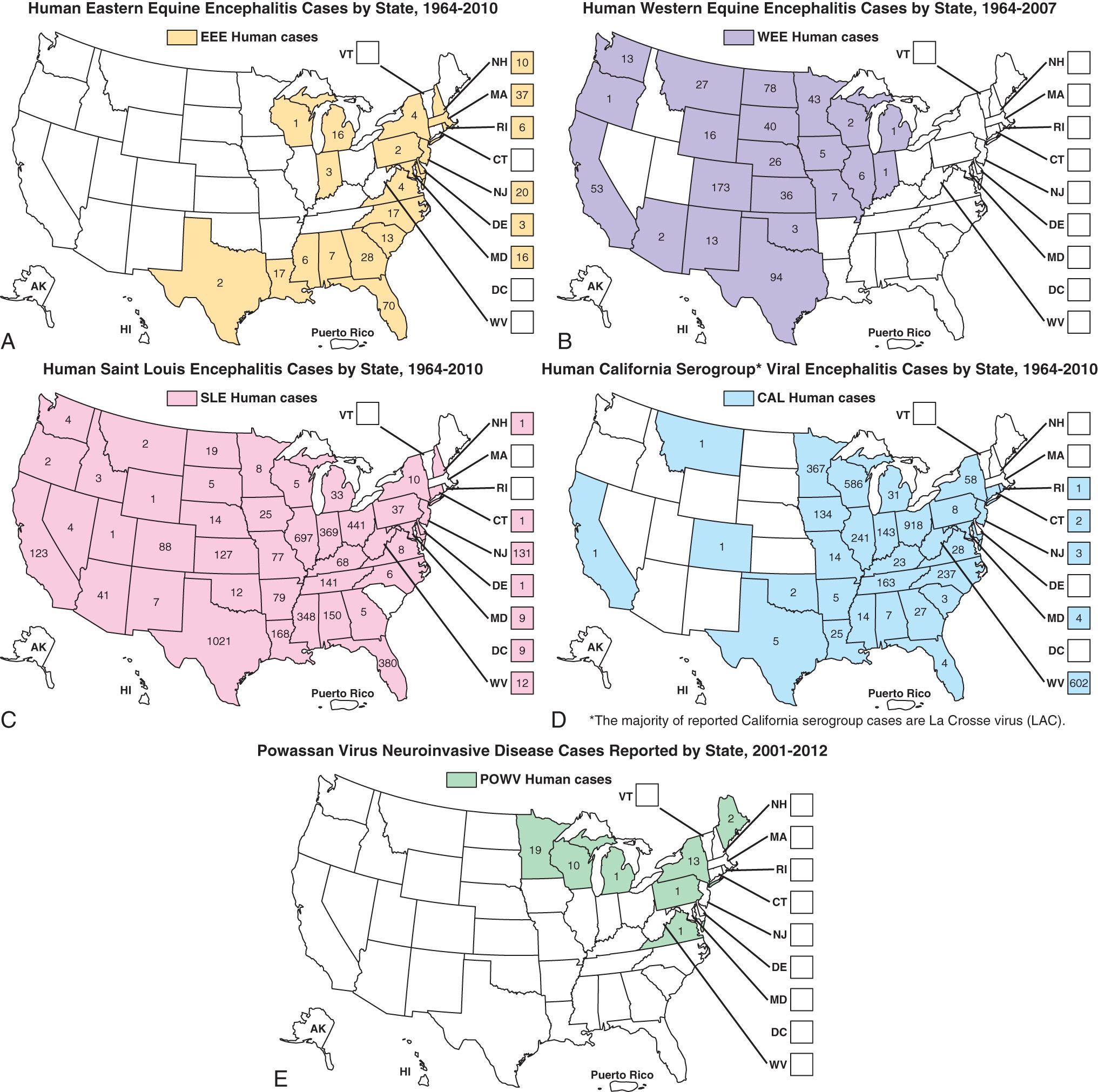
The principal arthropod-borne viral infections in North America are West Nile encephalitis (WNE), St. Louis encephalitis (StLE), Powassan (POW) encephalitis, a complex of California encephalitis group viruses, and, less frequently, western equine encephalitis (WEE), eastern equine encephalitis (EEE), and Colorado tick fever ( Fig. 294.1 ). In 2013, chikungunya virus (CHIK) emerged from its original African zoonosis via Asia into the Western Hemisphere, exposing many residents of the United States who were traveling in the region. A few cases occurred domestically in southern states. In 2015, Zika virus (ZIKV), a flavivirus also maintained in Africa zoonoses, was introduced into the Americas, again from endemic areas in Asia. Limited transmission occurred within the continental United States. The major source of infection among Americans for each of these viruses has been travel to tropical and subtropical countries.
Throughout the world outside North America, there are many arboviruses that pose major health problems ( Fig. 294.2 ). In descending order, these are the dengue viruses (DENV; Chapter 295 ), transmitted in all subtropical and tropical countries; Japanese encephalitis (JE), transmitted in northern, southern, and Southeast Asia; tick-borne encephalitis (TBE), transmitted across Europe and into northern and eastern Asia; yellow fever (YF; Chapter 296 ), transmitted from zoonotic cycles in Africa and South America; and Venezuelan equine encephalitis (VEE), transmitted in parts of South and Central America.
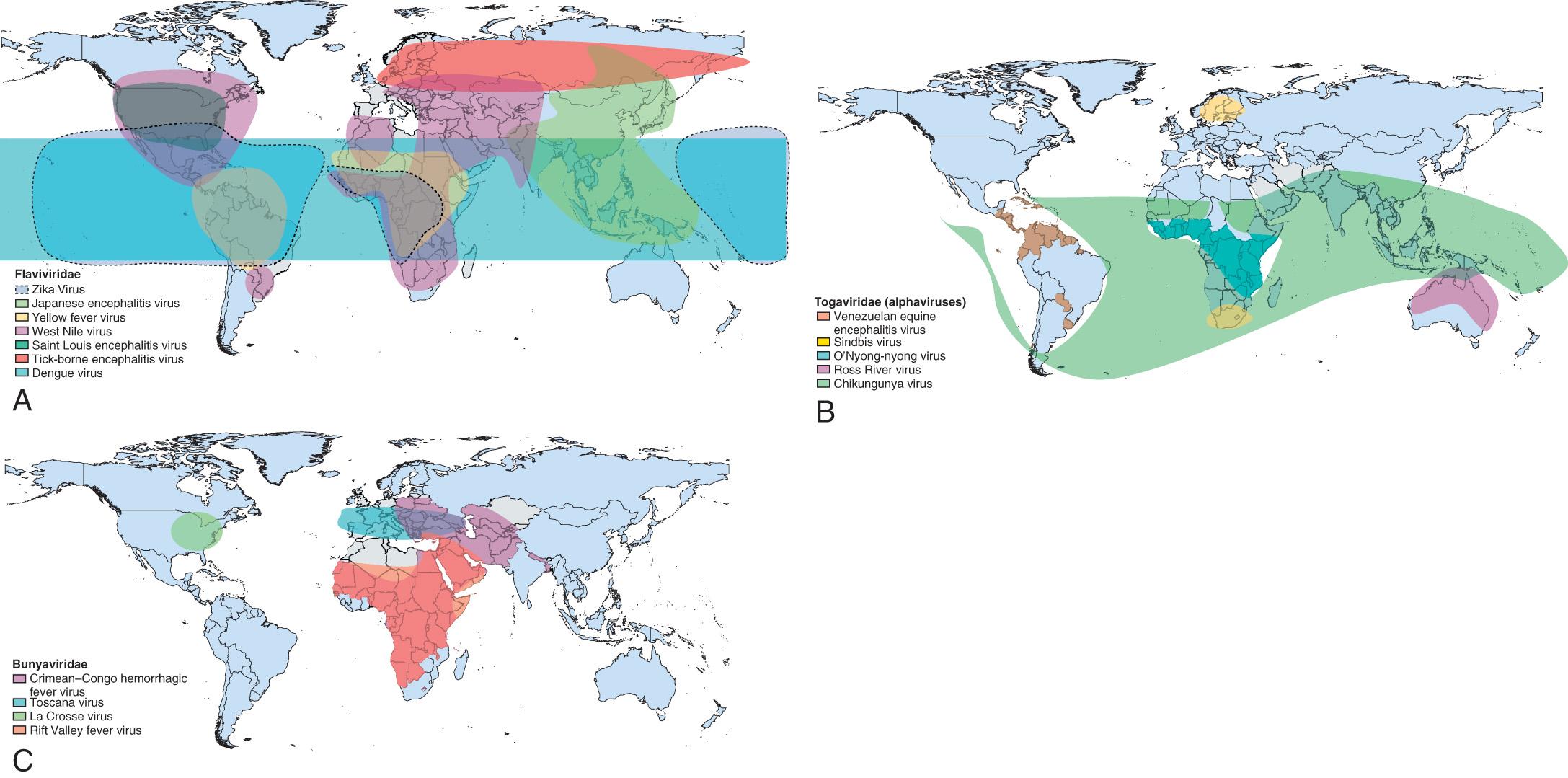
The etiologic agents belong to different viral taxa: alphaviruses of the family Togaviridae (CHIK, EEE, VEE, WEE), flaviviruses of the family Flaviviridae (DENV, JE, POW, STLE, TBE, WNE, YF, ZIKV), the California complex of the family Bunyaviridae (California encephalitis), and Reoviridae (Colorado tick fever virus). Alphaviruses are 69 nm, enveloped, positive-sense RNA viruses. Studies suggest that this group of viruses had a marine origin (specifically the southern ocean) and that they have subsequently spread to both the Old and New Worlds. VEE circulates in nature in six subtypes. Virus types I and III have multiple antigenic variants. Types IAB and IC have caused epizootics and human epidemics. Flaviviruses are 40- to 50-nm, enveloped, positive-sense RNA viruses that evolved from a common ancestor. They are mosquito-borne (WNE, STLE, JE, YF, DENV, ZIKV) and tick-borne (POW, TBE) agents, globally distributed, and responsible for many important human viral diseases. The California serogroup, 1 of 16 Bunyavirus groups, are 75- to 115-nm enveloped viruses possessing a three-segment, negative-sense RNA genome. Reoviruses are 60- to 80-nm double-stranded RNA viruses.
For arboviral infections not described separately, the etiologic diagnosis is established by testing either an acute-phase serum to detect the virus, viral antigen, or viral RNA (influenza-like illnesses or viral exanthems) or by recovery of virus from CNS tissue or CSF. More commonly, the diagnosis is established serologically. Serum obtained ≥ 5 days after the onset of illness is tested for the presence of virus-specific immunoglobulin (Ig) M antibodies using an enzyme-linked immunosorbent assay IgM capture test, an indirect immunofluorescence test, or a precipitin test. Alternatively, acute and convalescent sera can be tested for a four-fold or greater increase in enzyme-linked immunosorbent assay, hemagglutination inhibition, or neutralizing antibody titers. Commercial serologic diagnostic kits are marketed for DENV, CHIK, JE, TBE, WN, YF, or ZIKV viral infections. The serum and CSF should be tested for JE or WN virus–specific IgM. However, IgM may reflect past infection, because it may be present up to 12 mo after infection. For suspected flavivirus infections, including Zika virus, it may be possible to establish infection using a serologic test, calling upon the specificity of neutralizing antibodies. The most common of these is the plaque or focus-reduction neutralizing antibody test. Reference laboratories offer tests for all of the pathogenic flaviviruses. The diagnosis may also be established by the isolation of virus in cell cultures, by identification of viral RNA, or by detection of viral proteins (e.g., dengue NS1) from blood, brain tissue obtained by brain biopsy, or tissues obtained at autopsy.
Several vaccines for Japanese encephalitis and tick-borne encephalitis are licensed in endemic and nonendemic countries. An experimental vaccine for VEE is available to protect laboratory workers. Travelers who plan to be in rural areas of Asia during the expected period of seasonal transmission should receive JE vaccine. Similarly, travelers who plan to travel, camp, or picnic in rural areas of Europe and East Asia should consult local health authorities concerning the need to be vaccinated against TBE. An inactivated vaccine manufactured in Japan by intracerebral injection of young mice and available throughout the world has been taken off the market owing to a high incidence of adverse events. In 2008-2009, tissue culture–based JE vaccine (Ixiaro) was licensed in Europe, Australia, and the United States. In the United States, this vaccine is licensed for use in children and adults and is distributed by Novartis (Basel). This vaccine is administered intramuscularly as two doses of 0.5 mL each, 28 days apart. The final dose should be completed at least 1 wk prior to the patient's expected arrival in a JE endemic area. This vaccine contains alum and protamine sulfate and has exhibited only mild adverse events. A highly efficacious live-attenuated single-dose JE vaccine developed in China for children is licensed and marketed in Asian countries. This vaccine can be coadministered with live-attenuated measles vaccine without altering the immune responses to either vaccine. In humans, prior dengue virus infection provides partial protection from clinical JE.
No TBE vaccines are licensed or available in the United States. Two inactivated cell culture–derived TBE vaccines are available in Europe, in adult and pediatric formulations: FSME-IMMUN (Baxter, Austria) and Encepur (Novartis, Germany). The adult formulation of FSME-IMMUN is also licensed in Canada. Two other inactivated TBE vaccines are available in Russia: TBE-Moscow (Chumakov Institute, Russia) and EnceVir (Microgen, Russia). Immunogenicity studies suggest that the European and Russian vaccines should provide cross-protection against all three TBE virus subtypes. For both FSME-IMMUN and Encepur, the primary vaccination series consists of three doses. The specific recommended intervals between doses vary by country and vaccine. Because the routine primary vaccination series requires ≥ 6 mo for completion, most travelers to TBE-endemic areas will find avoiding tick bites to be more practical than vaccination.
For all viral diseases discussed in this chapter, personal measures should be taken to reduce exposure to mosquito or tick bites, especially for short-term residents in endemic areas. These measures include avoiding evening outdoor exposure, using insect repellents, covering the body with clothing, and using bed nets or house screening. Commercial pesticides, widely used by rice farmers, may be useful in reducing populations of vector mosquitoes or ticks . Fenthion, fenitrothion, and phenthoate are effectively adulticidal and larvicidal. Insecticides may be applied from portable sprayers or from helicopters or light aircraft.
In the United States, EEE is a disease with a very low incidence, with a median of eight cases occurring annually in the Atlantic and Gulf states from 1964 to 2007 ( Fig. 294.1 ). Transmission occurs often in focal endemic areas of the coast of Massachusetts, the six southern counties of New Jersey, and northeastern Florida. In North America, the virus is maintained in freshwater swamps in a zoonotic cycle involving Culiseta melanura and birds. Various other mosquito species obtain viremic meals from birds and transmit the virus to horses and humans. Virus activity varies markedly from year to year in response to still unknown ecologic factors. Most infections in birds are silent, but infections in pheasants are often fatal, and epizootics in these species are used as sentinels for periods of increased viral activity. Cases have been recognized on Caribbean islands. The case:infection ratio is lowest in children (1 : 8) and somewhat higher in adults (1 : 29).
EEE virus infections result in fulminant encephalitis with a rapid progression to coma and death in one third of cases. In infants and children, abrupt onset of fever, irritability, and headache are followed by lethargy, confusion, seizures, and coma. High temperature, bulging fontanel, stiff neck, and generalized flaccid or spastic paralysis are observed. There may be a brief prodrome of fever, headache, and dizziness. Unlike most other viral encephalitides, the peripheral white blood cell count usually demonstrates a marked leukocytosis, and the cerebrospinal fluid (CSF) may show marked pleocytosis. Pathologic changes are found in the cortical and gray matter, with viral antigens localized to neurons. There is necrosis of neurons, neutrophilic infiltration, and perivascular cuffing by lymphocytes.
The prognosis in EEE is better for patients with a prolonged prodrome; the occurrence of convulsions conveys a poor prognosis. Patient fatality rates are 33–75% and are highest in the elderly. Residual neurologic defects are common, especially in children.
The diagnosis of encephalitis may be aided by CT or MRI and by electroencephalography. Focal seizures or focal findings on CT or MRI or electroencephalography should suggest the possibility of herpes simplex encephalitis, which should be treated with acyclovir (see Chapter 279 ).
WEE infections occur principally in the United States and Canada west of the Mississippi River (see Fig. 294.1 ), mainly in rural areas where water impoundments, irrigated farmland, and naturally flooded land provide breeding sites for Culex tarsalis. The virus is transmitted in a cycle involving mosquitoes, birds, and other vertebrate hosts. Humans and horses are susceptible to encephalitis. The case:infection ratio varies by age, having been estimated at 1 : 58 in children younger than 4 yr of age and 1 : 1,150 in adults. Infections are most severe at the extremes of life; one third of cases occur in children younger than 1 yr of age. Recurrent human epidemics have been reported from the Yakima Valley in Washington State and the Central Valley of California; the largest outbreak on record resulted in 3,400 cases and occurred in Minnesota, North and South Dakota, Nebraska, and Montana, as well as Alberta, Manitoba, and Saskatchewan, Canada. Epizootics in horses precede human epidemics by several weeks. For the past 20 yr, only three cases of WEE have been reported, presumably reflecting successful mosquito abatement.
In WEE, there may be a prodrome with symptoms of an upper respiratory tract infection. The onset is usually sudden with chills, fever, dizziness, drowsiness, increasing headache, malaise, nausea and vomiting, stiff neck, and disorientation. Infants typically present with the sudden cessation of feeding, fussiness, fever, and protracted vomiting. Convulsions and lethargy develop rapidly. On physical examination, patients are somnolent, exhibit meningeal signs, and have generalized motor weakness and reduced deep tendon reflexes. In infants, a bulging fontanel, spastic paralysis, and generalized convulsions may be observed. On pathologic examination, disseminated small focal abscesses, small focal hemorrhages, and patchy areas of demyelination are distinctive.
Patient fatality rates in WEE are 3–9% and are highest in the elderly. Major neurologic sequelae have been reported in up to 13% of cases and may be as high as 30% in infants. Parkinsonian syndrome has been reported as a residual in adult survivors.
Cases of STLE are reported from nearly all states; the highest attack rates occur in the gulf and central states (see Fig. 294.1 ). Epidemics frequently occur in urban and suburban areas; the largest, in 1975, involved 1,800 persons living in Houston, Chicago, Memphis, and Denver. Cases often cluster in areas where there are ground water or septic systems, which support mosquito breeding. The principal vectors are Culex pipiens and Culex quinquefasciatus in the central gulf states, Culex nigripalpus in Florida, and C. tarsalis in California. STLE virus is maintained in nature in a bird–mosquito cycle. Viral amplification occurs in bird species abundant in residential areas (e.g., sparrows, blue jays, and doves). Virus is transmitted in the late summer and early fall. The case:infection ratio may be as high as 1 : 300. Age-specific attack rates are lowest in children and highest in individuals older than age 60 yr. The most recent small outbreaks were in Florida in 1990 and Louisiana in 2001. For the past 15 yr, there have been a mean of 18 cases annually.
Clinical manifestations of STLE vary from a mild flulike illness to fatal encephalitis. There may be a prodrome of nonspecific symptoms with subtle changes in coordination or mentation of several days to 1 wk in duration. Early signs and symptoms include fever, photophobia, headache, malaise, nausea, vomiting, and neck stiffness. About half of patients exhibit an abrupt onset of weakness, incoordination, disturbed sensorium, restlessness, confusion, lethargy, and delirium or coma. The peripheral white blood cell count is modestly elevated, with 100-200 cells/µL found in the CSF. On autopsy, the brain shows scattered foci of neuronal damage and perivascular inflammation.
The principal risk factor for fatal outcome of STLE is advanced age, with patient fatality rates being as high as 80% in early outbreaks. In children, mortality rates are 2–5%. In adults, underlying hypertensive cardiovascular disease has been a risk factor for fatal outcome. Recovery from STLE is usually complete, but the rate of serious neurologic sequelae has been reported to be as high as 10% in children.
West Nile (WN) virus was imported into the United States in 1999 and survives in a broad enzootic cycle across the United States and Canada. Every state in the continental United States plus nine provinces in Canada have reported mosquito, bird, mammalian, or human WN virus infection, most frequently during the summer or fall months. Through the end of 2015, 43,937 total cases had been reported in the United States, 40–50% of which were neuroinvasive, with 1,911 deaths ( Fig. 294.3 ). WN virus transmission cycles appear to resemble those of Japanese encephalitis with large epizootics and human cases every 5-10 yr. WN virus has entered the blood supply through asymptomatic viremic potential blood donors. Since 2003, blood banks screen for WN virus RNA. During the major outbreak of 2012, 597 viremic potential blood donors were identified and the donation was rejected. WN virus has also been transmitted to humans via the placenta, breast milk, and organ transplantation. Throughout its range, the virus is maintained in nature by transmission between mosquitoes of the Culex genus and various species of birds. In the United States, human infections are largely acquired from C. pipiens. Horses are the nonavian vertebrates most likely to exhibit disease with WN virus infection. During the 2002 transmission season, 14,000 equine cases were reported, with a mortality rate of 30%. Disease occurs predominantly in individuals >50 yr of age. WN virus has been implicated as the cause of sporadic summertime cases of human encephalitis and meningitis in Israel, India, Pakistan, Romania, Russia, Canada, the United States, and parts of Central and South America. All American WN viruses are genetically similar and are related to a virus recovered from a goose in Israel in 1998.
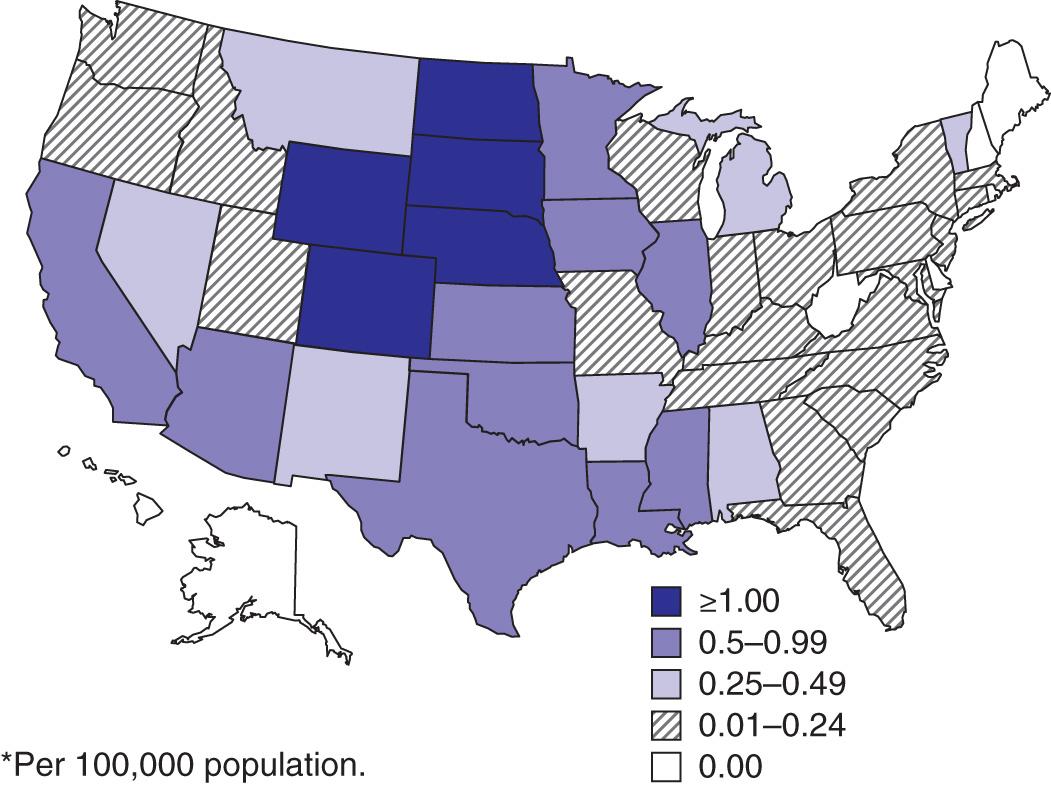
West Nile encephalitis (WNE) may be asymptomatic, but when clinical features appear, they include an abrupt onset of high fever, headache, myalgias, and nonspecific signs of emesis, rash, abdominal pain, or diarrhea. Most infections manifest as a flulike febrile illness, whereas a minority of patients demonstrate meningitis or encephalitis, or both. Rarely there may be cardiac dysrhythmias, myocarditis, rhabdomyolysis, optic neuritis, uveitis, retinitis, orchitis, pancreatitis, or hepatitis. WN virus disease in the United States has been accompanied by prolonged lymphopenia and an acute asymmetric polio-like paralytic illness with CSF pleocytosis involving the anterior horn cells of the spinal cord. A striking but uncommon feature has been parkinsonism and movement disorders (with tremor and myoclonus). WN virus infections have been shown to lead to chronic kidney disease in a small group of patients.
Cases of WNE and deaths due to the disease occur mainly in the elderly, although many serologic surveys show that persons of all ages are infected. In 2015, among a total of 2,175 human cases, 1,455 were neuroinvasive disease, which resulted in 146 deaths, a 10% mortality rate (see Fig. 294.2 ). Paralysis may result in permanent weakness.
POW virus is transmitted by Ixodes cookei among small mammals in eastern Canada and the United States; it has been responsible for 39 deaths in the United States since 2008 (see Fig. 294.1 ). Other ticks may transmit the virus in a wider geographic area, and there is some concern that Ixodes scapularis (also called Ixodes dammini ), a competent vector in the laboratory, may become involved as it becomes more prominent in the United States.
In a limited experience, POW encephalitis has occurred mainly in adults with vocational or recreational exposure and has a high fatality rate.
POW encephalitis has occurred mostly in adults living in enzootic areas with vocational or recreational exposure; it is associated with significant long-term morbidity and has a case-fatality rate of 10–15%.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here