Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Atopic dermatitis (AD) , or eczema, is the most common chronic relapsing skin disease seen in infancy and childhood. It affects 10–30% of children worldwide and frequently occurs in families with other atopic diseases. Infants with AD are predisposed to development of food allergy, allergic rhinitis, and asthma later in childhood, a process called the atopic march .
AD is a complex genetic disorder that results in a defective skin barrier, reduced skin innate immune responses, and polarized adaptive immune responses to environmental allergens and microbes that lead to chronic skin inflammation.
Acute AD skin lesions are characterized by spongiosis , or marked intercellular edema, of the epidermis. In AD, dendritic antigen-presenting cells (APCs) in the epidermis, such as Langerhans cells, exhibit surface-bound IgE molecules with cell processes that reach into upper epidermis to sense allergens and pathogens. These APCs play an important role in cutaneous responses to type 2 immune responses (see Chapter 166 ). There is marked perivenular T-cell and inflammatory monocyte-macrophage infiltration in acute AD lesions. Chronic, lichenified AD is characterized by a hyperplastic epidermis with hyperkeratosis and minimal spongiosis. There are predominantly IgE-bearing Langerhans cells in the epidermis, and macrophages in the dermal mononuclear cell infiltrate. Mast cell and eosinophil numbers are increased, contributing to skin inflammation.
AD is associated with multiple phenotypes and endotypes that have overlapping clinical presentations. Atopic eczema is associated with IgE-mediated sensitization (at onset or during the course of eczema) and occurs in 70–80% of patients with AD. Nonatopic eczema is not associated with IgE-mediated sensitization and is seen in 20–30% of patients with AD. Both forms of AD are associated with eosinophilia. In atopic eczema, circulating T cells expressing the skin homing receptor cutaneous lymphocyte-associated antigen produce increased levels of T-helper type 2 (Th2) cytokines, including interleukin (IL)-4 and IL-13, which induce isotype switching to IgE synthesis. Another cytokine, IL-5, plays an important role in eosinophil development and survival. Nonatopic eczema is associated with lower IL-4 and IL-13 but increased IL-17 and IL-23 production than in atopic eczema. Age and race have also been found to affect the immune profile in AD.
Compared with the skin of healthy individuals, both unaffected skin and acute skin lesions of patients with AD have an increased number of cells expressing IL-4 and IL-13. Chronic AD skin lesions, by contrast, have fewer cells that express IL-4 and IL-13, but increased numbers of cells that express IL-5, granulocyte-macrophage colony-stimulating factor, IL-12, and interferon (IFN)-γ than acute AD lesions. Despite increased type 1 and type 17 immune responses in chronic AD, IL-4 and IL-13 as well as other type 2 cytokines (e.g. TSLP, IL-31, IL-33) predominate and reflect increased numbers of Type 2 innate lymphoid cells and Th2 cells. The infiltration of IL-22–expressing T cells correlates with severity of AD, blocks keratinocyte differentiation, and induces epidermal hyperplasia. The importance of IL-4 and IL-13 in driving severe persistent AD has been validated by multiple clinical trials now demonstrating that biologics blocking IL-4 and IL-13 action lead to clinical improvement in moderate to severe AD.
In healthy people the skin acts as a protective barrier against external irritants, moisture loss, and infection. Proper function of the skin depends on adequate moisture and lipid content, functional immune responses, and structural integrity. Severely dry skin is a hallmark of AD. This results from compromise of the epidermal barrier, which leads to excess transepidermal water loss, allergen penetration, and microbial colonization. Filaggrin , a structural protein in the epidermis, and its breakdown products are critical to skin barrier function, including moisturization of the skin. Genetic mutations in the filaggrin gene (FLG) family have been identified in patients with ichthyosis vulgaris (dry skin, palmar hyperlinearity) and in up to 50% of patients with severe AD. FLG mutation is strongly associated with development of food allergy and eczema herpeticum. Nonetheless, up to 60% of carriers of a FLG mutation do not develop atopic diseases. Cytokines found in allergic inflammation, such as IL-4, IL-13, IL-22, IL-25, and tumor necrosis factor, can also reduce filaggrin and other epidermal proteins and lipids. AD patients are at increased risk of bacterial, viral, and fungal infection related to impairment of innate immunity, disturbances in the microbiome, skin epithelial dysfunction, and overexpression of polarized immune pathways, which dampen host antimicrobial responses.
AD typically begins in infancy. Approximately 50% of patients experience symptoms in the 1st yr of life, and an additional 30% are diagnosed between 1 and 5 yr of age. Intense pruritus , especially at night, and cutaneous reactivity are the cardinal features of AD. Scratching and excoriation cause increased skin inflammation that contributes to the development of more pronounced eczematous skin lesions. Foods (cow's milk, egg, peanut, tree nuts, soy, wheat, fish, shellfish), aeroallergens (pollen, grass, animal dander, dust mites), infection ( Staphylococcus aureus , herpes simplex, coxsackievirus, molluscum), reduced humidity, excessive sweating, and irritants (wool, acrylic, soaps, toiletries, fragrances, detergents) can trigger pruritus and scratching.
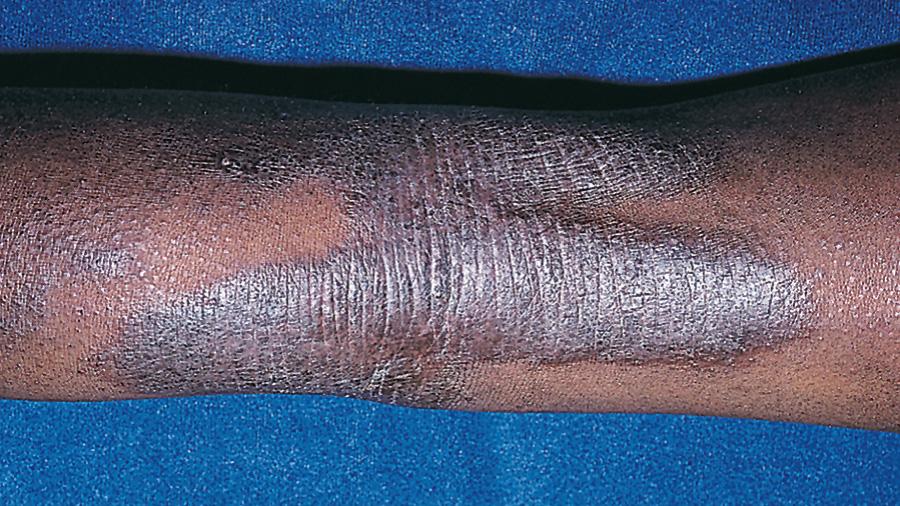
Acute AD skin lesions are intensely pruritic with erythematous papules ( Figs. 170.1 and 170.2 ). Subacute dermatitis manifests as erythematous, excoriated, scaling papules. In contrast, chronic AD is characterized by lichenification ( Fig. 170.3 ), or thickening of the skin with accentuated surface markings, and fibrotic papules . In chronic AD, all 3 types of skin reactions may coexist in the same individual. Most patients with AD have dry, lackluster skin regardless of their stage of illness. Skin reaction pattern and distribution vary with the patient's age and disease activity. AD is generally more acute in infancy and involves the face, scalp, and extensor surfaces of the extremities. The diaper area is usually spared. Older children and children with chronic AD have lichenification and localization of the rash to the flexural folds of the extremities. AD can go into remission as the patient grows older, however, many children with AD have persistent eczema as an adult ( Fig. 170.1 C ).


There are no specific laboratory tests to diagnose AD. Many patients have peripheral blood eosinophilia and increased serum IgE levels. Serum IgE measurement or skin-prick testing can identify the allergens (foods, inhalant/microbial allergens) to which patients are sensitized. The diagnosis of clinical allergy to these allergens requires confirmation by history and environmental challenges.
AD is diagnosed on the basis of 3 major features: pruritus, an eczematous dermatitis that fits into a typical pattern of skin inflammation, and a chronic or chronically relapsing course ( Table 170.1 ). Associated features, such as a family history of asthma, hay fever, elevated IgE, and immediate skin test reactivity, reinforce the diagnosis of AD.
Pruritus
Facial and extensor eczema in infants and children
Flexural eczema in adolescents
Chronic or relapsing dermatitis
Personal or family history of atopic disease
Xerosis
Cutaneous infections ( Staphylococcus aureus, group A streptococcus, herpes simplex, coxsackievirus, vaccinia, molluscum, warts)
Nonspecific dermatitis of the hands or feet
Ichthyosis, palmar hyperlinearity, keratosis pilaris
Nipple eczema
White dermatographism and delayed blanch response
Anterior subcapsular cataracts, keratoconus
Elevated serum IgE levels
Positive results of immediate-type allergy skin tests
Early age at onset
Dennie lines (Dennie-Morgan infraorbital folds)
Facial erythema or pallor
Course influenced by environmental and/or emotional factors
Many inflammatory skin diseases, immunodeficiencies, skin malignancies, genetic disorders, infectious diseases, and infestations share symptoms with AD and should be considered and excluded before a diagnosis of AD is established ( Tables 170.2 and 170.3 ). Severe combined immunodeficiency (see Chapter 152.1 ) should be considered for infants presenting in the first yr of life with diarrhea, failure to thrive, generalized scaling rash, and recurrent cutaneous and/or systemic infection. Histiocytosis should be excluded in any infant with AD and failure to thrive (see Chapter 534 ). Wiskott-Aldrich syndrome, an X-linked recessive disorder associated with thrombocytopenia, immune defects, and recurrent severe bacterial infections, is characterized by a rash almost indistinguishable from that in AD (see Chapter 152.2 ). One of the hyper-IgE syndromes is characterized by markedly elevated serum IgE values, recurrent deep-seated bacterial infections, chronic dermatitis, and refractory dermatophytosis. Many of these patients have disease as a result of autosomal dominant STAT3 mutations. In contrast, some patients with hyper-IgE syndrome present with increased susceptibility to viral infections and an autosomal recessive pattern of disease inheritance. These patients may have a DOCK8 (dedicator of cytokinesis 8 gene) mutation. This diagnosis should be considered in young children with severe eczema, food allergy, and disseminated skin viral infections.
| MAIN AGE GROUP AFFECTED | FREQUENCY * | CHARACTERISTICS AND CLINICAL FEATURES | |
|---|---|---|---|
| OTHER TYPES OF DERMATITIS | |||
| Seborrheic dermatitis | Infants | Common | Salmon-red greasy scaly lesions, often on the scalp (cradle cap) and napkin area; generally presents in the 1st 6 wk of life; typically clears within weeks |
| Seborrheic dermatitis | Adults | Common | Erythematous patches with yellow, white, or grayish scales in seborrheic areas, particularly the scalp, central face, and anterior chest |
| Nummular dermatitis | Children and adults | Common | Coin-shaped scaly patches, mostly on legs and buttocks; usually no itch |
| Irritant contact dermatitis | Children and adults | Common | Acute to chronic eczematous lesions, mostly confined to the site of exposure; history of locally applied irritants is a risk factor; might coexist with AD |
| Allergic contact dermatitis | Children and adults | Common | Eczematous rash with maximum expression at sites of direct exposure but might spread; history of locally applied irritants is a risk factor; might coexist with AD |
| Lichen simplex chronicus | Adults | Uncommon | One or more localized, circumscribed, lichenified plaques that result from repetitive scratching or rubbing because of intense itch |
| Asteatotic eczema | Adults | Common | Scaly, fissured patches of dermatitis overlying dry skin, most often on lower legs |
| INFECTIOUS SKIN DISEASES | |||
| Dermatophyte infection | Children and adults | Common | One or more demarcated scaly plaques with central clearing and slightly raised reddened edge; variable itch |
| Impetigo | Children | Common | Demarcated erythematous patches with blisters or honey-yellow crusting |
| Scabies | Children | Common † | Itchy superficial burrows and pustules on palms and soles, between fingers, and on genitalia; might produce secondary eczematous changes |
| HIV | Children and adults | Uncommon | Seborrhea-like rash |
| CONGENITAL IMMUNODEFICIENCIES (see Table 170.3 ) | |||
| Keratinization Disorders | |||
| Ichthyosis vulgaris | Infants and adults | Uncommon | Dry skin with fine scaling, particularly on the lower abdomen and extensor areas; perifollicular skin roughening; palmar hyperlinearity; full form (i.e., 2 FLG mutations) is uncommon; often coexists with AD |
| NUTRITIONAL DEFICIENCY–METABOLIC DISORDERS | |||
| Zinc deficiency (acrodermatitis enteropathica) | Children | Uncommon | Erythematous scaly patches and plaques, most often around the mouth and anus; rare congenital form accompanied by diarrhea and alopecia |
| Biotin deficiency (nutritional or biotinidase deficiency) | Infants | Uncommon | Scaly periorofacial dermatitis, alopecia, conjunctivitis, lethargy, hypotonia |
| Pellagra (niacin deficiency) | All ages | Uncommon | Scaly crusted epidermis, desquamation, sun-exposed areas, diarrhea |
| Kwashiorkor | Infants and children | Geographic dependent | Flaky scaly dermatitis, swollen limbs with cracked peeling patches |
| Phenylketonuria | Infants | Uncommon | Eczematous rash, hypopigmentation, blonde hair, developmental delay |
| NEOPLASTIC DISEASE | |||
| Cutaneous T-cell lymphoma | Adults | Uncommon | Erythematous pink-brown macules and plaques with a fine scale; poorly responsive to topical corticosteroids; variable itch (in early stages) |
| Langerhans cell histiocytosis | Infants | Uncommon | Scaly and purpuric dermatosis, hepatosplenomegaly, cytopenias |
* Common = approximately 1 in 10 to 1 in 100; uncommon = 1 in 100 to 1 in 1000; rare = 1 in 1000 to 1 in 10,000; very rare = <1 in 10,000.
| DISEASE | GENE | INHERITANCE | CLINICAL FEATURES | LAB ABNORMALITIES |
|---|---|---|---|---|
| AD-HIES | STAT3 | AD, less commonly sporadic | Cold abscesses Recurrent sinopulmonary infections Mucocutaneous candidiasis Coarse facies Minimal trauma fractures Scoliosis Joint hyperextensibility Retained primary teeth Coronary artery tortuosity or dilation Lymphoma |
High IgE (>2000 IU/µL) Eosinophilia |
| DOCK8 deficiency | DOCK8 | AR | Severe mucocutaneous viral infections Mucocutaneous candidiasis Atopic features (asthma, allergies) Squamous cell carcinoma Lymphoma |
High IgE Eosinophilia With or without decreased IgM |
| PGM3 deficiency | PGM3 | AR | Neurologic abnormalities Leukocytoclastic vasculitis Atopic features (asthma, allergies) Sinopulmonary infections Mucocutaneous viral infections |
High IgE Eosinophilia |
| WAS | WASP | XLR | Hepatosplenomegaly Lymphadenopathy Atopic diathesis Autoimmune conditions (especially hemolytic anemia) Lymphoreticular malignancies |
Thrombocytopenia (<80,000/µL) Low mean platelet volume Eosinophilia is common Lymphopenia Low IgM, variable IgG |
| SCID | Variable, depends on type | XLR and AR most common | Recurrent, severe infections Failure to thrive Persistent diarrhea Recalcitrant oral candidiasis Omenn syndrome: lymphadenopathy, hepatosplenomegaly, erythroderma |
Lymphopenia common Variable patterns of reduced lymphocyte subsets (T, B, natural killer cells) Omenn syndrome: high lymphocytes, eosinophilia, high IgE |
| IPEX | FOXP3 | XLR | Severe diarrhea (autoimmune enteropathy) Various autoimmune endocrinopathies (especially diabetes mellitus, thyroiditis) Food allergies |
High IgE Eosinophilia Various autoantibodies |
| Netherton syndrome | SPINK5 | AR | Hair shaft abnormalities Erythroderma Ichthyosis linearis circumflexa Food allergies Recurrent gastroenteritis Neonatal hypernatremic dehydration Upper and lower respiratory infections |
High IgE Eosinophilia |
Adolescents who present with an eczematous dermatitis but no history of childhood eczema, respiratory allergy, or atopic family history may have allergic contact dermatitis (see Chapter 674.1 ). A contact allergen may be the problem in any patient whose AD does not respond to appropriate therapy. Sensitizing chemicals, such as parabens and lanolin, can be irritants for patients with AD and are commonly found as vehicles in therapeutic topical agents. Topical glucocorticoid contact allergy has been reported in patients with chronic dermatitis receiving topical corticosteroid therapy. Eczematous dermatitis has also been reported with HIV infection as well as with a variety of infestations such as scabies. Other conditions that can be confused with AD include psoriasis, ichthyosis, and seborrheic dermatitis.
The treatment of AD requires a systematic, multifaceted approach that incorporates skin moisturization, topical antiinflammatory therapy, identification and elimination of flare factors ( Table 170.4 ), and, if necessary, systemic therapy. Assessment of the severity also helps direct therapy ( Table 170.5 ).
Maintain cool temperature in bedroom, and avoid too many bed covers.
Increase emollient use with cold weather.
Avoid exposure to herpes sores; urgent visit if flare of unusual aspect.
Clothing: Avoid skin contact with irritating fibers (wool, large-fiber textiles).
Do not use tight and too-warm clothing, to avoid excessive sweating.
New, nonirritating clothing designed for AD children is being evaluated.
Tobacco: Avoid exposure.
Vaccines: Normal schedule in noninvolved skin, including egg-allergic patients (see text).
Sun exposure: No specific restriction.
Usually helpful because of improvement of epidermal barrier.
Encourage summer holidays in altitude or at beach resorts.
Physical exercise, sports: no restriction.
If sweating induces flares of AD, progressive adaptation to exercise.
Shower and emollients after swimming pool.
Food allergens:
Maintain breastfeeding exclusively to 4-6 mo if possible.
Consider evaluation for early introduction of allergens (see Chapter 176 ).
Otherwise normal diet, unless an allergy workup has proved the need to exclude a specific food.
Indoor aeroallergens: House dust mites
Use adequate ventilation of housing; keep the rooms well aerated even in winter.
Avoid wall-to-wall carpeting.
Remove dust with a wet sponge.
Vacuum floors and upholstery with an adequately filtered cleaner once a week.
Avoid soft toys in bed (cradle), except washable ones.
Wash bedsheets at a temperature higher than 55°C (131°F) every 10 days.
Use bed and pillow encasings made of Gore-Tex or similar material.
Furred pets: Advise to avoid. If allergy is demonstrated, be firm on avoidance measures, such as pet removal.
Pollen: Close windows during peak pollen season on warm and dry weather days, and restrict, if possible, time outdoors.
Windows may be open at night and early in the morning or during rainy weather.
Avoid exposure to risk situations (lawn mowing).
Use pollen filters in motor vehicles.
Clothes and pets can vectorize aeroallergens, including pollen.
Clear —Normal skin, with no evidence of atopic eczema
Mild —Areas of dry skin, infrequent itching (with or without small areas of redness)
Moderate —Areas of dry skin, frequent itching, redness (with or without excoriation and localized skin thickening)
Severe —Widespread areas of dry skin, incessant itching, redness (with or without excoriation, extensive skin thickening, bleeding, oozing, cracking, and alteration of pigmentation)
Because patients with AD have impaired skin barrier function from reduced filaggrin and skin lipid levels, they present with diffuse, abnormally dry skin, or xerosis . Moisturizers are first-line therapy. Lukewarm soaking baths or showers for 15-20 min followed by the application of an occlusive emollient to retain moisture provide symptomatic relief. Hydrophilic ointments of varying degrees of viscosity can be used according to the patient's preference. Occlusive ointments are sometimes not well tolerated because of interference with the function of the eccrine sweat ducts and may induce the development of folliculitis. In these patients, less occlusive agents should be used. Several prescription (classified as a medical device) “therapeutic moisturizers” or “barrier creams” are available, containing components such as ceramides and filaggrin acid metabolites intended to improve skin barrier function. There are minimal data demonstrating their efficacy over standard emollients.
Hydration by baths or wet dressings promotes transepidermal penetration of topical glucocorticoids. Dressings may also serve as effective barriers against persistent scratching, in turn promoting healing of excoriated lesions. Wet dressings are recommended for use on severely affected or chronically involved areas of dermatitis refractory to skin care. It is critical that wet dressing therapy be followed by topical emollient application to avoid potential drying and fissuring from the therapy. Wet dressing therapy can be complicated by maceration and secondary infection and should be closely monitored by a physician.
Topical corticosteroids are the cornerstone of antiinflammatory treatment for acute exacerbations of AD. Patients should be carefully instructed on their use of topical glucocorticoids to avoid potential adverse effects. There are 7 classes of topical glucocorticoids, ranked according to their potency, as determined by vasoconstrictor assays ( Table 170.6 ). Because of their potential adverse effects, the ultrahigh-potency glucocorticoids should not be used on the face or intertriginous areas and should be used only for very short periods on the trunk and extremities. Mid-potency glucocorticoids can be used for longer periods to treat chronic AD involving the trunk and extremities. Long-term control can be maintained with twice-weekly applications of topical fluticasone or mometasone to areas that have healed but are prone to relapse, once control of AD is achieved after a daily regimen of topical corticosteroids. Compared with creams, ointments have a greater potential to occlude the epidermis, resulting in enhanced systemic absorption.
* Representative corticosteroids are listed by group from 1 (superpotent) through 7 (least potent).
Clobetasol propionate (Temovate) 0.05% ointment/cream
Betamethasone dipropionate (Diprolene) 0.05% ointment/lotion/gel
Fluocinonide (Vanos) 0.1% cream
Mometasone furoate (Elocon) 0.1% ointment
Halcinonide (Halog) 0.1% cream
Fluocinonide (Lidex) 0.05% ointment/cream
Desoximetasone (Topicort) 0.25% ointment/cream
Betamethasone dipropionate (Diprolene) 0.05% cream
Fluticasone propionate (Cutivate) 0.005% ointment
Halcinonide (Halog) 0.1% ointment
Betamethasone valerate (Valisone) 0.1% ointment
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here