Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Esophageal surgery in neonates includes the treatment of esophageal atresia, gastroesophageal reflux, congenital esophageal stenosis, esophageal duplication, and vascular rings.
Esophageal atresia, with or without tracheoesophageal atresia, is the most common congenital anomaly of the esophagus, occurring in 1 in 3500 live births.
The surgical treatment of esophageal atresia has undergone several advances including the introduction of thoracoscopic repair.
Management of long-gap esophageal atresia remains challenging even at high-volume centers.
Results from recent large, multicenter registries brought into question some aspects of traditional surgical management of esophageal atresia and are moving management toward an evidenced-based approach.
Esophageal pathology in the newborn period represents a broad range of disorders that affect feeding, swallowing, and airway protection. These conditions require the multidisciplinary care of neonatologists, pediatric surgeons, aerodigestive specialists, and many others and are just a part of the complex care delivered to infants in modern neonatal intensive care units. A detailed understanding of the surgical considerations of infants with congenital and acquired esophageal pathologies is advantageous to those who are focused on these most fragile patients, because the respiratory and developmental morbidities associated with esophageal pathology can be significant. Herein, we review the principles of surgical management of esophageal pathology in the newborn, including esophageal atresia (EA) and tracheoesophageal fistula (TEF), gastroesophageal reflux disease, and other congenital anomalies, with a focus on the evidence-based clinical approaches and evaluation of outcomes.
EA occurs in approximately 1 in 3500 live births and is a congenital anomaly that develops in utero. During normal development, the respiratory and digestive tracts separate into the anterior trachea and the posterior esophagus, a process that commences with the outgrowth of the trachea from the ventral foregut by the fourth week of gestation. , The precise mechanism by which EA/TEF occurs during organogenesis is incompletely understood, but several risk factors have been discovered that increase the odds of EA/TEF. EA occurs more commonly in twins, with a relative risk of EA in twins compared with singleton pregnancies of 2.56 (95% CI, 2.01–3.25). EA is also seen in a number of other congenital associations and genetic syndromes. Up to one in four infants born with EA will have an associated nonrandom anomaly as part of the VACTERL association—a spectrum that includes vertebral, anorectal, cardiac, tracheal, esophageal, renal, and limb anomalies. , Additionally, genetic syndromes occur in 10% of infants with EA and include trisomy (chromosomes 13, 18, or 21) and single-gene disorders (CHARGE syndrome [coloboma, heart defects, atresia of the choanae, retardation of growth and mental development, genital underdevelopment, esophageal atresia], DiGeorge syndrome, Feingold syndrome, Opitz syndrome, and Fanconi anemia).
A familiarity with the anatomic variations of EA/TEF is critical for the systematic assessment of an infant suspected of having the anomaly, an understanding of the physiologic consequences of uncorrected EA/TEF, and the formulation of a surgical strategy for correction. The most commonly used classification is that described by Gross, but a simple description of the anatomy of each arrangement can be equally helpful in understanding the presenting features and pathophysiology associated with each ( Fig. 86.1 ). Five common anatomic variants are described, in order of most to least frequent: (1) EA with distal TEF—Gross type C, (2) isolated EA with no fistula—Gross type A, (3) TEF with no EA—commonly referred to as “H” type fistula, (4) EA with a proximal TEF—Gross type B, and (5) EA with both proximal and distal TEFs—Gross type D.
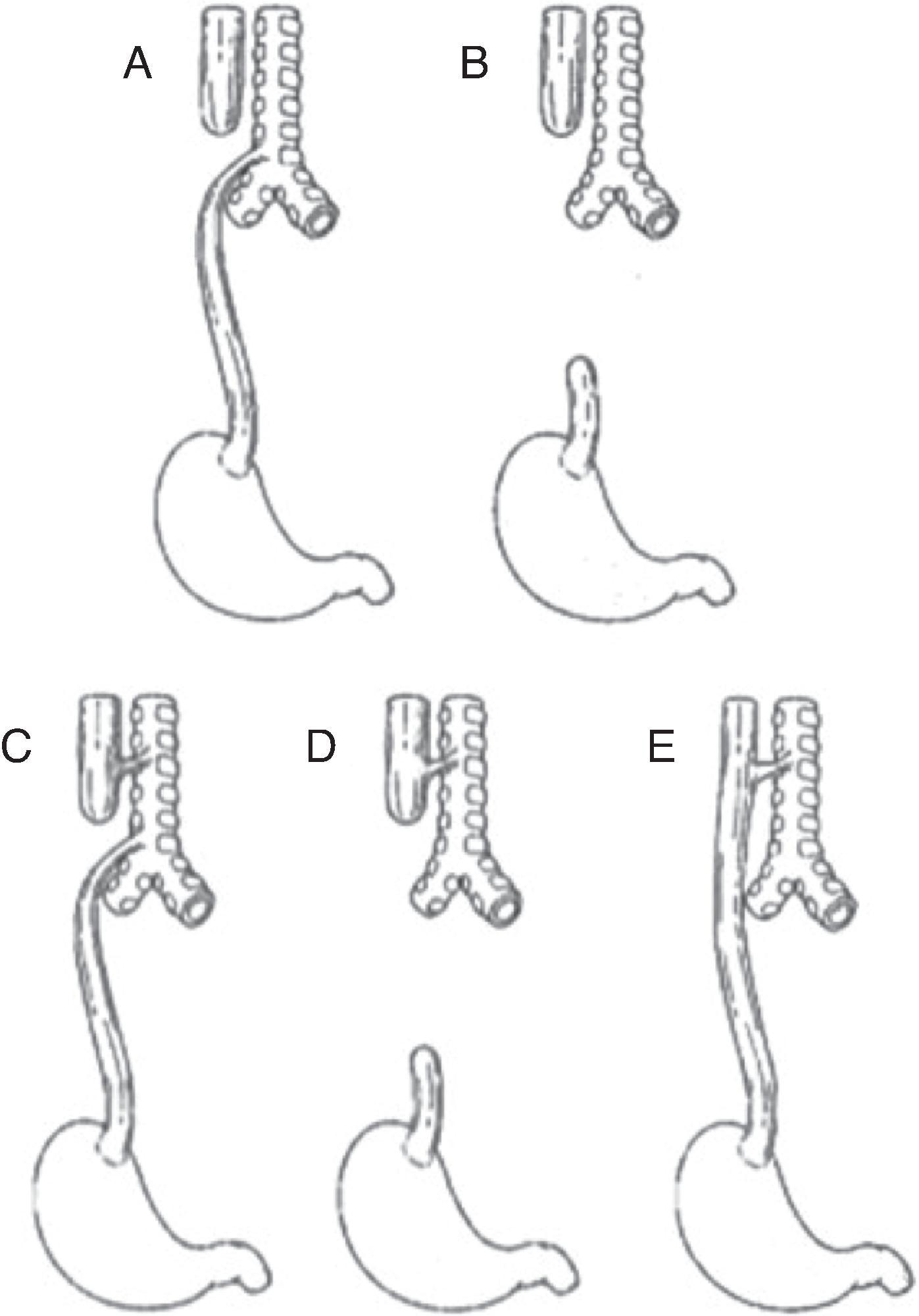
The most common conformation, EA with distal TEF, occurs in 86% of cases. In this arrangement, frequently referred to by its Gross classification “type C,” the proximal esophagus ends in a blind pouch in the superior mediastinum, typically at the level of the third to fourth thoracic vertebra. , The fistula occurs between the distal esophagus and the posterior wall of the trachea, usually within centimeters of the carina. This anatomy allows for the passage of air from the trachea into the low-resistance distal esophagus and intraabdominal intestinal tract, and thus intraluminal abdominal gas will be appreciated on plain radiograph. Additionally, because a TEF does exist, there is risk for gastric contamination of the respiratory tract with subsequent pneumonitis.
The next most common configuration occurs far less frequently, in 8% of cases, and involves an isolated EA with no fistula (type A). The absence of a distal fistula will result in a gasless abdomen, which can be seen on radiograph. In contrast to the gap distance in the typical type C defect, the distance between the blind upper and lower ends of the esophagus in type A is relatively far in isolated EA, thereby often precluding the possibility for a primary anastomosis shortly after birth.
The third most common type, TEF with no EA, occurs in only 4% of cases but presents some unique diagnostic challenges. , Although this is frequently referred to as an “H” type fistula, it has also been more accurately described as an “N” type due to the fistula running from the proximal orifice in the trachea to a distal orifice in the esophagus. Although there is no EA, most of these children can eat orally. The clinical presentation of this type is often more subtle and can involve difficulty feeding or, occasionally, excessive flatulence due to the increased passage of gas into the gastrointestinal tract. This condition can go undetected in the newborn period.
The abnormal anatomic arrangements that result from EA/TEF produce predictable physiologic patterns that are important to recognize and aid in operative planning. Without surgical repair or a temporizing operation, there is no spontaneous resolution of the pathophysiologic consequences of the enteral system discontinuity and/or fistulous connection between the aerodigestive tracts. Therefore, before surgical correction became a feasible operation, the condition was uniformly fatal. In the modern era, up-front surgical correction is successful in most cases, and when not possible (due to comorbidities, extreme low birth weight, or long-gap atresia), temporizing strategies to manage the fistula and to provide enteric feeding access can be implemented.
Without continuity of the digestive tract, neonates with EA are unable to feed orally and thus will develop oral aversion. Although enteral access can be attained surgically, neonates with uncorrected EA are also unable to handle oral secretions, which increases the risk of recurrent oropharyngeal aspiration. Additionally, abnormal development of the trachea and aspiration of gastric contents through a fistulous tract leads to pneumonitis and respiratory compromise.
Less than 20% of EA/TEF cases are detected prenatally. The characteristic findings on prenatal ultrasound suggestive of EA/TEF are polyhydramnios, an absent or small stomach bubble, and a “pouch sign” in which a fluid-filled, blind-ending esophagus is seen during fetal swallowing. However, these findings are usually not seen at the 20-week anatomic survey, are nonspecific, and can often be misleading, because there are several other and more common causes of polyhydramnios, and stomach size can vary even in cases of EA due to gastric secretion production. A recent meta-analysis of contemporary prenatal ultrasound estimated the sensitivity of detecting EA/TEF to be only 31.7%. The addition of fetal magnetic resonance imaging (MRI) has somewhat improved the ability to diagnose EA/TEF prenatally. When performed for cases of suspected EA/TEF, fetal MRI has a sensitivity of 94.7% and a specificity of 89.3%.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here