Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Muscle tissue in the body is classified into one of three major categories according to structure, function, and location. Skeletal muscle is the most common and characteristic type; the other two kinds are cardiac muscle and smooth muscle. Skeletal muscle produces purposeful movements of the skeleton. Cardiac muscle forms the myocardium and is responsible for beating of the heart to pump blood. Smooth muscle provides the motile force for many vital activities including peristalsis in the gut, emptying of the urinary bladder, pupillary constriction, and childbirth (parturition) by contraction of the uterus. The voluntary nervous system controls skeletal muscle function, whereas cardiac and smooth muscles are known as involuntary muscles and are innervated by the autonomic nervous system. Although most cells can undergo shape change and generate motile forces, contractility (cell shortening) is a property that is most highly developed in muscle cells. Under the microscope, the specialized contractile cells of skeletal muscle show an alternating series of transverse bands or striations, which result from the arrangement of contractile filaments; smooth muscle cells, which have a less orderly array of filaments and are found, for example, in internal organs and blood vessels, lack these striations. Cardiac muscle is striated and has characteristics that are intermediate between skeletal and smooth muscle. The principal cellular and functional unit of muscle tissue is the muscle fiber, an elongated and highly differentiated cell. Each fiber has a parallel array of cytoplasmic filaments containing the proteins myosin and actin. Filaments interact and slide past each other to cause contraction or shortening of muscle fibers.

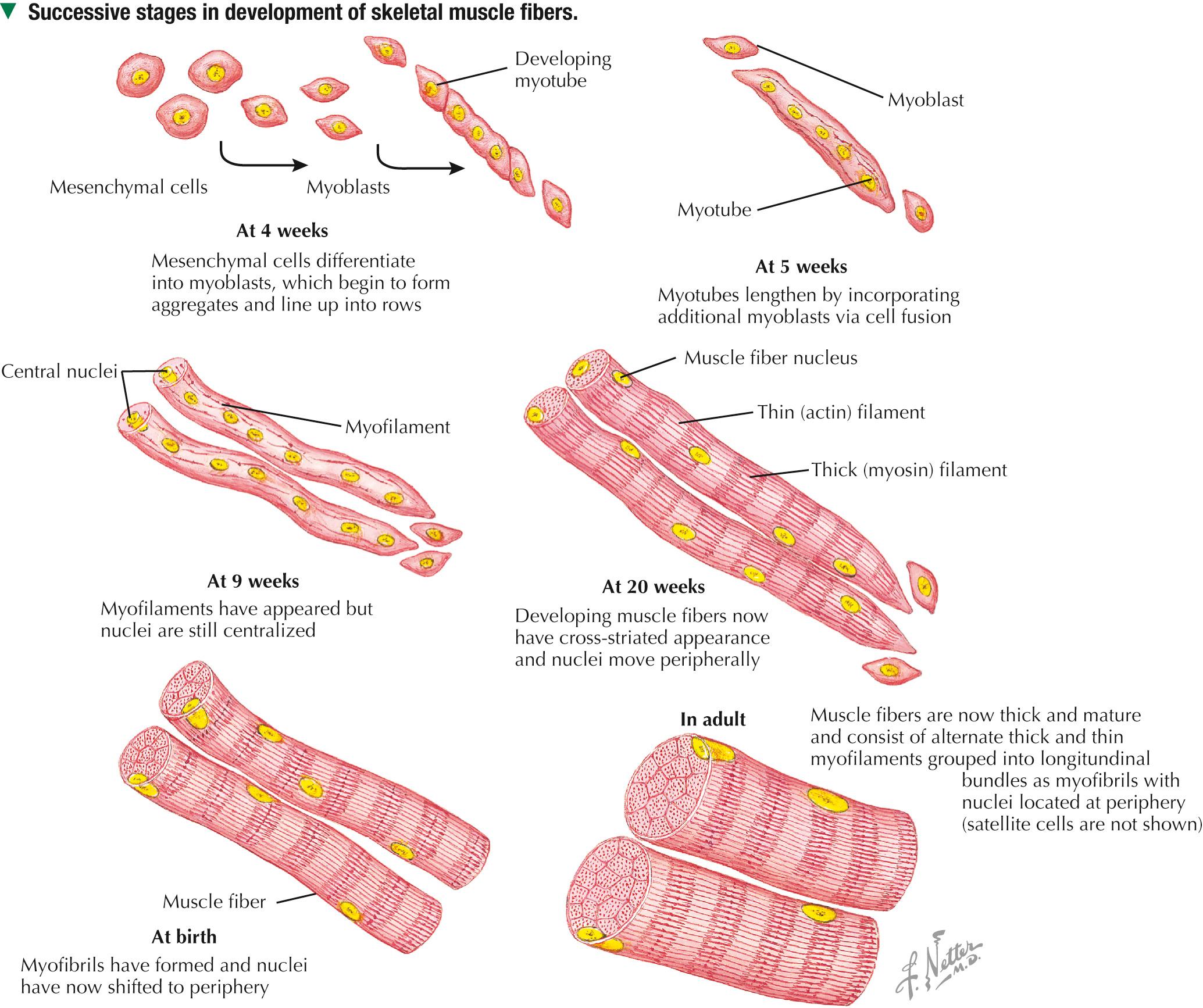
Muscle tissue originates from mesoderm (middle primary germ layer). In the human embryo, most skeletal muscles develop from segmented paraxial mesoderm, organized as myotomes of somites (each becoming innervated by a spinal nerve). Other muscles may develop from mesoderm of branchial arches on each side of the embryonic head (become innervated by cranial nerves), or in situ from local areas of condensed mesenchyme. In the 4-week embryo, mesenchymal cells at genetically predetermined sites proliferate, elongate, and differentiate into myoblasts. Aggregation and end-to-end fusion of myoblasts produce syncytial, multinucleated myotubes. At 5 weeks, myotubes lengthen by incorporating more myoblasts. Some myoblasts fail to fuse, retain an ability to undergo mitosis, and become future satellite (or myosatellite ) cells. At 9 weeks, myotubes synthesize two sets of longitudinally oriented myofilaments, which align in parallel arrays. By 20 weeks, myofilaments continue to proliferate, and nuclei become peripherally located as cells increase in both circumference and length. Myofilaments are arranged in alternating, overlapping bands, so muscle fibers have a cross-striated appearance. At birth, thick (myosin) and thin (actin) myofilaments have collected into cylindrical bundles called myofibrils, and muscle fibers can contract as they become innervated by motor neurons. A single motor neuron and all the skeletal muscle fibers it innervates are known as a motor unit. Myosatellite cells, capable of division, remain closely associated with the plasma membrane, or sarcolemma, of each muscle fiber throughout life.
Rhabdomyosarcoma is the most common soft tissue sarcoma in children and adolescents. Two histologic subtypes of this skeletal muscle neoplasm —the more common embryonal and less frequent alveolar —differ in clinical presentation, age of onset, metastatic behavior, and prognosis. Even though both are fast growing and malignant, long-term survival rates show improvement with advances in surgery combined with high-dose chemotherapy, stem cell rescue, and radiation. Tumor cells may arise directly from satellite cells or mesenchymal stem cells with capacity to become striated muscle; they resemble preinnervated fetal skeletal muscle. By immunocytochemistry, tumor cells express antibodies to myogenic transcription ( PAX3, PAX7 ) and regulatory ( MyoD1 ) factors. Electron microscopy is important for tumor diagnosis and staging and shows a wide spectrum of cellular differentiation with cells containing myofilaments, sarcomeres, and leptomeric fibrils.
Although individual skeletal muscles have remarkably diverse functions, their fundamental role is to produce movement and generate force. To do so, skeletal muscle contains elongated, thread-like multinucleated cells called muscle fibers. The ends of the fibers insert into tendons, which attach to bones across joints. A dense connective tissue sheath—the epimysium —surrounds the whole muscle externally. Connective tissue septa that make up the perimysium subdivide the muscle internally into bundles, or fascicles, each containing several muscle fibers. A more delicate, looser connective tissue—the endomysium —surrounds individual muscle fibers. The vascular supply follows the connective tissue into the muscle, where capillaries run parallel with the longitudinal axis of the muscle fibers. Each muscle fiber is invested by a thin external lamina (basement membrane), which also encloses satellite cells that closely adhere to the plasma membrane, or sarcolemma, of the muscle fibers. The most striking features of skeletal muscle fibers are an orderly arrangement of contractile myofilaments into myofibrils and a characteristic striation pattern. Each myofibril has alternating light ( I, for isotropic) and dark ( A, for anisotropic) bands along its length. Two sets of myofilaments, thick and thin, that make up each myofibril are organized into repeating units of contraction known as sarcomeres. Dark, transverse Z (Zwischenscheibe) bands mark the ends of each sarcomere and anchor the thin filaments. The center of the sarcomere contains the thick (myosin-containing) filaments, which form the A band; thin (actin-containing) filaments, which form the I bands, are at the ends of each sarcomere.
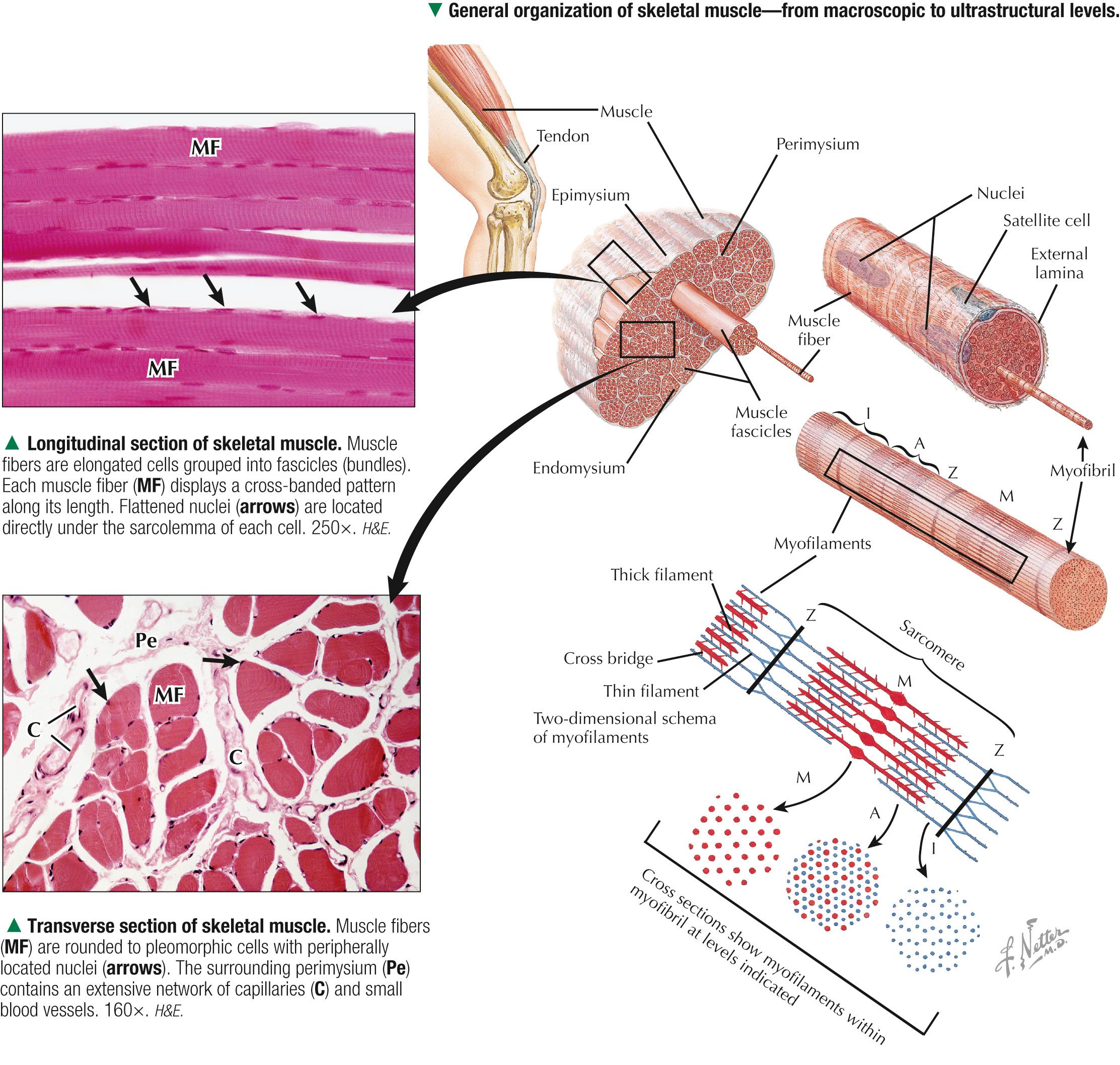
The sarcolemma is the plasma membrane of the muscle fiber enveloped by an outer layer of glycoproteins and a fine network of reticular fibers, all constituting an external lamina. The sarcolemma and its tubular invaginations, called transverse (T) tubules, play a role in initiation of contraction. The myofibrils, each of which is 1-2 µm in diameter, constitute about 80% of the cell volume. Each myofibril contains a closely packed, orderly, overlapping longitudinally arranged array of myofilaments with a highly regular banding pattern. Each sarcomere of a myofibril consists of an A band and half of two contiguous I bands. A relaxed sarcomere is about 2.5 µm long, with the A band being about 1.6 µm long, and the I band on each side of the Z band about 1 µm long. Organelles, including mitochondria and tubular elements of the smooth-surfaced sarcotubular system, are interposed between myofibrils. Mitochondria are pleomorphic, and their density and distribution vary markedly in different muscle fiber types. The sarcotubular system is composed of two separate and distinct membrane systems of the muscle fiber, known as the sarcoplasmic reticulum (SR) and the transverse tubular system. The SR, similar to the smooth endoplasmic reticulum of other cells, is an elaborate, anastomosing network of tubules and cisternae that surround the myofibrils. At regular intervals relative to the sarcomeres, two flattened sacs of the SR, known as terminal cisternae (lateral sacs), closely associate with a central transverse tubule and form a muscle triad, which is the main site for excitation-contraction coupling. The remaining sarcoplasm contains ribosomes, glycogen, and lipid droplets.
Duchenne muscular dystrophy is a genetic disorder caused by a deficiency of dystrophin, a large membrane-associated cytoskeletal protein. It is the most common of a group of muscular dystrophies characterized by rapid progression of skeletal muscle degeneration occurring early in life. Dystrophin is encoded by a gene on the short arm of the X chromosome (Xp21) and is linked to the cytoplasmic side of the sarcolemma of the muscle fiber. Dystrophin maintains mechanical integrity of the cell during contraction by anchoring cytoskeletal elements. Mainly young boys are affected, and symptoms, including muscle weakness and wasting and heart involvement, worsen with age.

Skeletal muscle constitutes 40%-50% of the body weight. As its name implies, it is mostly attached to the skeleton. It is also called striated, or voluntary, muscle, because its cells appear striated or cross-banded under the microscope. Skeletal muscle fibers are elongated, cylindrical cells, 50-200 µm in diameter, with tapered ends. They are multinucleated, the nuclei occupying peripheral positions in the cell. Their cytoplasm, known as sarcoplasm, is packed with myofibrils, which are cylindrical bundles of myofilaments along the length of the fiber. Each myofibril has a uniform diameter and consists of identical repeating units, called sarcomeres. Sarcomeres are composed of longitudinally oriented thick and thin filaments and perpendicular Z bands. In longitudinal section, skeletal muscle fibers show transverse striations because adjacent myofibrils are in lateral register with each other across the width of the fiber. The greater density of the A bands is due mainly to the presence of thick (myosin-containing) filaments, whereas the lighter density of the I bands is due to the prevalence of thin (actin-containing) filaments. In the center of each A band is a lighter H zone (the central part of thick filaments not overlapped by thin filaments), which is bisected by a thin, dark M band. The width of the I band and H zone in each sarcomere varies and depends on the extent to which the muscle fiber is contracted or stretched.

Adult skeletal muscle fibers are polygonal, although in infancy they tend to be rounded, as are those of the extrinsic eye muscles and some muscles of facial expression. In transverse sections stained with hematoxylin and eosin (H&E), the sarcoplasm of each fiber is intensely eosinophilic and looks punctate because of tightly packed myofibrils. Myofibrils constitute the bulk of each fiber and mostly contain the major contractile proteins myosin and actin, which make up the myofilaments. In cross section, myofibrils are often seen grouped together and form irregularly shaped Cohnheim's fields, which are probably artifacts of preparation caused by shrinkage. The surrounding endomysium supports a rich vascular and nerve supply, which consists of capillaries and nerve fascicles close to the muscle fibers. Electron microscopy clarifies further details of ultrastructure. Myofibrils are rounded to irregular in shape, and the intervening, intermyofibrillar sarcoplasm contains a variety of other organelles, mitochondria being the most conspicuous at low magnification.
Skeletal muscle biopsy —a valuable clinical procedure for diagnosis and management of many neuromuscular diseases—is removal of a small piece of muscle tissue under local anesthesia via open biopsy (surgical excision) or by less invasive percutaneous needle followed by microscopic evaluation. Serial frozen-sections treated with H&E and Gomori trichrome are used to assess morphology, periodic acid–Schiff ( PAS ) to detect glycogen, and histochemistry for muscle fiber typing (e.g., myofibrillar ATPase ) and oxidative enzyme profiles (e.g., succinic dehydrogenase, NADH-tetrazolium reductase ). Immunocytochemistry may help determine specific muscle protein deficiencies (e.g., dystrophin, sarcoglycan, merosin ) in different forms of muscular dystrophy. In some cases, electron microscopic evaluation of plastic-embedded sections may yield additionally useful diagnostic information (e.g., mitochondrial myopathy, inclusion body myositis, nemaline myopathy ).
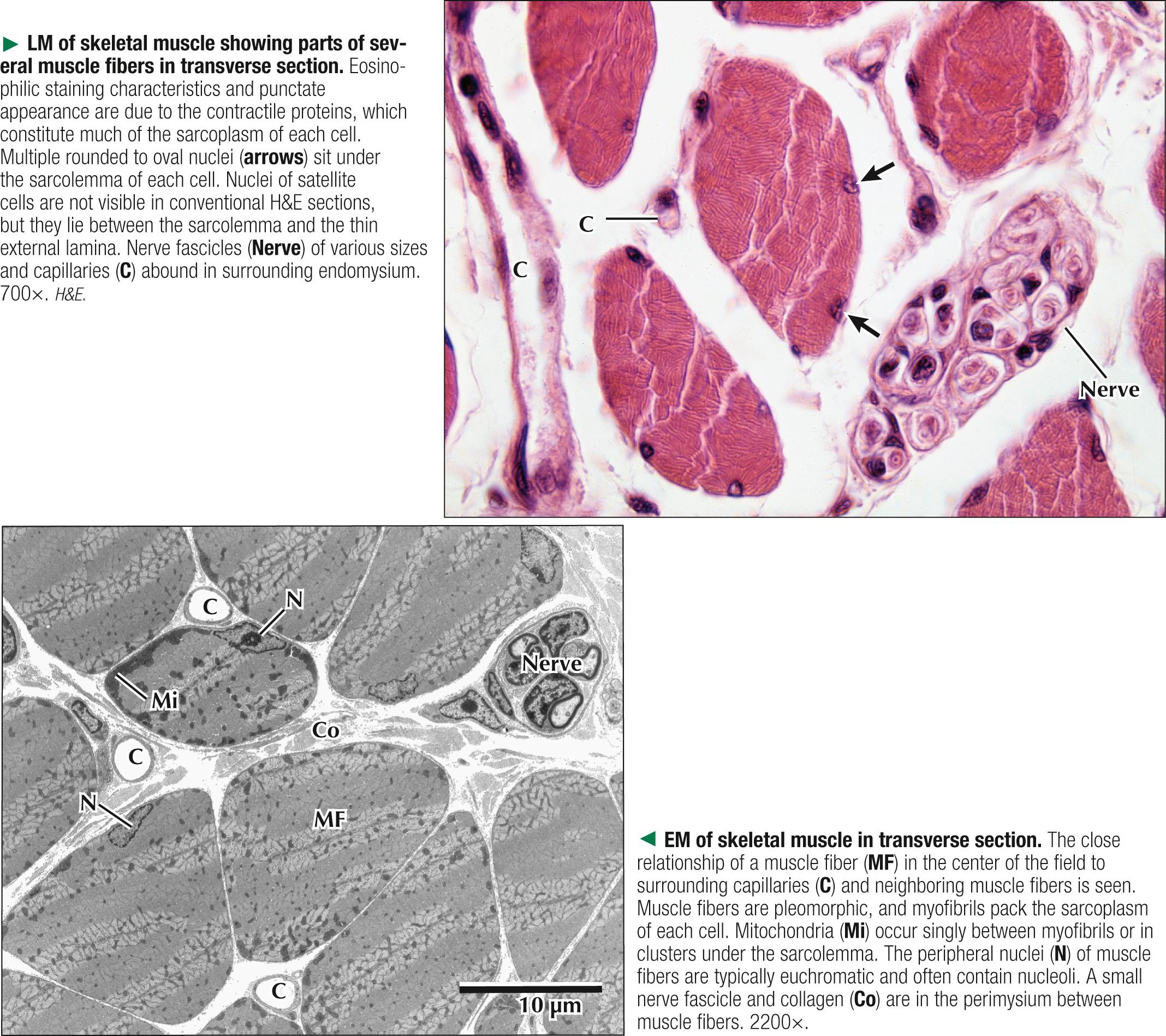
Two membranous components of skeletal muscle fibers, the sarcotubular system and mitochondria, are closely associated with myofibrils. The sarcotubular system, comprising the T tubules and the SR, plays a critical role in excitation-contraction coupling. These elements are not continuous with each other but are closely associated and have a specific orientation to the sarcomere. T tubules, a membrane system external to the muscle fiber at the junction of A and I bands, penetrate the fiber interior at regular intervals, mostly in a transverse plane. They rapidly convey electrical impulses from the sarcolemma to the cell interior. The SR, an internal membrane system that stores intracellular calcium ions, consists of a repeating array of longitudinally oriented tubules and flattened sacs, the terminal cisternae, surrounding the myofibrils. At A-I junctions, two terminal SR cisternae lie very close to a central T tubule, with an intervening gap of 15 nm, to form a tripartite complex called a muscle triad. Action potentials are propagated along T tubules. At the interface of the T tubule with the SR in the triad, calcium ions are released from the SR into the sarcoplasm, which initiates contraction. Junctional end-feet link the T tubule with the SR and are composed of a voltage-sensing dihydropyridine receptor on the T tubule connected to a ryanodine receptor calcium release channel. The elongated to pleomorphic mitochondria, an aerobic energy source, produce ATP, which is needed for contraction. They are aligned at strategic sites within muscle fibers; they usually occur at I band levels or in aggregates at the periphery of the fibers. Their density, location, and distribution in muscle fibers depend on muscle fiber type.

Parallel arrays of thin filaments span the I band and overlap thick filaments of the A band. A third filament system is made of single molecules of titin, one of the largest known proteins, that connects the Z band to the M band. Titin contains elastic elements that act as molecular springs and contribute to the passive elasticity of muscle. Nebulin, another giant protein, spans the length of thin filaments and forms a fourth filament system in skeletal muscle. At Z bands, thin filaments, nebulin, and titin are anchored to the protein α-actinin . Myosin molecules are polarized and have a globular head and a tail region. The globular head, or S1 region, contains an ATPase that facilitates binding to actin to move the head and produce a power stroke. Antiparallel association of myosin molecules forms thick filaments. In half of the thick filament, myosin heads are oriented in one direction; those in the other half are in the opposite direction. The tails of the myosin molecules overlap, which yields a bare central shaft. Each thick filament is 1.6 µm long and 15 nm in diameter. Each thin filament, about 1 µm long and 5 nm in diameter, consists of a double helix of filamentous actin. Two proteins associated with actin, tropomyosin and the troponin complex, respond to varying calcium ion concentrations by acting as a switch to enable or disable the interaction and formation of cross bridges between actin and the myosin heads. Myosin heads bind to actin and draw the thin filament a short distance past the thick filament. Then, linkages break and re-form farther along the thin filament to repeat the process. Filaments are thus pulled past each other in a ratchet-like action.
Myotonia is a neuromuscular condition characterized by slow or delayed relaxation of a muscle after voluntary contraction. It may arise in any muscle group by sudden exposure to cold temperature but usually improves after the muscles have been warmed up with brief exercise. Myotonia congenita (or myotonic muscular dystrophy ) is a genetic disorder that usually begins in infancy or early childhood. It is caused by mutations in the CLCN1 gene that controls voltage-gated chloride ion (Cl - ) channel proteins in the sarcolemma of skeletal muscle fibers. Subsequent interruption in Cl - flow triggers prolonged muscle contractions and stiffness. Clinical diagnosis is by genetic testing, electrodiagnostic procedures, and muscle biopsy. Although the disease severity differs among individuals, physical therapy and other rehabilitative procedures may facilitate muscle function.
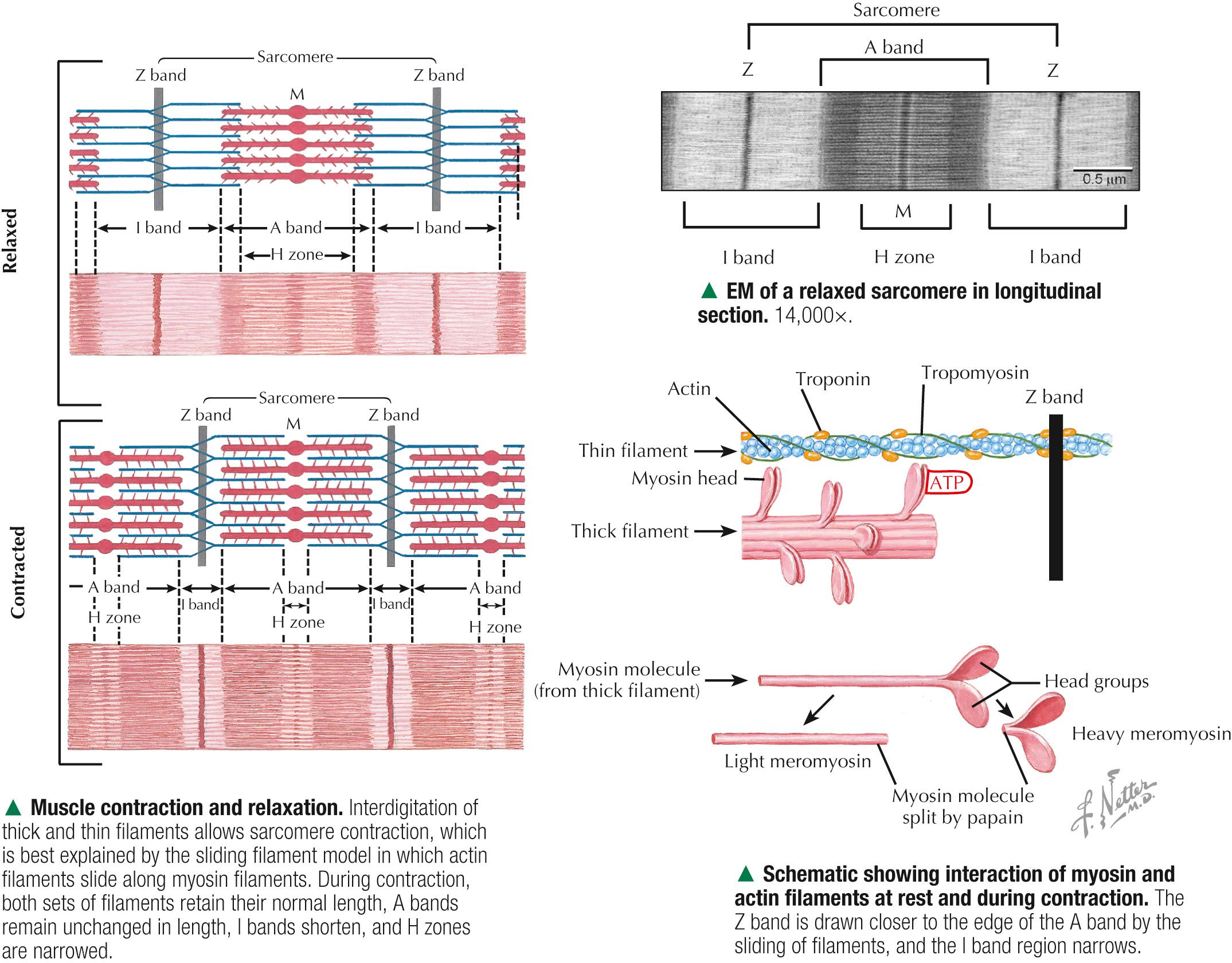
By high-magnification electron microscopy, transverse sections reveal the precise arrangement of two sets of myofilaments at different levels of the sarcomere. Myofilaments in cross section are electron-dense, punctate profiles; the diameter of thick filaments is more than twice that of thin filaments. Cross sections of I bands show only thin filaments, whereas A bands show both thick and thin filaments, which appear as hexagonal networks with myosin fixed at the M band. Where the two sets of filaments overlap, the networks mesh so that each thick filament is in the center of a hexagon made of six neighboring thin filaments. The interval between thick and thin filaments in the double hexagonal array is 10-20 nm. Regularly spaced globular heads of the myosin cross bridges radiate from each thick filament toward the thin filaments; however, cross bridges are not well resolved by routine electron microscopy, so thick filaments show a roughened surface. In pale H zones, the central segment of each thick filament lacks cross bridges. M bands, which are in the center of the H zones, show thick filaments with fine interconnections. Z bands in cross section show a typical square lattice pattern, and thin filaments in the immediate vicinity are organized in a regular array.

Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here