Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The complement system is a group of proteins that provide an important part of the immune defense against infection. Many components of the complement system circulate as inactive proteins in the plasma. Activation of the complement system generates peptide fragments that serve as ligands for several receptors and complete a multimeric complex (C5b-9) that forms pores in membranes resulting in cell lysis.
As with all components of the immune system, proper function of the complement system helps with the effective elimination of invasive pathogens while causing minimal inflammation or injury to host tissues. However, uncontrolled activation of the complement system can cause tissue injury, and there is clear evidence that the complement cascade is activated in many autoimmune and inflammatory diseases. The kidney is particularly susceptible to complement-mediated injury, and the complement system has been implicated in the pathogenesis of multiple kidney diseases. It is also evident that acquired and congenital defects in the complement system are important risk factors for several kidney diseases. For the most part, these disease-associated defects impair the body’s ability to regulate the complement system, thereby permitting overactivation or “dysregulation” of the complement cascade.
Uncontrolled complement alternative pathway activation appears to be central to the development of two kidney diseases: atypical hemolytic uremic syndrome (aHUS) and C3 glomerulopathy (C3G). These diseases are clinically and histologically distinct yet share similar risk factors. Our understanding of the pathogenesis of these diseases has been significantly advanced by recent discoveries. Although aHUS and C3G are both rare diseases, greater understanding of these diseases as extreme examples of complement dysregulation have provided insight into more common forms of glomerulonephritis that also involve complement activation.
The complement system is composed of soluble proteins, soluble regulatory proteins, cell surface regulatory proteins, and cell surface receptors. Activation proceeds through three different pathways: the classical pathway, the mannose-binding lectin (MBL) pathway, and the alternative pathway ( Fig. 22.1 ). Each pathway is activated by multiple different molecules. IgM- and IgG-containing immune complexes (ICs) activate the classical pathway, for example, but C-reactive protein, beta-amyloid fibrils, and other molecules also activate this pathway. The MBL pathway is activated when several different types of proteins, including MBLs, collectins, and ficolins, bind to sugars displayed on the surface of bacteria or damaged cells. All three pathways result in the cleavage of C3, forming C3b that can be covalently fixed to tissue surfaces.
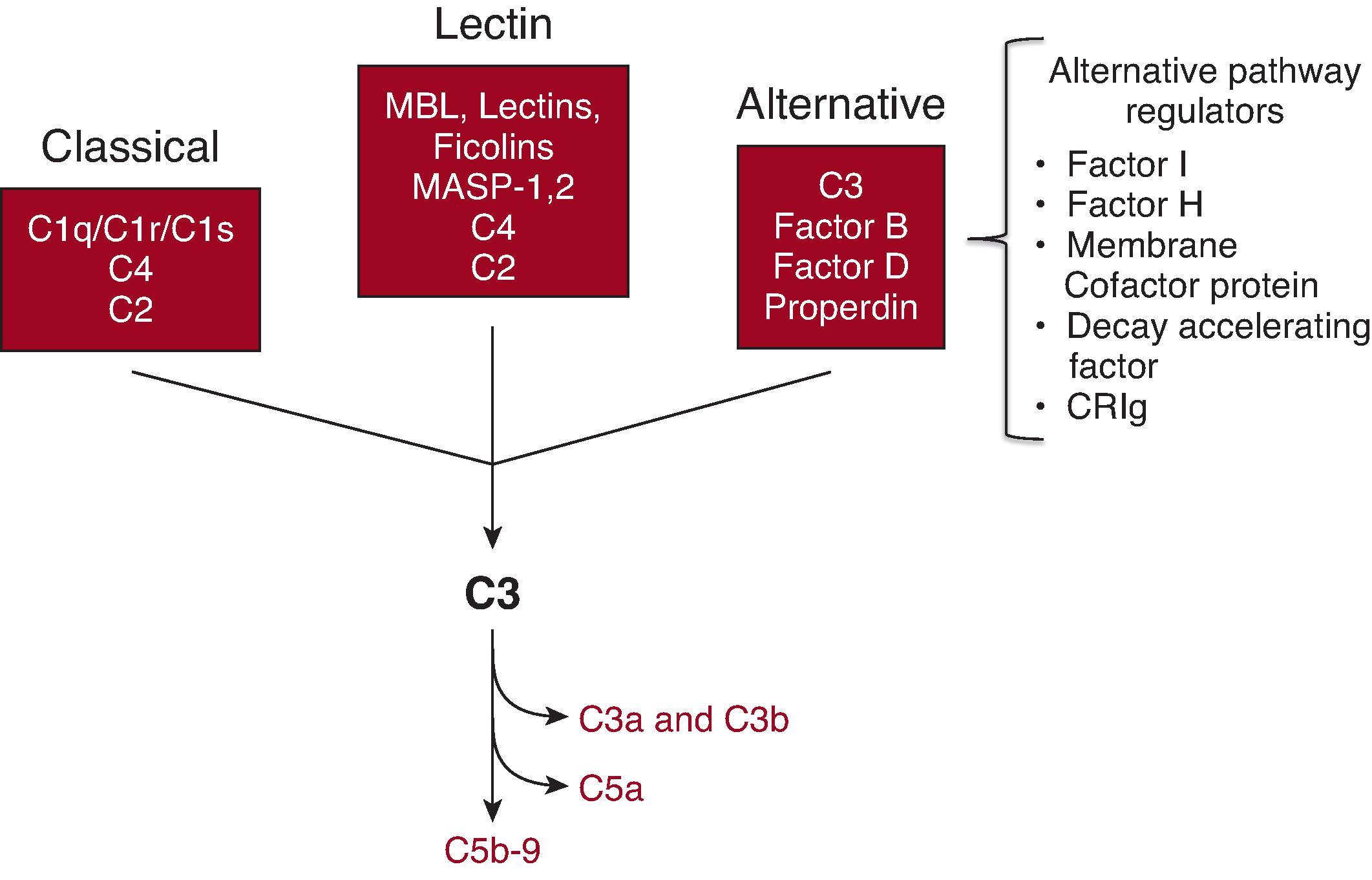
Full activation of the complement cascade generates C3a, C3b, C5a, and C5b-9. Receptors for C3a and C5a induce several different inflammatory responses. Leukocytes also express receptors for C3b and the C3b inactivation fragments (iC3b and C3d). C5b-9 is also called the membrane attack complex (MAC) or the terminal complement complex (TCC). The insertion of C5b-9 into cell membranes can result in cell activation or lysis.
Several features of the alternative pathway are notable and may explain the link between activation of this pathway and kidney disease. Like the classical and MBL pathways, some proteins, including IgA, directly activate the alternative pathway. Tissue-bound C3b can combine with a protein called factor B to form an alternative pathway-activating enzyme (C3bBb). Consequently, the deposition of C3b on tissue surfaces by the classical and MBL pathways can secondarily engage the alternative pathway, resulting in increased complement activation through an alternative pathway “amplification loop.” Finally, C3 in plasma is hydrolyzed at a slow rate, forming an enzyme complex that cleaves more C3 and activates the alternative pathway unless adequately controlled. This spontaneous “tickover” process continuously generates C3b that can also bind to surfaces and be amplified through the alternative pathway.
Because the alternative pathway of complement is continuously active in plasma and tends to self-amplify, it is crucial that the body adequately controls this process. Complement activation is controlled by a group of regulatory proteins expressed on the surface of cells or that circulate in plasma. The ability of a surface to regulate alternative pathway activation determines whether the process continues to self-amplify or is terminated ( Fig. 22.2 ). These regulatory proteins provide a shield that protects the host from complement-mediated injury. Pathogens do not express complement regulatory proteins, and expression of these proteins is decreased on damaged host cells.
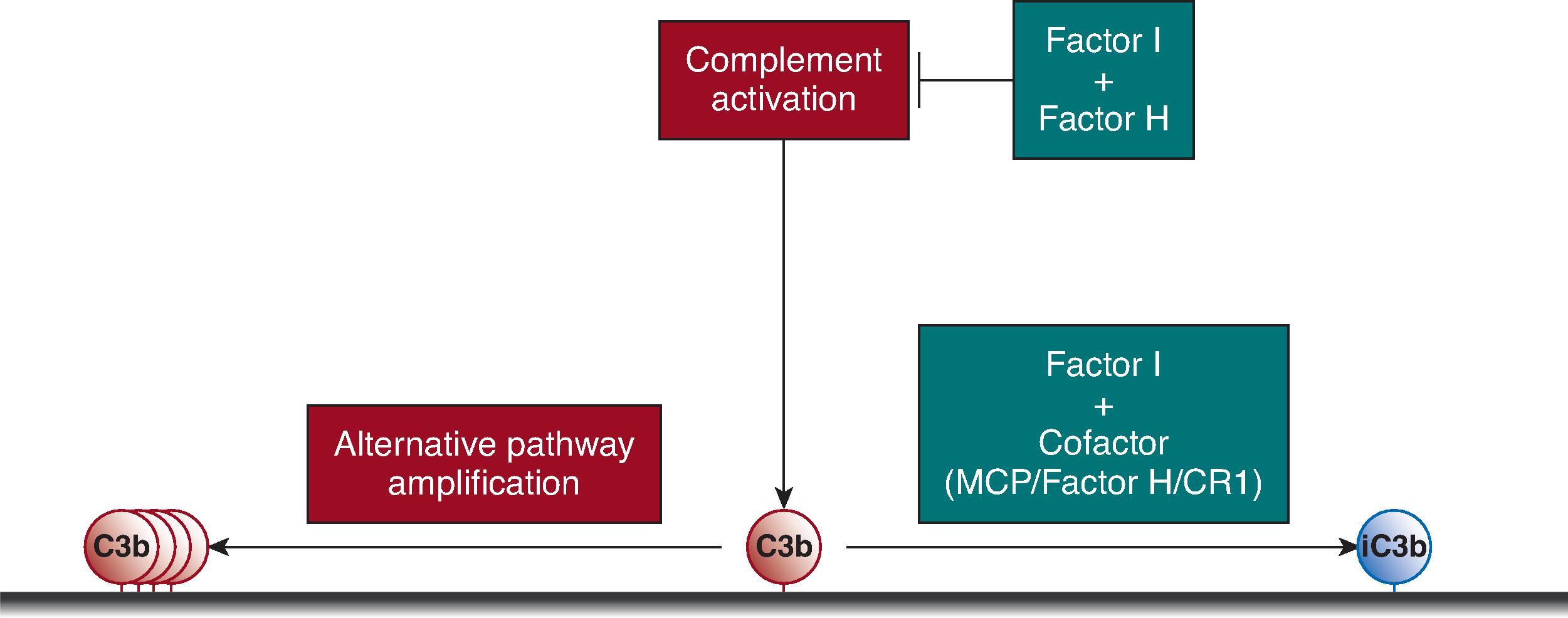
Specific activating proteins can trigger complement activation on a particular cell or surface, but the degree of activation is also determined by the local expression of complement regulatory proteins. Impaired regulation may lower the threshold for activation within a particular tissue, and local impairments of regulation may even be sufficient to permit spontaneous activation. Endothelial cells and podocytes each express several of the complement regulatory proteins, and ordinarily there is little evident complement activation within the glomerular capillary wall.
Several different proteins can regulate the complement system. Factor I is a circulating protein that cleaves (inactivates) C3b, forming iC3b (see Fig. 22.2 ). To function, however, factor I requires a “cofactor” protein. Several proteins with cofactor function are expressed on cell surfaces (e.g., membrane cofactor protein [MCP] and complement receptor-1 [CR1]). Factor H, a soluble alternative pathway inhibitor, also has cofactor activity. Other proteins regulate complement activation by reducing the half-life of the activating enzymes through a process termed decay acceleration. Decay-accelerating factor (DAF, or CD55) is a protein linked to the surface of cells that limits complement activation, and factor H also controls complement activation by this mechanism. Several additional proteins also control the complement system through other mechanisms. For example, carboxypeptidase N is a plasma enzyme that rapidly inactivates C3a and C5a, and CD59 is a protein that is linked to cell membranes to prevent the addition of C9 during formation of C5b-9.
Congenital or acquired defects can impair the body’s ability to regulate the complement system, and these defects can increase a patient’s risk of developing different autoimmune and inflammatory diseases. Mutations have been identified in the genes for the regulatory proteins factor I, factor H, and MCP. Gain-of-function mutations have also been identified in the genes for C3 and factor B. These mutations appear to reduce the ability of the regulatory proteins to inactivate the activating enzymes, so they are functionally similar to the loss-of-function mutations seen in the complement regulatory proteins. Autoantibodies to complement proteins have also been detected in patients with various diseases, and these autoantibodies tend to impair regulation of the alternative pathway.
Defects in factor H function are the most common complement abnormalities seen in aHUS, and impaired factor H function is also seen in some patients with C3G. Factor H is a soluble protein that is primarily produced by the liver and circulates in plasma at a concentration of 300 to 500 µg/mL. Factor H is made of up 20 repeating structures called short consensus repeats , or SCRs . Regulation of the alternative pathway is performed at the amino terminus of the protein in the first four SCRs, whereas the last two SCRs (19 and 20) mediate binding of factor H to molecules displayed on tissue surfaces such as glycosaminoglycans (GAGs) and sialic acid. Although it is not known why impairments in factor H so frequently manifest as injury within the glomerulus, one possibility is that the glomerular basement membrane (GBM) does not contain the other regulatory proteins and is completely dependent on factor H to control the alternative pathway.
The complement factor H–related proteins (CFHRs) are a group of five proteins that arose through reduplication of the gene for factor H with which they have high structural homology. The CFHRs all contain regions that are homologous to SCRs 19 and 20 of factor H, suggesting that they can bind similar molecules and surfaces. Various deletions and mutations in the CFHR genes have been identified in patients with aHUS and C3G. Experiments suggest that the mutant CFHRs competitively inhibit factor H. This can render patients functionally factor H deficient and cause complement dysregulation.
IgG and IgM containing ICs are activators of the classical pathway of complement, and there is strong evidence that the complement system is an important mediator of injury in diseases associated with glomerular IC deposition. The interaction between the complement system and ICs is complex. Some isotypes of IgG activate complement more efficiently than other isotypes. Also, the complement system helps solubilize ICs, mediating the downstream effects of ICs but also reducing their deposition in tissues. Moreover, complement activation fragments affect the adaptive immune response, so the complement system may also influence disease upstream of antibody formation.
In addition to the involvement of the classical pathway, evidence from studies examining large cohorts of patients with immune-complex glomerulonephritis have revealed in a substantial proportion of cases a dysregulation (both genetic and serological) of the alternative pathway of complement. This indicates that the presence of glomerular immune complexes does not rule out this pathway as a mediator of kidney injury.
C3G is a rare form of proliferative glomerulonephritis comprising two forms: C3 glomerulonephritis (C3GN) and dense-deposit disease (DDD; previously known as type II membranoproliferative glomerulonephritis [MPGN] or MPGN type II). The unifying feature of all forms of C3G is the presence of intense C3 staining with little or no immunoglobulin staining by immunofluorescence on kidney biopsy. However, as mentioned above, large retrospective studies have shown that the distinction between immune-complex mediated MPGN (so-called IC-MPGN) and C3G is not always clear cut, as a substantial number of patients with IC-MPGN also have dysregulation of the alternative pathway of complement. Clinically, these forms of primary alternative pathway-driven IC-MPGN are recognizable by low circulating C3 levels with normal circulating C4, setting them apart from typical secondary forms of MPGN in which C4 is also reduced.
Incidence of C3G is reported as one to two per million per total population. The age of onset is highly variable. The youngest reported patient was 1 year old, and about 40% of patients presented before 16 years of age in one large cohort. There was a slight prevalence of men in this series (60%), and a family history of glomerulonephritis was reported in about 11% of cases.
A large number of molecular causes of alternative pathway complement dysregulation have been identified in patients with C3G, including autoantibodies and genetic variants that encode dysfunctional complement proteins ( Table 22.1 ). Patients with monoclonal gammopathies can also develop C3G.
| Complement Protein | Abnormality |
| C3 convertase (C3bBb) | Autoantibody
|
| Factor H | Protein
Autoantibody
Mutations
|
| Factor B | Protein
Autoantibody
Mutations
|
| Factor I | Protein
Mutations
|
| C3 | Protein
Autoantibody
Mutations
|
| CFHR1 | Mutations
|
| CHFR2 | Mutations
|
| CHFR3 | Mutations
|
| CFHR5 | Mutations
|
The most common autoantibody associated with C3G is C3 nephritic factor (C3Nef). C3Nef can be detected in approximately 70% to 80% of patients with C3G. It is more common in DDD than in C3GN and correlates with lower levels of C3. The presence of C3Nef does not correlate with clinical outcomes, however, and can be detected in healthy control subjects.
A monoclonal immunoglobulin light chain that inhibited factor H was identified in a C3G patient. This antibody blocked the regulatory function of factor H, thereby impairing regulation of the alternative pathway. A similar mechanism may cause complement dysregulation in some patients over the age of 40 years with monoclonal gammopathies. Anti-factor H IgG antibodies have also been identified in additional patients with C3G. These antibodies bind to the amino terminus of the protein and block its regulatory function. Antibodies reactive to factor B and C3b have also been reported. These different autoantibodies are associated with increased alternative pathway activation, lending further support to the concept that alternative pathway activation is central to the pathogenesis of C3G. Recently, antibodies directed against factor B have been detected in a majority of children with acute postinfectious glomerulonephritis, suggesting that alternative pathway dysregulation plays a central but transient role in this acute form of glomerulonephritis. The detection of antibodies specific for the complement proteins is currently only performed in research labs and is not widely available.
Disease-associated mutations and rare variants have been identified in the genes for factor H, factor I, factor B, C3, and CFHRs 1, 2, 3, and 5 (see Table 22.1 ). An internal reduplication of a region in CFHR5 is associated with C3G, and a deletion causing formation of a hybrid CFHR2-CFHR5 protein was found in two related patients with C3G.
The different molecular causes of C3G increase alternative pathway activation or make this system resistant to regulation. It is not known, however, whether the disease is primarily caused by systemic complement activation in the plasma or local activation directly on the mesangium and glomerular capillary wall. Complement activation in the plasma could lead to deposition of complement proteins within the glomeruli as plasma is filtered by the kidney. The GBM may be dependent upon factor H for regulating the alternative pathway. Bruch’s membrane in the eye may be similarly dependent upon factor H, and patients with C3G develop retinal lesions and visual impairments.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here