Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Carotid disease is a common manifestation of focal atherosclerosis most often located at the origin of the internal carotid artery, and accounts for 10%–20% of ischemic strokes.
Intracranial carotid disease is more common than extracranial disease in Asian, black, and Hispanic populations.
The most common mechanism of stroke involves rupture of the atherosclerotic plaque with subsequent embolism to the brain hemisphere or eye.
Carotid stenosis can reliably be detected by noninvasive imaging, including CT and MR angiography as well as ultrasound. MRI and ultrasound are increasingly used to measure the composition of the atherosclerotic plaque which, together with clinical risk factors, may give clues to the risk of stroke and the necessity of invasive revascularizations.
Stroke risk in patients with carotid disease has been lowered with better medication, notably with widespread use of statins.
The most common disease of the carotid artery is atherosclerosis occurring at the carotid artery bifurcation, typically involving the distal common carotid and the proximal internal carotid artery, and to a lesser extent the proximal external carotid artery (ECA). Carotid artery disease accounts for between 10% and 20% of ischemic stroke, depending on the method of classification. The carotid arteries provide about 80% of the blood supply to the brain and thus the majority of emboli from the heart and the proximal aorta also pass through the carotid circulation, which makes it difficult to distinguish between carotid disease and these other sources as the cause for carotid territory stroke. Thus a carotid source for stroke or transient ischemic attack (TIA) tends to be inferred from the imaging finding of disease of the carotid artery in association with ipsilateral carotid territory symptoms in the absence of any other obvious cause. Most studies have required the finding of at least 50% stenosis of the carotid artery as indicating significant carotid disease, partly because detection of lesser degrees of stenosis was unreliable on Doppler ultrasound, and partly because mild disease of the carotid artery is very common. However, this does not mean that lesser degrees of disease will not necessarily cause stroke. Modern methods of carotid plaque imaging described below may alter this situation in the future.
Atherosclerotic carotid disease is more common in men than in women and its prevalence increases with age; ultrasound screening studies have shown a prevalence of moderate asymptomatic carotid stenosis narrowing the lumen by 50% or more in 2.3% of 60- to 69-year-old men, 6% of 70- to 79-year-old men, and up to 7.5% of ≥80-year-old men; in women the prevalence rates were 2%, 3.6%, and 5%, respectively. Severe carotid stenosis measuring 70% or more was present in 0.8%, 2.1%, and 3.1% in the same age groups in men, and in 0.2%, 1.0%, and 0.9% in women. Despite the high prevalence of carotid disease in the community, only a small proportion of these patients will develop TIA and stroke. Symptomatic carotid stenosis is less common in women, presumably reflecting the difference in prevalence in the community, and is more common in white Caucasian races than in black, Asian, or other races. Among white Caucasians, there appears to be considerable heritability of carotid plaque characteristics and individual gene variants promoting carotid atherosclerosis in stroke patients have been identified. ,
Risk factors for carotid atherosclerosis are similar to atherosclerosis at other sites and include raised cholesterol, systolic hypertension, and smoking, as well as increasing age. However, after adjustment for other risk factors, it appears that diabetes is not a strong risk factor. Among stroke survivors, smoking appears to be more strongly associated with the presence of carotid stenosis than other risk factors.
Longitudinal studies have observed a reduction in the proportion of ischemic strokes associated with carotid stenosis over the past years, along with a decrease in patients’ low-density-lipoprotein (LDL) cholesterol and blood pressure, perhaps related to better risk factor control in the community. A recent prospective study of consecutive patients admitted to a hyperacute stroke unit in London, UK reported that although carotid stenosis ≤50% was found in 19% of 1252 patients with ischemic stroke or TIA, the stenosis was only considered symptomatic in 7.9%.
The anatomy of the cerebral artery tree is presented in Fig. 22.1 . The bifurcation lies at the level of C3–4 in 55% of cases; the locations of the remainder are scattered from as high as C2–3 to as low as C5–6. In 85% the bifurcations are located at the same height on both sides. On average, women have smaller internal carotid arteries with a diameter of 4.7 mm, compared with 5.1 mm in men.
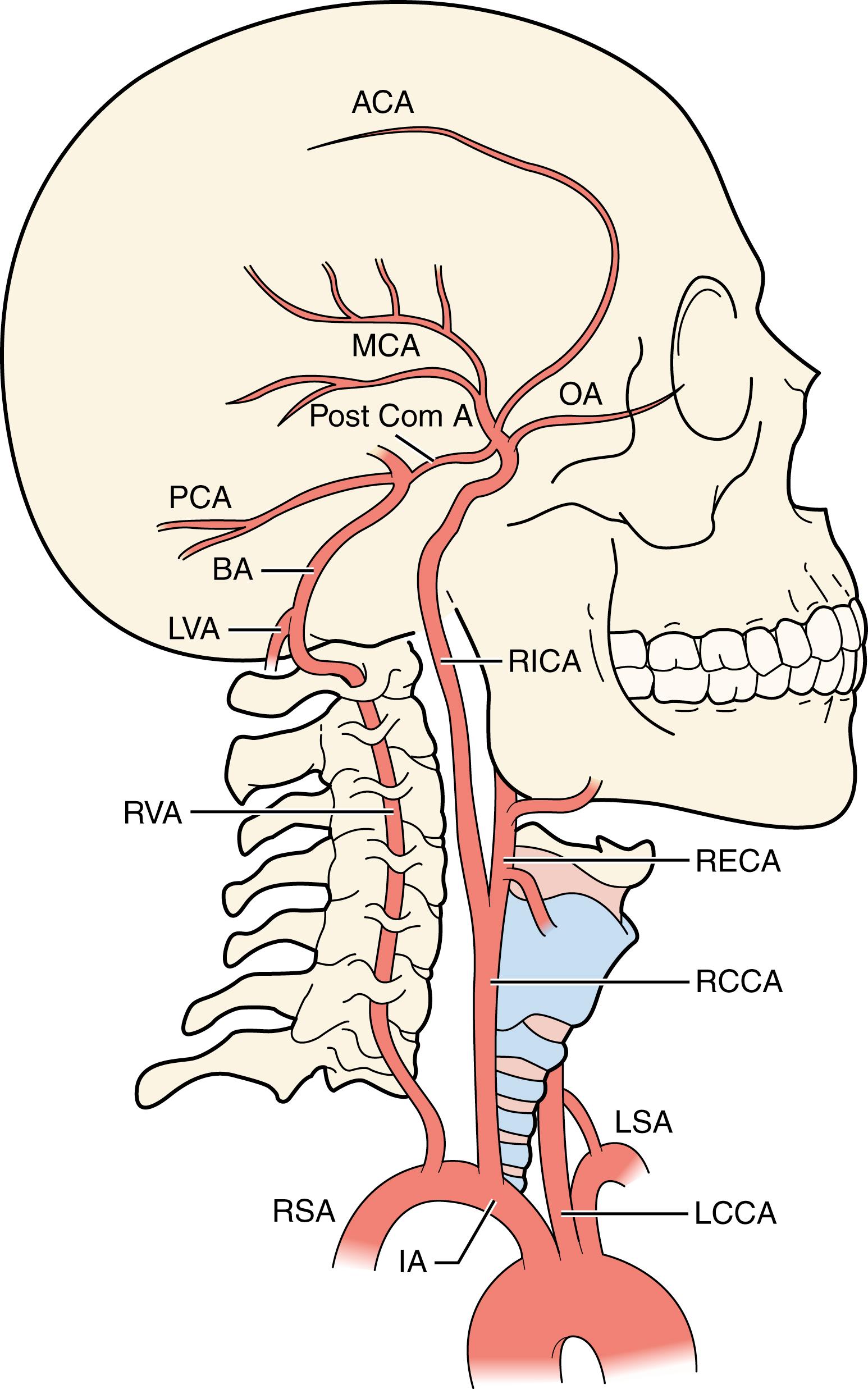
Atherosclerotic deposits are usually greatest in the distal common carotid artery, the bifurcation and the first few centimeters of the internal carotid artery, leaving the proximal common carotid artery and distal extracranial segment of the internal carotid artery relatively unaffected. In contrast, dissection typically occurs more distally along the course of the internal carotid artery close to the basis of the skull, where the artery is more vulnerable to traction during neck movement. Dissection is discussed in detail in Chapter 35 . Atherosclerosis is also seen at the origin of the common carotid artery and in the intracranial carotid syphon and cavernous portions of the internal carotid artery ( Fig. 22.2 ). The predilection for areas of flow disturbance supports the concept that wall shear stress plays an important role in the pathogenesis of atherosclerosis. Only about 30% of patients with stenosis of one carotid artery will have significant stenosis of the other carotid artery, and the degree of stenosis contralateral to a recently symptomatic carotid stenosis appears to be randomly distributed. When asymmetry of atheroma severity is found, early studies showed that atheroma is more common in the smaller carotid and the one more angulated. It is now recognized that hemodynamic factors related to the geometry of the carotid bifurcation play an important role in the development of carotid atheroma.
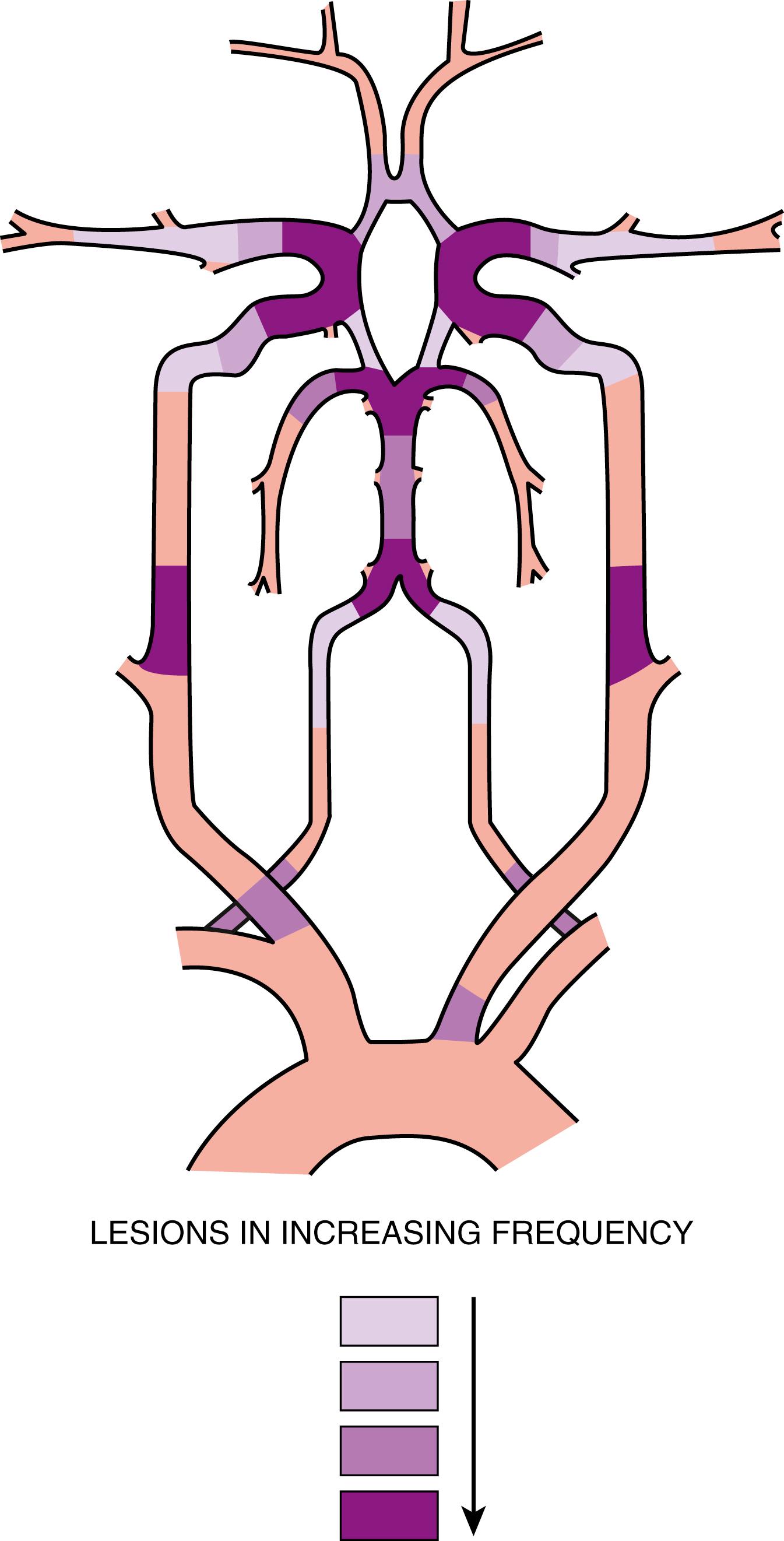
Atherosclerotic stenosis may on occasions develop swiftly over a few months. , Sudden stenosis can also develop very rapidly as a result of hemorrhage into the plaque. An apparent sudden progression of stenosis or the development of occlusion is, however, more commonly caused by thrombosis on the ruptured plaque surface. In general, the development of carotid stenosis from atherosclerosis is a very slow process, stretching over 50–60 years or more, starting with fatty streaks early in adolescent or early adult life and progressing to atheroma formation, and finally atherosclerosis and stenosis in middle or old age. Significant stenosis from atherosclerosis is rare under the age of 40, but earlier atheroma might still have a role to play in some younger cases of stroke, for example by promoting dissection. In many cases, after detection atherosclerotic stenosis may even remain static, especially if the lesion is heavily calcified, despite being hemodynamically significant.
In the Asymptomatic Carotid Stenosis and risk of Stroke (ACSRS) Study involving 1121 patients with asymptomatic carotid disease who were followed with 6-monthly duplex ultrasound examination for a mean duration of 4 years, the degree of stenosis remained unchanged in 76% of patients, stenosis progressed in 20% of patients, and regression of stenosis was observed in only 4% of patients. Male sex, diabetes mellitus, and active smoking were among the clinical predictors of progression identified in prospective ultrasound studies. ,
Statins reduce the progression of early-stage carotid atherosclerosis measured by intima-media-thickness on ultrasound. Small case-control and prospective cohort studies with ultrasound and magnetic resonance imaging (MRI) have shown that statins stabilize carotid plaque by reducing the lipid content (but not the total volume of developed plaques), decreasing plaque neovascularization and increasing the degree of calcification within the plaque. Patients not receiving lipid-lowering therapy in the ACSRS study were at increased risk of stenosis progression. Further, indirect evidence that intensive statin therapy slows plaque progression stems from randomized placebo-controlled secondary prevention trials: among patients with a history of cerebrovascular disease, the proportion requiring carotid endarterectomy or stenting was reduced from 2.3% in the placebo group to 1% in the group receiving 40 mg simvastatin per day in the Heart Protection Study. Similarly, 80 mg atorvastatin per day versus placebo reduced the carotid revascularization rate from 7.2% to 3.2% in patients with carotid atherosclerosis in the Stroke Prevention by Aggressive Reduction in Cholesterol Levels trial.
Pathologic studies indicate that the predominant mechanism underlying cerebral ischemia associated with carotid stenosis is rupture of the atherosclerotic plaque with subsequent embolism of locally formed thrombus or plaque debris to the brain. This has fostered the concept of the “vulnerable” or “unstable” plaque, which is one prone to rupture, as opposed to the “stable” plaque which may remain inert over many years. A growing body of data supports that inflammation plays a key role in the pathophysiology of atherosclerosis and particularly in plaque rupture. In the early phase of plaque formation, LDL accumulate in the vessel wall. These lipoproteins undergo oxidation and thereby acquire pro-inflammatory biologic functions unrelated to cholesterol homeostasis. Oxidized lipoproteins activate endothelial cells by inducing expression of adhesion molecules, chemokines, and pro-inflammatory cytokines. They also facilitate macrophage activation by triggering a variety of receptors of innate immune responses. Monocytes are attracted by chemokines and migrate into the plaque facilitated by expression of the vascular cell adhesion molecule-1 (VCAM-1) on endothelial cells. Within the plaque, monocytes differentiate into macrophages which take up oxidized lipoprotein particles and turn into so-called foam cells. Foam cells secrete pro-inflammatory cytokines and a large array of proteases, including matrix metalloproteases (MMPs), serine proteases, elastases, and cysteine proteases, that contribute to degradation of elastin and collagen in advanced lesions leading to destabilization of the plaque, thinning of its fibrous cap, and–ultimately–plaque rupture. Dysfunction of the endothelium is also an important contributor to the pathophysiology of atherothrombosis. When the plaque surface ruptures, platelets are exposed to sub-endothelial collagen, von Willebrand factor (vWF), and local agonists such as adenosine diphosphate (ADP). Activated platelets bind to fibrinogen and secrete various pro-coagulatory and pro-inflammatory factors, which mediate the binding of leukocytes and enhance fibrin formation. A local thrombus may form on the plaque surface, leading to embolization and occlusion of arteries in the eye or the brain manifesting as amaurosis fugax, TIA, or stroke.
A bruit in the neck is commonly encountered in routine clinical examination, found in as many as 4%–5% of the population aged 45–80 years. , A local cervical bruit can be detected in approximately 70%–89% of patients with an internal carotid stenosis of greater than or equal to 75% stenosis or less than or equal to 2 mm residual lumen. , In a population-based study of a stroke-free cohort of 686 subjects with a mean age of 68.2 years, the prevalence of carotid stenosis ≥60% detected by ultrasound was 2.2%; bruits had a prevalence of 4.1%. Bruit detection by auscultation had a sensitivity of 56%, a specificity of 98%, a positive predictive value of 25%, and a negative predictive value of 99%; the overall accuracy was 97.5%. Similar values demonstrating high specificity but low sensitivity were reported in a meta-analysis of 26 studies. Modern practice, therefore, dictates immediate imaging of the carotid as well as the brain in any patient with symptoms suggestive of ocular or cerebral ischemia, regardless of whether or not a carotid bruit is present.
The degree of luminal narrowing has traditionally been the most common parameter used to grade the severity of carotid disease. The standard method to determine degree of stenosis before the widespread availability of modern noninvasive techniques was the radiographic measurement of the diameter reduction of the carotid artery lumen on photographic film obtained after intra-arterial injection of contrast (catheter-based angiography). After the introduction of computerized imaging, the native radiographic x-ray image was subtracted from the contrast-enhanced view (digital subtraction angiography, DSA). An example of a very severe carotid stenosis shown by DSA is shown in Fig. 22.3 . Importantly, the two clinical trials that were pivotal in demonstrating efficacy of endarterectomy to prevent stroke in symptomatic carotid stenosis, the European Carotid Surgery Trial (ECST) and the North American Carotid Endarterectomy Trial (NASCET), used catheter-based angiography and DSA to measure stenosis severity. As atherosclerotic narrowing does not typically develop in a concentric way around the lumen, at least two perpendicular views, and often a third, oblique projection, were used in the trials as well as in clinical practice to show the maximum degree of narrowing. NASCET and ECST initially used different sites on the angiographic images to measure the normal reference diameter of the artery against which the diameter of the lumen at the site of maximum stenosis was compared: NASCET defined the diameter of the non-diseased ICA distal to the stenosis at a point where the artery became uniform in diameter as the reference, whereas ECST estimated likely normal diameter of the carotid bulb at the site of the stenosis by eye. Because the magnification of the angiograms was unknown, the degree of stenosis was expressed as percentage in diameter reduction using the formula ([1−minimum residual lumen/normal ICA diameter] × 100). A third trial, the Carotid And Vertebral Artery Transluminal Angioplasty Study (CAVATAS) compared the diameter at the site of the stenosis to the diameter of the common carotid artery. The different reference sites to measure the normal carotid diameter are shown in Fig. 22.4 . A lesion occurring in a typical location in the carotid bulb will result in a higher degree of stenosis using the ECST method than using the NASCET method, as the reference diameter of the normal artery is wider at the bulb than more distally along its cervical course. On average, 50% and 70% stenosis measured by the NASCET method are equivalent to 65% and 82% stenosis, respectively, measured by the ECST method. This methodologic difference partly explained the seemingly different long-term results of the two trials reported in 1998, with NASCET showing benefit of carotid endarterectomy for patients with 50% stenosis or greater, and ECST for patients with 80% stenosis or greater. However, re-assessment of ECST angiograms using the NASCET method demonstrated that the results of the two trials were broadly similar, the pooled analysis showing 4.6% absolute reduction in 5-year ipsilateral stroke-risk patients with symptomatic, moderate carotid stenosis (50%–69% NASCET narrowing), and 15.3% risk reduction in patients with symptomatic, severe stenosis (70%–99% NASCET narrowing, excluding patients with near-occlusion defined by collapsed lumen distal to the stenosis). Subsequently, the NASCET method has become widely accepted as the standard method to measure stenosis severity throughout the cerebral circulation.
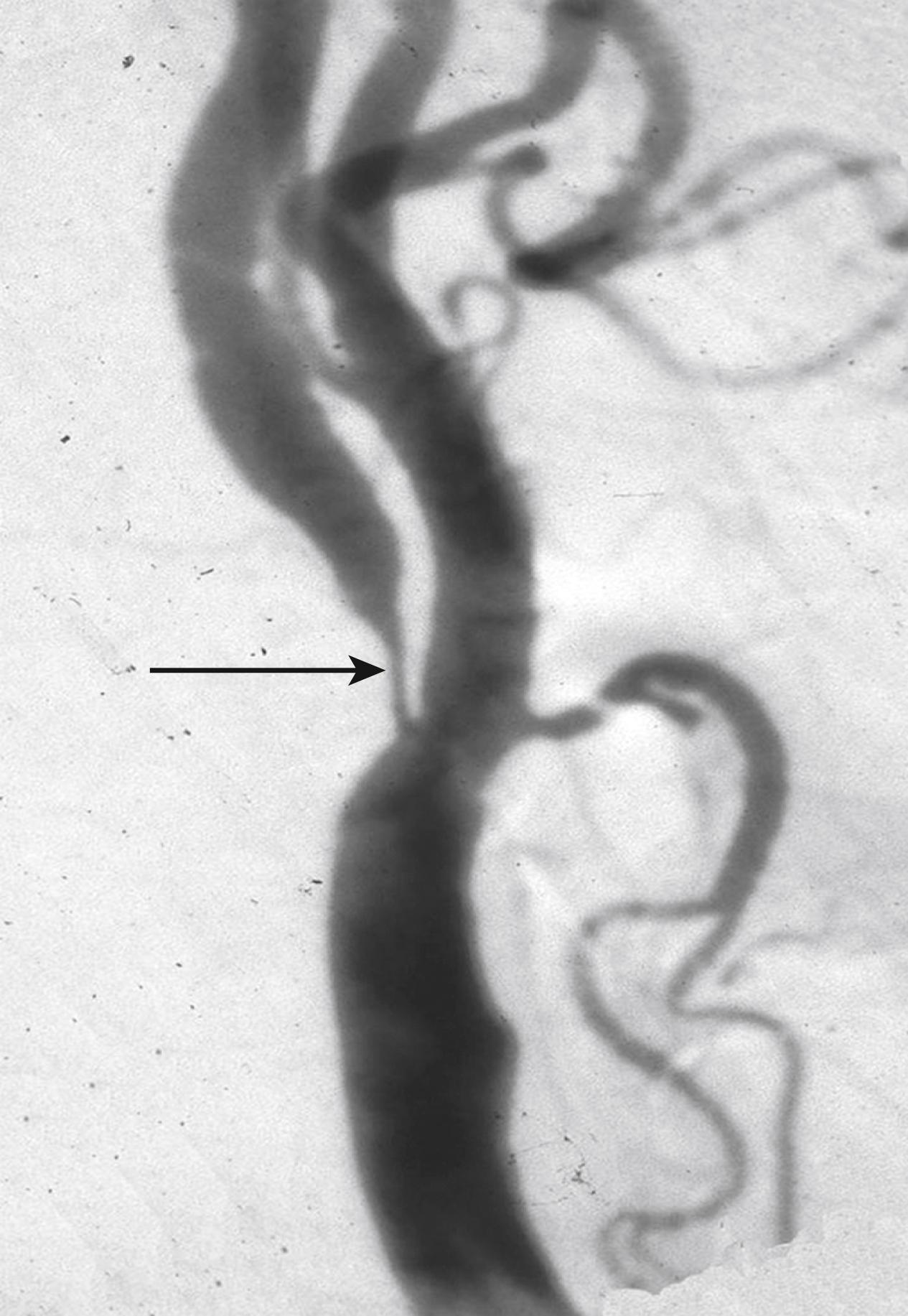
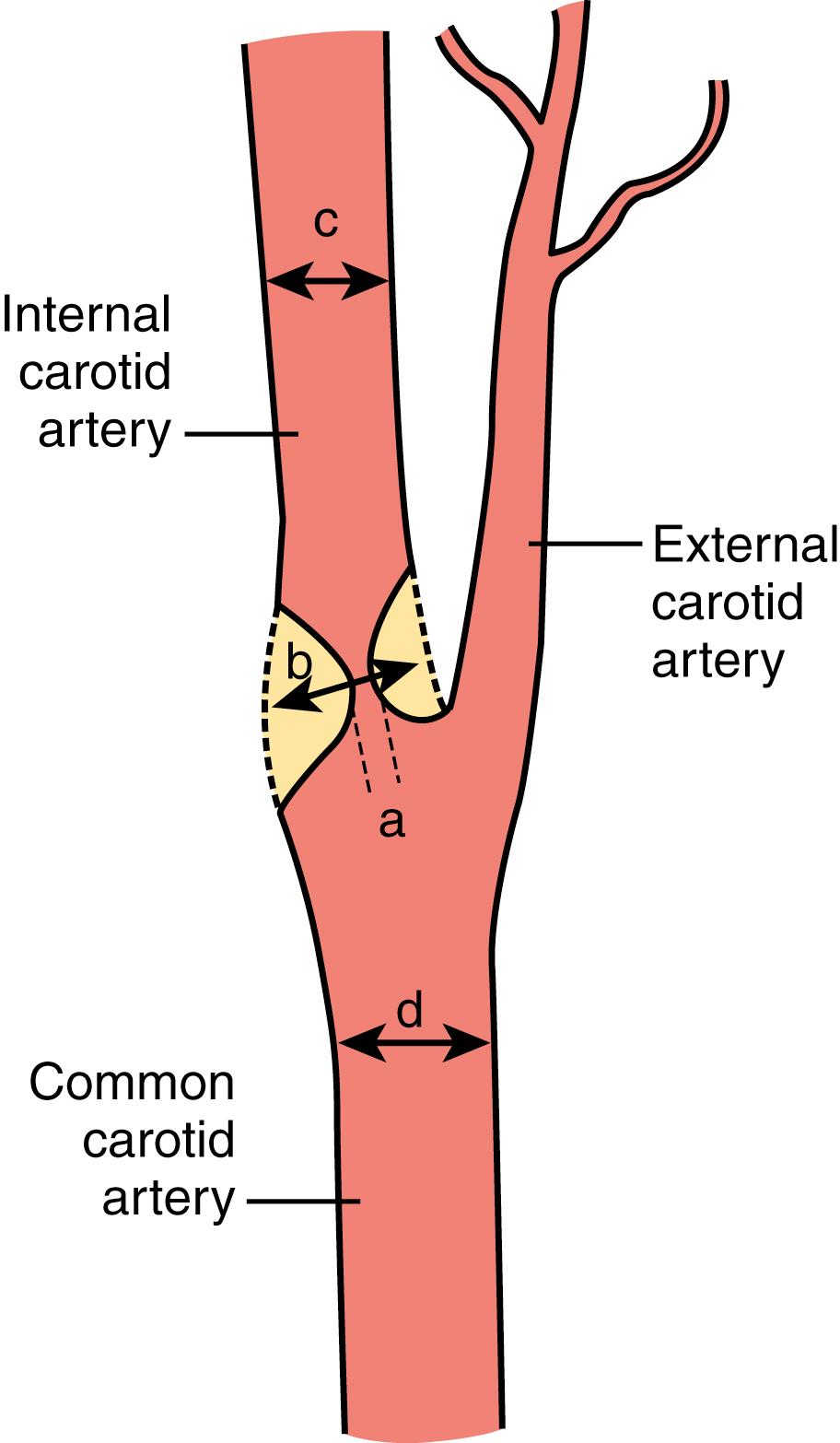
Since the time these trials were done, noninvasive imaging methods have largely superseded DSA. Using Doppler ultrasound and duplex sonography, the degree of stenosis is determined by a set of flow velocity criteria, most importantly the peak systolic and end-diastolic velocity in the ICA. Contrast-enhanced magnetic resonance angiography (CEMRA) or computer tomography angiography (CTA) after injections of intravenous contrast provide an anatomic image of the artery, albeit at lower resolution than DSA. It is standard practice to report the degree of stenosis measured by these noninvasive tests using measures regarded as giving results equivalent to the NASCET method used for DSA. Ultrasound laboratories use standard criteria that were originally calibrated against catheter-based angiographic measurements using the NASCET method. However, it should be borne in mind that few laboratories regularly audit the accuracy of their stenosis measurements given the risks of catheter-based angiography and there is a tendency for radiologists to believe they can simply “eyeball” a stenosis and report its severity, which can lead to errors in interpretation.
In a systematic review published in 2006, ultrasound, CTA, and MRA all had high sensitivity and specificity for diagnosing severe, 70%–99% carotid stenosis, with CEMRA appearing superior to ultrasound, CTA, and noncontrast MRA. In the interval since this report, the introduction of superior multislice CT machines has improved the resolution of cross-sectional CTA to the extent that, unless there is heavy calcification of the carotid plaque, the resolution of the lumen now exceeds that of standard CEMRA. None of the noninvasive techniques is entirely reliable and all suffer from technical limitations. Therefore, we recommend confirming the degree of stenosis by consistent findings from two different noninvasive tests in patients being considered for carotid revascularization, with DSA being reserved for patients in whom the noninvasive tests disagree or do not differentiate between total occlusion, near-occlusion, or high-grade stenosis.
The fact that the focus of carotid imaging has been limited to measuring the degree of stenosis is partly explained by the historical concept of flow impairment as the main mechanism of cerebral ischemia. Reduced cross-sectional area is the main factor in causing hemodynamic impairment. Other factors include the length of the stenosis, blood flow velocity, and blood viscosity. Limitation of intracranial flow requires a cross-sectional area of 4–5 mm 2 along a length of 3 mm ( Fig. 22.5 ); this area corresponds to less than 2 mm in the smallest 2D profile seen on an angiogram and occurs when 84% diameter stenosis and 96% area stenosis are achieved. Until these severities of stenosis are reached, any reduction in diameter is compensated for by an increase in blood flow velocity across the stenosis, which is the basis of using Doppler ultrasound to determine stenosis severity. Flow in the carotid artery is predominantly the result of ejection of a bolus of a column of blood from the heart and is influenced by the distensibility of the arterial wall, thus it is not laminar and does not obey Poiseuille’s law defining the relationship between flow velocity and radius. Only a small minority among symptomatic patients with carotid disease has such severely narrowed arteries that significant impairment of cerebral blood flow can be inferred, suggesting that other mechanisms may play a part in the strong association between stenosis severity and risk of recurrent stroke in medical-treated patients.
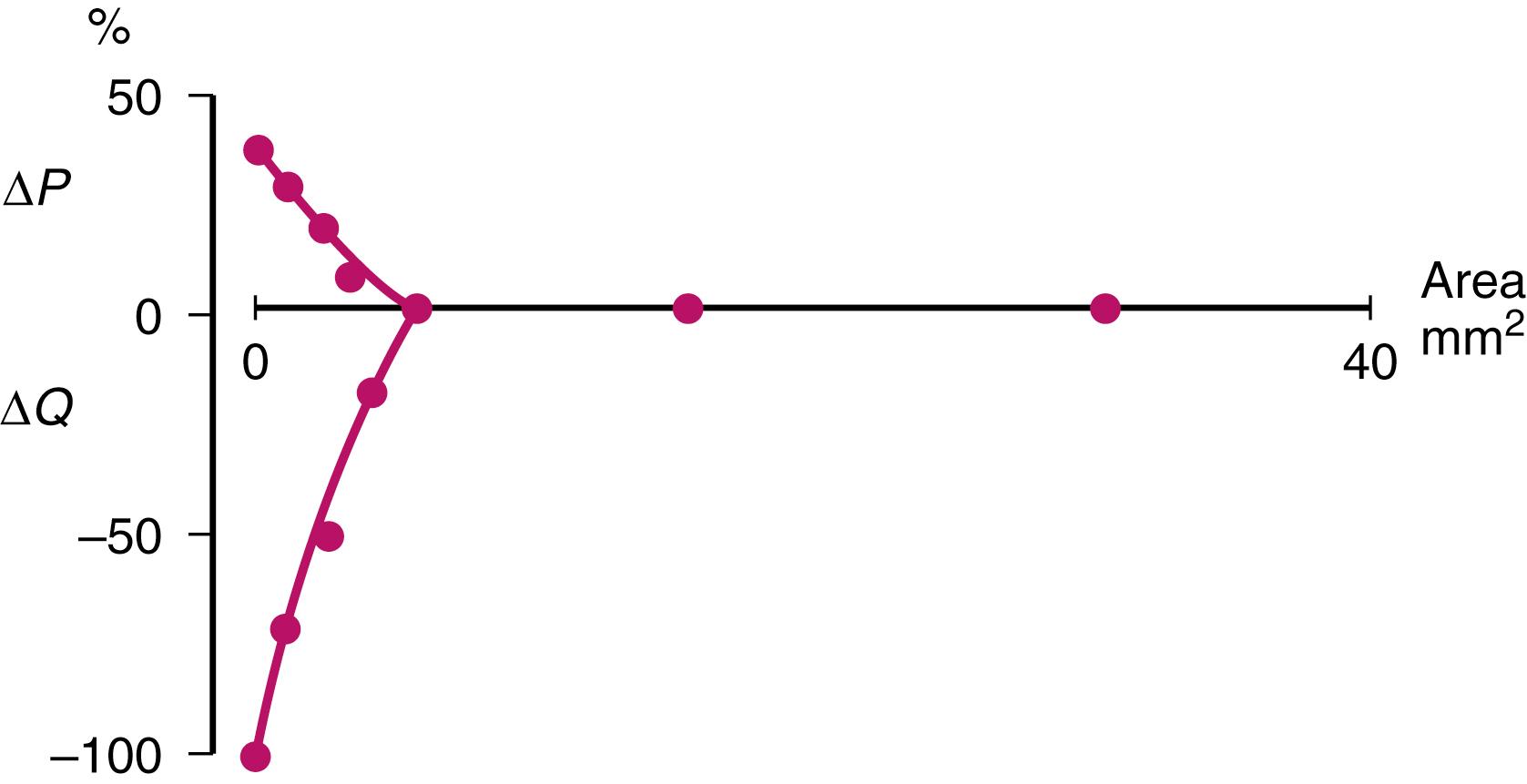
Another piece of evidence arguing against impairment of blood flow being the main mechanism of carotid stroke is that in ECST and NASCET, patients with near-occlusion or collapse of the carotid artery beyond the stenosis (generally patients with at least 95% stenosis), had a lower rate of stroke on medical therapy than patients with lesser degrees of stenosis and did not benefit from endarterectomy. This might in part reflect selection of patients in the trial who had sufficient intracranial collateral supply to the ipsilateral carotid territory to protect them against major stroke: major stroke was an exclusion criteria for the trials. An alternative explanation is that rather than directly affecting blood flow to the brain, increasing stenosis is associated with increasing turbulence beyond the stenosis, hence activating platelets and contributing to thrombus formation in symptomatic patients. In near-occlusion, flow velocity in the carotid artery is very low and hence might not activate platelets in the same way.
Despite the observation that the degree of stenosis was an important predictor of stroke risk under medical therapy and, hence, benefit from carotid endarterectomy in patients with recently symptomatic stenosis, the same relationship could not be demonstrated in trials of carotid endarterectomy for patients with asymptomatic disease. , Some authors explained this by the fact that mostly ultrasound instead of DSA was used for inclusion of patients in these trials, which is less accurate in determining the degree of stenosis. Certainly in a large community study of the prevalence of carotid stenosis there was a strong association between the severity of carotid stenosis measured on ultrasound and a history of previous stroke or TIA.
Given our current understanding that embolism is the most important mechanism causing stroke in patients with extracranial carotid stenosis, it is counterintuitive that luminal narrowing should be the only or, in fact, the most important determinant of stroke risk. Indeed, early studies already seemed to suggest that TIAs could be attributable to any degree of stenosis by means of microembolization. Evidence in support of this view arose from individual case reports, to which whole series of cases were later added. , In more recent research, carotid imaging has, therefore, focused on the morphology of the lesion and the composition of the atherosclerotic plaque. Already at the time of NASCET and ECST, irregular or ulcerated plaque surface was identified as an important predictor of stroke risk under medical therapy. Fig. 22.6 shows a large ulcer within a carotid plaque shown using the early catheter-based angiography technique common at that time. Ulcers are often found in surgical specimens. , Small ulcers are difficult to demonstrate angiographically, and considerable interobserver variation exists in the diagnosis of ulceration. As many as 40% of small ulcers may be missed on routine angiograms, but whether these are clinically relevant is uncertain.
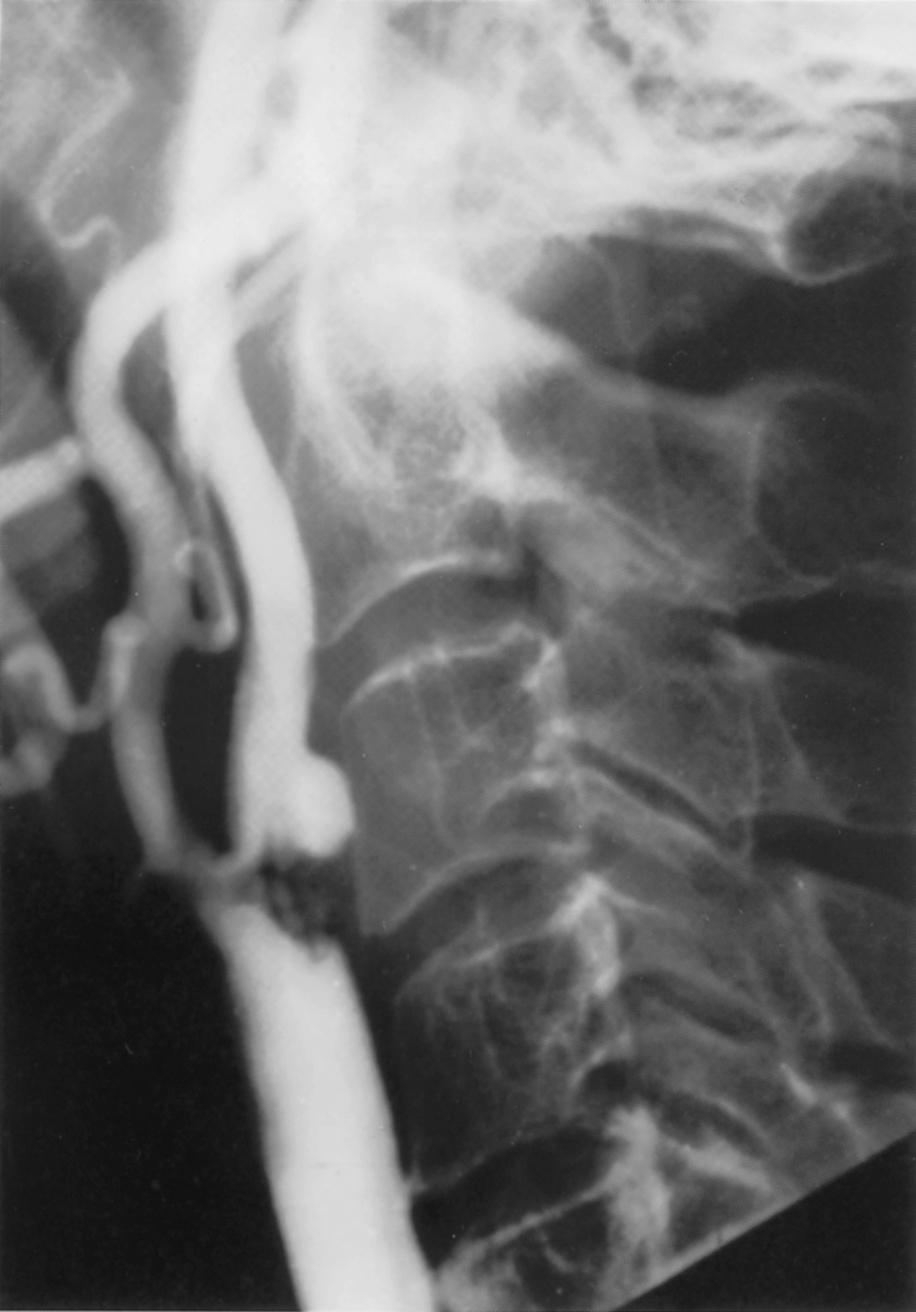
After the introduction of ultrasound and noninvasive angiography as the methods of imaging carotid atherosclerosis, the importance of ulceration was neglected and often not reported routinely, probably because earlier techniques had a low resolution and, therefore, were not very sensitive to ulceration. However, modern ultrasonography using B-mode imaging may be as sensitive as conventional angiography to the presence of plaque irregularity and ulceration ( Fig. 22.7 ). The use of a bolus of micro-bubble contrast during the examination increases the sensitivity of ultrasound to ulceration, while the yield of modern CTA is better than either conventional angiography or ultrasound because of the ability to view the entire circumference and length of the plaque. However, CEMRA remains poorly sensitive to ulceration compared to the other techniques.
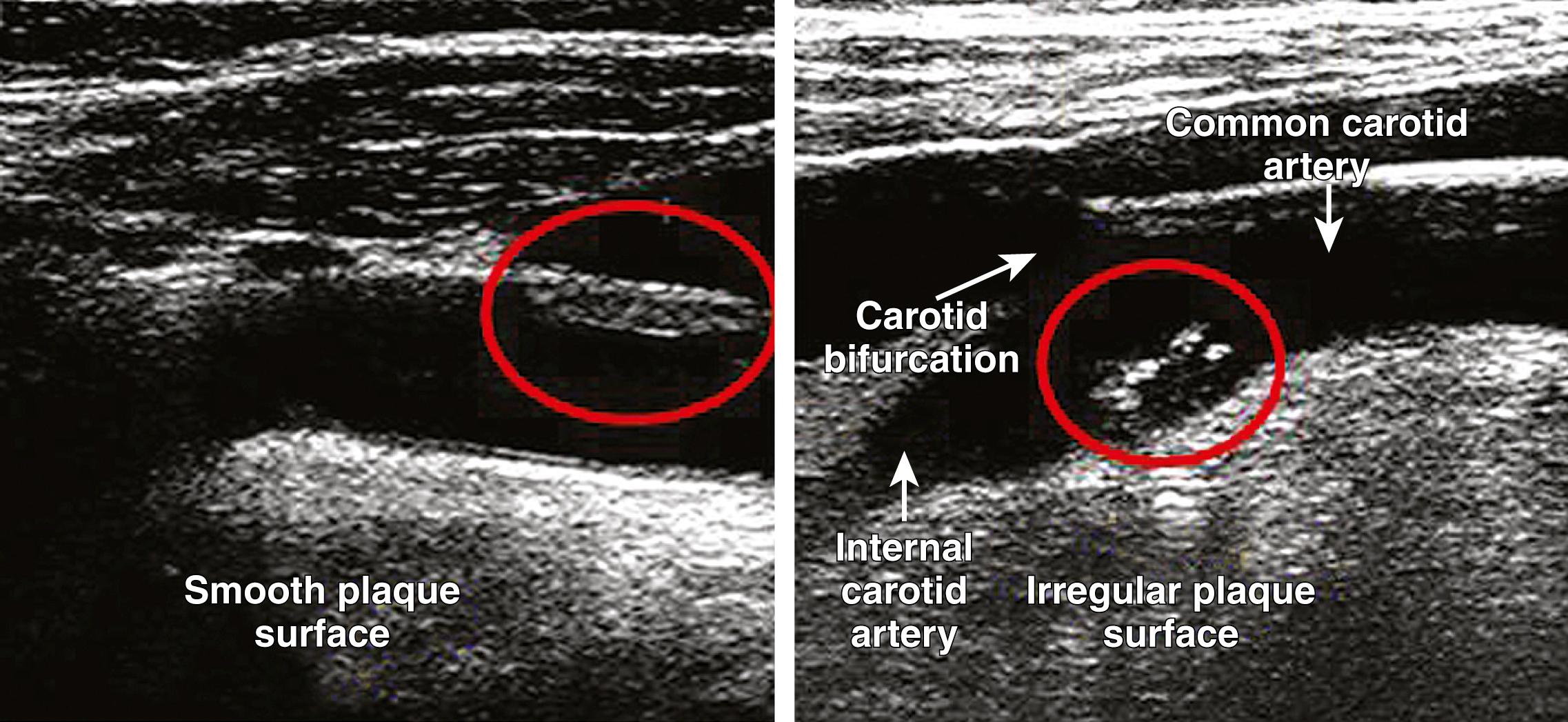
One advantage of the noninvasive diagnostic modalities in contrast to DSA is their ability to image the arterial wall and composition of the atherosclerotic plaque itself. On B-mode ultrasound of carotid bifurcation plaques, parts appearing dark (echolucent) correspond to necrotic areas with increased lipid content and plaque hemorrhage in histologic examination. , Interrater reliability on qualitative interpretation of carotid plaque morphology has in the past been poor. More recently, computerized analysis of B-mode ultrasound images has allowed for normalization against reference tissues and, therefore, a quantitative and objective assessment of plaque echolucency. The most common quantitative measure of plaque morphology on ultrasound is the so-called grayscale median (GSM), that is, the median grayscale value in the plaque. Several prospective studies have demonstrated that patients with echolucent plaques , and low GSM , are at increased risk of stroke, independently from degree of stenosis. More recently, an Echographic Risk Index based on a combination of degree of stenosis and proportion of echolucent area near the plaque surface, has been demonstrated to be superior to degree of stenosis and GSM alone in predicting cerebrovascular ischemic events and brain infarction on MRI. Echolucent plaque area at the surface also independently predicted stroke in the ACSRS study.
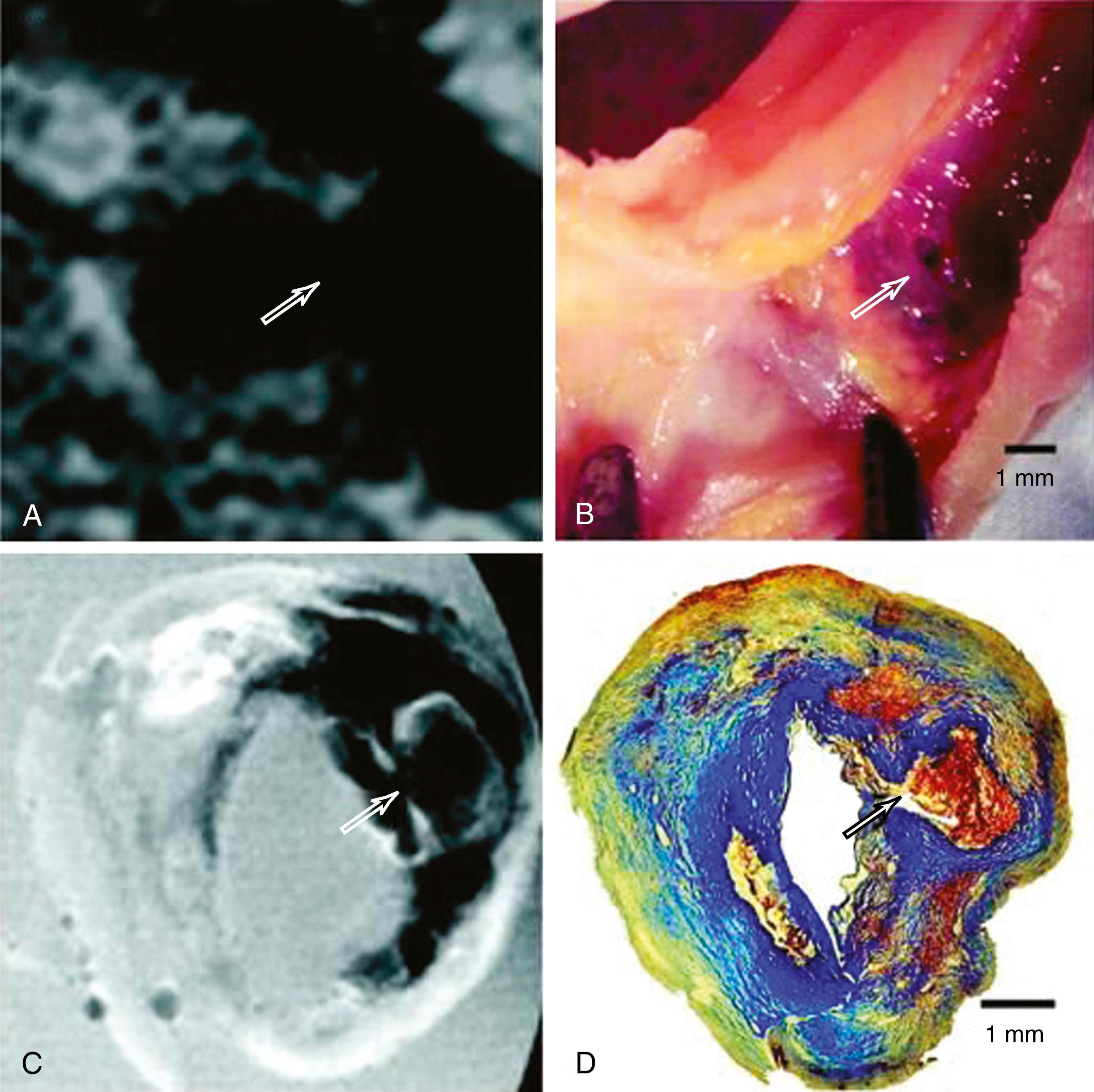
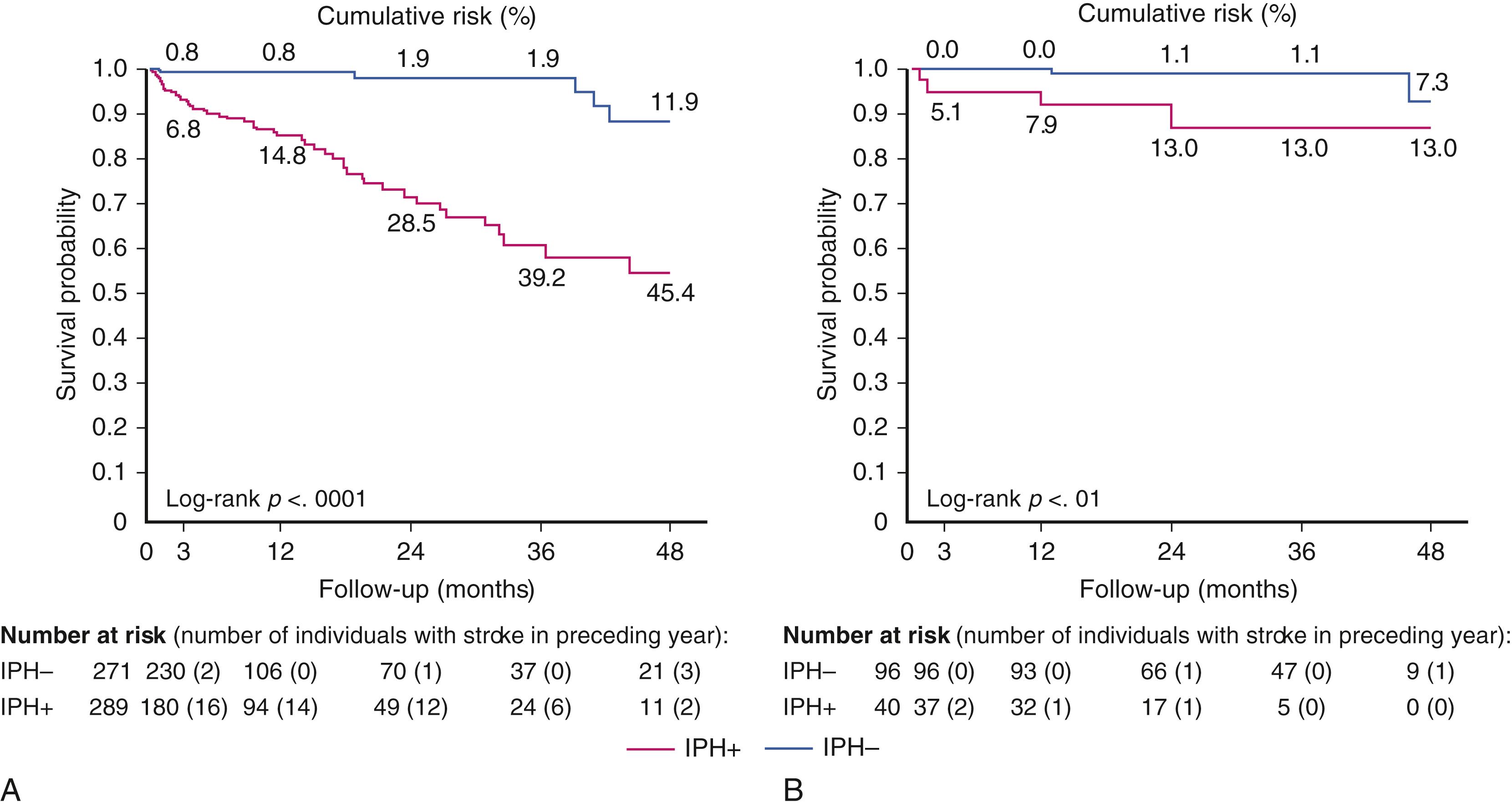
MRI is able to visualize key structural correlates of carotid bifurcation plaque instability, including lipid-rich necrotic core, intra-plaque hemorrhage (IPH), and disruption of the luminal surface ( Fig. 22.8 ). , These findings are more common in patients with symptomatic than with asymptomatic stenosis. The presence of these features fundamentally alters the biology and natural history of carotid atherosclerosis: plaques with MRI evidence of IPH may exhibit rapid progression despite statin therapy, and are associated with a 6- to 11-fold increase in risk of ipsilateral cerebrovascular events compared to plaques without IPH ( Fig. 22.9 ). , In these studies, IPH was a better predictor of recurrent events in recently symptomatic patients than any other clinical risk factor.
Positron emission tomography (PET) studies have shown that radiolabeled 18F-fluorodeoxyglucose ( 18 F-FDG) accumulates in carotid plaques, particularly at sites with high inflammatory activity. Increased update of 18 F-FDG appears to be associated with recent ipsilateral cerebral ischemia irrespective of the degree of carotid stenosis. In a prospective cohort study of 60 patients with symptomatic carotid stenosis higher plaque FDG uptake at baseline was also associated with increased risk of recurrent stroke, independently from degree of stenosis.
On a physiologic level, hemodynamic impairment resulting from carotid stenosis may best be understood as a reduced capacity of the cerebral vasculature to increase flow when required, rather than as a reduction in blood flow per se. This autoregulatory capacity can be assessed as the so-called cerebrovascular reserve, that is, the increase in blood flow to stimuli such as carbon dioxide (CO 2 ). This has been most often assessed by measuring blood flow in the middle cerebral artery (MCA) by transcranial Doppler (TCD) or single-photon emission CT (SPECT), before and after induction of hypercapnia by breath-holding, inhalation of hyperbaric CO 2 , or administration of acetazolamide. PET can also be used to directly identify patients with exhausted cerebrovascular reserve by demonstrating increased oxygen extraction in areas of impaired blood flow. Prospective cohort studies demonstrated that reduced cerebrovascular reserve measured by TCD was associated with an increased risk of recurrent stroke or TIA independently from degree of stenosis, both among patients with symptomatic and asymptomatic carotid stenosis. , A meta-analysis of 13 prospective studies found that reduced cerebrovascular reserve was associated with an almost fourfold increase in stroke risk (odds ratio [OR], 3.9; 95% confidence interval [CI], 2.0–7.5), without material differences in the strength of this association between patients with symptomatic and asymptomatic carotid stenosis. However, it has not been shown that measuring cerebrovascular reserve consistently identifies patients who will benefit from revascularization. In the most recent trial of extracranial–intracranial bypass surgery, patients with symptomatic carotid occlusion and exhausted cerebral reserve identified by PET scanning had no improvement in cognitive function after bypass surgery and had a significantly higher rate of stroke than those treated with medical therapy alone. ,
TCD is able to detect emboli passing along the insonated portion of the MCA as short, high-intensity signals (HITS) which are characterized by a chirping sound. The presence of these so-called microembolic signals during a 1-hour TCD recording at baseline predicted the risk of stroke, and stroke and TIA combined, in a cohort of 200 patients with symptomatic carotid stenosis. The association with cerebrovascular events was independent from other risk factors including degree of stenosis. A systematic literature review and meta-analysis published in 2009 found that the presence of HITS increased the risk of stroke in patients with symptomatic carotid stenosis by an OR of 9.6 (95% CI, 1.5–59.4) and in patients with asymptomatic carotid stenosis by an OR of 7.5 (2.2–24.9). This finding was later confirmed in a large prospective cohort of patients with asymptomatic carotid stenosis (Asymptomatic Carotid Emboli Study, ACES). However, a more recent systematic review from 2016 concluded that the evidence was relatively weak and the sensitivity of a positive TCD examination was poor. Thus, the clinical relevance of microembolic signals has not been convincingly established.
In summary, qualitative, quantitative, and functional imaging of the carotid plaque and assessment of microembolization hold promise of identifying patients with carotid disease at risk for stroke. This evidence is further discussed in the section “Stroke Risk in Patients with Carotid artery Disease.” Whether these tests add to clinical decision making in improving the selection of patients who benefit from carotid revascularization remains to be proven in randomized controlled trials.
In ethnically mixed populations, relevant stenosis of large intracranial arteries (including the intracranial internal carotid, middle cerebral, and vertebrobasilar arteries) has been identified as the cause of acute ischemic stroke in as many as 8% of patients. The intracranial ICA, especially the cavernous part, is the second site of predilection of carotid atherosclerosis, but in Western populations, intracranial stenosis is far less common than at the extracranial bifurcation. In contrast, symptomatic intracranial atherosclerosis is generally reported as being much more common among Asians, Hispanics, and blacks than among white Caucasian patients, and its incidence exceeds that of symptomatic extracranial atherosclerosis in those populations. Although recognized as an important cause of stroke in white Caucasians in postmortem studies performed early in the 20th century, intracranial atherosclerosis, therefore, came to be considered a rare cause of stroke in white Caucasian races compared to carotid stenosis. However, more recent postmortem studies and community imaging studies have once again drawn attention to the frequent finding of intracranial carotid atherosclerosis in white Caucasian patients with stroke, suggesting that it may be a more common cause of stroke than currently recognized. , Metabolic syndrome and diabetes mellitus as risk factors are more strongly associated with intracranial disease than with extracranial disease. ,
There is no clear evidence to indicate that a siphon stenosis (tandem stenosis) increases the risk of occlusive thrombosis of a stenosis at the ICA origin or vice versa, nor do tandem stenoses appear to raise the risk involved in carotid endarterectomy, although most trials of carotid revascularization excluded patients with significant tandem lesions.
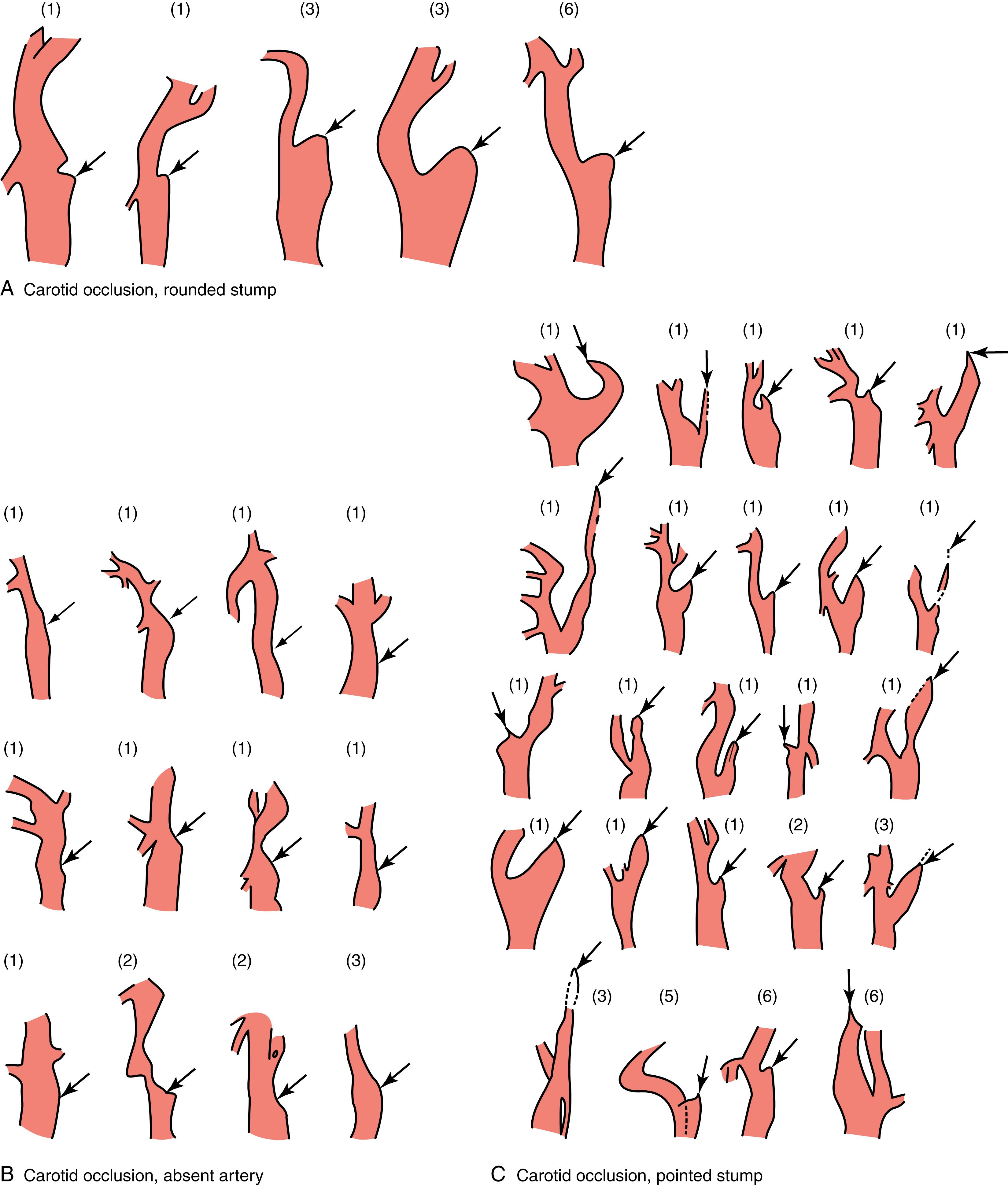
Anterograde and retrograde secondary thromboses of the intracranial ICA are subjects for which only scanty literature exists. In general, occlusive thrombus arising on atheromatous plaque at the carotid bifurcation extends rapidly from the origin of the internal carotid artery at least to the origin of the ophthalmic artery, the first branch beyond the bifurcation. The distal intracranial internal carotid may then remain patent either as a result of reversed flow in the ophthalmic artery maintained by ECA collaterals or reversed flow down the distal internal carotid artery to the ophthalmic artery. However, in many cases, angiography will show an occlusion extending all the way from the bifurcation to the tip of the intracranial carotid artery and even beyond into the MCA. It is then difficult in such a case to know whether the thrombosis started at the bifurcation and extended intracranially or vice versa. Pathologically well-studied instances of intracranial ICA thrombotic occlusion are rare, and the role of anterograde or retrograde extension of an associated thrombus is unclear. , Retrograde thrombi developing down to the ICA origin are probably not rare, and the angiographic appearance of the proximal end of carotid occlusion does not predict the age of the occlusion, at least within the first 6 days after stroke onset ( Fig. 22.10 ). Discontinuous occlusions (i.e., the presence of a patent segment of the ICA between the extracranial occluded ICA and the intracranial ICA) have been reported at autopsy, and it may be difficult or impossible to decide whether the distal plug is the result of local thrombosis or embolic, the lack of knowledge of which poses some difficulties for surgeons or interventionalists attempting removal of what is inferred to be an acute occlusion at the origin or the siphon of the ICA. , , ,
Progressive occlusion of the siphon and distal intracranial ICA has been reported in a setting of essential thrombocythemia and in mucormycosis, and encasement by nasopalatine carcinoma (carcinoma of the fossa of Rosenmüller) and inflammatory pseudotumors is also well-known. Progressive occlusion of the distal intracranial ICA, often bilateral, is also a feature of moyamoya disease, where it is associated with contiguous involvement of the MCA and the development of characteristic basal and pial collateralization. In such cases, the proximal internal carotid artery usually remains patent, but because of reduced run-off intracranially will often appear small or collapsed throughout its course. Similar collapse of the proximal internal carotid artery can also occur in cases of severe atheromatous stenosis of the cavernous carotid artery. Moyamoya disease is described in detail in Chapter 40 .
Described in more detail in Chapter 46, Chapter 47, Chapter 48 , transcranial ultrasound and noninvasive angiography (MRA and CTA) have advanced enough to allow investigation of the orbit and siphon in many individuals, decreasing the former dependency on catheter angiography. The calcium score correlates with atheromatous disease severity in the siphon. ,
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here