Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Portal vein embolization (PVE) is the most common portal flow modulation procedure performed preoperatively to reduce the risk of extensive liver resection in patients with a small anticipated future liver remnant (FLR). PVE redirects portal blood flow to the intended FLR and is expected to produce a shift in hepatic functional reserve resulting from atrophy of the embolized liver and compensatory hypertrophy of the FLR (see Chapters 5 and 6 ). PVE can reduce perioperative morbidity and allow for safe, potentially curative hepatectomy for patients previously considered ineligible for resection because of a small anticipated FLR , , (see Chapters 101 , 102A , and 102B ). Therefore PVE is currently performed at many hepatobiliary centers worldwide before major hepatectomy. In this chapter, indications, technical details and clinical outcomes of PVE are reviewed and discussed.
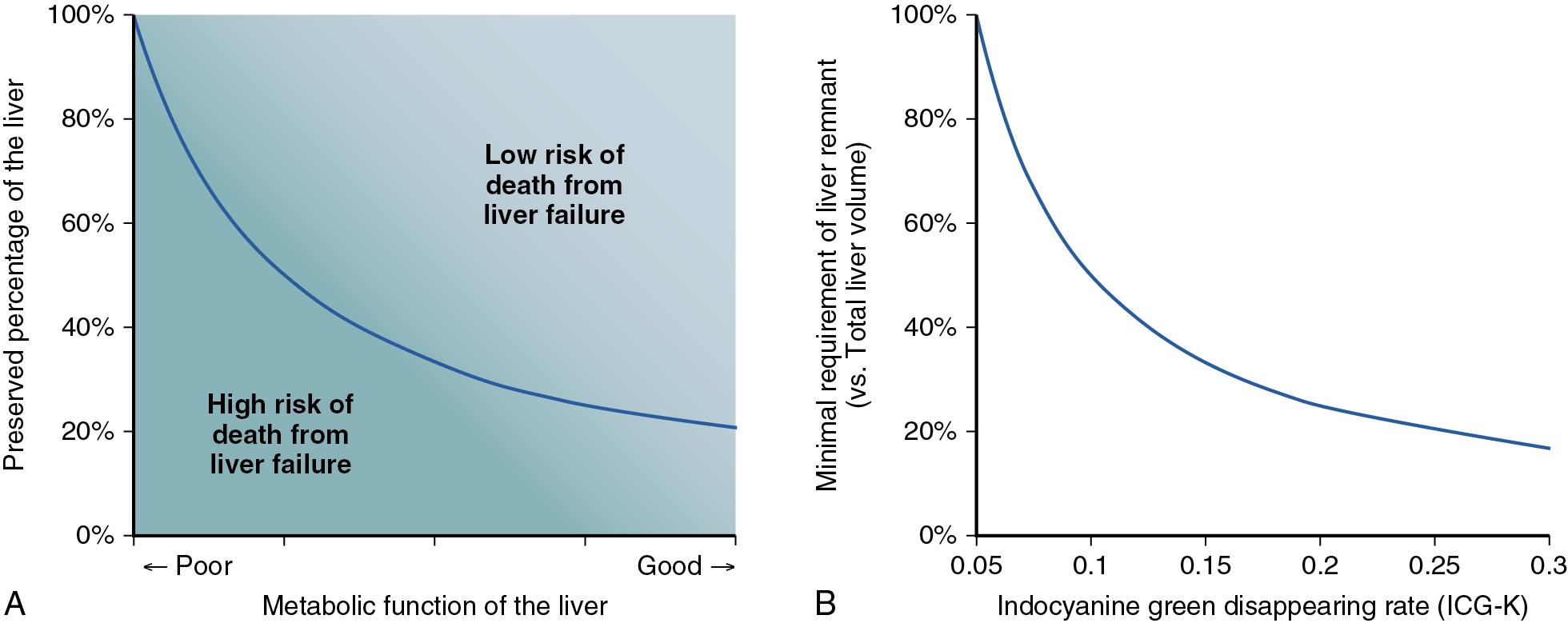
The safety of extensive liver resection is highly dependent on the net functional reserve of the FLR. The FLR volume and the regenerative capacity of the liver , are important predictors of postoperative morbidity and mortality after major hepatectomy. Therefore systematic volumetry and estimation of hepatic functional reserve are needed to assess the risk of major hepatectomy ( Fig. 102C.1 ).
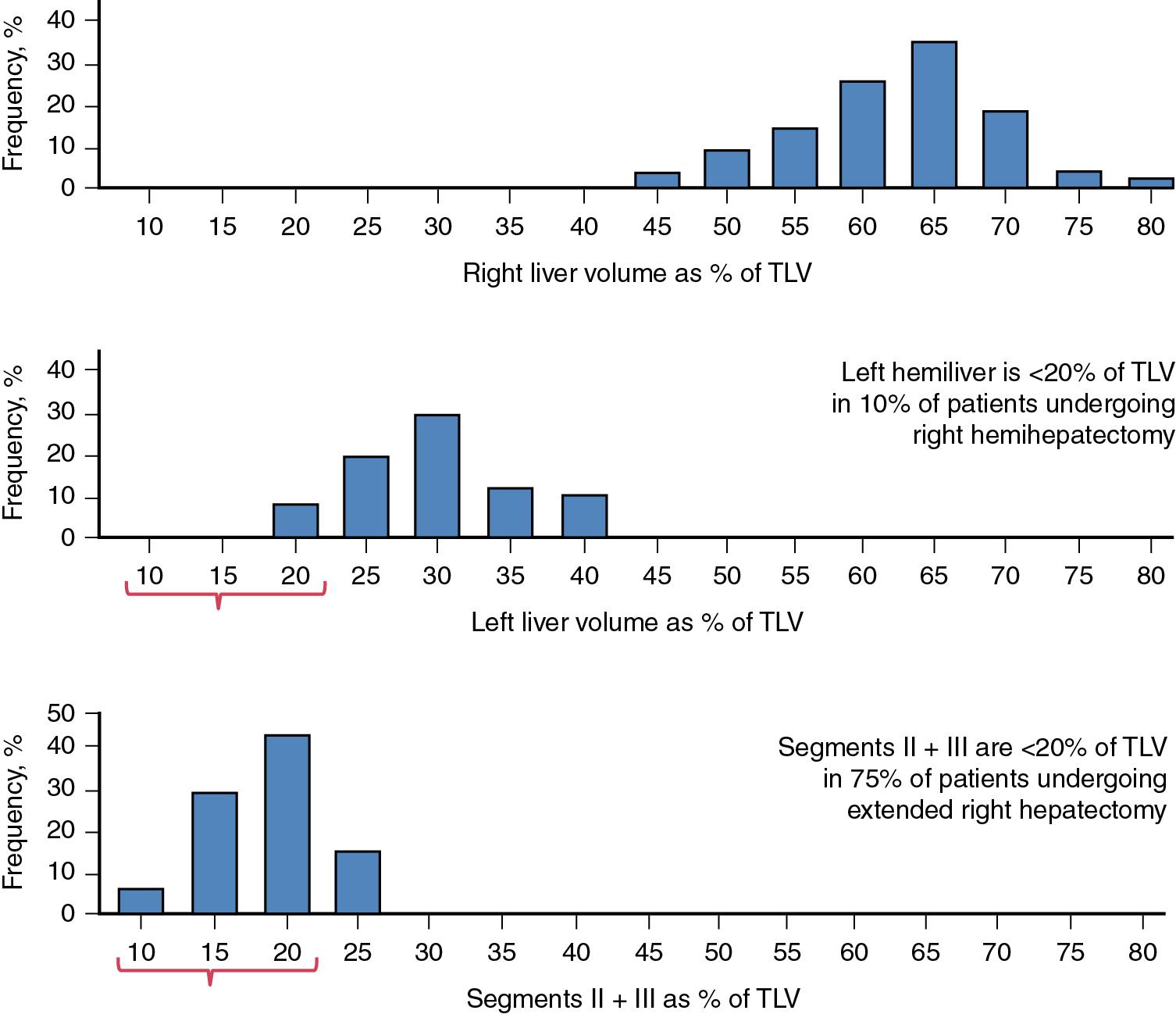
A previous anatomic study revealed that only 25% of patients would have a sufficient FLR volume (i.e., >20% of the estimated standard liver volume) for safe extended right hepatectomy; the same study revealed that 90% of patients would have a sufficient FLR volume for right hemihepatectomy ( Fig. 102C.2 ). Thus routine volumetry using a validated method is recommended, especially in patients for whom extended right hepatectomy is being considered.
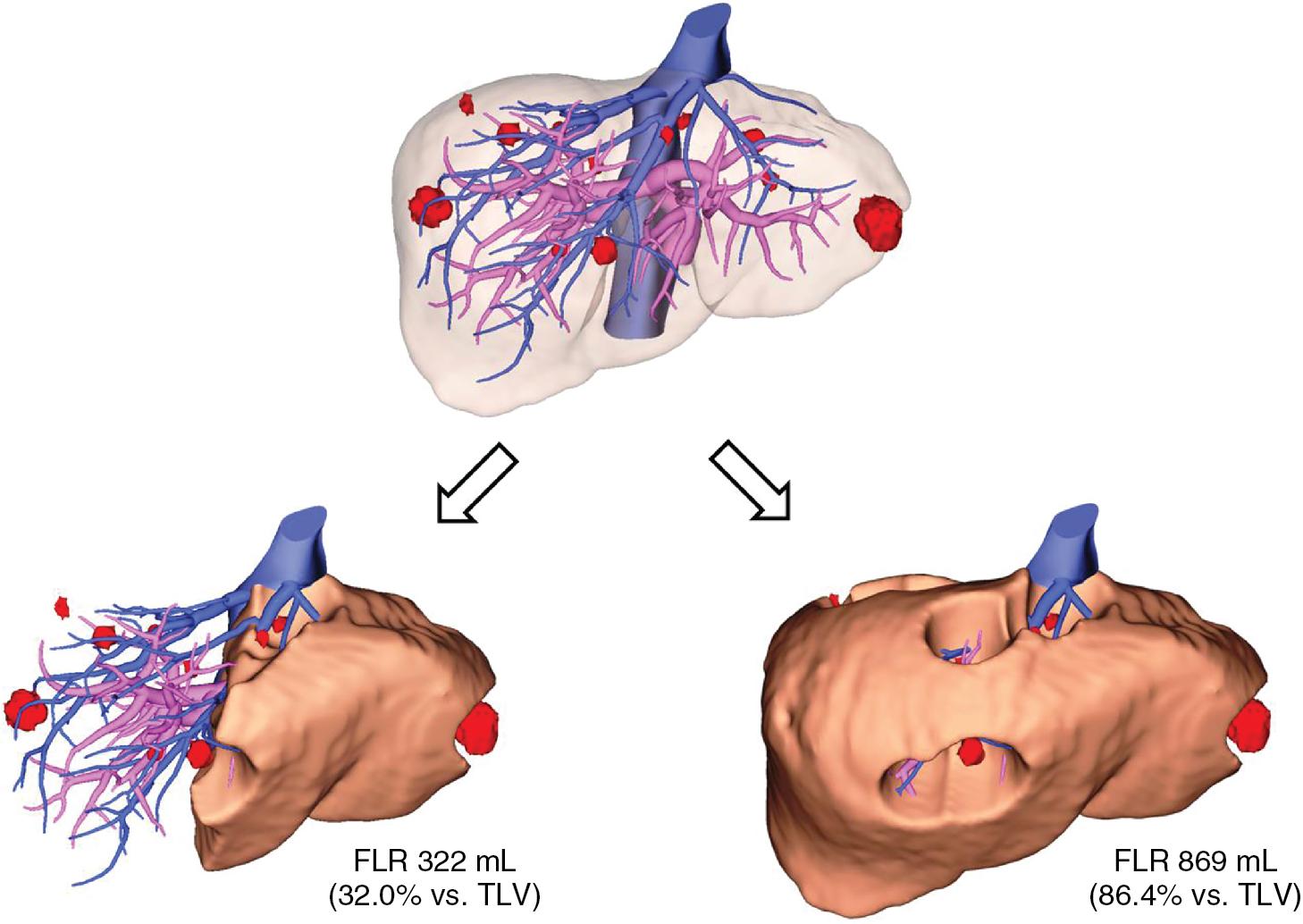
For volumetric calculations, the FLR volume should be calculated as the absolute volume of the fully functioning part of the liver after resection; in other words, the part of the liver that will have adequate inflow and outflow. When a hepatic vein draining a specific part of the liver is removed, the corresponding part of the liver will become congested and will atrophy as a result of losing its normal function. , Therefore, in calculating the FLR volume for patients undergoing extended right hepatectomy, in which the middle hepatic vein will be removed or compromised, segment IV volume should not be included. This is because in the absence of middle hepatic vein drainage, most of segment IV will become congested and lose its normal function unless an anatomically significant umbilical fissure vein or a segment IV vein directly draining into the left hepatic vein is present.
Several methods have been described to evaluate the risk of major hepatectomy based on the volume of the FLR. In Japanese high-volume centers, volumetry is currently performed using three-dimensional (3D) image reconstruction systems ( Fig. 102C.3 ), which allow direct measurement of the liver parenchymal volume per se, excluding vascular structures and tumors; they provide reliable results based on semiautomatic calculations of the vascular territories. Conventionally, the minimum FLR volume required was calculated on the basis of the functional reserve of the liver determined by the indocyanine green (ICG) clearance test, and recent reports have clarified that an estimated ICG clearance rate of the FLR (ICG-Krem) of 0.05 or greater is predictive of the maximum limit of liver resection. , Overall, it is important to obtain a reliable result for the required FLR volume when determining the surgical indications based on the safety limit for hepatectomy. Although the 3D simulation technique mentioned earlier has recently been reported to allow precise volumetry of the liver and direct measurement of the metabolic function of the liver through an ICG clearance test and is a reliable method, the limitations of this approach include the requirement of high-quality thin-slice CT images for the 3D image reconstruction and ICG clearance test is not always available in many countries.
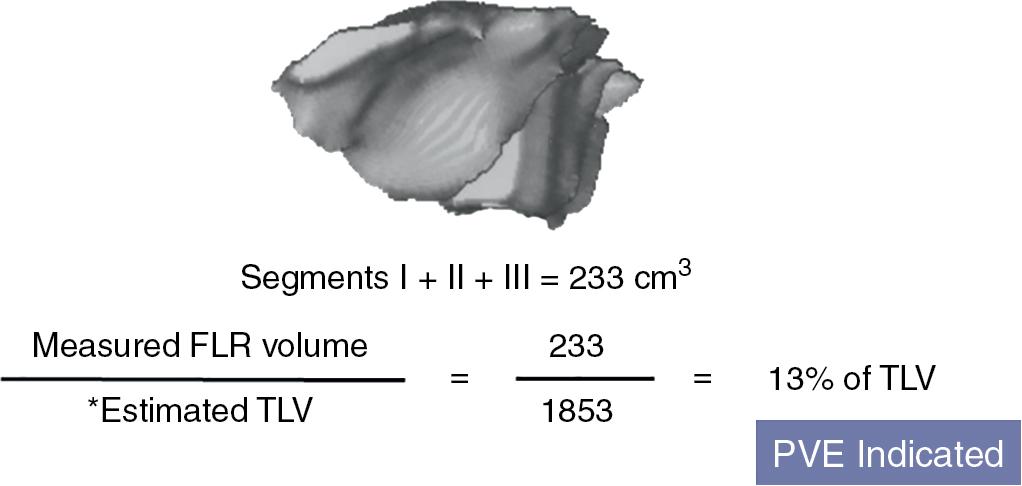
At The University of Texas MD Anderson Cancer Center, the standardized FLR (sFLR) volume is used to evaluate regeneration after PVE and to estimate the surgical risk. The sFLR is defined as the measured FLR volume divided by the estimated total liver volume (TEL) ( Fig. 102C.4 ). At MD Anderson Cancer Center, the FLR volume is obtained from 3D reconstruction of the CT images of the FLR. TEL is estimated using a formula that relies on the linear correlation between the total liver volume and body surface area (BSA), as follows:
This method of estimating the FLR has several advantages: (1) It avoids direct 3D computed tomography measurement of the portion of the liver to be removed, which might not reflect adequate hepatic function (dilated bile ducts, hypertrophy from steatosis, or atrophy from cirrhosis). (2) It avoids the cumulative error associated with the measurement of multiple tumors that need to be subtracted from the total liver volume. (3) It is based on the actual hepatic physiologic needs of the individual patient as reflected in the patient’s BSA. (4) The standardization corrects for patient size: small patients need a small liver remnant, and large patients need a large liver remnant.
In a series of 301 patients without chronic liver disease or hepatic injury undergoing extended right hepatectomy, an sFLR of less than 20% was a risk factor for postoperative liver insufficiency and 90-day postoperative mortality. However, functional reserve per liver volume depends on the quality of the hepatic parenchyma. Therefore different sFLR cutoff values apply for safe liver resection in patients with hepatic injury or chronic liver disease. Patients with chemotherapy-induced liver injury after prolonged chemotherapy (e.g., >3 months) require an sFLR of approximately 30%, and patients with cirrhosis require an sFLR of at least 40%. , , ( Fig. 102C.5 )
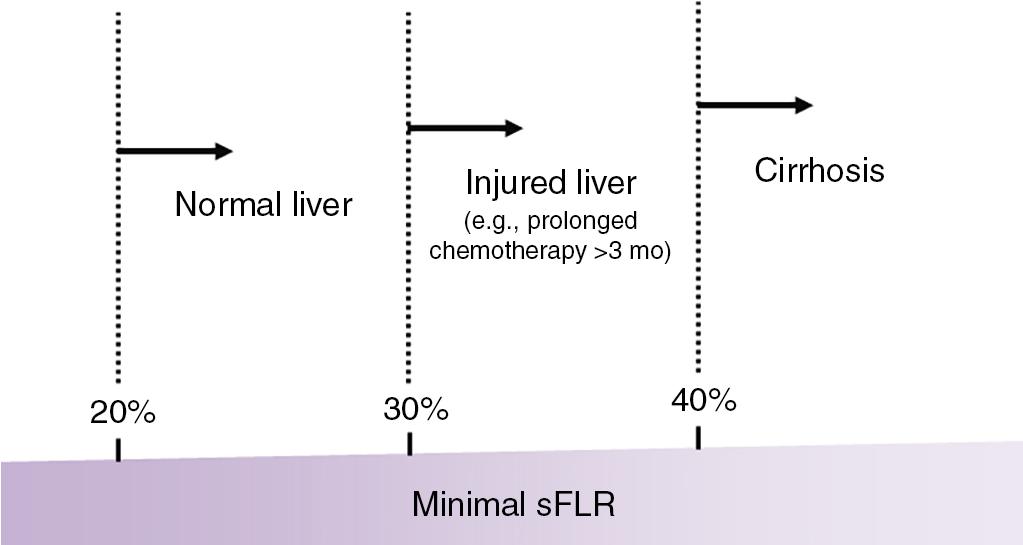
PVE should be considered when the FLR or sFLR is not large enough to permit safe hepatectomy given the quality of the patient’s underlying liver and the planned extent of surgery.
There are two absolute contraindications for PVE: established portal hypertension and extensive tumor thrombus of the ipsilateral portal vein. For patients with mild subclinical portal hypertension, PVE is not always contraindicated. However, PVE may increase portal vein pressure and worsen the clinical symptoms associated with portal hypertension (e.g., esophageal bleeding). Therefore considerable attention should be given to patients requiring PVE who have clinical evidence of portal hypertension.
PVE can be performed by any of three standard approaches: the transileocolic venous approach, transhepatic contralateral approach (percutaneous portal vein access via the FLR), and transhepatic ipsilateral approach (percutaneous portal vein access via the liver to be resected). It is important to note that the approach to the portal vein is chosen at the discretion of the operator, and the decision may be based on multiple factors, including the extent of the embolization and surgery, the operator’s preference for a specific embolic agent, tumor burden within the liver, and the operator’s level of experience with one technique over another.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here