Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
This work was supported in part by an award from Soy Health Research Program (SHRP, United Soybean Board, Chesterfield, MO, USA), University of South Carolina School of Medicine Research Development Fund (USC SOM RDF, Columbia, SC, USA), South Carolina Spinal Cord Injury Research Fund (SC SCIRF-2015-I-01, Columbia, SC, USA), and earlier grants (R01 CA91460 and R01 NS057811) from the National Institutes of Health (Bethesda, MD, USA).
The Krüppel-like factor (KLF) family in humans includes 17 members, which are transcription factors and have homology with their founding member Krüppel (“crippled” in German), the deletion of which is associated with a crippled-like phenotype in Drosophila melanogaster . It is now well recognized that some of the members of the KLF family have very important roles in human health and diseases including tumor progression or suppression . As such, there is a great interest in the deeper understanding of their structural features, regulation of expression, and molecular mechanism of action for controlling the growth or death of malignant neuroblastoma, which is a deadly solid tumor occurring mostly in children. Among all the members of the KLF family, now KLF4 is of special interest because its transcription regulatory roles (as transactivator or transrepressor) may result in oncogenic or tumor suppressive effects ( Fig. 16.1 ). Dual roles of KLF4 as oncogene and tumor suppressor are due to its interaction with the binding elements on promoters of the target genes in different cellular contexts . Results from recent studies strongly suggested that indirect upregulation of KLF4 was associated with suppression of growth, invasion, and metastasis in human malignant neuroblastoma cells in vitro and in vivo . A thorough understanding of the level of expression and function of KLF4 may help in the diagnosis, treatment, and prognosis of malignant neuroblastoma.
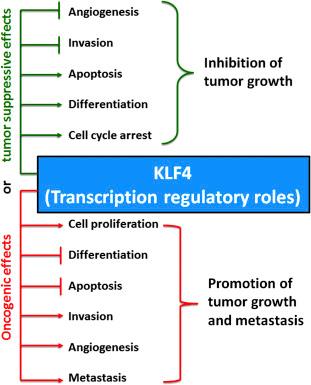
The transcription factors of both KLF family and specificity protein (Sp) family have key roles in critical biological processes such as stem cell maintenance, proliferation, differentiation, development, and metabolism, and thereby, impairment in their regulation is associated with many diseases including cancers in humans . Results from the phylogenic studies show that members of the KLF family share a common structural feature with the members of the Sp family. All members of these two protein families have zinc finger (ZnF) motifs at their C-terminal ends . Nevertheless, the proteins of the KLF family are structurally different as they have three highly conserved and distinct ZnF motifs with additional conserved residues between each ZnF . To act as transcription factors, proteins of the KLF family employ their conserved ZnF motifs to recognize and bind to the nearly identical GC/GT box or CACCC element consensus sequences: even thereafter, the specificity of their activities is imposed by their varying N-terminals and/or by their expression in distinctive tissues .
Because of abundant expression of KLF4 in the gut, it is every so often called the gut-enriched KLF . Moreover, KLF4 has been highly found to be expressed in the epithelial tissues. Conspicuously, KLF4 is highly expressed in terminally differentiated epithelial cells of the intestinal mucosa. Considering its customary roles and ample expression in epithelial cells of the gut, KLF4 certainly has key roles to play in maintaining cellular homeostasis in the intestinal epithelium. Studies show that colon cancer has a notable alteration in expression of KLF4 . Many other solid tumors also exhibit alterations in the expression of KLF4 . Because it can function as an oncoprotein or a tumor suppressor protein depending on the cellular, tissue, and genetic contexts, the pathobiology of KLF4 is very complicated . Results accumulated so far from various studies almost convincingly show that the transcription regulatory roles of KLF4 make it a tumor suppressor in human malignant neuroblastoma.
Human malignant neuroblastoma is one of the deadliest pediatric solid tumors, as it currently accounts for more than 15% of all cancer deaths in children . Although the pathogenesis of neuroblastoma involves upregulation of many oncogenes and downregulation of some tumor suppressors, many other key factors remain unknown. Proper diagnosis of neuroblastoma is problematic. Neuroblastoma follows an unpredictable clinical course. Some of these tumors undergo regression spontaneously without any treatment or respond quickly to the right treatment strategies . But some other tumors grow relentlessly due to drug resistance, resulting in poor therapeutic outcomes and eventually leading to the death of many pediatric patients . It is increasingly becoming evident that new and reliable prognostic biomarkers must be identified by the basic scientists for helping clinical scientists devise the most appropriate treatment strategies for saving many children who are currently under the dark spell of malignant neuroblastoma.
To predict the clinical outcomes of neuroblastoma patients, currently, there exist several prognostic markers such as the age of the patient at diagnosis, amplification of N-Myc, and level of expression of TrkA . Studies suggest that the more than 1 year age of the patient is associated with poor prognosis and amplification of N-Myc in neuroblastoma is also an indication of poor prognosis . There are no doubts that the age of the patient and amplification of N-Myc in neuroblastoma are currently useful in determining the stages of a progressive disease and predicting the gloomy therapeutic outcomes . On the other hand, the functional TrkA and EphB signaling pathways have been associated with a good prognosis of neuroblastoma patients . The functional TrkA and EphB signaling pathways are known to block the growth of neuroblastoma, respectively, by induction of terminal differentiation and inhibition of proliferation, angiogenesis, and metastasis in neuroblastoma cells . A later study shows that recruitment of histone deacetylase 1 (HDAC1) by a potent repression complex containing N-Myc at the TrkA promoter represses TrkA expression directly and thereby neuroblastoma cell response to the nerve growth factor, resulting in inhibition of terminal differentiation and promotion of aggressive growth of neuroblastoma .
A relatively recent study showed that expression of KLF4 at a low level in primary neuroblastoma in patients was a harbinger of unfavorable outcomes, while expression of KLF4 at a high level in human malignant neuroblastoma SH-SY5Y cells profoundly prevented cell proliferation due to direct upregulation of the cell cycle inhibitor protein p21Waf1/Cip1 and promoted loss of their neuroblastic phenotypes making them resemble epithelial cells, ardently adherent to the substrate, express smooth muscle marker, and non-tumorigenic cells, suggesting that the expression of KLF4 at a high level triggered terminal differentiation of neuroblastoma cells paving their path to spontaneous death . This study in neuroblastoma patients as well as in preclinical models of human neuroblastoma revealed the signaling pathways for strongly supporting the contention that KLF4 acts as a tumor suppressor in human malignant neuroblastoma. Expression of KLF4 at a high level in neuroblastoma patients seems to be a new and reliable biomarker for prediction of favorable outcomes.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here