Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
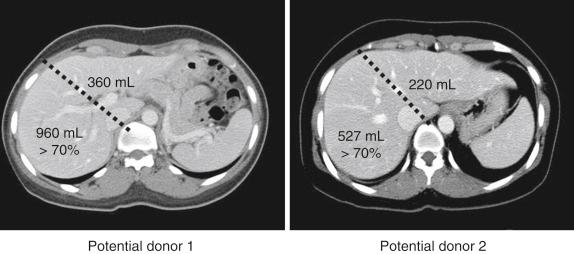
The shortage of cadaveric liver grafts has stimulated the development of innovative surgical procedures with various forms of living donor liver transplantation (LDLT), especially in adult patients, which produce satisfactory results comparable to whole-liver deceased donor liver transplantation (DDLT). For adult LDLT, insufficient graft size has been a major obstacle for an expansion of this procedure when the liver graft is confined to the left lobe (LL). To expand adult LDLT, right-lobe (RL) grafts have been used, with a rapid increase in the number of cases, but the risk to the donor increases with the extent of donor hepatectomy. It is reported that operative mortality for the RL donor is estimated to be as high as 0.5% to 1%. Regarding the recipients, the most important factor for a successful outcome after adult LDLT is the size of the liver graft. Meanwhile the safe remnant liver volume after RL donation has been reported to be greater than 30% to 35% of the total liver volume (TLV) of the donor, whereas the safe limit of the graft volume is greater than 40% to 50% of the recipient’s standard liver volume. Leelaudomlipi et al reported that in 25% of donors evaluated the volume ratio of RL was over 70% of TLV, or in other words, the remnant liver volume after RL donation was less than 30% of TLV. Even though the donor has a sufficiently large RL that it has adequate volume as a liver graft for an adult recipient, the remnant LL is too small to ensure donor safety in these procedures. Undoubtedly the safety of donors must have top priority when considering LDLT, and the inevitable risk to the donor should not be allowed in an LDLT procedure. The donor demonstrating RL to LL volume disproportion (RL > 70% of TLV) on preoperative volumetric computed tomography should not be allowed to donate the RL for a large-size adult recipient.
As an alternative, dual LL or left lateral segment (LLS) transplantation can be an option to avoid the small-for-size graft (SFSG) problem caused by a LL liver transplantation (LT) alone and the critical right lobectomy posing an independent donor risk. The ethical issue of putting two donors at risk simultaneously for one recipient can be debatable. However, because the clear relationship between donor mortality and the extent of resection has been demonstrated, the sum of potential risks from two donors’ left lobectomies will be much lower than the risk of an inappropriate right hepatectomy. To alleviate the SFSG problem by summing the suboptimal volumes of two LL grafts, dual LL grafts LT preserving the donors' safety is realistic and justified. Although LDLT with dual LL grafts is a technically complex and elaborate procedure, dual-graft LT can solve problems related to SFSG. It can also help expand the donor pool for LDLT and even for split-liver DDLT by application of two RL and one dual LL/LLS split transplantation to three recipients from two deceased donors. Furthermore, if a large-size recipient requires a bigger graft liver volume than the volume of a RL from a small-size donor when a RL harvest from one of two potential donors is decided to be safe, one RL and one LL graft from two independent donors can be transplanted to a large-size recipient to avoid the SFSG problem. This chapter introduces the technical aspects and the issues of dual-graft (two LLs or a RL and a LL) transplantation by reviewing our 300 dual-graft LTs.
Not all potential living donors can donate their RL because safe donation is possible only when the estimated remnant liver volume is more than 30% of the donor’s TLV. At the Asian Medical Center the minimally accepted remnant liver volume in RL donors is individualized by age and degree of steatosis ( Fig. 57-1 ). If the remnant LL hepatic mass is fully functioning without congestion injury by middle hepatic vein (MHV) preservation, the lower limit of 30% remnant liver volume is acceptable if the donor’s age is 35 years or less and preoperative donor liver biopsy reveals no steatosis. However, when donor age increases, the degree of steatosis increases, and congestion injury of liver segment IV by extended RL hepatectomy (MHV is included in RL graft) is expected, the lower limit of the remnant liver volume in donors should be increased.
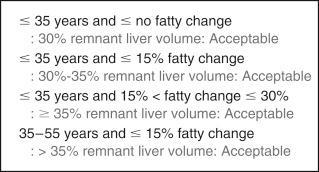
Selection criteria for dual LL or LLS transplantation requires two concurrent situations. First, the volume disproportion between RL and LL (RL > 70% of TLV) in the donor liver exists, and the right hepatectomy endangers donor safety ( Fig. 57-2 ). Second, the donor’s LL is too small to meet the metabolic demands of the recipient. Under these circumstances, dual-graft LDLT using two LLs can provide an adequate graft volume to the recipient while leaving a safe remnant liver volume in the donor ( Fig. 57-3 ). Besides the graft-to-recipient size mismatching and the unacceptable right-to-left-lobe volume discrepancy, severely steatotic (>60% steatosis) LLS or LL grafts have been successfully transplanted to low (<20) Model for End-Stage Liver Disease (MELD) patients in nonurgent dual-graft LDLT.
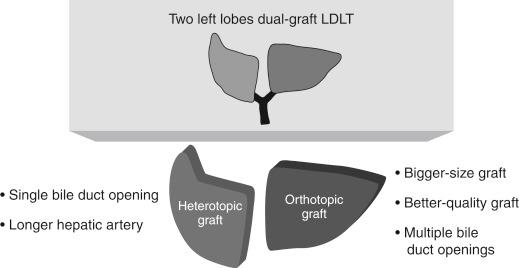
This new procedure needs a few technical modifications during implanting the heterotopic right-positioned LL graft. The rotation of the heterotopic liver graft through 180 degrees of sagittal orientation brings hilar structures into reversed position. The bile duct lies behind the portal vein (PV) and hepatic artery. If the PV anastomosis is performed before the bile duct anastomosis, biliary reconstruction of the heterotopic liver graft will be difficult or impossible in such a limited and hidden area. Thus the first technical modification is that the bile duct is reconstructed by duct-to-duct anastomosis before the PV anastomosis lying ventrally. Therefore the liver graft with a single duct opening is the first choice for the heterotopic right-sided graft to facilitate an easy and fast duct-to-duct anastomosis and to decrease the graft ischemic time. In addition, a longer length of hepatic artery is needed for tension-free anastomosis compared to the orthotopic left-sided graft because the recipient’s right hepatic artery arises from the most dorsal position. As the orthotopic left-sided graft, the bigger-size and better-quality graft is commonly preferred because it has less possibility of vascular complications as a result of its natural position. In addition, it has no strict limitation on the number of bile duct openings because biliary reconstruction with hepaticojejunostomy after hepatic artery (HA) anastomosis does not need to be hurriedly performed ( Fig. 57-4 ).
The thickness of the LL or LLS graft is too thin compared to that of the RL of the recipient’s resected liver. Therefore the second technical modification is that tissue expander filled with saline solution (from 200 to 450 mL) is inserted underneath the graft. This relieves undue tension on the hilar anastomoses of the heterotopic liver graft because the LL graft is usually too small to replace the right upper quadrant space after total hepatectomy in the recipient. The tissue expander is gradually deflated after the fifth postoperative day and removed after 2 weeks, when the undue tension on the hepatic hilum is relieved by sufficient regeneration of the right-sided liver graft.
In the recipient operation both the right and left branches of the PV and hepatic artery are dissected free from the surrounding tissue as peripherally as possible to obtain enough length for future bilateral vascular anastomoses. The bile duct is similarly dissected to the level of the hilum without disturbing the course of the right hepatic artery behind the common hepatic duct. The proximal part of the recipient’s vena cava should be mobilized from its retroperitoneal attachment because venoplasty of the recipient’s hepatic veins and graft implantation are performed under clamping of the suprahepatic and infrahepatic vena cava. Venovenous bypass (Bio-Pump, Bio-Medicus, Inc) is necessary when the recipient hemodynamics is unstable during the test clamping of the vena cava. It is also helpful to reduce bowel edema for future hepaticojejunostomy to the left-sided graft when more than 2 hours of anhepatic phase is expected.
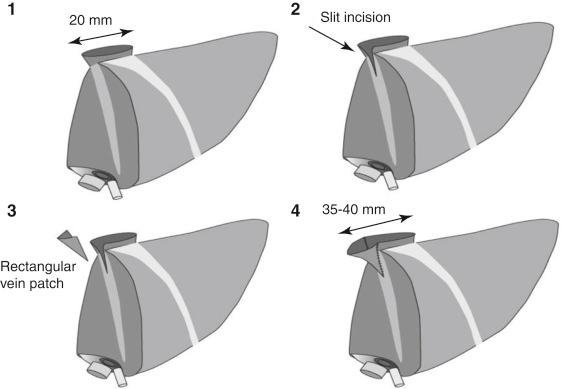
Which graft will be positioned heterotopically is determined by consideration of the number of bile duct openings, the length and number of hepatic arteries, the degree of steatosis, and the expected graft volume of each liver graft (see Fig. 57-4 ). Harvest of liver grafts and the recipient’s hepatectomy are usually performed simultaneously. Before implantation of the liver grafts, venoplasty of the hepatic veins in the recipient and/or the liver grafts at the bench should be performed to make wide outflow orifices of approximately 35 to 40 mm in diameter for prevention of the occasional outflow obstruction during liver graft regeneration. At the back table the hepatic vein of the liver graft is enlarged by augmentation venoplasty if its diameter is less than the target diameter of 35 to 40 mm. The venoplasty is performed using a rectangular vein patch after slit incision at one or both corners of the graft hepatic vein ( Fig. 57-5 ). In the recipient the right hepatic vein (RHV) is enlarged and elongated by longitudinal incision at the inferior corner with or without augmentation venoplasty. The left and middle hepatic veins (LMHV) are converted to a single common opening by division of the septum between the MHV and the LHV. The common hepatic vein opening is then enlarged and elongated by an additional transverse incision at the right corner and circumferentially fenced with a vein patch to facilitate hepatic vein anastomosis under redundancy to prevent the tearing of the posterior wall anastomosis. Venoplasty corresponding to the size of the hepatic vein between the liver graft and the recipient is an essential step so that the surgeon can perform engraftment without difficulty during hepatic vein anastomosis and can also avoid postoperative outflow disturbance. An autologous bisected great saphenous vein segment is the preferred augmentation and fencing material at our department because it has a thicker and stronger wall than other homologous vascular grafts.
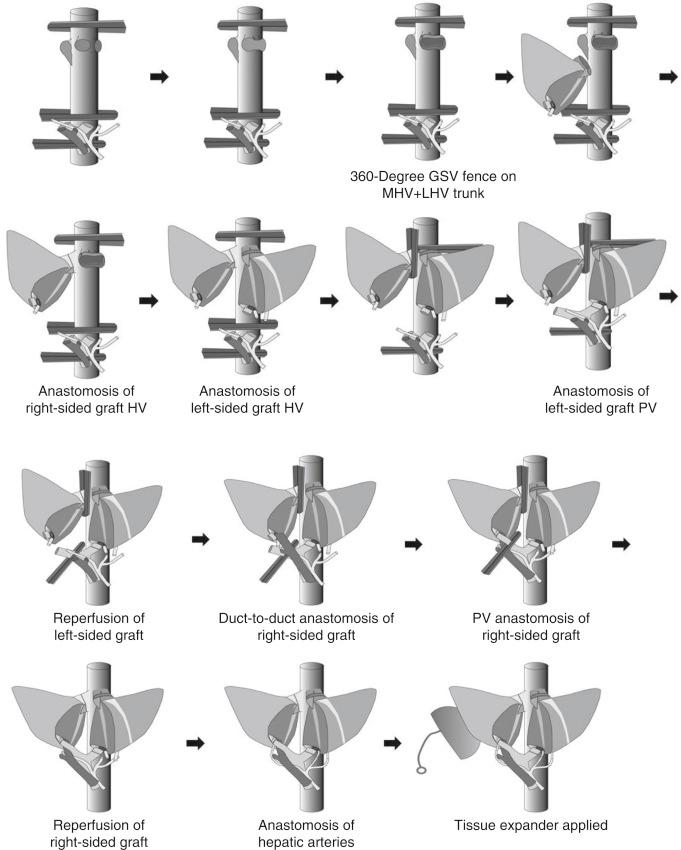
Engraftment procedures using two LL grafts are as follows. First, a 180-degree rotated LL graft is heterotopically placed into the right upper quadrant space, and its hepatic vein anastomosis to the recipient RHV is performed. Second, the hepatic vein of the orthotopically positioned LL graft is anastomosed to the common opening of the LMHV. Third, the PV anastomosis of the orthotopic graft is performed to the recipient’s left PV. Fourth, the orthotopic left-sided graft is reperfused earlier to avoid bowel congestion and reduce the graft ischemic time. Before reperfusion of the orthotopic liver graft proceeds, a vascular clamp is applied to the recipient’s RHV to prevent regurgitation of caval flow into the heterotopic right-sided graft after release of the caval clamps. Vascular clamps to the vena cava and the PV trunk are then removed together under new clamping of the recipient’s right PV. Fifth, bile duct anastomosis of the right-sided (heterotopic) graft to the recipient’s bile duct is performed in duct-to-duct fashion earlier than PV anastomosis because of the reversed hilar structures of the graft. Sixth, the PV of the right-sided heterotopic liver graft is anastomosed to the recipient’s right PV and is reperfused after removal of the vascular clamps on the recipient’s RHV and right PV. Seventh, hepatic artery anastomoses are performed, and finally a Roux-en-Y hepaticojejunostomy to the left-sided (orthotopic) liver graft is performed. As an additional procedure, a tissue expander filled with saline solution should be inserted underneath the right-sided (heterotopic) graft to relieve undue tension on the hilar anastomoses ( Fig. 57-6 ).
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here