Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
![]() Please access Videos to view the corresponding videos for this chapter.
Please access Videos to view the corresponding videos for this chapter.
Historically, cerebrospinal fluid (CSF) leak rates of approximately 20% were observed after translabyrinthine craniotomy. With refinement of closure technique, CSF leak rates have decreased significantly to 0% to 3%. The egress of CSF from the subarachnoid space into surgical wounds may result in the leakage of CSF from the wound, ear canal (if the tympanic membrane is not intact), or nose (via the eustachian tube). CSF follows the path of least resistance. Any procedure that encounters the subarachnoid space can be complicated by a postoperative CSF leak. These leaks result from failure to obtain watertight dural closure; an inadequate seal of dural defects; or inadequate packing of the eustachian tube, petrous apex air cells, or retrofacial air cells.
Although CSF leaks are less frequently seen than in the past, CSF leaks are a concern because the defect provides a potential portal of entry for infection to seed the leptomeninges. In the setting of postoperative CSF leak, the rate of meningitis has been shown to be as high as 14%. Meningitis is accompanied by significant morbidity and mortality.
Various reports have found that operative time, tumor size, , BMI, , , younger age, and male sex , may predispose a patient to postoperative CSF leak. Reports of similar quality have found that operative time, tumor size, , , BMI, age, , , , and sex do not increase the risk for CSF leak. Medical comorbidities have been shown not to play a role in CSF leak rate. Postoperative facial nerve dysfunction, cases performed at a teaching hospital, and cases performed at hospital with a lower surgical volume have all been correlated with a higher rate of CSF leak.
Intraoperative treatment of the sigmoid sinus may lead to venous thrombosis, which has been shown to triple the odds of a postoperative CSF leak. The sigmoid sinuses are co-dominant in 50% of cases, right-dominant in 40%, and left-dominant in 10%. Lateral sinus thrombosis has been shown to lead to hydrocephalus in 5% to 17% of cases. This subsequent rise in intracranial pressure increases the strain on operative closure. In addition, the degree of mastoid and petrous apex pneumatization can lead to an increased rate of CSF leak given the routes for CSF egress through the eustachian tube.
Deliberate closure techniques including proper fat packing, layered closure, eustachian tube packing, and waxing air cells are the most important steps to prevent postoperative CSF leaks. Despite the skull base surgeon’s best intentions and techniques, postoperative CSF leaks will occur. This chapter describes the various techniques for treating postoperative CSF leaks.
For procedures in which an abdominal fat graft has been used during closure to plug dural defects, a pressure dressing can be applied to control CSF leaks. The pressure dressing works by pushing the fat back into the dural defect, sealing off the subarachnoid space. In patients in whom fat has been used, most leaks stop with a pressure dressing. If a subcutaneous CSF collection (pseudomeningocele) is present, it may be aspirated before the placement of the pressure dressing. If performed, aspiration should be done with a sterile technique. Procedures involving incisional wound leaks may be treated by a simple suture overclosure of the leaking portion of the incision and a pressure dressing.
The dressing is applied in a manner similar to the initial postoperative dressing. First, a vertical gathering tie is placed in the temporal fossa. Dressing sponges (4×4) are folded in half and placed directly over the surgical site and in the postauricular sulcus to support the auricle ( Fig. 55.1A ). Next, fluffed Kerlix is placed over the 4×4 sponges and the auricle ( Fig. 55.1B ). A tight wrap of roller gauze is applied ( Fig. 55.1C ). The direction of the wrap should be from the ear toward the occiput, which ensures that the auricle is not damaged by anterior folding. In addition, the vertical tie must be as lateral as possible in the temporal fossa to avoid a pressure point on the forehead. Pressure necrosis of the forehead skin can develop easily if attention is not given to this point.
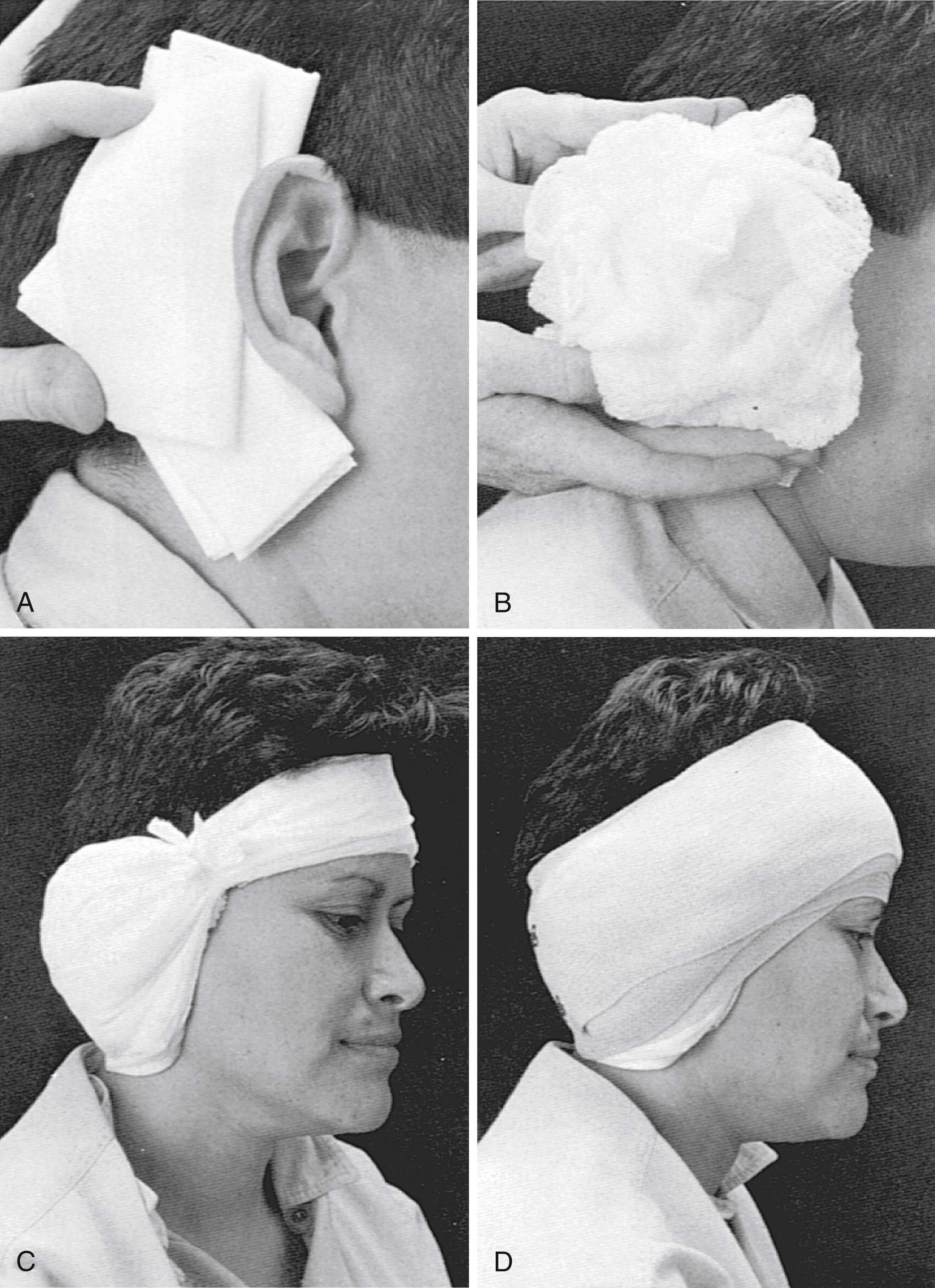
The last layer of this dressing is a 3-inch (7.62-cm) elastic bandage, which provides the final compression ( Fig. 55.1D ). The elastic bandage should be wrapped firmly, but patient comfort must be accommodated. Usually, the last several turns can be altered to adjust the exact amount of compression. Ideally, a pressure dressing should be left in place for 3 to 5 days, which allows time for healing of the CSF leak site to occur. A minimum of 48 hours without leakage must pass before the dressing is removed.
In addition to the pressure dressing, other conservative actions should be undertaken. All these measures are directed at decreasing CSF pressure. Straining (Valsalva maneuver) is strictly avoided and the patient is kept at bed rest with the head elevated 45 degrees. Limited activity, such as bathroom privileges or brief periods of sitting up in a chair, is at the surgeon’s discretion. Stool softeners and cough suppressants can be used. Acetazolamide (Diamox) may be administered to decrease CSF production.
The next step in CSF leak treatment is using a lumbar subarachnoid spinal fluid drain. By removing CSF from the system at a site away from the dural defect, CSF pressure is decreased, and healing can occur. Some surgeons routinely place a lumbar drain at the time of surgery to assist with intraoperative CSF removal and decompression in the postoperative period.
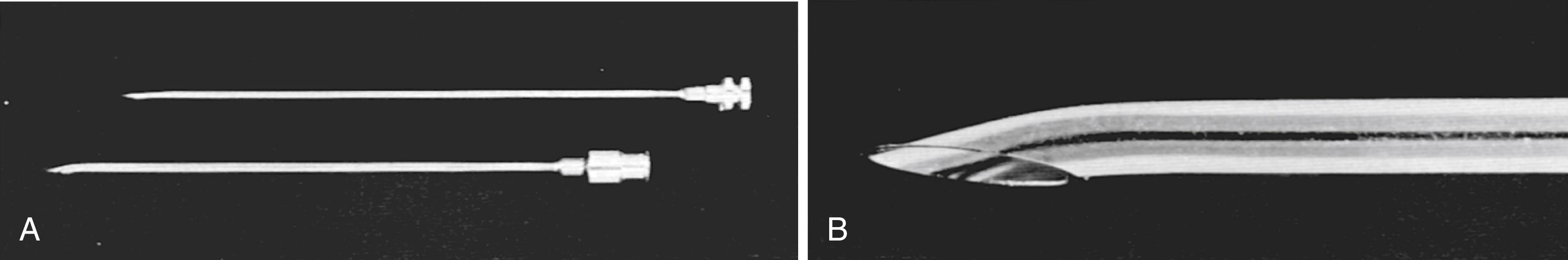
To place the catheter, a lumbar puncture is performed at the L4–L5 level. The patient is placed in either a sitting or lateral decubitus position. The spinous processes are palpated and L4–L5 is identified at the level of the iliac crest. The patient is asked to flex the back and bring the knees and chin to the chest. A prepared catheter kit contains all the necessary supplies for preparing, draping, and anesthetizing the site. A 14-gauge Tuohy needle is introduced in the midline with a slightly superior angle to traverse the space between the spinous processes ( Fig. 55.2 ). The obturator is removed at intervals so that the surgeon can look for a flow of CSF. When a good flow of CSF is established, the epidural catheter is introduced through the needle and threaded into the epidural space. The opened side of the bevel of the needle faces the patient’s left or right side on penetrating the spinous ligaments and arachnoid. Before the catheter is threaded, the bevel is turned to open superiorly, facilitating directing the catheter cephalad. With a flow of CSF established, the needle is withdrawn, and the connector is placed on the end of the catheter so that a connection to intravenous tubing can be made. An empty intravenous fluid bag is attached to the tubing, and all connections are secured with tape ( Fig. 55.3A ).
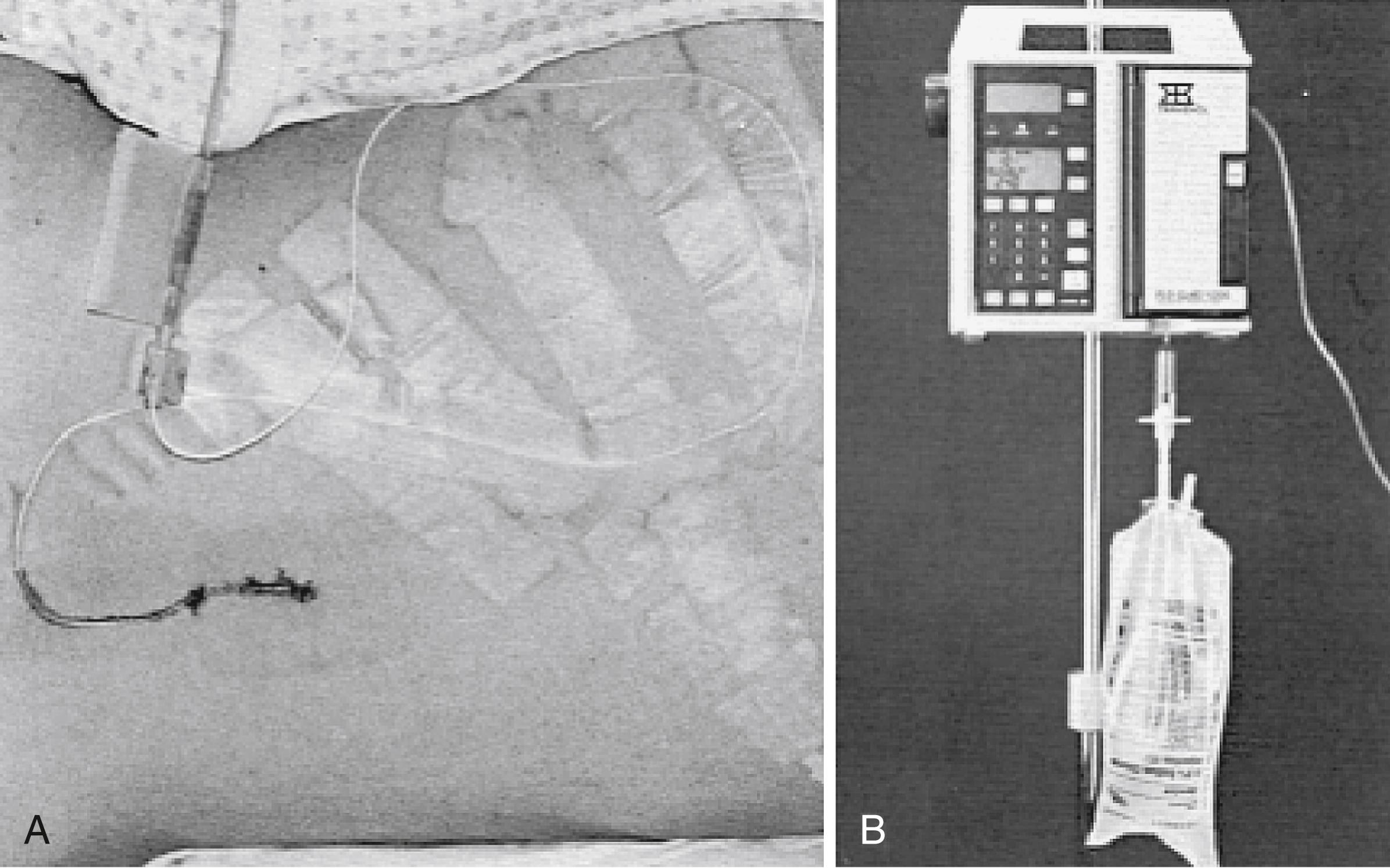
Several methods of regulating CSF output exist. This regulation is important because tension pneumocephalus and brain herniation can occur if CSF is removed too rapidly. Alternatively, if an inadequate amount of CSF is removed, the purpose of the drain is defeated. Humans produce approximately 18 mL of CSF per hour. The natural mechanisms of CSF absorption reduce some CSF; the lumbar drain should remove less than 18 mL of CSF per hour. The common neurosurgical practice is to order 50 mL of CSF removed every 8 hours.
Gravity is the standard method of effecting CSF drainage. The collection bag is placed at the level of the patient’s heart or below to create increasing output of CSF as needed. This method requires extremely attentive nursing care to ensure the proper amount of drainage. Drains placed to respond to gravity have highly variable output, and a change in patient position may significantly increase or decrease the flow. Establishing an even flow of CSF is difficult, and a bolus of fluid is usually removed, after which the drain is clamped. A limiting device, such as the LimiTorr volume limiting CSF drainage system (Integra LifeSciences), may be employed to halt drainage at a predetermined volume (i.e., 30 mL). The drainage system is at risk of occluding and, subsequently, removing the amount of CSF ordered becomes difficult.
While the lumbar drain is in place, the patient must be closely observed for signs of infection. The temperature, surgical wound, lumbar drain site, and white blood cell count must be monitored. Samples of CSF may be examined at any time. Headache during CSF drainage is common, but meningismus should not be present. Prophylactic antibiotics are controversial but are administered by some surgeons. As with a pressure dressing, the lumbar drain should be left in place for 4 or 5 days. We often clamp the drain for the last 24 hours so that the drainage can be resumed if the leak recurs.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here