Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Gonadotropin-releasing hormone (GnRH) analogs are synthesized by substitution of amino acids in the parent molecule at the 6 and 10 positions for GnRH agonists and at the 2 or 3 position for GnRH antagonists. The various analogs have greater potencies and longer half-lives than the parent GnRH and are used clinically for suppression.
LH and FSH have the same α subunit as thyroid-stimulating hormone (TSH) and human chorionic gonadotropin (HCG). The β subunits of all these hormones have different amino acids and carbohydrates, which provide specific biologic activity.
Kisspeptin (KISS1) plays a key role in the regulation of GnRH release.
Ovulation occurs about 24 hours after the estradiol peak and 32 hours after the initial rise in LH, as well as about 12 to 16 hours after the peak of LH levels in serum. Serum progesterone is less than 1 ng/mL before ovulation and reaches midluteal levels of 10 to 20 ng/mL.
Four characteristics of hormone assays establish their reliability: sensitivity, specificity, accuracy, and precision.
The endocrine regulation of the reproductive system is very complex. This chapter presents only the basic information required to understand this complex subject. More detailed and in-depth information is found in the several books that have been dedicated to this subject.
Successful function of the reproductive system requires the involvement of several organs, none of which acts independently. In this chapter we discuss the physiology of the hypothalamic-pituitary-ovarian (HPO) axis. For ease of understanding, each organ will be discussed first as an individual unit; information will include the central nervous system control of gonadotropin-releasing hormone (GnRH), the primary neurohormone controlling the whole reproductive endocrine axis; the GnRH action on the anterior pituitary and the resultant secretion of the gonadotropins; the gonadotropins’ action on the ovaries and the release of gonadal steroids; and finally the action of these sex steroids on the uterus and cervix. Although it is fair to state that the HPO axis is driven by the hypothalamus and its release of GnRH, it is important to point out that normal function of the HPO-endocrine axis requires a remarkable information flow and coordination between each of these organs, as exemplified by the existing inhibitory and stimulatory feedback loops. Their relevance will become obvious in the discussion of the menstrual cycle, which closes the chapter.
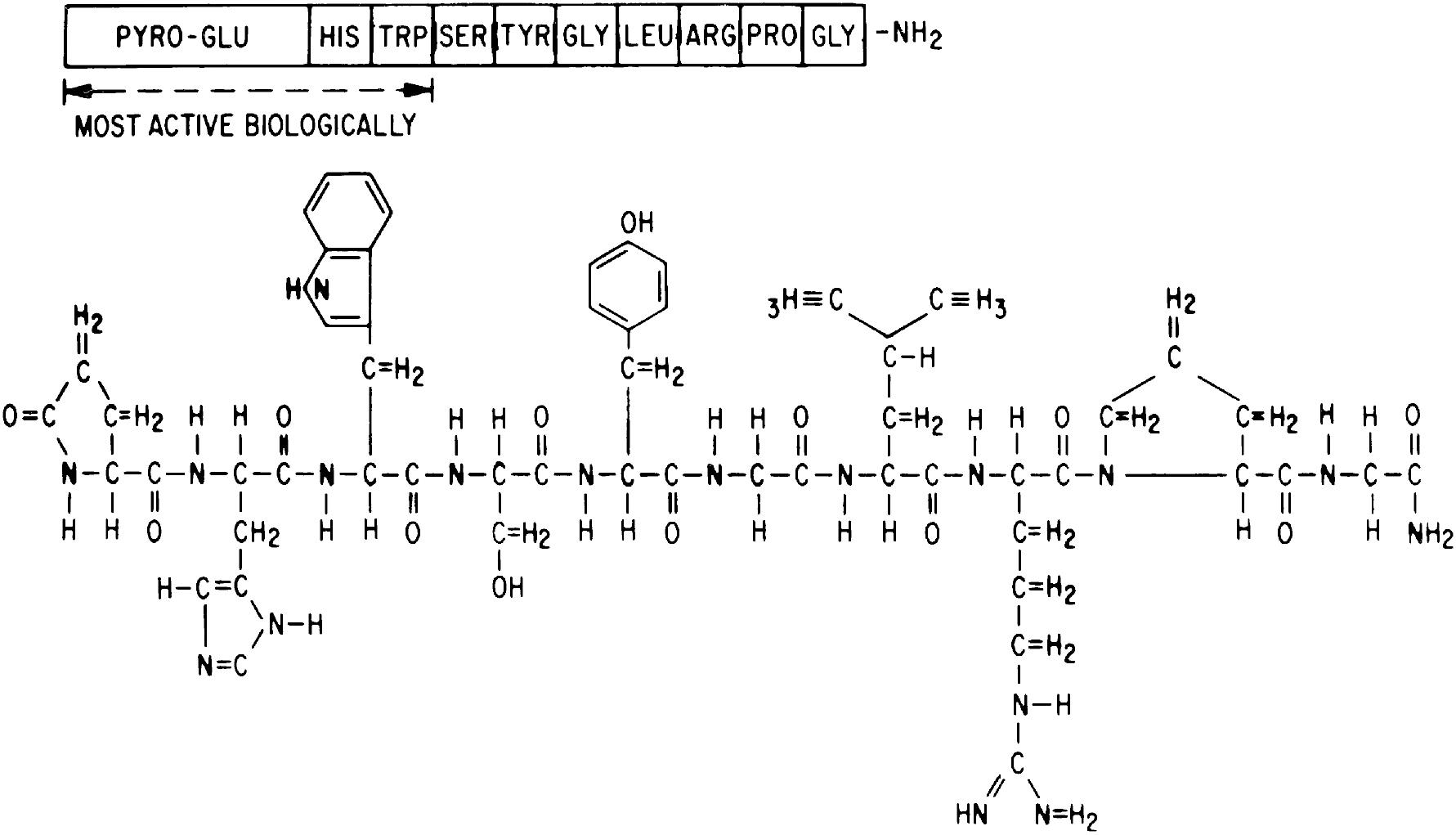
The reproductive process starts in the brain, through the activation of the initial hormonal signal that will release the gonadotropins from the pituitary gland. This hormone released by the hypothalamus is GnRH, a decapeptide (10 amino acids) ( Fig. 4.1 ). The GnRH gene (GNRH1, situated on the short arm of chromosome 8) encodes for a 92 amino acids precursor molecule, composed of a signal peptide sequence, the GnRH sequence itself, a posttranscriptional processing signal (3 amino acids long), and a 56-amino acid peptide known as GnRH-associated peptide (GAP). (Several forms of the GnRH decapeptide have been identified, the principal of which is GnRH-2, which differs from GnRH by 3 amino acids. It is found in several areas of the body, where it may subserve functions unrelated to those of GnRH.) Its role in fertility, if any, remains to be determined.
Surprisingly, GnRH-synthesizing neurons do not originate within the brain, like the majority of all neurons. Rather, GnRH neurons derive from progenitor cells in the embryonic olfactory placode where they develop. In a particular journey unique for a neuron, GnRH neurons migrate toward the brain during early fetal life to reach the locations that they will occupy during adult life . This migration of GnRH neurons over long distances and through changing molecular environments suggests that numerous factors, local and possibly external, influence this process at its different stages. Such factors play critical roles, such as mediating the adhesion of GnRH neurons to changing surfaces along their voyage, promoting cytoskeleton remodeling, and modulating axonal guidance.
Functional connections between GnRH neurons and the hypophyseal portal system that will transport GnRH to the anterior pituitary gland are established by about 16 weeks of fetal life. Migration failure of GnRH neurons and the resultant lack of the establishment of functional connections are characteristic of patients with the Kallmann syndrome, who show hypogonadotropic hypogonadism accompanied by anosmia ( ). In the 19-week-old fetus with X-linked Kallmann syndrome, the GnRH neurons accompanying the olfactory nerves have been shown to be arrested in their voyage within the meninges, and therefore contact with the brain and the hypophyseal portal system is not established.
In adults, neurons producing GnRH are present in several hypothalamic nuclei and other parts of the brain. However, the majority of GnRH neurons controlling the HPO axis are located within the anterior hypothalamus and primarily within the medial basal hypothalamus, with the greatest number in the primate within the arcuate nucleus . The 92 amino acid GnRH precursor is released into the axons of these neurons and cleaved during transport to yield GnRH and GAP. (The biologic function of GAP or fragments thereof remains to be clarified.)
A substantial number of GnRH axons terminate within the external zone of the median eminence (infundibulum) where GnRH is released . This area is the site of an important capillary plexus, with a fenestrated epithelium similar to that of peripheral capillaries, which allows passage of large molecules. (These capillaries differ from brain capillaries, which are not fenestrated. Thus the median eminence is viewed as an area outside the blood-brain barrier.) This pathway is the most relevant one in regard to the control of the pituitary-ovarian axis ( Fig. 4.2 , A). Another substantial projection of GnRH axons is through circumventricular organs, the major of which is the organum vasculosum of the lamina terminalis (OVLT). These areas are also outside the blood-brain barrier. (The function of GnRH release into these areas remains somewhat unknown. One role may be to enable the release of GnRH into cerebrospinal fluid [CSF], perhaps to facilitate actions of GnRH in other areas of the brain. GnRH levels have been found to be elevated in CSF as opposed to being minimal in peripheral blood.) Another possible route of GnRH release may involve specialized ependymal cells, referred to as tanycytes . These have been found to extend from the lumen of the third ventricle to the external zone of the median eminence.
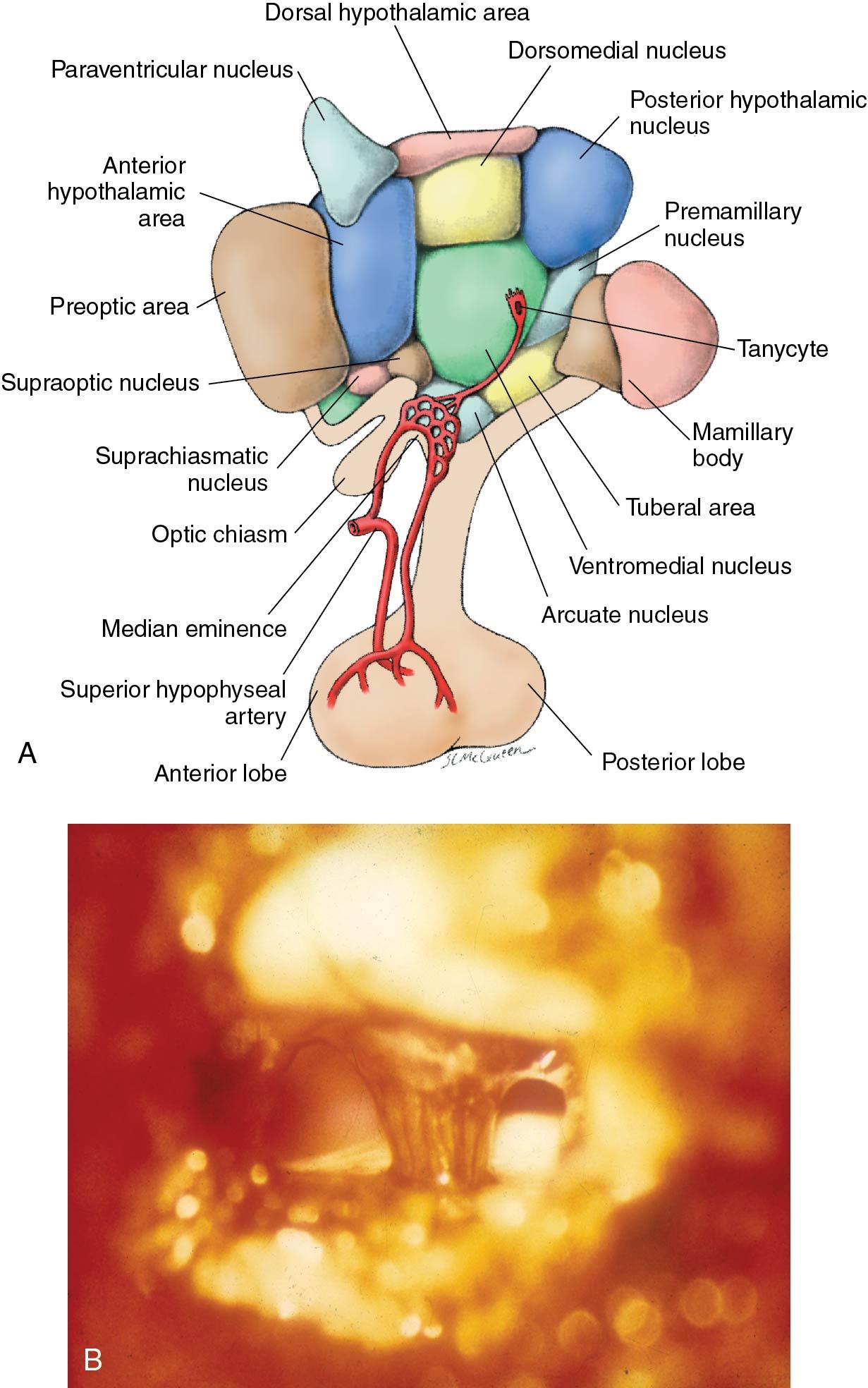
The capillary plexus of the external median eminence, into which GnRH is released, collects into several hypophyseal portal vessels, which descend along the pituitary stalk to terminate within another capillary plexus (hence the term portal ) within the anterior lobe of the pituitary (see Fig. 4.2 A). (Unlike the posterior lobe of the pituitary, also referred to as the neurohypophysis, the anterior lobe has no direct blood supply and receives all of its vascularization from this portal system.) The vascular arrangement whereby GnRH and other neurohormones reach the anterior pituitary is very important to the proper function of the endocrine system: It allows for the rapid (within minutes) and undiluted transport of relatively small amounts of neurohormones to the pituitary. This is especially crucial to GnRH because this neurohormone has a short half-life of about 2 to 4 minutes (it is rapidly degraded by peptidases in blood; as a consequence, GnRH is not measurable in peripheral blood) and because of its pulsatile mode of release (discussed later).
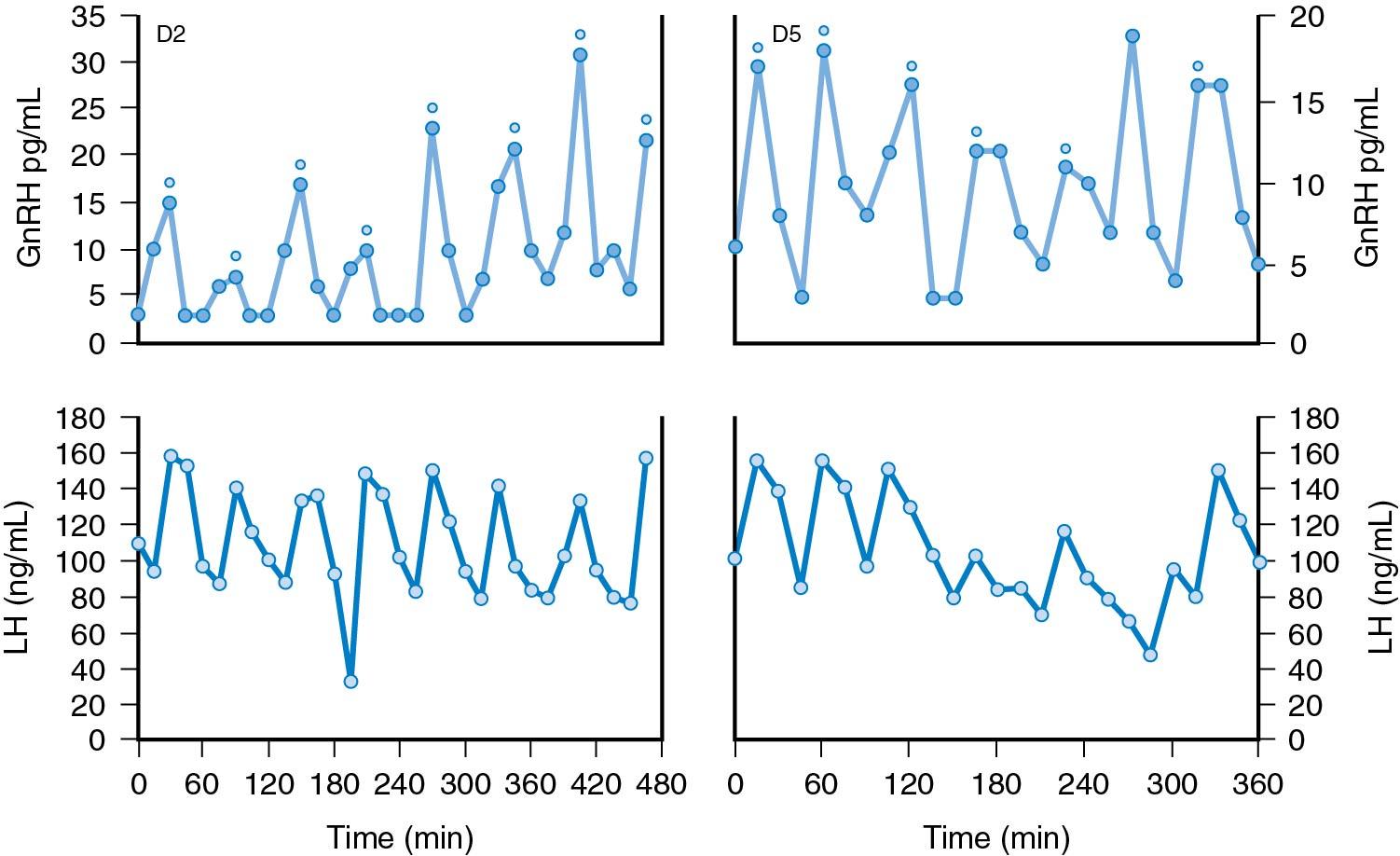
Studies have shown that GnRH is characteristically released intermittently, in a pulsatile fashion. From this comes the concept of the “GnRH pulse generator” responsible for the pulsatile release of the hormone ( ). GnRH pulses occur at about hourly intervals ( Fig. 4.3 ). The rising edge of each GnRH pulse is abrupt, such that GnRH can increase by a factor of 50 within 1 minute. Each GnRH pulse is preceded by an increase in multiunit activity within the area of the arcuate nucleus.
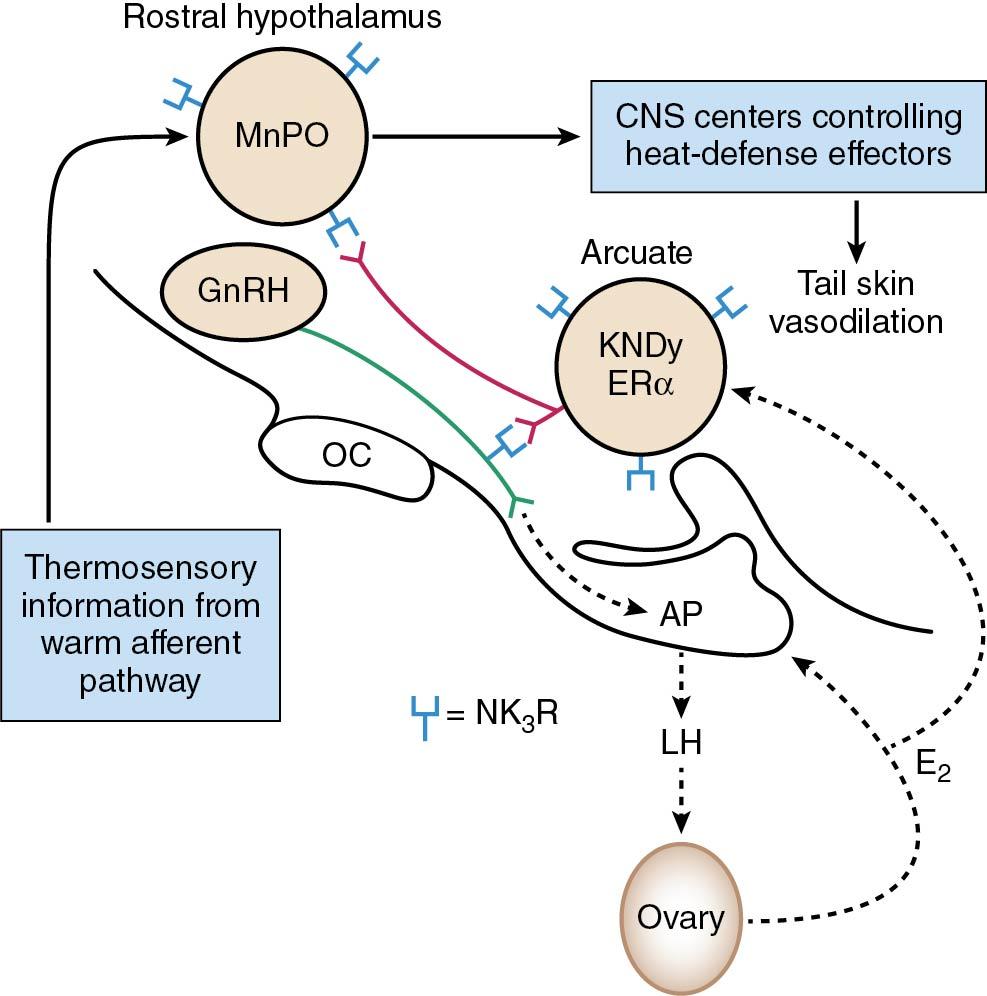
The cellular basis and the mechanisms that determine the timing of the increase in multiunit activity resulting in pulsatile GnRH activity are still under study. First, there is a growing consensus that pulsatile activity originates from an inherent pace-making activity of the GnRH neuron itself; in vitro data have shown that individual neurons have the capacity of spontaneous oscillations in activity. In this case such activity would also require a synchronized action from enough neurons to provide a discrete GnRH pulse. Intercommunication between GnRH neurons may occur through gap junctions between such neurons, which have been demonstrated, and through synaptic forms of interaction between cells. Second, evidence identifies a key role of kisspeptin (KISS1), a product of the KISS1 gene, and its receptor (GPR54 or KISS1R) in the regulation of GnRH release ( ). KISS1 neurons have been found to directly innervate and stimulate GnRH neurons. In humans, mutations or targeted deletions of KISS1 or of its receptor cause hypogonadotropic hypogonadism. Patients with these mutations, however, do not have anosmia, unlike those with Kallmann syndrome, suggesting that there are no major deficits in the embryonic migration of olfactory or GnRH neurons. KISS1 neurons within the arcuate nucleus (rodents) and infundibular nucleus (humans) express the estrogen, progesterone, and androgen receptors , and KISS1 signaling in the brain is implicated in mediating sex steroids feedback loops, especially during the preovulatory GnRH/luteinizing hormone (LH) surge. Kisspeptin has been used to induce egg maturation in women undergoing in vitro fertilization therapy, with subsequent fertilization of the mature eggs, embryo transfer into the uterus and successful human pregnancy ( ). There is the potential for routine use of kisspeptin to induce ovulation during fertility treatment cycles. KISS1 has also been shown to play a role in the initiation of puberty . A subpopulation of kisspeptin neurons in the human infundibular nucleus coexpress neuropeptides neurokinin B and dynorphin (an opioid inhibitor) ( ). These kisspeptin-neurokinin-dynorphin (KNDy) neurons express neurokinin B and dynorphin receptors, as well as estrogen and progesterone receptors . KNDy neurons coordinate inputs, including sex steroid feedback, to regulate pulsatile kisspeptin secretion. The interaction of KISS1 neurons with other neurotransmitter systems is currently being studied. Fig. 4.4 depicts the hypothalamic circuitry between the KNDy neurons and GnRH release, as well as the newly appreciated connections to the thermoregulatory areas in the hypothalamus ( ). Thus blockade of the neurokinin (NK3) receptor with a specific antagonist has been found to control hot flushes in postmenopausal women by blocking the pathways of the thermoregulatory center.
The foremost modulatory influence on the frequency and amplitude of GnRH pulses is exerted by the ovarian steroid hormones through their feedback loop actions. In general, estradiol is known to decrease GnRH pulse amplitude, whereas progesterone decreases GnRH pulse frequency (see the discussion presented later for details).
Numerous other studies suggest that the spontaneous activity of the GnRH pulse generator may also be modulated by a variety of additional stimulatory and inhibitory afferent neural signals. Stimulatory inputs to GnRH release may originate from neurons using the biogenic amine neuroepinephrine (NE), the amino acid glutamate, and the peptide neuropeptide Y (NPY). Inhibitory inputs may come from the amino acid gamma-aminobutyric acid (GABA), the biogenic amine dopamine (DA), the endogenous opioid β -endorphin, and the neurosecretory peptide corticotropin-releasing hormone (CRH) neurons. These systems may affect the GnRH pulse generator either tonally or conditionally.
In the first category we find, for example, NE as a potential tonal stimulator and GABA as a tonal inhibitor of GnRH release. Administration of alpha-adrenergic blockers has been shown to reduce pulse frequency in animals, in accord with the postulated tonal stimulatory role for NE. The role of GABA as a tonal inhibitor may be more prominent during the prepubertal period, at which time a diminishing inhibitory GABA tone may activate puberty and the resumption of GnRH pulsatile release. Glutamate’s role is more uncertain, although it also is suspected in the initiation of pulses at puberty. Dopamine infusions in women are associated with a decrease in circulating LH and prolactin (dopamine is also known as the prolactin-inhibitory neurohormone). The effect on LH is thought to be mediated through GnRH because, in patients with hypothalamic amenorrhea in whom there appears to be an excess of dopaminergic tone, administration of a dopamine blocker may return the LH pulse frequency to normal. It should also be remembered that specific effects of neurotransmitters on GnRH neurons may be altered by the administration of certain drugs, which may interfere with the proper synthesis, binding, storage, or receptor function of these neurotransmitters. Thus on treatment with such drugs (e.g., methyldopa; reserpine; tricyclic antidepressants such as propranolol, phentolamine, haloperidol, and cyproheptadine; selective serotonin reuptake inhibitors [SSRIs]; and serotonin-norepinephrine reuptake inhibitors [SNRIs]), patients may develop disorders such as oligomenorrhea or galactorrhea, the result of alterations in GnRH secretion or hyperprolactinemia . Other studies also suggest that hypothalamic prostaglandins may also modulate the release of GnRH; for instance, the midcycle surge of LH (see The Menstrual Cycle later in this chapter) can be abolished in animals by the administration of aspirin or indomethacin, which blocks the synthesis of prostaglandins.
In the second category, other systems may affect the GnRH system only conditionally —that is, under specific hormonal or physiologic conditions. One example is the endogenous opioid β -endorphin, which exerts an inhibitory action on GnRH pulsatile activity that depends largely on the endogenous endocrine milieu. This is related to the fact that ovarian hormones control the release of β -endorphin within the brain, which is lowest in the absence of estradiol, such as in the ovariectomized nonhuman primate, and highest in the presence of both estradiol and progesterone, such as during the luteal phase of the menstrual cycle. Experimental administration of the opiate antagonist naloxone during the luteal phase increases GnRH/LH pulse frequency, significantly suggesting the reversal of a inhibitory action under the endocrine milieu that characterizes the luteal phase. No effect on LH pulsatility follows naloxone injection in postmenopausal or ovariectomized women, unless they are treated with an estrogen-progesterone therapy. Another example is CRH, the main neuropeptide controlling the adrenal endocrine axis, which is released in greater amount during stress. In this condition, increased CRH release negatively affects the GnRH pulse generator, which results in a decrease in GnRH pulse frequency . This action is indirect through the release of β -endorphin and is prevented by the administration of the opiate antagonist naloxone, as demonstrated in studies in nonhuman primates and in patients with hypothalamic amenorrhea, many of whom have elevated levels of cortisol.
There is good clinical evidence linking energy homeostasis and reproductive function in the human. A functional reproductive system requires an accurate integration of energy balance, and a significant imbalance may lead to reproductive dysfunction and amenorrhea. Nutritional deprivation and abnormal eating habits are known to interfere with the normal reproductive process. Anorexia nervosa is a well-known and extreme example of how alteration in food intake can result in the suppression of the menstrual cycle. Obesity also may contribute to menstrual disorders.
Growing evidence indicates that complex and extensively integrated physiologic mechanisms connect an active reproductive axis to the metabolic state. The brain, and in particular the hypothalamus and the GnRH pulse generator, function as the center for the integrative metabolic response process. The nature of the afferent signals that provide information about energy metabolism to the reproductive axis is presently under intense study, and data have shown possible roles for several energy-related proteins.
One such example is leptin, an anorexigenic protein that is the product of the ob gene and that is primarily produced by adipocytes ( ). Leptin levels are reduced when body fat stores are decreased by fasting. Besides conveying metabolic information to several parts of the brain through its own receptors, leptin also appears to function as one of the metabolic cues regulating the GnRH pulse generator. High leptin levels are interpreted as conducive to reproduction, and the administration of leptin stimulates the secretion of GnRH and of the gonadotropins, with the effects most pronounced in individuals showing signs of reproductive impairment. Peripheral injections of leptin can prevent the reduction in GnRH/gonadotropins and the disturbances in cyclicity that accompany caloric reduction. Ob/ob mice, which are leptin deficient because of a mutated leptin gene, besides exhibiting a pronounced obesity, show a complete failure to display normal estrous cycles because of absent GnRH secretion . The latter can be reversed by leptin administration. Evidence suggests a role for kisspeptin in modulating metabolic leptin signals on the hypothalamus and pituitary ( ). Forty percent of kisspeptin neurons in the mouse arcuate nucleus express leptin. Leptin-deficient mice show decreased expression of Kiss1 messenger RNA (mRNA). However, leptin administration only partially restores Kiss1 mRNA levels, indicating that other mediators are involved in inhibiting kisspeptin signaling in leptin deficiency ( ). Another example is the orexigenic peptide NPY , which is synthesized in the arcuate nucleus. During fasting, expression of NPY mRNA increases in this nucleus and intracerebroventricular injection of NPY stimulates food intake . NPY has been shown to affect pulsatile GnRH/LH activity in the nonhuman primate, but this occurs in two apparently contradictory modes, one excitatory and one inhibitory. It was shown in the ovariectomized monkey and rodent that a pulsatile intracerebroventricular infusion of NPY stimulates GnRH release, whereas a continuous infusion clearly decreases the pulsatile electric activity of GnRH neurons, as well as pulsatile LH release. In accord with this observation of an inhibitory effect of NPY is that, whereas fasting decreases LH secretion in normal mice, fasting mice lacking NPY Y1 receptor have a higher pituitary LH content than wild-type ones. What these data suggest is that although a supportive effect of NPY on the GnRH pulse generator may occur within a limited window of normalcy (i.e., within a normal background of basic and pulsatile NPY release), in physiopathologic situations (i.e., in circumstances mimicking increased endogenous NPY activity such as in undernutrition) an inhibitory effect of NPY on the GnRH pulse generator can be observed. Evidence suggests that kisspeptin neurons can sense and convey information about energy status to GnRH neurons . In rodents, hypothalamic levels of Kiss1 mRNA are reduced in metabolic conditions, such as undernutrition, uncontrolled diabetes, and immune/inflammatory challenge, which are associated with suppressed gonadotropins. Administration of kisspeptin in rodent models with disrupted metabolism and energy reserves restores gonadotropin secretion, suggesting a potentially important central role of KISS1 neurons in the regulation of reproduction by metabolic factors ( ).
Overall, in regard to the reproductive system, the GnRH pulse generator actually acts as the link between the environment, the internal milieu, and the reproductive axis . Its overall activity most probably reflects the summation of simultaneous stimulatory and inhibitory inputs. It is evident that events, disorders, or drug administration may tip the physiologic balance, cause disruption or cessation of GnRH pulse activity, and lead to disruptions of the menstrual cycle and to reproductive disorders such as oligomenorrhea and hypothalamic amenorrhea.
The anterior pituitary (also referred to as the adenohypophysis ) derives from Rathke’s pouch, a depression in the roof of the developing mouth in front of the buccopharyngeal membrane. It originates at about the third week of life. Origin of the adenohypophysis contrasts with that of the posterior pituitary (neurohypophysis), which develops as a direct extension of the brain. It should also be noted that whereas the neurohypophysis receives a direct arterial blood supply from the hypophyseal arteries, the only vascularization to the adenohypophysis is through the hypothalamic-hypophyseal portal system (into which GnRH and several other neuropeptides are secreted; discussed earlier).
The gonadotropes are the specialized cells within the adenohypophysis that produce the gonadotropins . On stimulation of the gonadotropes by GnRH, two gonadotropins are released into the general circulation and regulate endocrine function in the ovaries and testes.
Pulses of GnRH released by the GnRH neurons in the arcuate nucleus reach the gonadotropes in the anterior pituitary via the hypophyseal portal circulation. These GnRH pulses then act on GnRH receptors (GnRH-Rs) on the gonadotropes to stimulate both the synthesis and release of both gonadotropins, LH and follicle-stimulating hormone (FSH). Women with GnRH-R mutations typically present with incomplete or absent pubertal development and primary amenorrhea. Although reproductive function is compromised, conception may be successfully obtained after gonadotropin treatment.
On the cell membranes of the gonadotrope, GnRH interacts with high-affinity GnRH-Rs. The gene encoding the GnRH-R is located on chromosome 4q13.2-13.3, spanning 18.9 kb. This receptor belongs to a large family of G protein–coupled receptors . These contain seven transmembrane helices connected by six alternating intracellular and extracellular loops, with the amino-terminus located on the extracellular side. In contrast to other protein receptors (see Fig. 4.10 , presented later in the chapter), the GnRH-R lacks a carboxy-terminus located on the intracellular site.
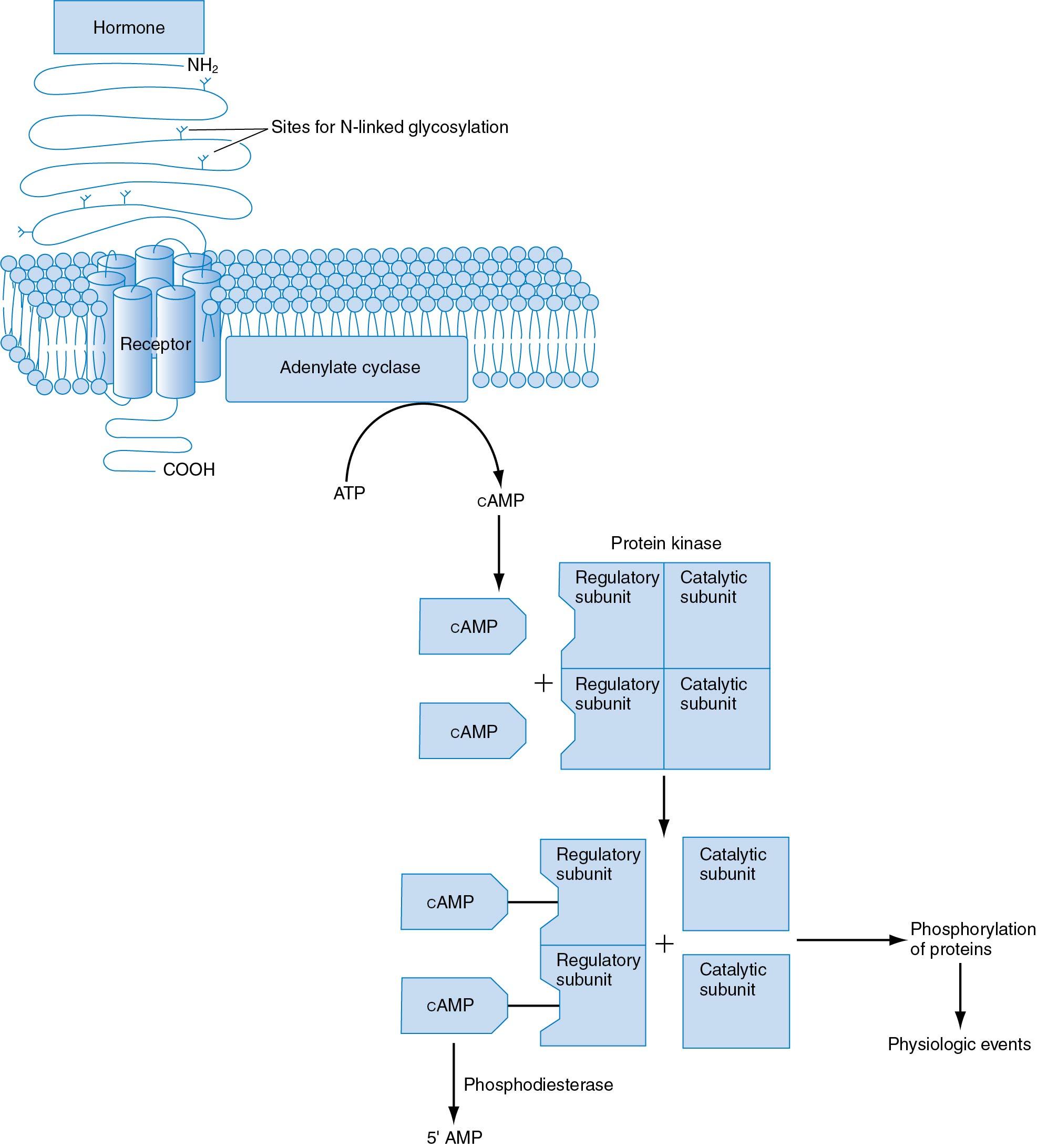
GnRH activation of the receptor requires the release of constraining intramolecular bonds, which maintain the receptor in an inactive configuration. Once activated, the GnRH-R stimulates cellular production of specific membrane-associated lipid-like diacylglycerols, which, acting as a second messenger, activate several cellular proteins. Among these are the enzyme protein kinase C (PKC) and mitogen-activated protein kinase (ERK; also called MAPK). Phosphorylated ERK activates transcription factors, the end result being gene transcription of gonadotropin subunits and the synthesis of both gonadotropins.
Binding of GnRH to its receptor also rapidly mobilizes transient intracellular calcium , which triggers a burst of exocytosis to rapidly release LH and FSH. It also provokes a rapid influx of Ca + + into the cell from the extracellular pool, which in turn activates calmodulin, a calcium-binding protein, maintaining gonadotropin release. Diacylglycerols amplify the action of Ca + +-calmodulin, thereby synergistically enhancing the release of gonadotropins. Administration of a calmodulin antagonist has been shown to decrease GnRH-stimulated gonadotropin release.
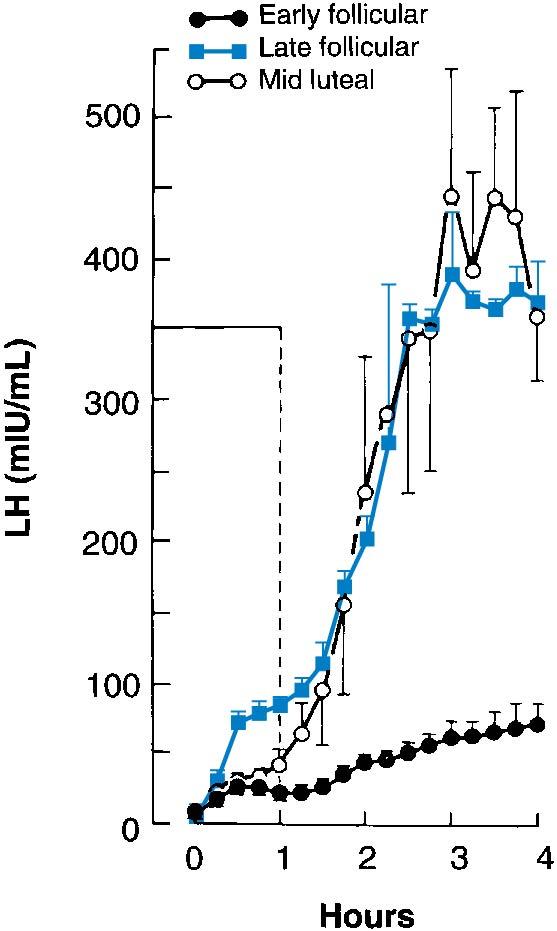
Pulsatile GnRH increases GnRH-R gene expression and the number of GnRH-Rs on the gonadotrope’s cell surface. The number of GnRH-Rs also varies with the hormonal environment, with highest number of receptors expressed when high concentrations of estrogens are present. This leads to an increase in the overall Ca2 + + response and a significantly amplified gonadotropin response to a GnRH pulse. This action explains the variations in the gonadotropin response to GnRH at various times of the menstrual cycle : GnRH pulses of similar amplitude elicit greater gonadotropin responses during the late follicular phase and luteal phase when estradiol levels are highest, but the responses are lower during the early follicular phase when estradiol levels are lowest ( Fig. 4.5 ).
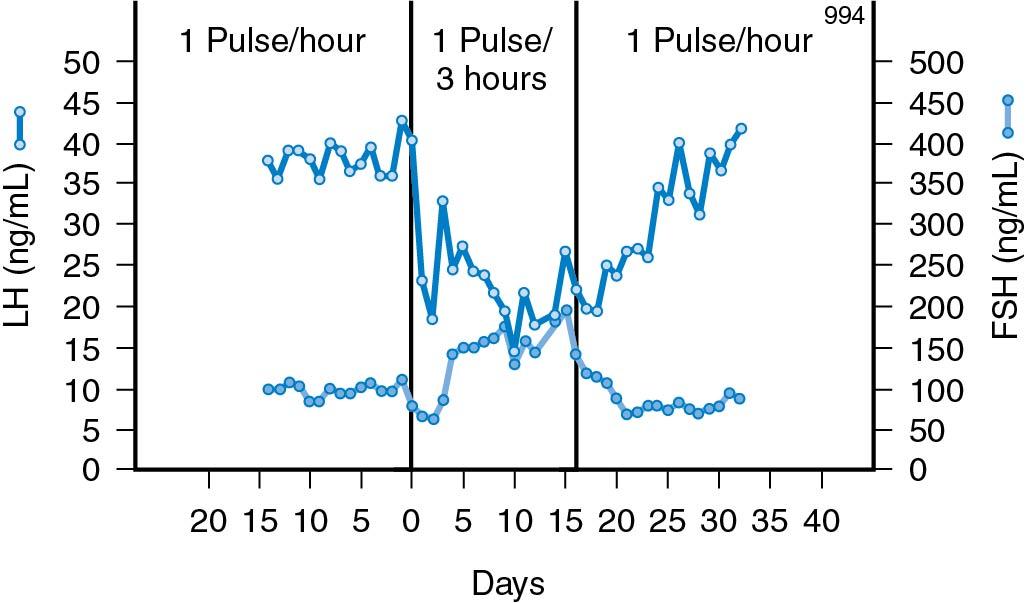
It is also important to note that varying frequencies of the GnRH pulse signal regulate gonadotropin subunit gene transcription differentially. Overall, a low GnRH pulse frequency favors FSH synthesis, whereas a high GnRH pulse frequency favors LH synthesis . This is well demonstrated experimentally where changing a pulsatile infusion from a high to a low pulse frequency results in a matter of days in an increase in the FSH/LH ratio ( Fig. 4.6 ). This phenomenon may play a role during the luteal phase of the menstrual cycle and in the changing FSH/LH ratio that occurs during the passage from one menstrual cycle to another (see The Luteal Phase, discussed later). It is also reflected in patients known to have a high GnRH pulse frequency, such as in women with the polycystic ovary syndrome, a high proportion of whom have a characteristically elevated LH:FSH ratio .
Gonadotropin release after a GnRH pulse is rapid: Within minutes, both FSH and LH are released. It is important to recognize that the pulsatile release mode of GnRH is essential for the maintenance and control of normal gonadotropin secretion.
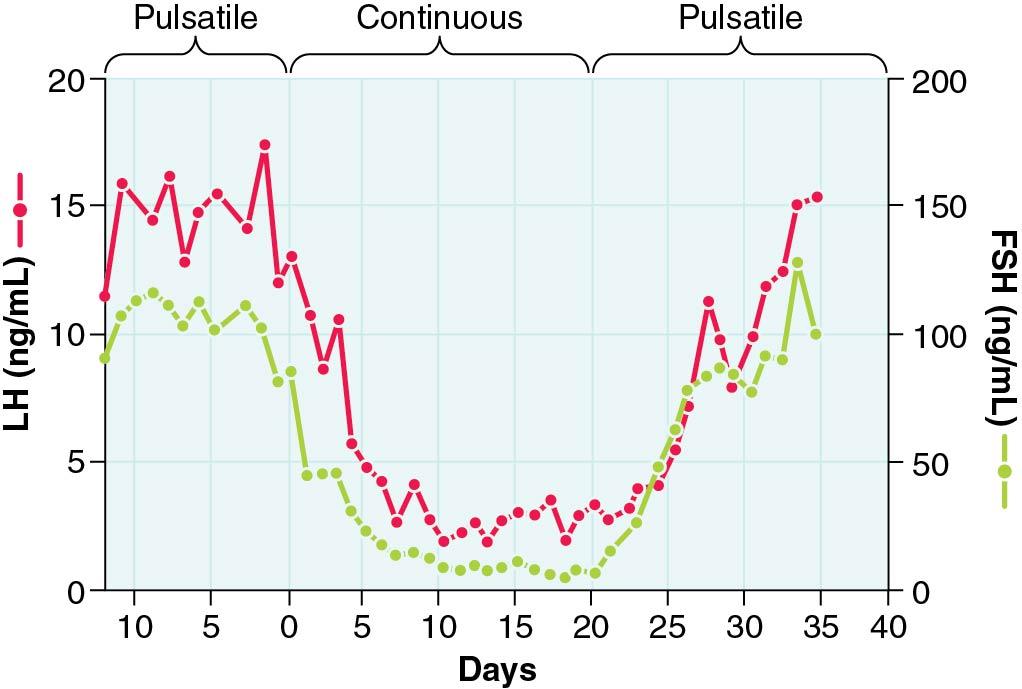
In contrast to the response to the normal pulsatile mode of GnRH release, sustained exposure of the GnRH-R to constant GnRH concentrations drastically reduces the response of the gonadotrope to subsequent stimulation with GnRH. This phenomenon is referred to as homologous desensitization or downregulation of the receptor, which denotes a reduction in the ability of GnRH to elicit gonadotropin release after prior continuous exposure to GnRH. This phenomenon is well illustrated in a classic experiment in ovariectomized monkeys lacking endogenous GnRH secretion after lesion of the arcuate nucleus ( Fig. 4.7 ). As illustrated, 6-minute pulses administered once an hour restored normal LH levels in these animals. In contrast, when a continuous mode of GnRH infusion was substituted to the pulsatile mode, a profound inhibition of LH concentrations occurred, which reflects desensitization of the GnRH-R. This phenomenon, which takes a few days to be established, may reflect a loss of active cell surface receptors and be maintained by a loss of functional Ca + + channels. However, the mechanism of desensitization is still under investigation, and additional intermediary changes remain to be characterized.
The GnRH half-life in the peripheral circulation is very short because peptidases rapidly degrade naturally occurring GnRH by cleaving the decapeptide molecule at the Gly 6 to Leu 7 and at the Pro 9 to Gly 10 bonds. However, by substituting amino acid 6 in the natural GnRH molecule with a d-amino or replacing amino acid 10 with an N-methylamide (Na-CH2-CH3) or Aza-Gly (NHNHCO) moiety, GnRH analogs were synthesized and shown to have acquired a greater resistance to enzymatic proteolysis and hence a longer half-life (hours vs. 2 to 4 minutes). After administration of these GnRH agonists, there is an initial stimulation of gonadotropin release (flare), followed by the process of desensitization blocking the releasing effect on the gonadotropins . This observation has led to the clinical application of the functional desensitization property of GnRH:GnRH analogs; they have been used to induce a “medical castration” state by shutting down the pituitary-gonadal axis in a variety of clinical conditions. In contrast to GnRH agonists, GnRH antagonists act by competing with GnRH for receptor sites and thereby never activating a stimulatory signal. Many of these result from the substitution of amino acids at the 2 or 3 position. Thus GnRH antagonists have the advantage over the GnRH agonists of a rapidly decreasing LH and FSH release, without the flare . Clinical applications for both GnRH agonists and antagonists are listed in Box 4.1 . Although GnRH agonists have been in use longer and have several Food and Drug Administration (FDA)–approved indications, the GnRH antagonists, though theoretically useful for the same conditions, are only approved for a few indications at present. An orally active GnRH antagonist, elagolix (Orilissa), is available in the United States and has been approved in two dosages, 150 and 200 mg twice daily, for the treatment of severe endometriosis.
Activation of pituitary–gonadal function (GnRH)
Delayed puberty
Cryptorchidism
Functional hypothalamic amenorrhea
Hypogonadotropic hypogonadism (Kallmann syndrome)
Pituitary–gonadal inhibition (agonists)
Precocious puberty
Hormone-dependent tumors
Endometriosis
Uterine leiomyoma
Breast cancer
Prostatic cancer
Suppression of ovarian function in polycystic ovary syndrome and in vitro fertilization
Premenstrual syndrome
Abnormal uterine bleeding including clotting disorders
Contraception
Suppression of spermatogenesis
Ovulation inhibition
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here